Abstract
In the present study, the effects of 60Co-γ irradiation on the diversity of fungal microflora in blueberries during cold storage have been explored in detail. The effects of irradiation on the fungal microflora in blueberries could be observed at the lowest effective dose of 1.0 kGy. The genome library of fungal microflora in blueberries was evaluated by tusing Shannon, Simpson, Chao1, and ACE indices, presenting good coverage of blueberry samples. Principal coordinate analysis clarified the differences in the evolution and clustering of fungi in blueberries under different irradiation levels. The composition and diversity of fungal microflora in blueberries treated with different doses of radiation exhibited significant differences under cold storage of 4°C. However, most of the fungi belonged to the Ascomycota at the phylum. The fungal microflora was relatively stable at the initial stage of cold storage, however, the diversity of fungal species in irradiated blueberries varied greatly in the middle stage around the 15th day. This comprehensive study provides deeper insight about the effects of irradiation on the evolution of fungal microflora in blueberries during cold storage. The findings of this study lay a good foundation for post-harvest storage and preservation of blueberries.
Introduction
Blueberry is a healthy berry fruit with high nutrition and unique flavor. Blueberries have been demonstrated to possess various biological properties, including antioxidation, visual protection, anti-tumor activities, promoting cardiovascular health, and other physiological effects [1–3]. However, due to the special physical and chemical properties of blueberry, the post-harvest storage and freshness preserving of blueberries are challenging tasks [4, 5]. Nowadays, various preservation methods, including modified atmosphere packaging, film-forming system and low temperature conditions, have been applied for the post-harvest storage of fruits [6, 7]. However, these fresh-keeping technologies are not able to meet the high efficiency, and safety requirements for blueberry storage.
Irradiation technology has been reported as a more convenient and efficient preservation method for fruits, livestock and poultry meat, and aquatic products [8–10]. However, the maximum dose of irradiation is set according to the actual situation, which vary greatly depending on the geographical region or type of food. As for fruits, irradiation preservation has been applied for storage of strawberries, blueberries, cherries, mangoes, date fruit, citrus fruit and other fruits [11]. Irradiation treatment can change the appearance quality, nutrient content, microorganism load, and shelf-life of fruits. Results of some researches indicated that γ-irradiation could significantly reduce the population of microorganisms in pomegranate, grape tomatoes, and mango pulp [12–14]. γ-irradiation could can also affect the growth of microorganisms, and has various bacteriostatic properties. Studies have reported with the 25% inhibition in the growth of
In this study, fresh blueberries were subjected to different doses of 60Co-γ radiations to analyze the impact of irradiation on fungal microflora. A fungal genomic library was constructed and analyzed by high-throughput sequencing technology. The composition and diversity of fungal microflora in irradiated blueberries during cold storage were further explored to elaborate the growth characteristics of fungi in blueberries exposed to different irradiation.
Materials and methods
Reagents, materials and apparatus
The blueberries were picked from local blueberry planting base in Heyuan city in China. DNA extraction kit, DNA purification kit, and PCR amplification kit were purchased from Tiangen Biotech Co., Ltd. (Beijing, China). Rose Bengal media for fungal growth was purchased from Guangdong Huankai Microbial Sci. & Tech. Co., Ltd (Guangzhou, China). The Novaseq 6000 platform was (Illumina Inc., San Diego, CA, USA) used for genome sequencing.
Irradiation treatment and low temperature storage of blueberries
The blueberry samples were picked and quickly transported to Guangzhou Furui High Energy Technology Co., Ltd., and the irradiation treatment was conducted in the special γ-irradiation device of 60Co. The 60Co-γ irradiation treatments were carried out in five groups (A, B, C, D, and E group) with doses (0, 0.5, 1.0, 1.5, and 2.0 kGy), respectively. The actual radiation doses measured by ferrous sulfate dosimeter were 0, 0.32, 0.72, 1.02, and 1.48 kGy, respectively. After irradiation, treated blueberries were sealed in fresh-keeping bags and further stored at 4°C. The diversity and composition of fungal microflora in treated blueberries were assessed at different cold storage. The irradiated blueberry samples from five dose groups (A, B, C, D, and E) were collected at 1st, 5th, 10th, 15th, and 20th days, and labelled as: A1, A2, A3, A4, and A5; B1, B2, B3, B4, and B5; C1, C2, C3, C4, and C5; D1, D2, D3, D4, and D5; and E1, E2, E3, E4, and E5.
Amount analysis of fungal microflora in blueberries
On 1st, 5th, 10th, 15th, and 20th days of cold storage at 4°C, the irradiated blueberry samples were collected to analyze the fungal microflora. 25 g blueberries were placed in 225 mL PBS buffer, and the mixture were homogenized by using a bag mixer for 1 min. Afterwards, the homogenized samples were centrifuged at 10000 rpm for 2 min to obtain the supernatants for further use. After adjusting the pH to 7.4, the sample solutions were diluted in gradients and spread on the solid agar plate of rose Bengal media. The plates were incubated at 37°C overnight and the fungal colonies on plates were counted to evaluate the fungal microflora in blueberries.
Composition and diversity evaluation of fungal microflora in blueberries
Irradiated blueberries with different storge time (1, 5, 10, 15, and 20 days) were sampled and stored in dry ice. The genome libraries of fungal microflora of blueberry samples were constructed as described in the previous study [21]. More specifically, the genomic fungal DNA was extracted and purified by using commercial kits, and then the sample DNA was amplified by using ITS1 primers ITS5-1737-F and ITS2-2043-R to construct the genome library [21]. After quantitative analysis of the library, the fungal microflora of different blueberries were evaluated by high throughput sequencing technology using the Illumina Novaseq 6000 platform. The sequencing data were evaluated to explore the composition and diversity of fungal microflora in blueberries by assessing the relative abundance of fungal taxa as well as classification and clustering analysis [22].
Results and discussion
Fungal growth characteristics of irradiated blueberries during cold storage
Figure 1 shows the fungal growth of irradiated blueberries during cold storage. It was indicated that higher dose of irradiation at the five gradients could keep blueberries fresh for a longer period during cold storage without obvious changes in quality. The growth rates of fungal growth were greatly effected by the irradiation dose. Fungal in the non-irradiated blueberries showed the highest growth rate, and the growth rate of fungal in the E group was the lowest one. Compared to non-irradiated blueberries, the irradiation dose in effectively destroyed most of the fungal microflora in blueberries. Thus, irradiation treatment could inhibit the growth of fungal microflora and greatly prolong the storage life of blueberries under 4°C. These results suggest that 60Co γ-irradiation treatment at proper doses can effectively maintain the storage quality of blueberry fruits. Moreover, previous studies have shown that the irradiation at dose below 1 kGy has no significant effect on the characteristics of blueberries[23, 24]. The maximum dose of 1.0 kGy has been approved by United States Food and Drug Administration for irradiation of fresh fruits and vegetables [25]. Considering the growth characteristics of fungi and the appearance quality of blueberries after irradiation, the irradiation dose of 1.0 kGy can be considered more suitable for preservation of blueberries.
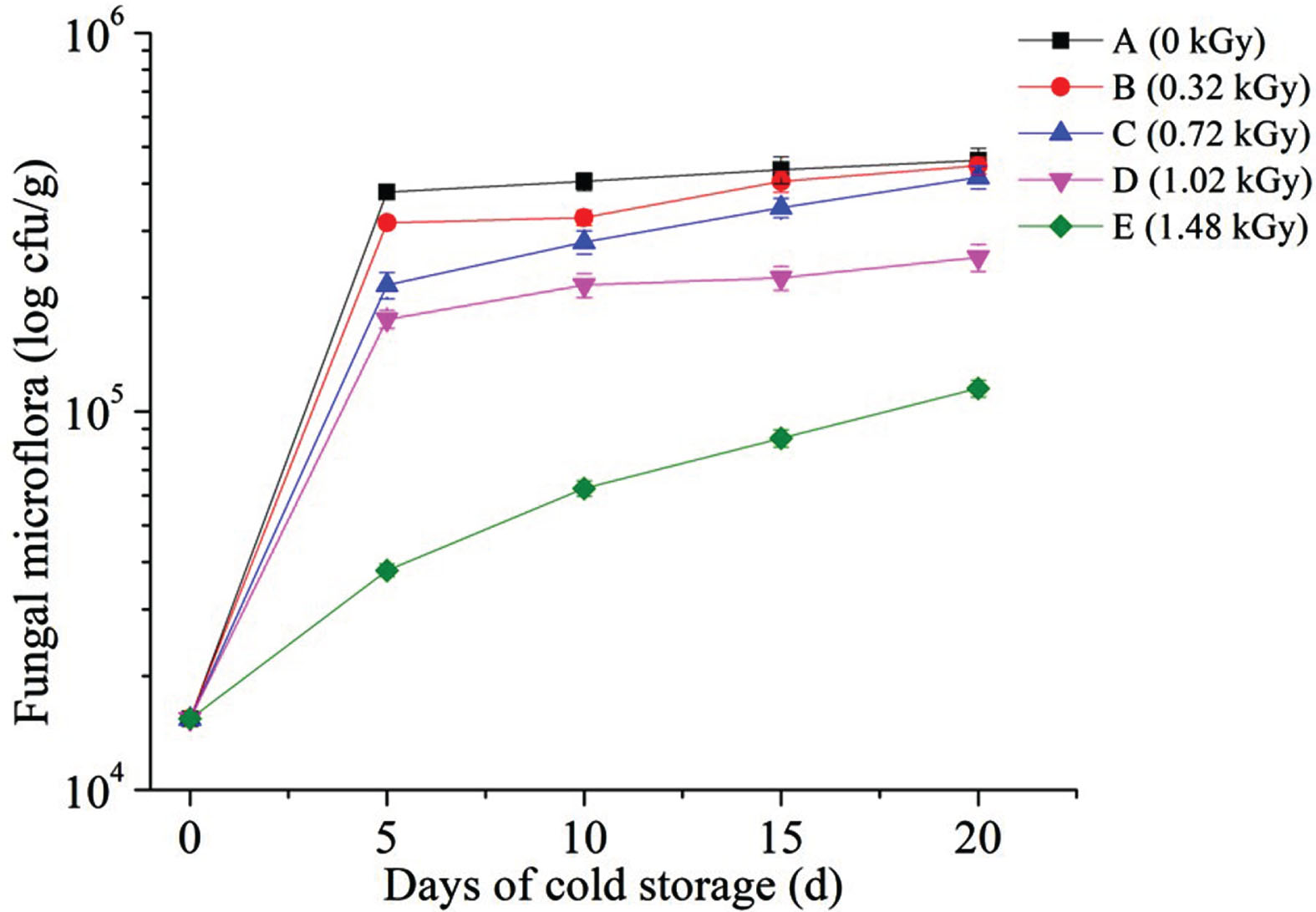
Fungal growth of irradiated blueberries with different doses during cold storage (
The genome of fungal microflora in blueberries was constructed through genome sequencingby NovaSeq 6000. The rarefaction curves in Fig. 2 A showed the sharp increase in fungal growth in irradiated blueberries with the extension of storage period. These results were consistent with the previously reported growth trend of microbes in fruits during post-harvest storage [26, 27]. However, there were significant differences in the diversity of fungal microflora in blueberries treated with different irradiation doses (Fig. 2B). The various diversity indices (i.e., Shannon, Simpson, Chao1, and ACE) were used to evaluate the genome library of fungal microflora in blueberries (Table 1), which could meet the analysis requirements of fungal by high throughput sequencing [28]. The results showed that the average values of these four indices increased continuously in general with the extension of cold storage, indicating the rising trends of abundance and diversity of fungal microflora in irradiated blueberries showed a rising trend. The genome library possessed high coverage of samples with the values of goods_coverage over 0.99. Overall, the diversity and abundance of fungi in blueberry samples showed increasing trend with the extension of storage time at 4°C.
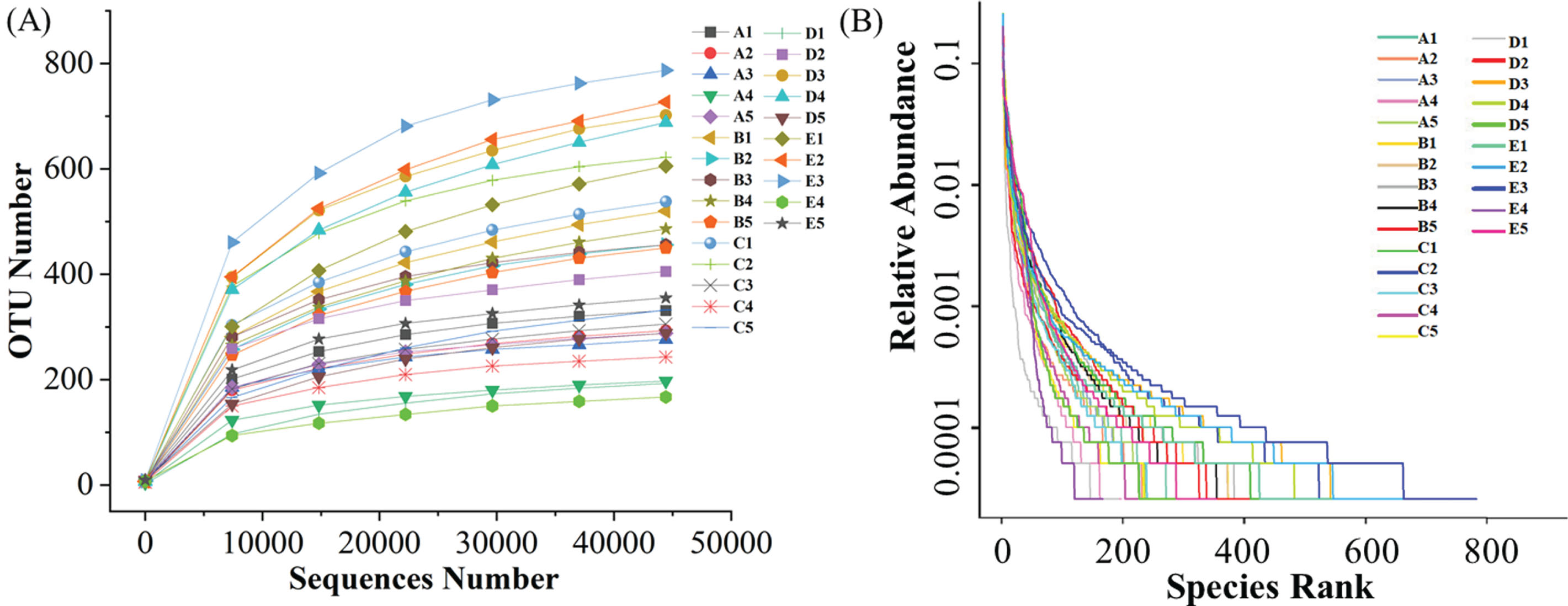
Rarefaction curves (A) and rank abundance curves (B) of fungal microflora in irradiated blueberries during cold storage.
Parameters of fungal microflora in irradiated blueberries during cold storage
The operational taxonomic units (OTUs) generated from the processed sequence data were used to assess the characteristics of fungal microflora in irradiated blueberries. The common and specific OUTs among the fungal microflora in irradiated blueberries were evaluated by Venn diagrams shown in Fig. 3. The common fungi OUTs of blueberries treated with different irradiation doses for 1, 5, 10, 15, and 20 days were 74, 159, 79, 80, and 86. However, significant quantity of specific OUTs were also observed in irradiated blueberry during cold storage. The results illustrated the similarities in OTUs, as well as the growth characteristics of fungal microflora in irradiated blueberries under cold storage. The fungal flora of irradiated blueberries was observed to be in a dynamic state during storage, which was similar to the reported studies [29–31].
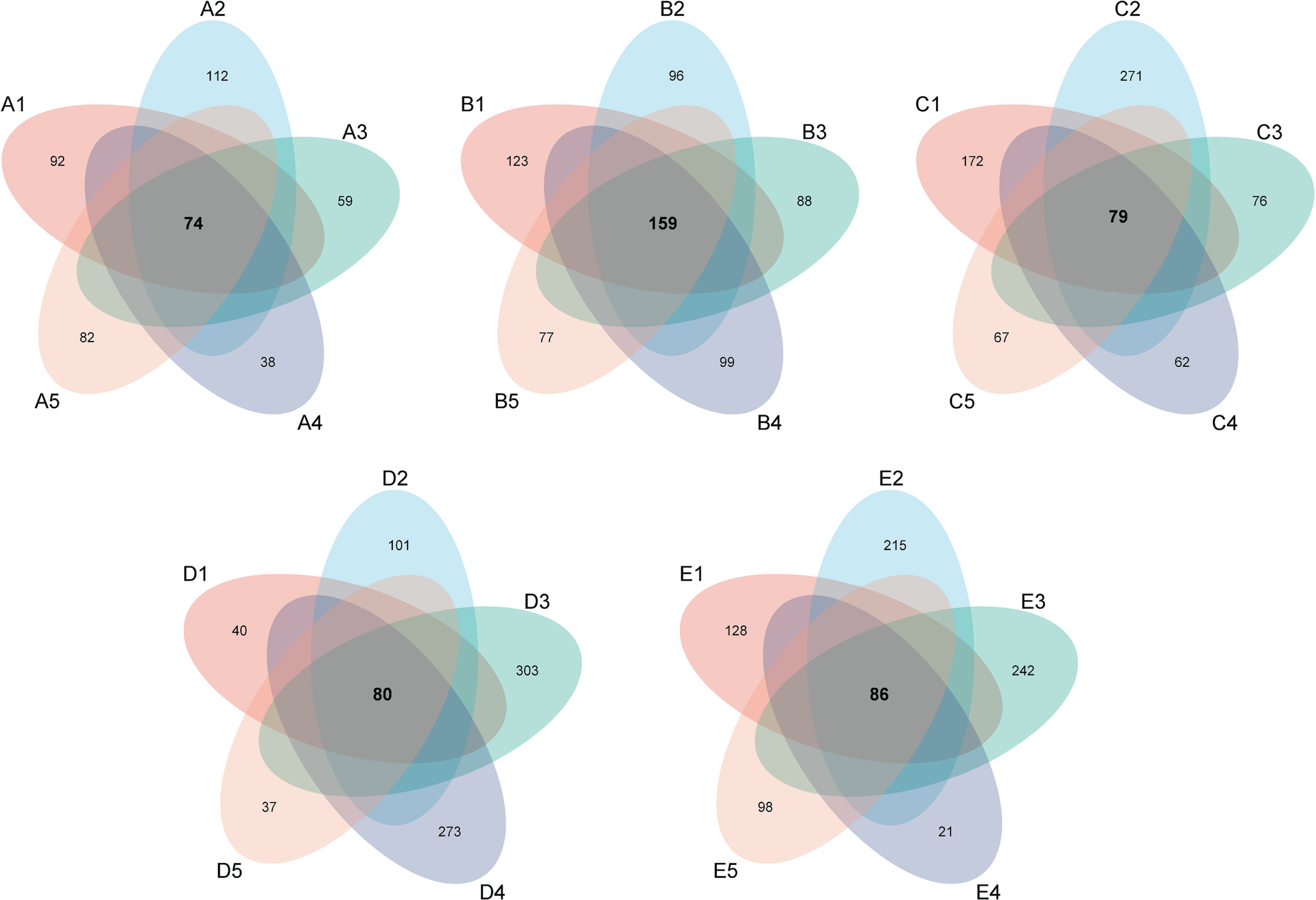
Venn diagrams of fungal microflora in irradiated blueberries during cold storage.
The microbial community and overall fungal diversity in fruits may reflect the impact of storage period and deterioration characteristics [32, 33]. Therefore, the composition of fungal microflora in irradiated blueberries was further assessed, and the OTU sequences were used for species annotation. The relative abundances of fungal taxa were determined at different classification levels (i.e., phylum, class, order, family, genus, specie). As shown in Fig. 4, the dominant fungal phyla in irradiated blueberries were Ascomycota and Basidiomycota, while the dominant fungal classes were Dothideomycetes and Saccharomycetes, At order level, fungal microflora was dominated by Capnodiales, Saccharomycetales, and Helotiales, while Mycosphaerellaceae, Saccharomycetales_fam_incertae_sedis, Unidentified_helotiales_sp, Botryosphaeriaceae, and Cladosporiaceae were found to be the dominant fungal families. The results further showed that the fungal microflora in irradiated blueberries of different groupsvaried greatly at genus level and species level. The prominent fungal genera were
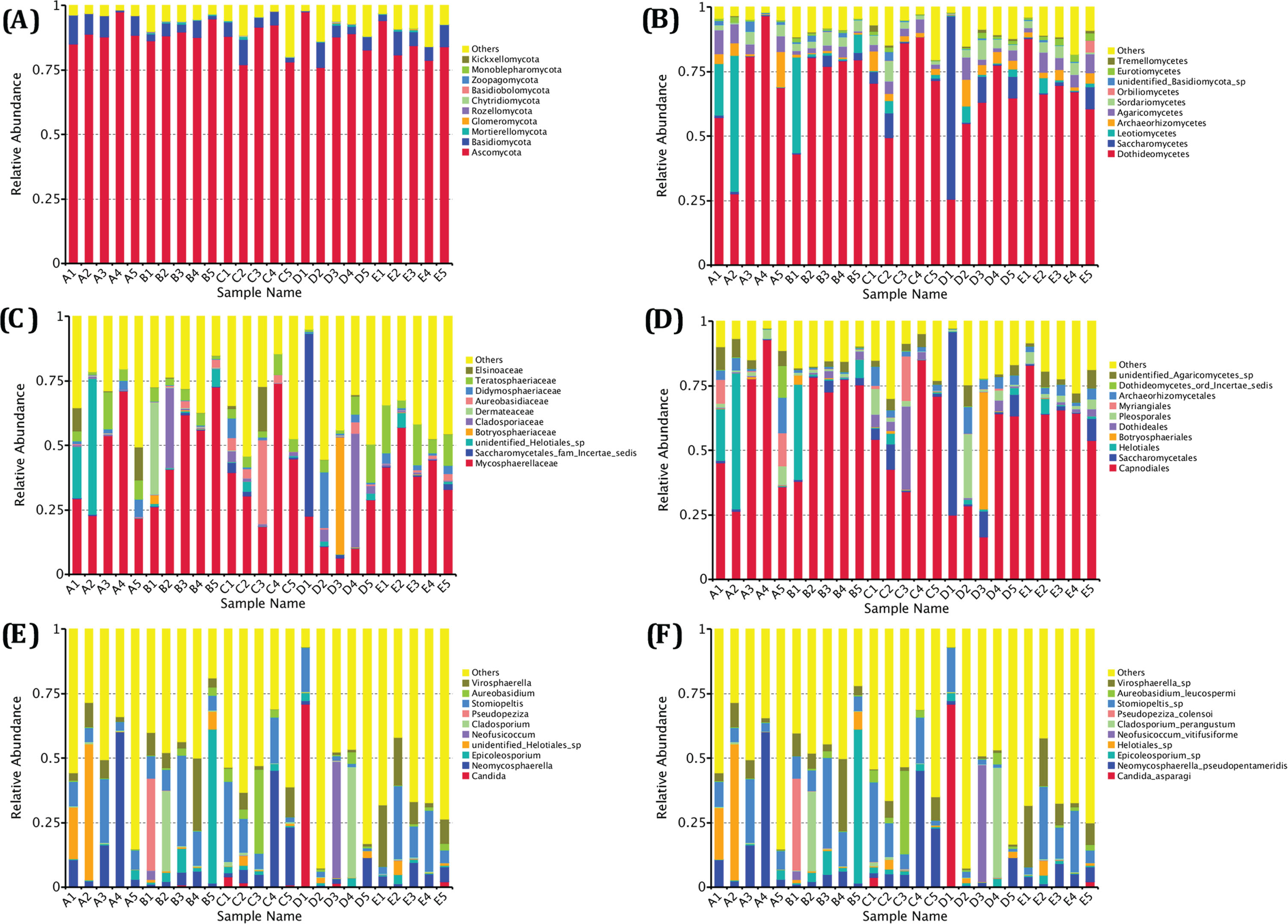
Analysis of fungal phylum, class, order, family, genus and species level in irradiated blueberries during cold storage (A–F).
Cluster analysis is often used to annotate and assess the characteristics of microbial population [33]. The top 35 fungal genera were clustered together based on the species annotation and relative abundance data to generate the heat map shown in Fig. 5. The results revealed that fungal microflora in irradiated blueberries remained stable during 1st to 5th day. However, significant changes in the composition of fungal microflora were observed on the 15th day. These results were consistent with the above-mentioned results of diversity evaluation of fungal microflora in irradiated blueberries. In general, the fungal microflora in irradiated blueberries during cold storage indicated that 90% fungal species belonged to Ascomycota, while 10% fungal species belonged to Basidiomycota phylum. Therefore, Ascomycota was considered to be the most abundant phylum in irradiated blueberries, and this finding was consistent with the previously reported studies [36, 37].
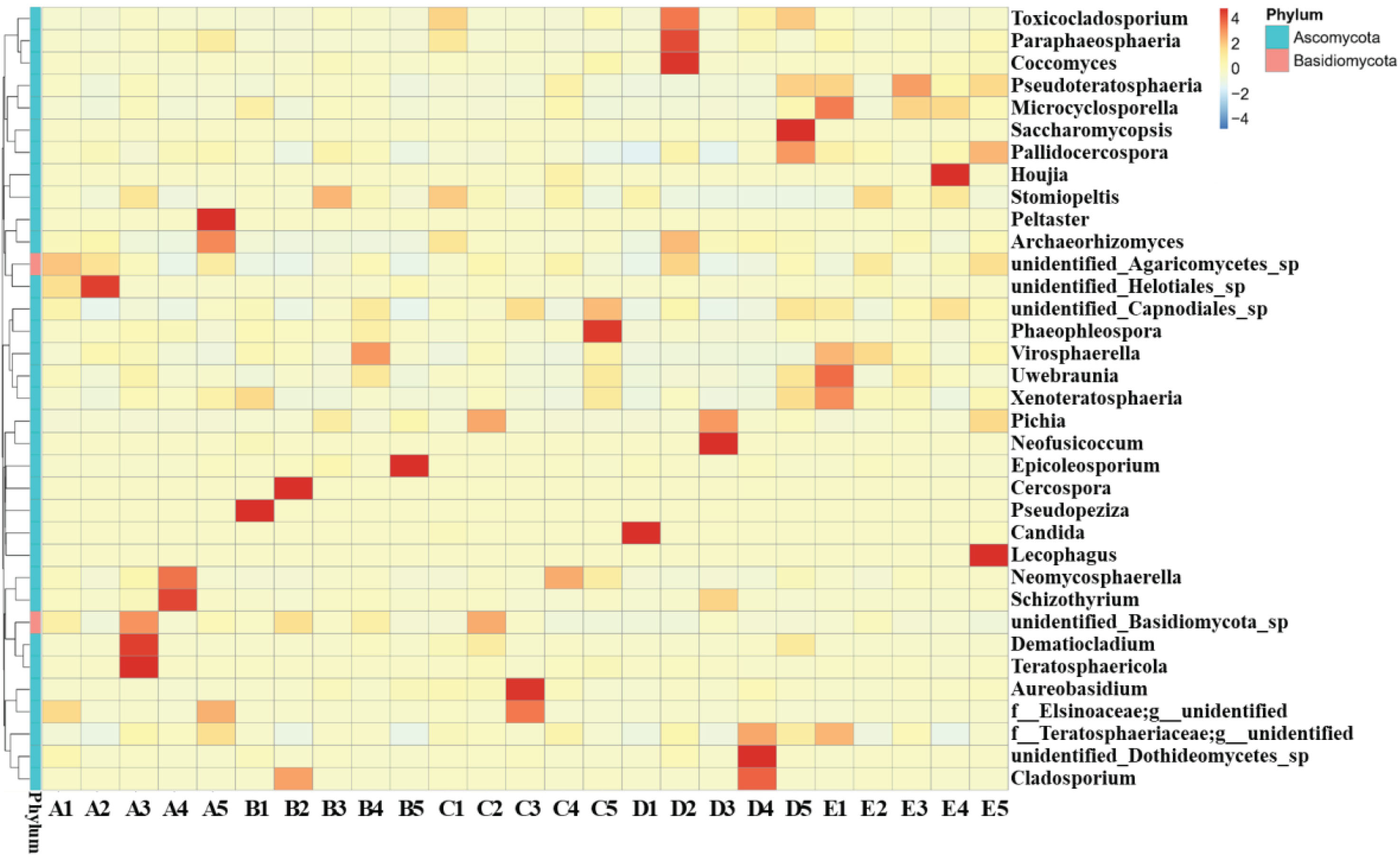
Cluster analysis of fungal microflora with genus level in irradiated blueberries during cold storage.
After the irradiation treatment at different doses, the blueberry samples were stored at 4°C to explore the evolution of fungal microflora during cold storage. As shown in Figs. 4 and 5, the composition and diversity of fungal microflora in irradiated blueberries varied dynamically. Principal coordinates analysis (PCoA) was further conducted to elaborate the effect of irradiation on the fungal diversity in blueberries during cold storage. In Fig. 6, blueberry samples with 60Co-γ irradiation treatment of B, C, D, and E treatment groups during different storage periods were observed to be located far away from the control group (A group). Moreover, the composition of fungal flora varied significantly across in all the treatment groups. With the prolongation of storage time, the fungal composition in different groups changed greatly, and all the samples in each irradiation treatment group showed a partial cross. The results of PCoA revealed multiple obvious separations in different groups of irradiated blueberries. The analysis showed significant differenc in the characteristics of five irradiation groups of blueberries, and the diversity of fungal microflora was significantly correlated with the properties of irradiated blueberries. These results further demonstrated that irradiation treatment had a significant impact on the microbial communities in blueberries at different time of cold storage, which was similar to the earlier reported studies [38–40]. Results indicated that 60Co-γ irradiation of blueberries could not only greatly reduce the total number of molds, but also effectively inhibit the dominant spoilage bacteria during cold storage, which can be very beneficial for the preservation of blueberries.
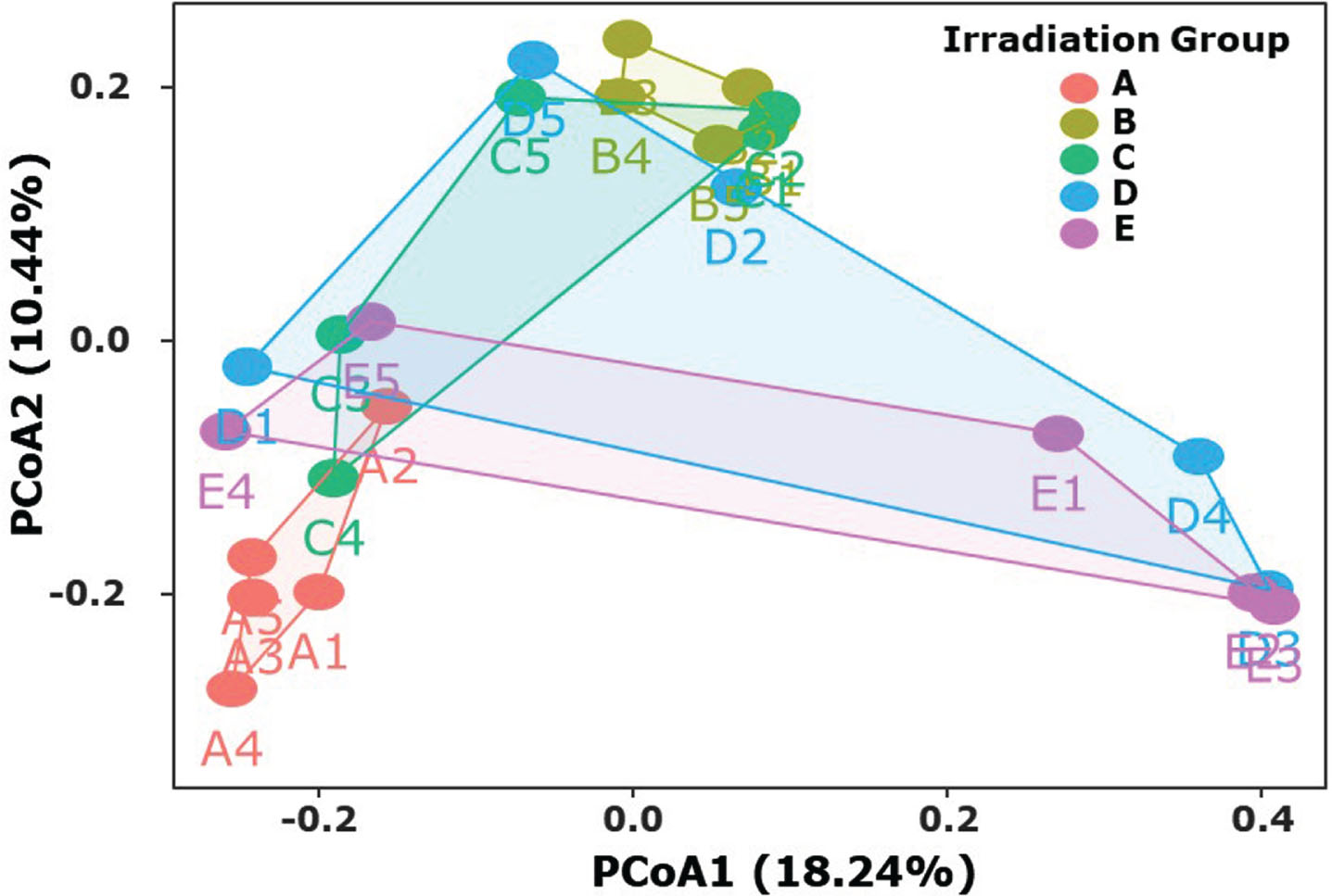
PCoA of the fungal diversity and the irradiation treatment for irradiated blueberries during cold storage.
The functional guild (FUNGuild) has been used for functional prediction of the fungal microflora in some studies [41–43]. As shown in Fig. 7, the fungal species in the irradiated blueberries were categorized into saprotroph, pathotroph, pathotroph-symbiotroph and the “unassigned”. The “unassigned” category accounted for the vast majority of the fungal community, which needs to be explored further. Saprotroph was the second most abundant trophic mode for fungi in irradiated blueberries. With the extension of storage time, the relative abundance of saprotroph in irradiated blueberries was remained relatively stable, while the relative abundance of pathotroph showed an increasing trend. These results indicated that pathotroph fungi were very sensitive to 60Co-γ irradiation, even at lower doses. After irradiation, the relative abundance of pathotroph remained at a low levle during cold storage.
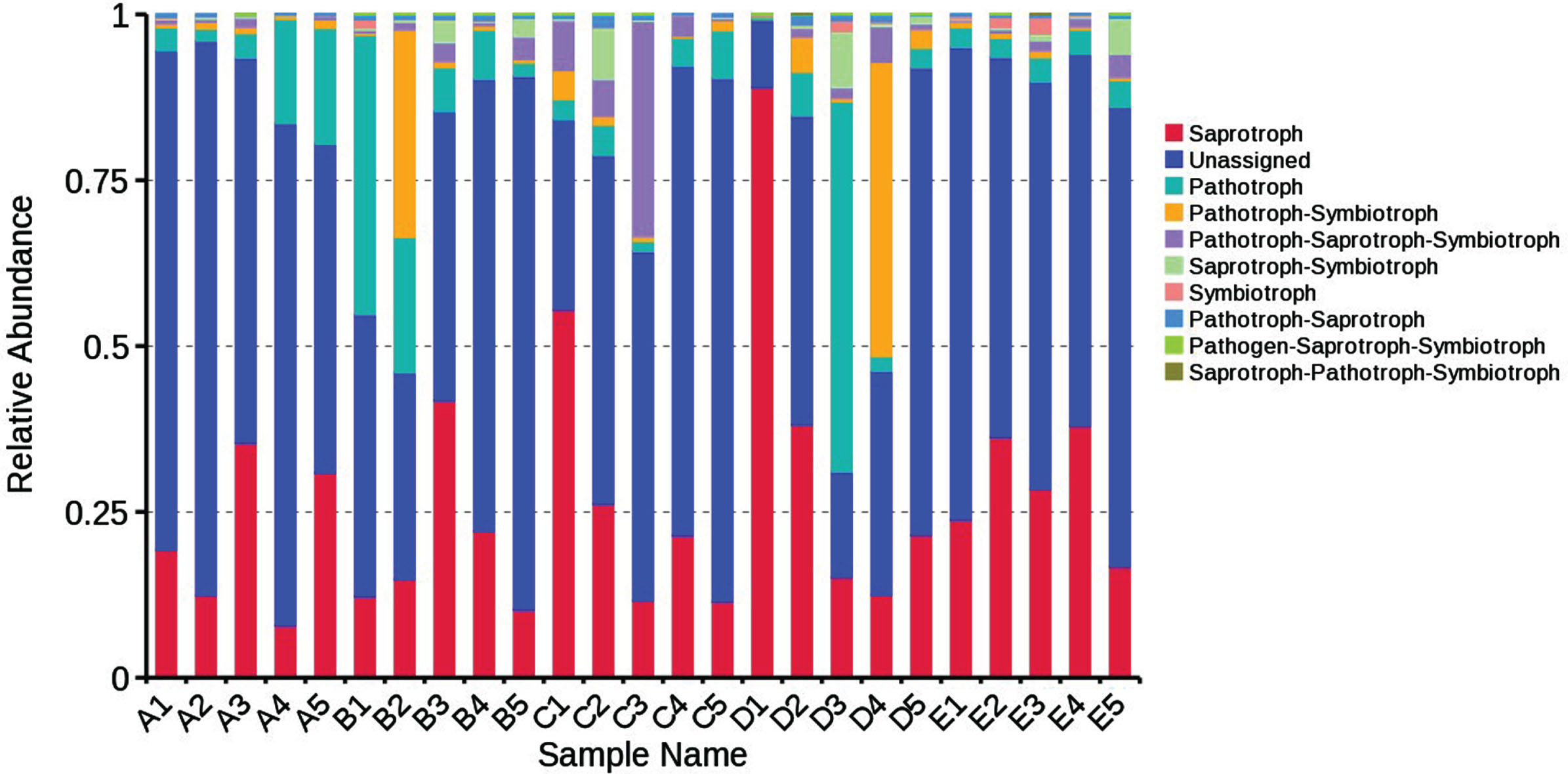
Predicted trophic mode of fungal microflora in irradiated blueberries during cold storage.
In this study, the effects of 60Co-γ irradiation on the diversity of fungal microflora in blueberries during cold storage were elaborated in depth. Different doses of irradiation exhibited significant differences in the characteristics of fungal microflora in blueberries, which greatly affected the composition and diversity of fungi in irradiated blueberries. The blueberries treated with different irradiation doses showed significant clustering effects based on fungal characteristics. In conclusion, this study provides substantial technical basis for post-harvest preservation of blueberries via irradiation.
Author contributions
Conceptualization, F.W. and D.L, methodology, F.W., and P.Y, software, F.W, validation, F.W. and P.Y, formal analysis, F.W. and P.Y, investigation, F.W. and P.Y, resources, C.M, data curation, F.W. and P.Y, writing— original draft preparation, F.W. and D.L, writing— review and editing, F.W., D.L. and Q.W, visualization, Q.W. and G.X, supervision, Q.W., D.L. and G.X, project administration, Q.W. and G.X, funding acquisition, F.W., Q.W., D.L. and G.X. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
The authors have no acknowledgments.
Funding
This work was supported by Natural Science Foundation of China (32202175, 31901734), the Guangdong Basic and Applied Basic Research Foundation (2021A1515110608), the Characteristic Innovation Project of the Higher Education Universities of Guangdong Province (2023KTSCX049), the Guangdong Province Key Construction Discipline Research Ability Improvement Project (2021ZDJS003, 2022ZDJS021), the Guangdong Province Agricultural Science and Technology Innovation Ten Main Direction of the “Top” Project (2022SDZG04), the Project of Guangdong Provincial Key Laboratory of Lingnan Specialty Food Science and Technology (2021B1212040013), and the Innovation and Entrepreneurship Training Projects for College Students in Zhongkai University of Agriculture and Engineering (X202211347160).
Conflict of Interest
The authors have no conflict of interest to report.
Notes
The authors declare no competing financial interest.
