Abstract
Coronary atherosclerotic heart disease is an important threat to human health. The pathological basis is atherosclerosis, and foam cell formation is the key link in the initiation of atherosclerosis. Here, foam cell models were established using 50 ng/ml oxidized low-density lipoprotein (ox-LDL) to stimulate in vitro cultures of THP-1 cells for 72 h. The expression of ZNF580, a Cys2–His2 (C2H2) zinc finger protein containing 172 amino acids that was originally cloned by screening a human aortic cDNA library, was measured in foam cells, and its interaction with various regulatory factors during foam cell formation was investigated. Oil red O (ORO) staining was used to observe cell morphology and intracellular lipid levels. Lentivirus transfection was used to induce high ZNF580 expression (Ad-ZNF580) and low ZNF580 expression (Si-ZNF580) in THP-1 cells, and a fluorescent inverted microscope was used to observe the distribution of ZNF580 immunofluorescence to deduce the transfection rate. RNA and total protein were extracted, and the expression levels of ZNF580, cluster of differentiation 36 (CD36), peroxisome proliferator activated receptor-γ (PPAR-γ), ATP-binding cassette transporter A1 (ABCA1) and apolipoprotein E (ApoE) were measured by real-time quantitative PCR. The protein levels were examined by western blotting to evaluate the interaction between ZNF580 and associated regulatory factors. ZNF580 can significantly increase the expression levels of ApoE and ABCA1 and significantly decrease the expression levels of CD36 and PPAR-γ, suggesting that ZNF580-mediated inhibition of foam cell formation is associated with the PPAR-γ-CD36 signalling pathway. Based on these findings, ZNF580 might be a potential therapeutic candidate for the treatment of coronary atherosclerotic heart disease.
Introduction
Cardiovascular disease (CVD) is an important threat to the health of middle-aged and elderly individuals and leads to a decline in quality of life, and coronary atherosclerotic heart disease is an important part of CVD [1]. The pathological basis of coronary atherosclerotic heart disease is the development of coronary atherosclerosis, in which the production and accumulation of foam cells are important [2]. Foam cells are fatty macrophages that cause artery hardening and may further lead to heart disease. Foam cells are the first visible lesion feature in atherosclerotic diseases [3]. Circulating monocytes are recruited to the damaged vascular endothelium from the blood, penetrate the subintima through the endothelial space and differentiate into macrophages. Macrophages oxidize accumulated lipoproteins under the intima. A large amount of oxidized lipoprotein enters macrophages, which form foam cells, and smooth muscle cells also participate in foam cell formation [4]. The accumulation of foam cells forms the inner core and lipid streaks of coronary atherosclerotic plaques. In the late stage of atherosclerosis, foam cells undergo necrosis or apoptosis, resulting in lipid leakage and formation of a necrotic core, which increases plaque instability. The occurrence and development of foam cells contributes to the pathophysiological process of atherosclerosis [5]. Therefore, understanding the mechanism by which foam cells form and reducing foam cell formation factors can effectively reduce the formation of atherosclerotic plaques in coronary arteries.
Scavenger receptors (SRs) play an important role in foam cell formation and are a significant mechanism by which macrophages identify, ingest and engulf lipoprotein oxide [6]. SRs are present on the cell surface in a variety of molecular forms and are mainly divided into five types: A, B, C, D, and E. Cluster of differentiation 36 (CD36) is a member of the Class B SR family and is the main receptor by which macrophages consume lipoprotein oxide. When oxidized low-density lipoprotein (ox-LDL) enters macrophages, which form foam cells, CD36 activation is necessary [7]. Since CD36 is not regulated by negative feedback from intracellular cholesterol esters during lipoprotein uptake, the formation of tissue foam cells must be regulated by modulating the regulatory factors upstream of CD36. Macrophage-produced factors, such as interleukin-4 and peroxisome proliferator activated receptor-γ (PPAR-γ), are upstream regulatory factors that can regulate CD36 expression, and PPAR-γ is widely distributed in adipose tissue, vascular smooth muscle, and cardiomyopathy tissue. There it participates in lipid metabolism, inflammatory reactions, atherosclerosis and other pathological processes [8]. Studies have shown that PPAR-γ activation inhibits foam cell formation by increasing foam cell apoptosis while also regulating the activation of ATP-binding cassette transporter A1 (ABCA1). This occurs by increasing the expression of liver X receptor-α to affect cholesterol outflow and delay the formation of atherosclerotic plaques [9]. In addition, studies have shown that apolipoprotein E (ApoE) has a similar function as ABCA1, which transports excess cholesterol esters from within cells to the extracellular space, reducing lipid build-up in cells and delaying foam cell formation [10]. Therefore, the aim of this study was to find drugs that act on these targets to prevent foam cell formation.
ZNF580 (GenBank: AF184939) is a Cys2–His2 (C2H2) zinc finger protein containing 172 amino acids that was originally cloned by screening a human aortic cDNA library. The protein contains a highly conserved C-terminus, three tandemly repeated C2H2 zinc finger domains, and a proline-rich N-terminus. The protein structure of ZNF580 is similar to Sp1-like or Krüppel-like transcription factors, and it is also characterized by three tandemly repeated C2H2 zinc fingers at the C-terminus [11]. A previous investigation showed that ZNF580 could protect against CVD through multiple signalling pathways: ZNF580 regulates eNOS expression via the TGF-β1/ALK5/Smad2 pathway and mediates vascular endothelial inflammation though the H2O2/NF-κB signalling pathway [12, 13]. Single-core macrophages or smooth muscle cells are usually used to replicate foam cell models; the available cells are human leukaemic monocyte-1 (THP-1) cells, rat celiac macrophages, animal aortic smooth muscle cells, and THP-1 cells are the most commonly used [14, 15]. THP-1 is a human mononucleotic cell line that is induced by phorbol 12-myristate 13-acetate (PMA) to differentiate into macrophages to build a foam cell model. Moulding is usually used with oil red O (ORO) staining for model identification. However, whether ZNF580 affects the initial link in atherosclerotic plaque formation has not yet been studied. Therefore, this study used lentiviruses to express and silence ZNF580 to identify genes related to foam cell formation and clarify the role of ZNF580 in foam cell formation. The experimental results provide a new theoretical basis for the treatment of coronary atherosclerotic heart disease.
Materials and methods
Cell culture
THP-1 cells were obtained from the Tianjin Key Laboratory of Hepatopancreatic Fibrosis and Molecular Diagnosis & Treatment (Tianjin, China), and the cells were cultured in Roswell Park Memorial Institute-1640 culture medium (RPMI-1640, Gibco) supplemented with 10% foetal bovine serum (FBS, Gibco) and 1% penicillin/streptomycin. THP-1 cells were grown in an incubator with 100% humidity containing 95% air and 5% CO2 at 37 °C. Depending on cell growth, the medium was changed once every 2–3 days and passaged every 3 days. The model was established with third-generation cells.
Cell transfection
THP-1 cells in the logarithmic growth phase were uniformly inoculated at 2×105 cells/well in a 6-well plate. According to the manufacturer’s instructions, THP-1 cells were transfected with 20μl/well ZNF580 lentivirus using Lipofectamine 2000 (Thermo Fisher Scientific, Inc.) in an incubator with 100% humidity containing 95% air and 5% CO2 at 37 °C for 96 h. The negative control groups were experimented with no-loaded lentivirus in the same environment. FBS-free RPMI-1640 medium was used during transfection. The transfection efficiency was monitored by fluorescence microscopy.
Cell model
THP-1 cells were inoculated at 1×105 cells/well in a 6-well plate at 37 °C with 5% CO2 for 12 h. After stimulation with 10 ng/ml PMA, THP-1 cells adhered to the wells and became macrophages. After the RPMI-1640 medium was changed, 50 ng/ml ox-LDL was added, and the cells were cultured for 24 h, 48 h, and 72 h to establish the model [16, 17].
Cell groupings
THP-1 cells were grown on plates and randomly divided into six groups: 1) in the control group, THP-1 cells were grown in an incubator containing 95% air and 5% CO2 at 37 °C after stimulation with 10 ng/ml PMA, and no additional treatment was performed; 2) in the control+ox-LDL (Model group) group, after 10 ng/ml PMA was added to control group cells, 50 ng/ml ox-LDL was added, and the cells were cultured in an incubator containing 95% air and 5% CO2 at 37 °C for 72 h; 3) in the Ad-ZNF580 group, THP-1 cells in the control group were transfected with 20μl/well Ad-ZNF580 lentivirus, and no additional treatment was performed; 4) in the Ad-ZNF580 + ox-LDL (Ad-ZNF580 + Model group) group, cells in the Ad-ZNF580 group were administered 50 ng/ml ox-LDL and cultured in an incubator containing 95% air and 5% CO2 at 37 °C for 72 h; 5) in the Si-ZNF580 group, THP-1 cells in the control group were transfected with 20μl/well Si-ZNF580 lentivirus, and no additional treatment was performed; and 6) in the Si-ZNF580 + ox-LDL (Si-ZNF580 + Model group) group, the cells in the Si-ZNF580 group were administered 50 ng/ml ox-LDL and cultured in an incubator containing 95% air and 5% CO2 at 37 °C for 72 h.
Observation of cell morphology and intracellular lipid levels
ORO staining was used to observe cell morphology and intracellular lipid levels. The cells in the different groups were sequentially washed with phosphate-buffered saline (PBS) and deionized water 2-3 times and incubated with ORO fixative for 20 min. After the cells were washed 2-3 times, ORO stain was added and incubated for 20 min, and the cells were then placed in Yin Hong solution for 1 min. Finally, the cells were dried in a ventilated place, and cell morphology and intracellular lipid levels were observed using an optical microscope.
RNA extraction and reverse transcription-quantitative PCR (RT–qPCR)
A total of 1×105 THP-1 cells per well were inoculated into 6-well plates and cultured at 37 °C with 5% CO2 for 24 h. RT–qPCR was used to measure the expression of ABCA1, CD36, ApoE, PPAR-γ, and ZNF580 in each group. TRIzol reagent (Invitrogen, USA) was used to extract total RNA, and a UV spectrophotometer was used to measure RNA purity. Then, the RNA was reverse transcribed into cDNA using a HIFIScript cDNA Synthesis Kit (ComWin Biotech, Beijing, China) according to the manufacturer’s instructions. The cDNA templates were analysed using RT–qPCR with UltraSYBR Mixture Low ROX (ComWin Biotech, Beijing, China) under the following conditions: 40 cycles of 10 s at 95 °C, 30 s at 60 °C, and 32 s at 72 °C. The nucleotide sequences of the forward and reverse primers are shown in Table 1. The relative expression level of each mRNA was calculated using the 2-ΔΔCq method [18].
Protein preparation and western blot analysis
THP-1 cells were washed three times with PBS. Samples from the different groups were lysed in complete RIPA buffer (R0020, Solarbio, China) at 4 °C for 15 min. The total protein concentrations were determined using a BCA kit (A045-4-2, Jiancheng, Nanjing). Equal amounts of protein were loaded onto 10% polyacrylamide gels and transferred to nitrocellulose membranes (Millipore, Billerica, MA, USA). Then, the membranes were blocked with Tris-buffered saline plus Tween-20 (TBST) containing 5% skim milk for 3 h at 25 °C, after which the membranes were incubated with the following primary antibodies overnight at 4 °C: ZNF580 (PA5-62904, 1 : 1000, Thermo), ApoE (cat. no. 18254-1-AP, 1 : 2000, Proteintech), CD36 (cat. no. 18836-1-AP, 1 : 2000, Proteintech), ABCA1 (AB7360, 1 : 1000, Abcam), PPAR-γ (cat. no. 16643-1-AP, 1 : 2000, Proteintech) and GAPDH (cat. no. 5174; 1 : 1000, Cell Signalling Technology, Inc.). The membranes were then incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies (cat. no. 7074 V; 1 : 5000; Cell Signalling Technology, Inc.) for 1 h at 25 °C. Signals were observed using ECL reagents (Thermo Fisher Scientific) according to the manufacturer’s instructions. Band densities were measured using ImageJ 1.52a software (National Institutes of Health, USA).
Statistical analysis
The data are presented as the means±standard deviation (n = 10). Statistical comparisons between groups were assessed using a paired t-test or one-way ANOVA followed by Tukey’s post hoc test with SPSS version 25.0 statistical software (IBM Corp.). A value of P < 0.05 was considered significant. All experiments were repeated at least three times.
Results
Foam cell model verification
Under normal conditions, THP-1 cells grow in suspension, and the cells are round or oval in shape. After PMA stimulation, the cells developed a long spindle-like morphology with pseudopodia, grew adherently, and differentiated into macrophages (Fig. 1A). When stimulated by 50 ng/ml ox-LDL, macrophages developed into foam cells, and intracellular lipid droplets could be stained with ORO (Fig. 1B–D). With prolonged ox-LDL stimulation, the number of ORO-positive cells increased significantly. Furthermore, the degree of ORO dye aggregation increased significantly, and the colour darkened, suggesting that the levels of lipid droplets in macrophages increased and foam cells formed more fully with prolonged ox-LDL incubation time (Fig. 1E).
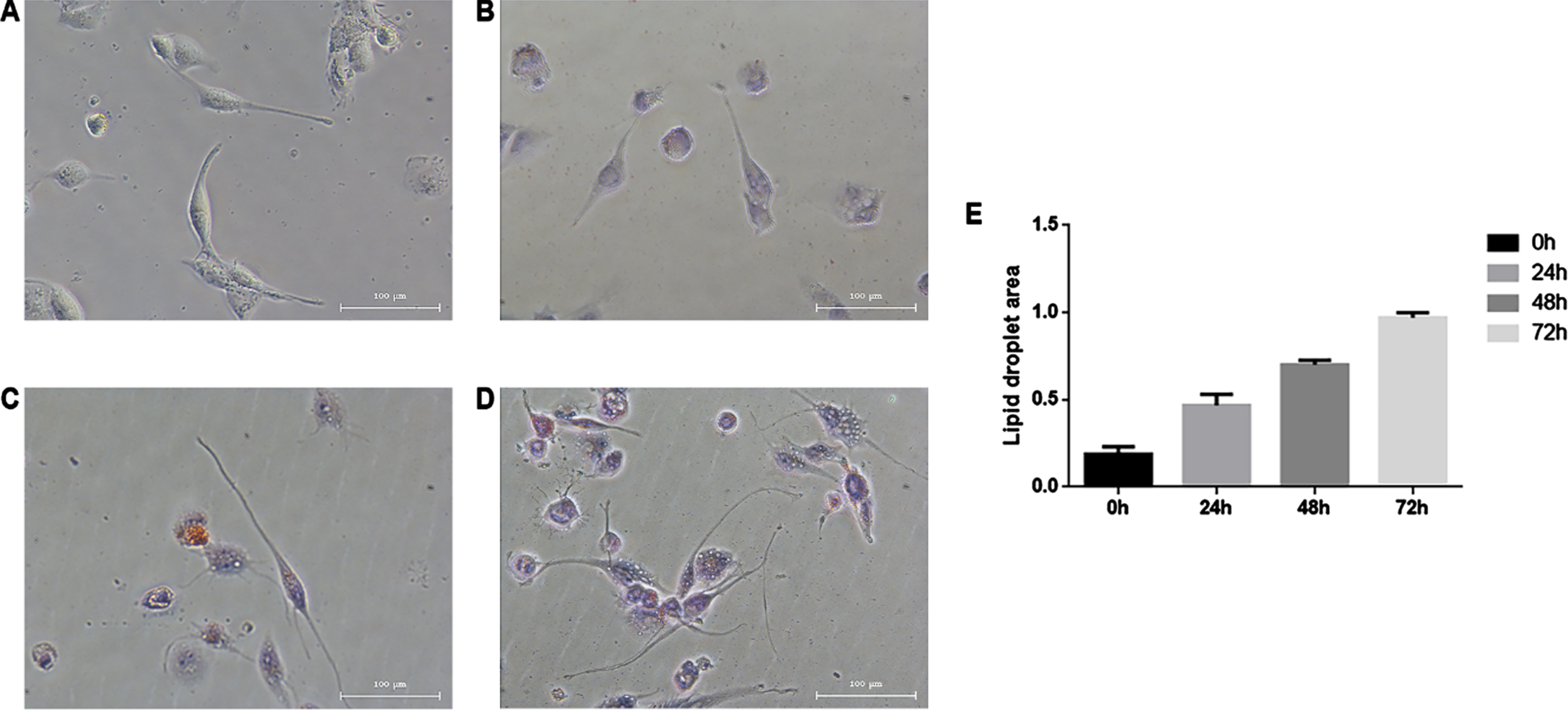
Ox-LDL mediates the transformation of macrophages into foam cells. The cells in the control group (A) had a long spindle-like morphology with full cytoplasm and pseudopodia and grew adherently. After incubation with ox-LDL for 24 h (B), 48 h (C), and 72 h (D), the lipid droplet levels in macrophages gradually increased, and the number of ORO-stained cells increased significantly (E). Magnification,×100.
ORO staining confirmed that ox-LDL-stimulated macrophages could successfully establish a foam cell model. To determine the optimal stimulation time, 50 ng/ml ox-LDL was used to stimulate foam cell formation in macrophages for different times. The expression of CD36 mRNA increased in the control group with increasing ox-LDL induction times, and CD36 expression was highest at 72 h, suggesting a time-dependent effect on CD36 expression in normal macrophages. In the model group, CD36 mRNA expression levels were consistent with those in the control group, and the growth trends from 24 h to 48 h were comparable. However, there were no significant differences in expression levels between 48 h and 72 h. At 72 h, the mRNA expression of CD36 in the control group was 1.5 times that in the model group, and the difference was statistically significant (P < 0.05; Fig. 2A). The mRNA expression of PPAR-γ did not differ significantly at different times in the control group, while the mRNA expression of PPAR-γ increased with prolonged ox-LDL induction time in the model group; the mRNA expression of PPAR-γ was highest at 72 h and was significantly higher than that in the control group (P < 0.05; Fig. 2B). The mRNA expression of the lipid flow-related genes ABCA1 and ApoE was consistent in the control group and the model group. The mRNA expression levels of ABCA1 and ApoE increased with prolonged ox-LDL induction time; the mRNA expression of ABCA1 and ApoE was the highest at 72 h, and the difference was statistically significant (P < 0.05; Fig. 2C-D). Therefore, 72 h was chosen as the optimal time for ox-LDL to induce foam cell formation in macrophages.
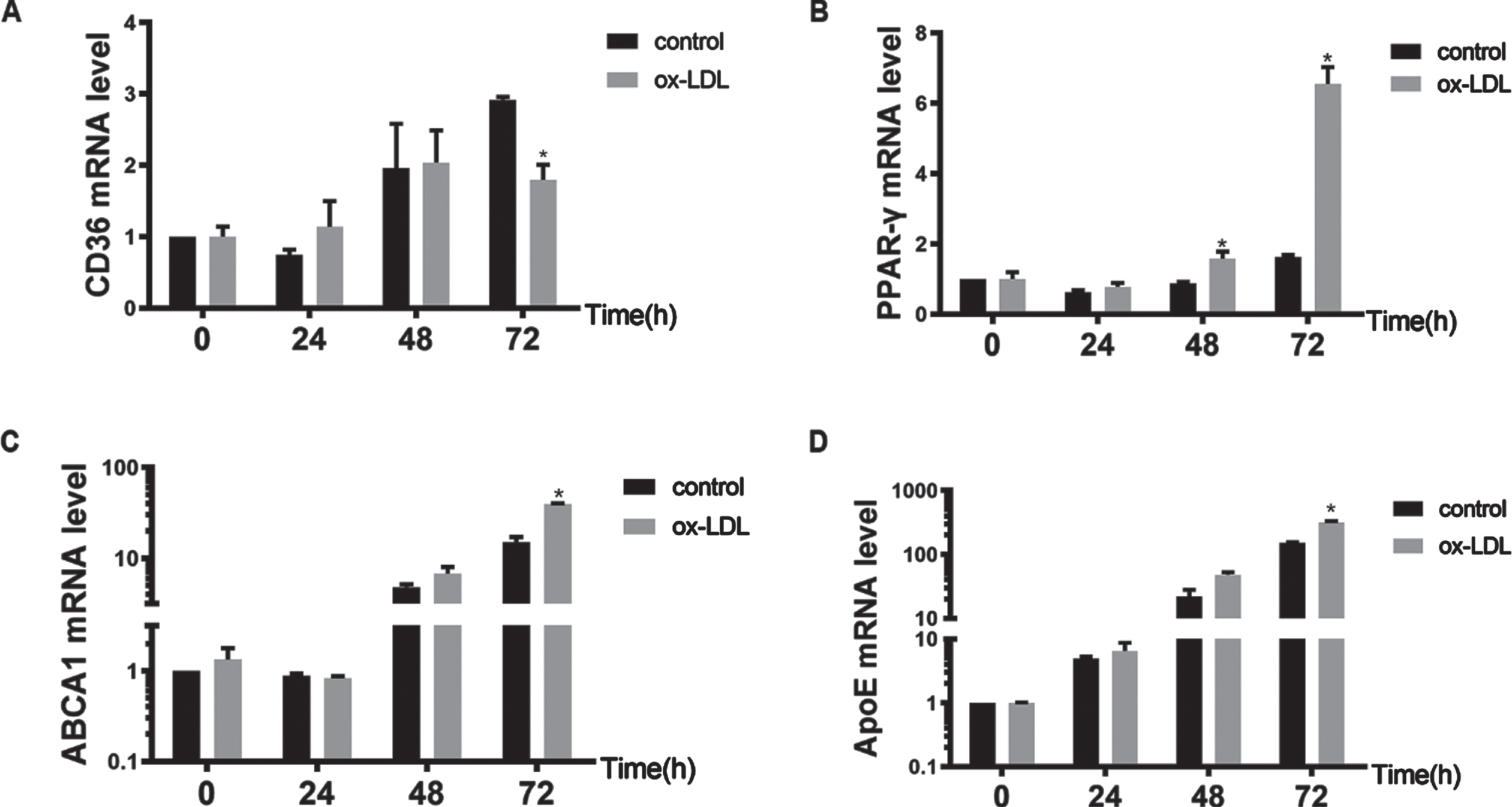
Model foam cells show changes in lipid metabolism-related mRNA expression at different time points. The mRNA levels of lipid metabolism-related genes, including CD36 (A), PPAR-γ (B), ABCA1 (C) and ApoE (D), were measured by RT-qPCR. The mRNA expression levels of CD36, PPAR-γ, ABCA1 and ApoE increased with prolonged ox-LDL induction time compared with those of the control group. At 72 h, the mRNA expression of CD36 in the control group was 1.5 times that of the model group, and the mRNA expression of PPAR-γ, ABCA1 and ApoE was highest at 72 h. Therefore, 72 h was chosen as the optimal time for ox-LDL to induce macrophages to form foam cells. *P < 0.05 compared with the control group. ox-LDL, oxidized low-density lipoprotein; CD36, cluster of differentiation 36; PPAR-γ, peroxisome proliferator activated receptor-γ; ABCA1, ATP-binding cassette transporter A1; ApoE, apolipoprotein E.
The expression of ZNF580 in each group was analysed by RT-qPCR and western blotting to examine whether foam cell formation was associated with ZNF580 expression (Fig. 3). There was no significant difference between the control group and the model group when the ox-LDL induction time was 24 h; when the induction time was 48 h and 72 h, the mRNA expression level of ZNF580 in the model group was significantly higher than that in the control group (P < 0.05; Fig. 3A). The western blot results (Fig. 3B) were consistent with the RT-qPCR results, suggesting an increase in ZNF580 expression upon foam cell formation. Compared with that in the control group, the mRNA expression of ZNF580 peaked in the model group at 72 h, and the difference was statistically significant (P < 0.05; Fig. 3C). The western blots results (Fig. 3D) were consistent with the PCR results, suggesting an increase in ZNF580 expression when foam cells formed at 72 h.
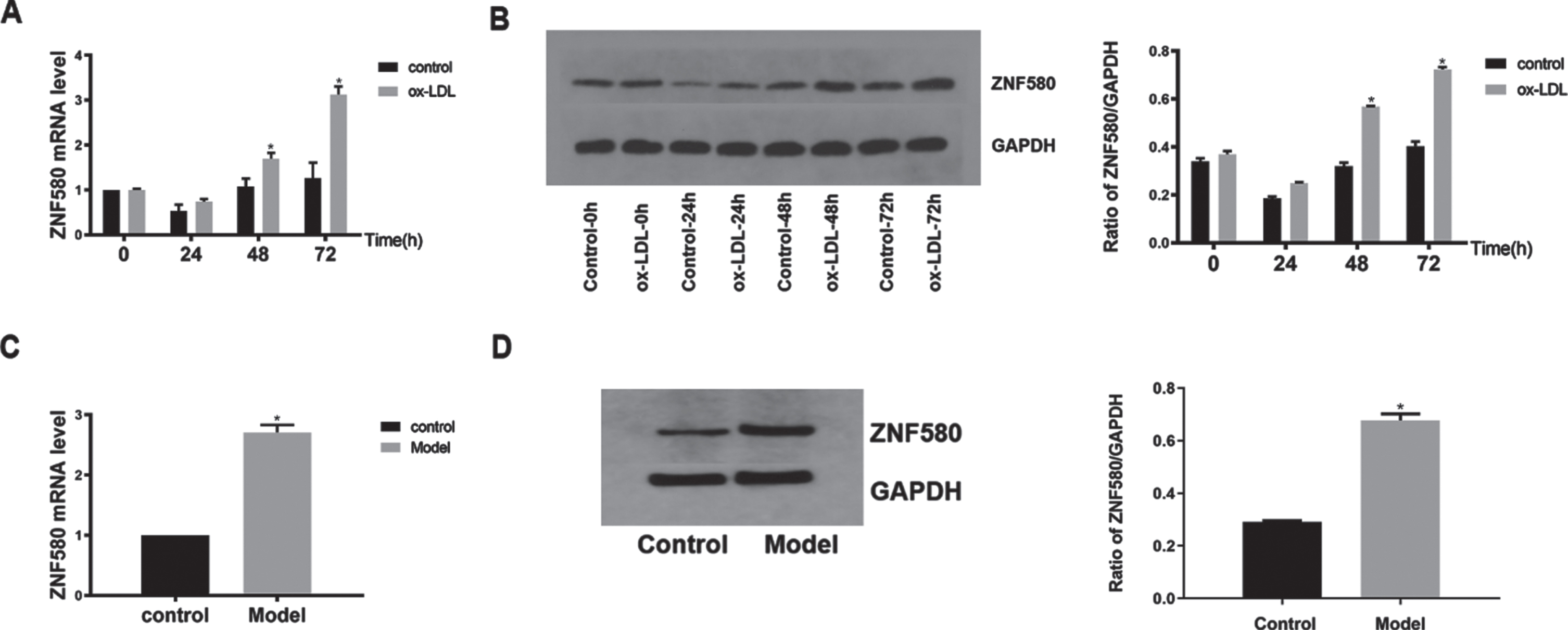
The expression of ZNF580 in foam cells at different times. (A) After ox-LDL induction for different times, the levels of ZNF580 mRNA in each group were measured using RT–qPCR. (B) The levels of proteins involved with ZNF580 were measured using western blotting. (C) After ox-LDL induction for 72 h, the mRNA levels of ZNF580 in the control group and model group were measured using RT–qPCR. (D) The levels of proteins involved with ZNF580 in the control group and model group after 72 h of ox-LDL induction were measured by western blotting. *P < 0.05 compared with the control group. ox-LDL, oxidized low-density lipoprotein; ZNF580, zinc finger gene 580.
Fluorescence microscopy showed green fluorescence in the cytoplasm and around the nucleus in THP-1 cells after successful transfection, while untransfected cells had no green fluorescence (Fig. 4A-B). The expression of ZNF580 in each group was analysed by RT-qPCR and western blotting to examine whether the cells were successfully transfected (Fig. 4). Compared with that in the control group, ZNF580 mRNA was significantly increased by Ad-ZNF580 and was significantly decreased by Si-ZNF580 (P < 0.05; Fig. 4C). There was no significant difference between the control virus and control groups, suggesting that the lentivirus had no effect on the cells. The protein expression levels of ZNF580 in the different groups were analysed by western blotting (Fig. 4D), and the results were consistent with the mRNA levels, indicating that THP-1 cells were successfully transfected with the Ad-ZNF580 and Si-ZNF580 lentiviruses.
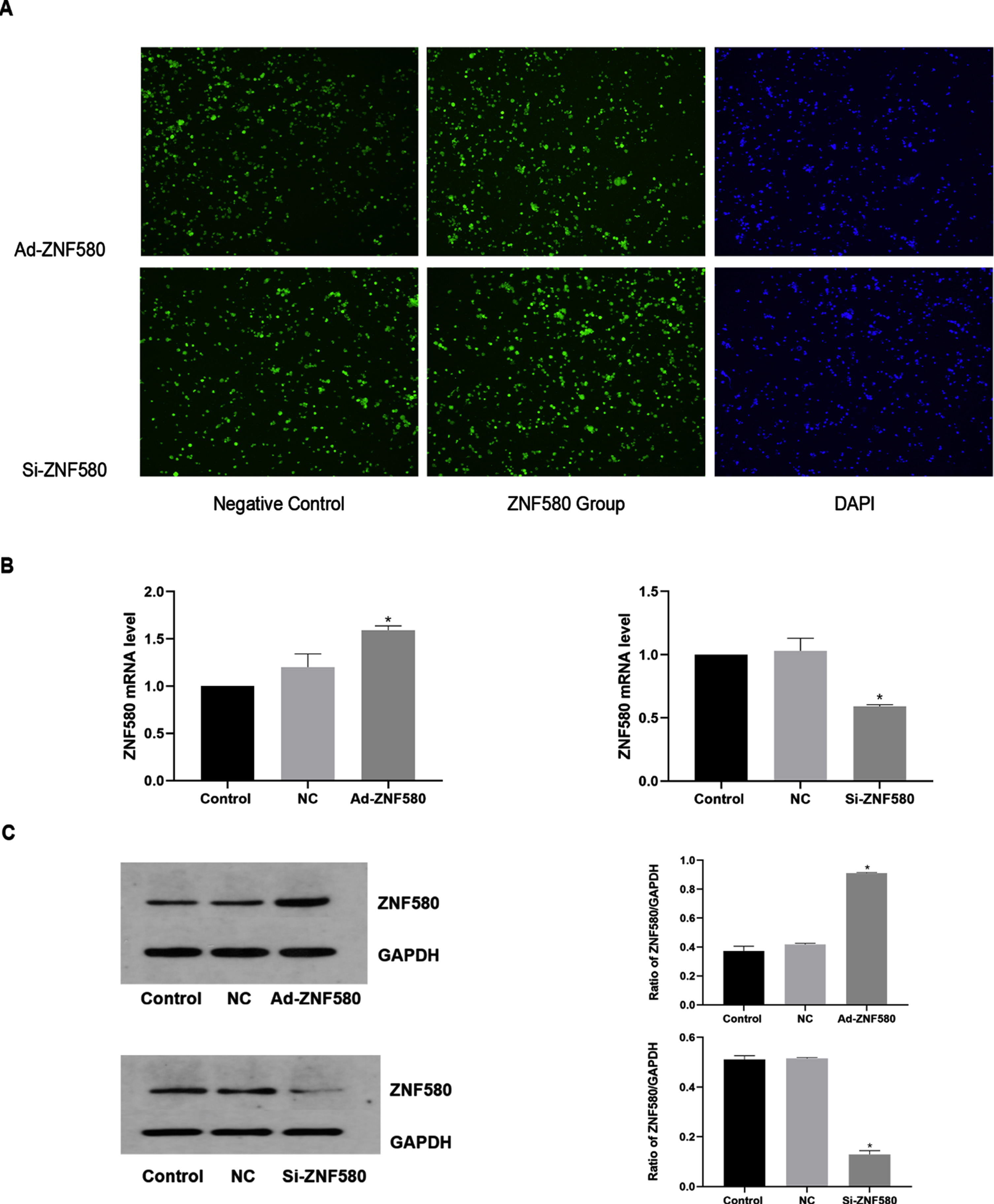
Ad-ZNF580 and Si-ZNF580 lentivirus transfection. (A) Fluorescence microscopy was used to examine the conversion efficiency of Ad-ZNF580 lentivirus (×200). Green fluorescence was visible in the cytoplasm and around the nucleus in THP-1 cells. Fluorescence microscopy was used to examine the conversion efficiency of Si-ZNF580 lentivirus (×200). Green fluorescence was visible in the cytoplasm and around the nucleus in THP-1 cells. (B) The level of ZNF580 mRNA expression was measured by RT–qPCR. (C) The levels of ZNF580 protein in THP-1 cells transfected with lentivirus were measured by western blotting. *P < 0.05 compared with the control group. THP-1, human leukaemic monocyte-1; ZNF580, zinc finger gene 580; Ad-ZNF580, high expression of ZNF580; Si-ZNF580, low expression of ZNF580; NC, negative control.
As shown in the ORO staining images (Fig. 5A), the Ad-ZNF580 model group had decreased numbers of ORO-stained cells compared to the model group, and the lipid droplet area was smaller than that of the model group (Fig. 5B). Compared with those in the model group, the numbers of ORO-stained cells increased in the Si-ZNF580 model group (Fig. 5A), and the lipid droplet area was larger than that of the model group (Fig. 5C), indicating that ZNF580 inhibited foam cell formation.
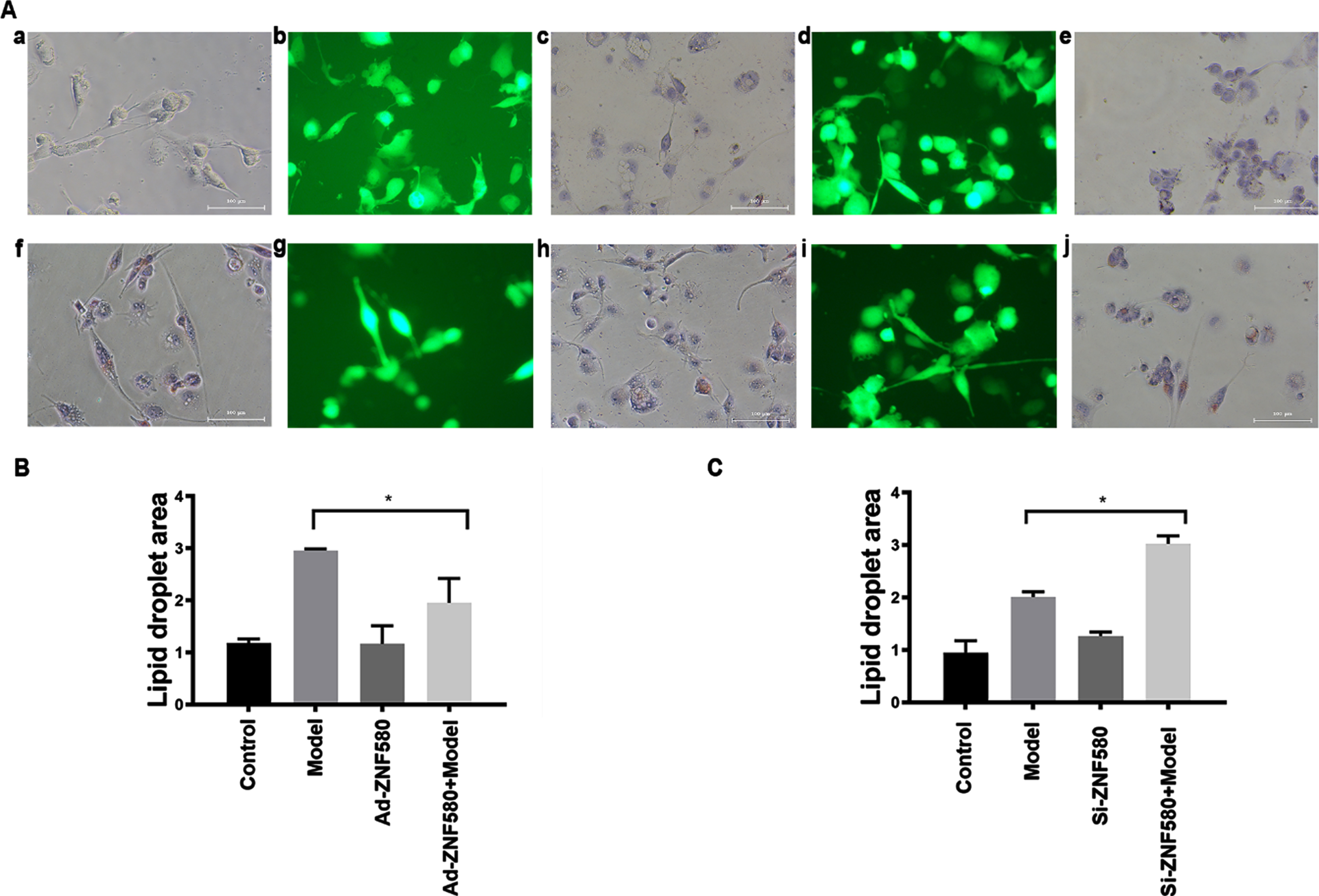
The establishment of foam cells in Ad-ZNF580- and Si-ZNF580-transfected cells. (A) Morphological changes in cells in the (a) control, (b) Ad-ZNF580 (fluorescence microscopy), (c) Ad-ZNF580 (ORO staining), (d) Si-ZNF580 (fluorescence microscopy), (e) Si-ZNF580 (ORO staining), (f) model, (g) Ad-ZNF580 + model (fluorescence microscopy), (h) Ad-ZNF580 + model (ORO staining), (i) Si-ZNF580 + model (fluorescence microscopy) and (j) Si-ZNF580 + model (ORO staining) groups. (B) Changes in the lipid droplet area in Ad-ZNF580 foam cells, as determined by ORO staining. (C) Changes in the lipid droplet area in Si-ZNF580 foam cells, as determined by ORO staining. *P < 0.05 compared with the model group. Magnification,×100. ZNF580, Zinc finger gene 580; Ad-ZNF580, high expression of ZNF580; Si-ZNF580, low expression of ZNF580.
To determine whether ZNF580 could prevent foam cell formation by regulating ApoE transcription and increasing cholesterol outflow (Fig. 6A), RT–qPCR was performed. The results showed that the mRNA expression of ApoE in the model group was higher than that in the control group. Compared with the model group, the mRNA expression level of ApoE increased significantly in the Ad-ZNF580 model group, the mRNA expression level of ApoE decreased significantly in the Si-ZNF580 model group, and the difference was statistically significant (P < 0.05). The western blot results were consistent with the mRNA levels, indicating that ZNF580-mediated inhibition of foam cell formation was associated with the regulation of ApoE (P < 0.05).
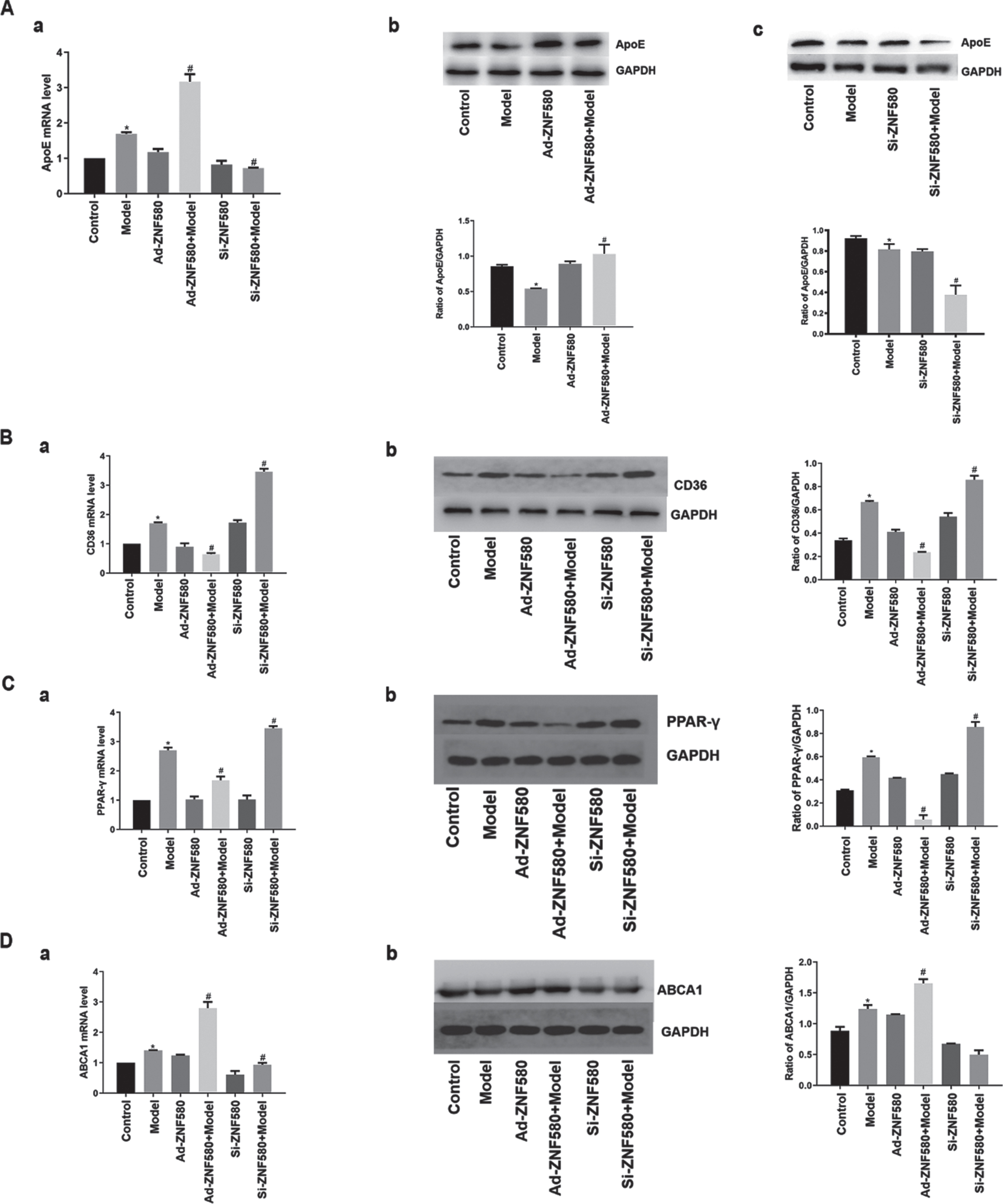
ZNF580 inhibits foam cell formation by regulating the expression of lipid-related genes. (A) The expression of ApoE in Ad-ZNF580 and Si-ZNF580 foam cells was measured by RT–qPCR (a) and western blotting (b). The expression of CD36 (B), PPAR-γ (C) and ABCA1 (D) in Ad-ZNF580 and Si-ZNF580 foam cells was measured by RT–qPCR and western blotting. *P < 0.05 compared with the control group; #P < 0.05 compared with the model group. ZNF580, zinc finger gene 580; Ad-ZNF580, high expression of ZNF580; Si-ZNF580, low expression of ZNF580; CD36, cluster of differentiation 36; PPAR-γ, peroxisome proliferator activated receptor-γ; ABCA1, ATP-binding cassette transporter A1; ApoE, apolipoprotein E.
The expression of lipid-related genes, such as CD36, PPAR-γ and ABCA1, in each group was analysed using RT-qPCR and western blotting to examine the relationship between ZNF580 and lipid-related genes during foam cell formation (Fig. 6B–D). Compared with the model group, the mRNA expression levels of CD36 and PPAR-γ decreased and that of ABCA1 significantly increased in the Ad-ZNF580 model group, while the mRNA expression levels of CD36 and PPAR-γ increased and that of ABCA1 significantly decreased in the Si-ZNF580 model group. The western blot results were consistent with the mRNA levels, indicating that ZNF580-mediated inhibition of foam cell formation was associated with the regulation of ABCA1, CD36 and PPAR-γ.
CVD has become the most serious threat to human health among major diseases and is the leading cause of death worldwide; thus, the prevention and treatment of CVD has become the focus of global public health concern. Among CVDs, ischaemic heart disease (IHD), mainly coronary heart disease (CHD), threatens human life [19]. The main pathological change in CHD is coronary atherosclerosis, the pathological basis of which is atherosclerosis, a chronic inflammatory disease associated with lipid metabolic disorders. Lipids and other substances circulating in the blood are deposited in the endometrium of blood vessels, promoting the growth of local fibrous tissue and causing changes in the endometrial structure, bulging and expansion of the tube cavity, and constantly forming lipid stripes and necrotic cores under the influence of persistent chronic inflammation. Lipid stripes and necrosis kernels begin with foam cell formation and are the structural basis for atherosclerosis [20]. Therefore, preventing excessive foam cell production and accumulation is critical for the treatment of coronary atherosclerosis. Based on lipid metabolism in macrophages, this study examined the receptors and proteins associated with lipid internalization, fat decomposition, and cholesterol and phospholipid efflux and explored the possible correlation between the zinc finger gene ZNF580 and foam cell formation.
The class B SR CD36 plays an important role in lipid uptake [21]. CD36 is a single-stranded transmembrane glycoprotein with a long chain that mostly extends outside the cell; thus, the regulation of nonnegative feedback is not limited by recognition, and the intake of ox-LDL accelerates foam cell formation [22]. After CD36 gene deletion, the formation of intravascular foam cells was significantly lower than that in mice without CD36 gene deletion, suggesting that reducing the expression of CD36 could effectively reduce the risk of developing atherosclerosis [23]. In this study, ox-LDL-stimulated macrophages increased CD36 expression, confirming that CD36 is involved in the process of foam cell formation by macrophages. The mRNA expression of CD36 increased with time in ox-LDL-induced macrophages, indicating that CD36 affected foam cell formation by macrophages in a time-dependent manner. Therefore, it is important to effectively inhibit foam cell formation to study the regulatory factors that affect CD36 expression.
PPARs are members of the ligand-activated nuclear transcription factor superfamily and are divided into three types (PPAR-α, PPAR-β/δ, and PPAR-γ). PPAR-γ is an important regulatory factor that can effectively regulate CD36 expression [24]. Some scholars have found that inhibiting PPAR-γ activation can effectively reduce the expression of CD36, thereby reducing the accumulation of lipids in high sugar-induced THP-1 cells [25]. Therefore, by suppressing expression of the upstream factor PPAR-γ, the expression of CD36 can be indirectly reduced. In this experiment, RT–PCR was used to measure the expression level of PPAR-γ, and the results showed that the trend was consistent with the trend in CD36 mRNA expression, supporting the conclusions of the original study. Meanwhile, other studies have shown that activating PPAR-γ directly inhibits the migration and proliferation of single-core macrophages and indirectly inhibits foam cell formation [26]. The PPAR-γ agonist pyrethroidone is an important insulin allergen that is widely used to treat insulin resistance caused by elevated blood sugar. It can induce the expression of lipoprotein esterase to further promote fat breakdown and reduce plasma cholesterol and triglyceride levels. In addition, PPAR-γ can competitively inhibit the expression of inflammatory pathways and inflammatory factors to inhibit the occurrence and expansion of inflammation and reduce inflammation associated with endothelial damage in blood vessels [27, 28]. Therefore, it is urgent to find a suitable way to modulate PPAR-γ. PPAR-γ activation is also beneficial in reducing the risk factors associated with atherosclerotic plaque formation. Whether activating or suppressing PPAR-γ, it is extremely important to find a suitable regulatory point to treat coronary atherosclerosis.
In the normal state, macrophages can remove lipids to reduce cholesterol in and around the microenvironment and reverse the transfer of cholesterol to the liver for excretion. They also use cell surface ABCA1, a transmembrane protein capable of transporting cholesterol and phospholipids to the outside of cells, in order to form a high-density lipoprotein precursor to achieve cholesterol reuse and excretion, reduce the accumulation of cholesterol esters in cells, and inhibit foam cell formation [29]. This protects vascular walls and cardiovascular health. Furthermore, ABCA1 can delay the progression of atherosclerotic plaques and the erosion of plaques by inhibiting inflammatory factors involving Toll-like receptors at the atherosclerotic plaque formation site [30]. Therefore, it is critical to explore the regulatory factors that affect ABCA1 to prevent and treat CHD. In this study, RT–PCR was used to measure gene expression, and ABCA1 mRNA increased with ox-LDL induction time, indicating that the cellular demand for cholesterol efflux increased. Another study confirmed that PPAR-γ activation could promote the transport efficiency of ABCA1 to facilitate efflux, suggesting that, by regulating PPAR-γ, the expression of ABCA1, a gene associated with cholesterol efflux, can be indirectly regulated to reduce atherosclerotic plaque formation [31].
ApoE is an alkaline protein that is rich in arginine; is found in high-density lipoproteins, very low-density lipoproteins, and celiac particles; and plays an important role in maintaining the structure of the abovementioned lipoproteins [32]. Furthermore, ApoE is an independent risk factor for the formation of atherosclerotic heart disease because of its gene polymorphism, which is involved in various lipid metabolic processes, such as cholesterol efflux, transport and storage. Since studies have shown that ApoE deficiency can quickly lead to hyperlipidaemia in lipid metabolism disorders in the body, most experiments have modelled atherosclerotic plaques by knocking out this gene [33]. In foam cells, ApoE has a function similar to that of ABCA1, which can transfer excess cholesterol esters from the cell to the extracellular space, reduce the accumulation of lipids in foam cells, and reduce and delay the formation and accumulation of foam cells. The results showed that ox-LDL stimulated macrophages, leading to the upregulation of CD36, which regulates PPAR-γ-mediated phagocytosis, and an increase in the mRNA expression of ApoE upon increased ox-LDL uptake by macrophages.
LDL is the most common cholesterol lipoprotein and is mainly responsible for the transfer of cholesterol to organs and tissues. Because natural LDL cannot induce foam cell formation in macrophages, acetylation and other modifications can only induce mononuclear cells to form macrophages and achieve lipid uptake, accumulation and eventually foam cell formation when LDL is oxidized [34]. Oxidation or other modifications to LDL have a higher risk of progression to atherosclerotic-like plaques; thus, reducing ox-LDL damage to blood vessel walls, ox-LDL accumulation in mononuclear cells and macrophage uptake of ox-LDL are new prevention strategies to prevent plaque formation [35].
ZNF580 is a nuclear transcription factor that is similar in protein structure to the Sp/KLF family, is widely distributed in human organs and regulates downstream target genes to participate in physiological and pathological processes in the body in a variety of ways. ZNF580 plays an important role in the disease-causing mechanism of CHD, and a previous study showed that ZNF580 could participate in ischaemic reperfusion damage mediated by umbilical venous endotropic cell growth factor-15 [36]. After establishment of the experimental foam cell model, ZNF580 mRNA levels were measured, and the results showed that the expression of ZNF580 was higher than that in the normal control group, indicating that ox-LDL-stimulated macrophages exhibited changes in ZNF580 expression. To further study the effect of ZNF580 on foam cell formation, ZNF580 mRNA and protein levels were measured in the control group at 24 h, 48 h, and 72 h, and the results confirmed that ZNF580 expression increased with increasing foam cell formation over time. This trend was largely the same as that of CD36, PPAR-γ, ABCA1 and ApoE, which are associated with lipid uptake and efflux in macrophages. Based on the results of this experiment, we believe that ZNF580 may block foam cell formation by affecting relevant lipid regulatory molecules, possibly in relation to the PPAR-γ-CD36 signalling pathway. However, the more specific relationship will be explored further in future work. Lentivirus transfection was then used to induce high expression or silence the ZNF580 gene in THP-1 cells, after which the foam cell model was established and the above indicators examined. The results showed that CD36 and PPAR-γ mRNA expression in the high-expressing ZNF580 group was lower than that in the model group, and CD36 and PPAR-γ mRNA expression in the ZNF580 silencing group was significantly higher than that in the model group. These data indicate that ZNF580 can inhibit the expression of PPAR-γ mRNA, thereby reducing expression of the downstream molecule CD36 and cattle-LDL intake, hindering the formation of foam cells. The genes associated with lipid outflow were examined, and it was found that high expression of ZNF580 could induce an increase in ABCA1 and ApoE mRNA expression that was significantly higher than that in the model group. Additionally, silencing ZNF580 could inhibit the mRNA expression of ABCA1 and ApoE, which was significantly lower than that in the model group, suggesting that ZNF580 can hinder foam cell formation by increasing cholesterol efflux. Therefore, our group hypothesized that ZNF580 may be upstream of PPAR-γ, directly modulating PPAR-γ, thereby affecting downstream CD36 expression and reducing lipid build-up.
In summary, our study provides new insight into the positive effects of ZNF580 on the prevention of atherosclerosis. High expression of ZNF580 can decrease lipid uptake by macrophages and increase cholesterol efflux, reduce the possibility of lipid accumulation in foam cells, reduce the proportion of foam cell formation, reduce the risk of plaque formation, and delay the progression of atherosclerotic plaques. However, our experiment also has some shortcomings, and future in vivo studies will be conducted to more fully explore the role of ZNF580 in atherosclerosis. Based on these findings, ZNF580 may be a potential target for preventing atherosclerosis.
Footnotes
Acknowledgments
Not applicable.
Funding
The present study was supported by the Key project of tianjin natural science foundation (approval no.16JCZDJC31900), Fundamental research project of logistics university of People’s Armed Police Force (approval no. WHJ202108) and Applied research project of logistics university of People’s Armed Police Force (approval no. WHJ202104).
Availability of data and materials
All datasets generated or used during the study are included in this article.
Author contributions
ZZB, QXT and ZM designed the experiments and revised the manuscript. ZZB, CHX, LYC and YM performed RT-qPCR. CJX, XHW, WZH and LYC performed ORO staining. ZZB, QXT, CHX, LC and YM performed western blotting. ZZB, LC, WZH, CJX and ZM analyzed the datasets and supervised the project. All authors read and approved the final manuscript, and confirm the authenticity of all the raw data.
Patient consent for publication
Not applicable.
Conflict of interest
The authors have no competing interests to declare.
