Abstract
BACKGROUND:
Due to the presence of wide-range phytochemicals, particularly anthocyanins, and their health-promoting activities, black goji fruits (Lycium ruthenicum Murr., LRf) gained attention as functional foods or nutraceuticals. However, data on the phytochemical composition and bioactivities of LRf from outside Asian countries are very limited.
OBJECTIVE:
This study reports the polyphenols profile and in vitro biological activities of LRf cultivated in the southern part of Serbia.
METHODS:
The total content of polyphenols, flavonoids and anthocyanins of LRf was determined spectrophotometrically, while the polyphenol profile was evaluated by LC-DAD-ESI-MS. Antioxidant and enzyme-inhibitory potentials against α-amylase, α-glucosidase, acetylcholinesterase, tyrosinase, and 15-lipoxygenase properties were determined spectrophotometrically. The prebiotic potential was assessed by the broth microdilution method.
RESULTS:
Chromatographic analysis confirmed that hydroxycinnamic acid derivatives and acylated anthocyanins were the predominant polyphenols of LRf. In addition to antioxidative activities, Lycium ruthenicum methanol extract (LRe) showed inhibitory effects against all tested enzymes (IC50 = 5.28–7.28 mg/mL). Prebiotic potential on the growth of various microorganisms’ species was observed, with a marked effect on the growth of yeast Saccharomyces boulardii.
CONCLUSIONS:
The polyphenol pattern of LRf cultivated in Serbia corresponds with the literature data. Observed bioactivities support further phytochemical characterizations of LRf and investigations of their health-promoting effects, especially prebiotic properties.
Introduction
Black goji or wolfberry (Lycium ruthenicum Murr., Solanaceae) is a perennial shrub widely distributed among Asian countries. The elliptical, purple-black goji berries have been used in the traditional diet and ethno-medicine for thousands of years [1]. These berries are mainly used fresh or dried, but also as a raw material for producing herbal products (tinctures, teas, juices, jams) with medicinal properties [2]. Based on the folk experience, L. ruthenicum fruits (LRf) could be beneficial for many diseases, including cardiac, renal, and skin diseases, menstrual abnormalities, and symptoms of menopause [3]. Previous phytochemical analyzes of LRf have revealed the presence of diverse phytochemicals, including anthocyanins (glycosides and acylglycosides of anthocyanidins), phenolic acids, alkaloids (spermidine derivatives), and coumarins. In addition, fruits contain polysaccharides, unsaturated fatty acids, amino acids, vitamins and minerals [2, 5]. LRf are known as “soft black gold” because they contain more than ten times anthocyanins than raspberries and grapes. Acylated anthocyanins have also been shown to have better stability than non-acylated ones found in other berry species, such as blackberries and blueberries [6, 7]. Based on previous studies, these compounds are responsible for antioxidant [8, 9], anti-inflammatory, hypolipidemic [7], antimicrobial [10], gut microbiota modulatory [11], and hypoglycemic effects [12]. In addition, anthocyanins from the LRf are gaining attention as natural antioxidants and colorants [13].
There is evidence that LRf are cultivated worldwide. However, the chemical composition and bioactivities of LRf could be affected by geographic, climate, cultivation, and processing factors [14, 15]. Liu et al. [4] reported significant differences in polyphenols among LRf from seven regions of western China. However, cultivated LRf from north Macedonia displayed a similar polyphenol pattern to native berries from China [5].
We previously reported significant differences in chemical compositions among different Lycium fruits cultivated in Serbia, highlighting the most intense antioxidant properties of LRf compared to other goji berries [16]. This study reports the polyphenols profile and in vitro bioactivities of the LRf, including antioxidative, enzyme inhibitory activities, and prebiotic potential.
Materials and methods
Plant material
Full-ripened fruits of L. ruthenicum were harvested in September 2020 from a plantation in Niš, southern Serbia (coordinates 43°19’29” N; 21°54’11 “E). Samples were transported to the laboratory in plastic bags. Due to high water content and limited storage time, the fruits were freeze-dried for two days at –40°C under a pressure of 0.02m Bar using an LS500 model (Elbi Valjevo, Serbia). Then, the samples were pulverized and stored in a hermetic container at –20°C until analysis.
Extraction procedure
The samples were macerated with methanol (1 : 10). After 24 hours at room temperature, the solvent was removed by Rotavapor RII vacuum evaporator (Buechi Labortechnik AG, Flawil, Switzerland) at 45°C. Obtained dry L. ruthenicum methanol extract (LRe) was stored at 4°C until further analysis.
Determination of the total phenolic, total flavonoid, and total anthocyanin contents
The total phenolic content (TPC) was determined by the Folin-Ciocalteu microplate method at 630 nm [17]. The calibration curve was constructed using a gallic acid ranging from 10 to 80 mg/L, and results were expressed as milligrams of gallic acid equivalents (GAE) per gram dry weight of the sample.
Total flavonoid content (TFC) was carried out using the AlCl3 method with slight modifications, as described by Zhishen et al. [18]. The standard curve was prepared using catechin in concentrations ranging from 100 to 800 mg/L, and the results were expressed as mg of catechin equivalents (CE) per g of dry weight.
The total anthocyanin content (TAC) of black goji berries was evaluated using the pH differential method [19]. Absorbance values were measured at 520 nm and 700 nm. Results were expressed as mg of cyanidin-3-glucoside (C3G) per g of dry weight.
Qualitative and quantitative analysis of polyphenols
Qualitative analysis of phenolic compounds in the LRe was performed by the LC-MS method (Agilent LC-MS System 1260/6130, Agilent Technologies, Waldbronn, Germany), as previously described by Marčetić et al. [20] with slight modifications. 5μL of the samples (10 mg/mL) were separated on a reversed-phase column (Zorbax SB -Aq, 3×150 mm, particle diameter 3.5μm, Agilent Technologies) using the mobile phase composed of 0.1% aqueous formic acid (A) and acetonitrile (B). The flow rate was 0.35 mL/min and the gradient program: 5–25% B (0–20 min), 25–90% B (20–24 min), 90% B (24–25 min), and 90–5% B (25–30 min). UV chromatograms were recorded at 210, 270, 320, 350 and 530 nm. The negative mode was used for the analysis of hydroxycinnamic acid derivatives. The analyses of anthocyanins were performed in positive mode. The polyphenols were identified by comparing their recorded UV and MS spectra with literature data [5, 21–24].
The quantitative analysis of anthocyanin aglycones, which represent the sum of free and glycosylated aglycones, and hydrocinnamic acids in the LRe, was carried out according to the procedure described by Djordjevic et al. [25], with slight modifications. In the first step, a twenty-minute ultrasound-assisted extraction of lyophilized plant material (1 g) was performed using 10 mL of a mixture of methanol, water and 37% HCl (50 : 33 : 17, v/v/v). The resulting extract was filtered through a membrane filter (pore size 0.45μm) into a Teflon bottle and heated for 60 min at a temperature of 100°C. The cooled samples were injected into an HPLC system (HP series 1090 chromatograph) with a DAD detector. Analytes separation took place on a reverse phase analytical column (Lichrospher RP-18; particle diameter 5μm; column dimensions 250×4 mm i.d.). The flow rate of the mobile phase was set to 1 mL/min. Gradient elution was performed with a mixture of acetonitrile and 10% aqueous formic acid. The proportion of acetonitrile in the mobile phase was changed as follows: 1%, 0–0.5 min; 1–7%, 0.5–1 min; 7%, 1–4 min; 7–10%, 4–7.5 min; 10–14%, 7.5–11.5 min; 14%, 11.5–15.5 min; 14–18%, 15.5–18.5 min; 18%, 18.5–22 min. The described HPLC-DAD conditions were also applied for the determination of the chlorogenic acid concentration in the unhydrolyzed sample. The contents of delphinidin, petunidin, chlorogenic acid, p-coumaric acid, and caffeic acid were calculated using calibration curves of commercial standards (Extrasynthese, France). The content of anthocyanin aglycones and phenolic acids was expressed as mg per g of the dry weight.
In vitro bioactive properties of LRe
Antioxidant activity
A set of five in vitro microassays were conducted to assess the antioxidant properties of LRe. These assays are performed on three distinct mechanisms of action: radical scavenging (2,2’-azino-bis(3-ethylbenzothiazoline)-6-sulfonic acid (ABTS) and diphenylpicrylhydrazyl (DPPH)), reducing power (copper ion reducing antioxidant capacity (CUPRAC) and ferric reducing antioxidant potency (FRAP)), and inhibition of lipid peroxidation (β-carotene/lionoleic acid bleaching assay). Absorbance measurements were taken in 96-well microplates at different wavelengths against a blank sample using a microplate reader (ELx800, BIOTEK, Santa Clara, CA, USA).
DPPH radical scavenging ability was done as previously described by Norma et al. [26], while ABTS radical scavenging ability was estimated according to the procedure outlined by Re et al. [27]. The FRAP assay was performed according to Bolanos de la Torre et al. [28], and the CUPRAC assay was determined using a microassay previously described by Apak et al. [29]. The Trolox calibration curve was used for quantification, and results were expressed in μM Trolox equivalents (TE) per g dry weight of fruit.
The β-carotene/linoleic acid bleaching assay was conducted using a spectrophotometer based on the procedure previously described by Reis et al. [30]. The extent of oxidation in the β-carotene emulsion was measured at 490 nm at the initial time point (0 min) and after 120 minutes, using an emulsion without β-carotene as a blank. The results were expressed as the % of inhibition of oxidative degradation of β-carotene.
Anti-inflammatory activity
The inhibitory activity of soybean 15-lipoxygenase was used to evaluate the anti-inflammatory activity of LRe, according to the Cayman Chemical test protocol, No. 760700. In this assay, the samples were dissolved in a Tris-HCl buffer with a pH of 7.4 and incubated with the enzyme solution. The reaction was initiated by adding the substrate solution (linoleic acid) and chromogen. The colored products in the microtiter plate were read at 490 nm, and the following equation (1) was used to calculate the percentage of inhibition:
Where Ac represents the average absorbance of wells with 100% enzyme activity and As represents the average absorbance of wells containing both, sample and enzyme. The results were expressed as the sample concentration (mg/mL) required to inhibit half of the enzyme activity, known as IC50 value.
The inhibitory enzyme activities of the LRe were investigated against α-amylase, α-glucosidase, tyrosinase and acetylcholinesterase. The percentages of enzyme inhibition in all assays were calculated according to the following formula (2):
α-Amylase (α-Amy) inhibitory activity was performed using the method previously described by Ahmed et al. [31], with slight modifications. The LRe was dissolved in a phosphate buffer with a pH of 6.9. The tubes containing serial dilutions of the samples (200μL) were mixed with 200μL of α-amylase solution (1 mg/mL), followed by the same volume of colour reagent (3,5-dinitrosalicylic acid)and a starch solution (1%). After dilution, the absorbance values of the samples were measured at 540 nm. Acarbose was used as a reference standard.
The inhibitory activity against α-glucosidase (α-Gls) was evaluated using a previously described method [32]. Briefly, the sample dissolved in phosphate buffer with a pH of 5.0 was mixed with substrate (p-nitrophenyl-α-D-glucopyranoside). After incubation, an enzyme solution was added, and after 30 minutes, phosphate buffer pH 10.0 to stop the reaction. The absorbance of the reaction mixture was measured at 405 nm. Acarbose was used as a reference standard.
Acetylcholinesterase (AChE) inhibitory activity was assessed by a slightly modified method reported previously [33]. Briefly, the test sample (40μL) dissolved in Tris buffer pH 8.0 was pipetted into a tube. Subsequently, an enzyme solution (60μL) and buffer (400μL) were added. After incubation at 37°C for 10 minutes, 100μL of substrate (acetylcholine iodide) and 400μL of chromogen (5,5’-dithiobis-2 nitrobenzoic acid) were added. The absorbance of the resulting yellow-coloured solution was measured at 412 nm after 30 minutes of incubation at 37°C. Galantamine was used as a reference standard.
A tyrosinase inhibition activity (TIA) was determined by the method described by No et al. [34]. In this assay, the test sample was dissolved in phosphate buffer with a pH of 6.6 and 25μL pipetted into a 96-well plate. Then, an enzyme solution (40μL) and buffer (100μL) were added. After incubation at 30°C for 10 minutes, the L-tyrosine solution (40μL) was added to initiate the reaction. The absorbance was measured at 490 nm after 30 minutes of incubation at 30°C. Kojic acid was used as a positive control.
The potential prebiotic activity of LRe was investigated on four probiotic strains of Gram-positive bacteria (Lactobacillus plantarum Lp 299v; Limosilactobacillus reuteri Protectis (DSM 17938) (formerly Lactobacillus reuteri); Lactobacillus rhamnosus GG (LGG) and Streptococcus salivarius subsp. thermophilus ST-21), two strains of yeast Saccharomyces boulardii, and two mixtures of probiotic lactobacilli/bifidobacterium strains Lactobacillus helveticus, L. rhamnosus, and Bifidobacterium longum (MIX 1) and their combination with Saccharomyces boulardii (MIX 2). The prebiotic activity was tested as previously described [20]. In short, overnight cultures of probiotic strains cultivated on SDA (Sabouraud Dextrose agar, Oxoid Ltd., Basingstoke, Hampshire, UK) (S. boulardii strains), or MRS Lactobacillus agar (de De Man Rogosa and Sharpe agar, Oxoid) were suspended in saline (0.5 per McFarland standard) (Bio-Merieux, France) and further diluted to an inoculum containing 5x105 CFU/mL of microorganisms. Stock solutions of black goji berries methanol extracts were prepared in DMSO and double diluted in Sabouraud Dextrose broth (Oxoid) and MRS broth (Oxoid) to concentrations 0.312–5 mg/mL. Each dilution was set in triplicate in a 96-well microtiter plate with the addition of 5×105 CFU/mL of probiotic strains. Each plate contained triplicates of positive control (growth of microorganisms in medium) and negative controls (medium with extracts). Plates were incubated for 20–48 h at 35°C in aerobic/microaerophilic conditions, and optical density was measured (600 nm for bacteria; 530 nm for yeasts) with a microtiter plate reader (Multiskan™ FC Microplate Photometer, ThermoFisher Scientific USA). Results were calculated as percent of growth stimulation compared to positive controls using the following equation (3):
All analysis for the studied sample was performed in triplicate, and the results were expressed as mean values with standard deviation.
Results and discussion
Bioactive compounds
Based on the nutrients and bioactive compounds contents, goji berries are recognized as super fruits [35]. Although they have been less studied than the fruits of other Lycium species [36], the available literature data suggest that LRf are present source of nutrients (amino, and fatty acids, carbohydrates, including dietary fibers) and non-nutritive bioactive compounds [1, 4]. Anthocyanins, a subclass of flavonoids, determine the black colour of LRf, distinguishing them from other fruits of Lycium species [16]. In addition to anthocyanins, LRf contains other polyphenolic compounds [4, 37]. The total contents of the main classes of polyphenols in LRf obtained in this study are shown in Table 1.
Content of bioactive compounds in the LRe
Content of bioactive compounds in the LRe
GAE–gallic acid equivalents; CE–catechin equivalents; C3G–cyanidin-3-glucoside; CAE–caffeic acid equivalents.
The obtained TPC content was about 1.4 times higher than dried LRf from Turkey [38] and 5 times higher than fresh LRf from Serbia, as we previously reported [16]. Moreover, the TPC was higher than in the acetone/water/acetic acid extracts (2.17–9.01 mg GAE /g) of dried LRf cultivation among different regions of China [39]. The higher TPC in this study might be related to the favourable effects of freeze-drying on TPC compared to sun-drying and hot air-drying [15]. Moreover, methanol is a more efficient solvent for total phenol extraction than water and acetone [5].
Polyphenol compositions varied among goji species. The TFC was lower, but the TAC was higher in the LRf than in the fruits of the other Lycium species [16, 37]. The TFC value determined in this study is comparable to previous literature data (0.27–12.32 mg/g DW) [40]. Anthocyanins, the most abundant flavonoid compounds in LRf, comprise about 2.3% of the dried fruit [1, 9]. Our result for TAC was comparable to anthocyanin content in freeze-dried samples of LRf from China (7.9 mg C3G/g DW) [23] and slightly higher compared to dried samples of goji berries from the Trans-Himalayan region (6.08 mg C3G/g DW) [41].
The HPLC-DAD-ESI-MS analysis revealed the presence of six anthocyanins: four were petunidin glycosides, one was delphinidin glycoside, and one was malvidin glycoside (Table 2).
Phenolic compounds in the LRe identified by LC-MS analysis
t R –retention time.
They consisted of aglycones and sugar moieties acylated with p-coumaric or caffeic acid. Based on peak areas of the recorded chromatograms, the petunidin derivative (retention time 19.5 min) could be considered the quantitatively dominant constituent (Supplemental Materials, Fig. S1). These results align with previous studies on the chemical composition of LRf originating from Asian countries [9, 23]. Limited data are available on the polyphenol profile of LRf cultivated in European countries. The anthocyanin profile of LRf in this study was very similar to that of LRf cultivated in North Macedonia [5].
Our results are consistent with previous studies reporting that petunidin derivatives constitute more than 95% of black goji anthocyanins. They are characterized by 3,5-diglycosylation and acylation by phenolic acids, such as ferulic, p-coumaric, and caffeic acid, and the coexistence of cis- and trans-isomers [9, 42]. Among them, petunidin 3-p-coumaroylrutinoside-5-glucoside was the major phenolic in the LRf [2]. In addition to improving photo- and thermo stability, acylation influences the biological activity of anthocyanins [43]. Previous studies highlighted the effects of processing factors on the anthocyanin composition of black goji [44, 45]. In general, air- and oven-dried LRf exhibit significant qualitative and quantitative variations in anthocyanins compared to fresh and freeze-dried LRf. In contrast to fresh and freeze-dried samples, air- and oven-dried LRf did not contain 5-O-glucoside derivatives. Some authors also identified pelargonidin in oven-dried LRf [44]. In previous research, Wang et al. [46] performed anthocyanins analysis in freeze-dried LRf after a storage period. Among the five identified anthocyanidins, delphinidin was predominant (about 51%), followed by cyanidin, petunidin, malvidin, and peonidin. In addition to processing factors, the degree of ripeness and variety could also influence the LRf anthocyanin profile [43]. Besides anthocyanin glycosides, weidentified the hydroxycinnamic acid derivatives (chlorogenic acid and dicaffeoylquinic acid) and spermidine derivatives. Analysis of the sample before and after hydrolysis showed that hydroxycinnamic acids were present in the LRf, mostly in bound form. The amount of caffeic acid and its derivatives found in complex metabolites was notably higher than the corresponding quantity of p-coumaric acid (Table 1). During passage through the gastrointestinal tract, under the influence of enzymes and variable pH values, it is possible that hydroxycinnamic acids are released from their conjugates, thereby becoming available for absorption or for activity at the local level. Therefore, from a nutritional point of view, for estimating the hydroxycinnamate intake, not only free phenolic acids but also their glycosides, depsides, and other related compounds should be considered.
In line with previously reported data [47–49], our results indicated a high level of chlorogenic acid in the LRf. The presence of chlorogenic acid is, at least partly, responsible for antioxidative, anti-inflammatory and many other health-protective effects of LRf [48]. However, the content of chlorogenic acid in this study is higher than that reported in other studies [47–49], which could be influenced by the origin and genetics of the plant material, environmental, harvest, and post-harvest factors, extraction procedures and analytical technique used.
Spermidine derivatives occurred as conjugates of spermidine and dihydrocaffeic acid or caffeic acid. They were detected in free form or as complexes with hexose. It was proposed that the hexose linkage to spermidine derivatives contributes to their stability [5].
The antioxidant properties of LRe was evaluated in terms of free radical scavenging ability (DPPH, ABTS), reducing power (FRAP, CUPRAC), and ability to inhibit lipid peroxidation (β-carotene bleaching test). The results obtained are shown in Table 3.
Antioxidant and anti-inflammatory effects of LRe
Antioxidant and anti-inflammatory effects of LRe
TE—Trolox equivalents, 15-LOX—15-lipoxygenase, aexpressed as IC50 value.
Liu et al. [4] reported that phenolics accounted most for the antioxidant activity of LRf. Also, previous studies reported that LRf has higher antioxidant potential than red goji berries due to higher anthocyanin content [50, 51] and that the TPC positively correlated with antioxidant potential assessed by different assays [4, 9].
Lipoxygenases produce inflammatory leukotrienes and lipoxins from arachidonic acid [52]. Evidence shows that extracts from various plants can be considered weak to excellent LOX inhibitors [53]. In the present study, the anti-inflammatory potential of LRe was assessed by tinhibiting 15-LOX, and the results were expressed as IC50 values (Table 3). To our knowledge, this is the first data on in vitro 15-LOX inhibitory activity of LRe. However, several studies have demonstrated the anti-inflammatory effects of polysaccharides and crude anthocyanins from LRf [54, 55]. Furthermore, recent research conducted by Magãlhaes et al. [56] showed that LRe exhibited significantly more potent anti-inflammatory effects compared to red goji berry extract, based on the results of the inhibition of COX-2, modulation of nitric oxide release, and the influence on gene expression of proinflammatory cytokines in BV2 microglial cells stimulated with LPS.
As shown in Table 4, LRe exhibited in vitro inhibitory effects on α-amylase, α-glucosidase, acetylcholinesterase, and tyrosinase.
Enzyme inhibitory effects (IC50) of LRf
Enzyme inhibitory effects (IC50) of LRf
Positive controls: acarbose ( α-Amylase and α-Glucosidase), galantamine (acetylcholinesterase), kojic acid (tyrosinase).
α-Amylase and α-Glucosidase play crucial roles in the digestion of dietary carbohydrates into glucose. Therefore, inhibiting these enzymes can potentially slow down the increase in blood glucose levels, leading to the suppression of postprandial hyperglycemia, which could be helpful in the management of type 2 diabetes mellitus. Our data indicates that the LRe exhibited a greater efficacy in inhibiting α-Amy than α-Gls. However, inhibitory activities against both enzymes were lower than acarbose (a positive control). Previous studies have demonstrated in vitro antidiabetic properties of fruit extracts from L. barbarum and L. chinense [59]. To the best of our knowledge, there are no data regarding the impact of LRe on α-Amy and α-Gls. However, it was shown that LRf anthocyanins have inhibitory effects on α-Gls in Saccharomyces cerevisiae and Caco-2 cells. The IC50 values for these inhibitory effects were 1.32–1.57μg/mL and 25.3μg/mL, respectively, comparable to acarbose, a known α-Gls inhibitor [12, 60].
Furthermore, it was demonstrated that the effects of anthocyanins against α-Gls were augmented through in vitro gastrointestinal digestion. Anthocyanidins form a tertiary complex known as anthocyanidin-α-glucosidase substrate, further enhancing their inhibitory activity [60]. Also, non-anthocyanin compounds in LRf, such as the phenylpropanoid derivative ethyl-p-trans-coumarate, demonstrated α-Gls inhibitory effects comparable to those of the positive control (acarbose) [61]. Wu et al. [62] demonstrated that pectins from LRf, obtained by various extraction procedures, exerted inhibitory activities on α-Gls. However, their inhibitory potential was much lower than that of acarbose.
The inhibition of acetylcholinesterase (AChE), which breaks down the neurotransmitter acetylcholine, is a promising approach to treating neurodegenerative disorders [63]. Although we observed inhibition of AChE by LRe, the inhibition rate was much lower than galantamine as a positive control (Table 4). In contrast to LRf, it previously demonstrated that L. barbarum berries inhibited AChE [58]. Ruffo et al. [64] suggested the role of carotenoids, especially zeaxanthin, in AChE inhibition. Zhao et al. [61] performed an in vitro screening of the bioactivity of 26 phenylpropanoid derivatives isolated from LRf. However, none of the compounds showed AChE inhibitory activity.
Due to its role in melanin synthesis, increased tyrosinase activity has been linked to excessive skin pigmentation, skin cancers and neurological disorders. Therefore, potential tyrosinase inhibitors, especially from natural sources, attract attention for cosmetic and medical purposes [65]. The results of inhibitory activities of LRe and kojic acid (positive control) are present in Table 4. Like against other enzyme activities, LRe was a less potent anti-tyrosinase inhibitor than the positive control. Compared to our result, Shen et al. [66] reported about 1.8-fold stronger TIA of the aqueous hydrochloric acid extract of commercial dried LRf (IC50 = 2.95±0.02 mg/mL), and more than 2-fold potent TIA for purified anthocyanins (IC50 = 1.48±0.06 mg/mL), but less than kojic acid.
Prebiotics are indigestible compounds that selectively stimulate the gastrointestinal microbiota’s composition and/or activity [67]. Probiotic bacteria have favourable health benefits, including reducing the risk of inflammation, ulcers, colon cancer, gastroenteritis, and metabolic syndrome [68]. In addition to the prebiotic properties of carbohydrates, many studies have highlighted the ability of polyphenols and their metabolites to modulate the intestinal microbiota [69, 70]. These effects are linked to their structures; as electron donors, they could prevent oxidative stress, and reduce inflammation [71]. It was demonstrated that methanol extract from black goji berry (0.125–2 mg/mL) did not inhibit the growth of Gram-positive and Gram-negative bacteria and the yeast Candida albicans [16]. As shown in Fig. 1, LRe (0.3–5.0 mg/mL) showed dose-depending growth-promoting effects on all tested microorganisms and their mixtures. Addition of LRe resulted in 11.1–129% higher growth stimulation in each strain compared to the positive control (red line).
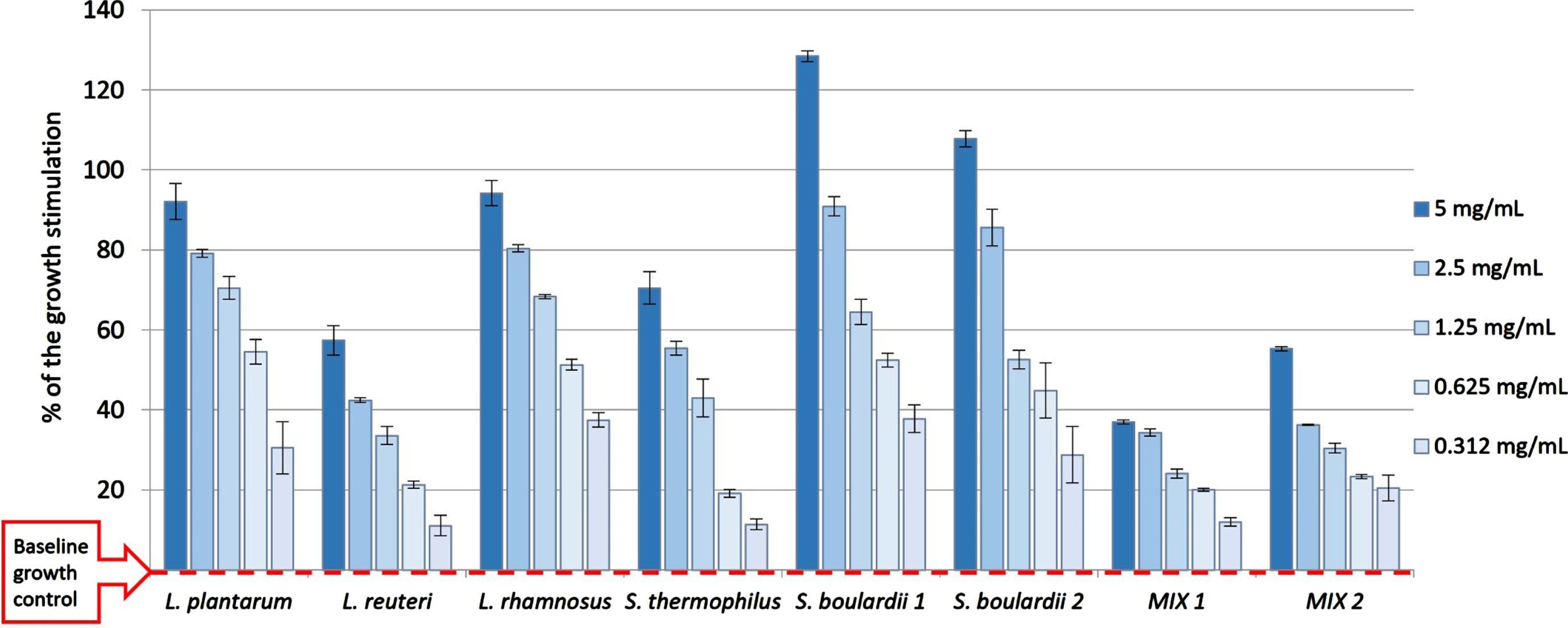
The prebiotic potential of the LRe on various probiotic strains of microorganisms. The data in the figure are presented as the mean percent of growth stimulation±SD compared with the baseline growth of the positive control for each strain (red line).
Among all tested microorganisms, LRe the most stimulated the growth of the yeast Saccharomyces boulardii, 2.8 times more than the control (Supplementary Materials, Table S2). The observed prebiotic properties could be attributed to anthocyanins, as the major functional ingredients of LRf. Namely, Peng et al. [72] recently demonstrated in vitro modulatory effects of LRf anthocyanins and their metabolites on probiotic (Lactobacillus and Bifidobacterium) and pathogenic bacteria (Escherichia/Shigella species). One proposed mechanisms for prebiotic properties included the selective fermentation of anthocyanins to some short-chain fatty acids. Several recent studies reported that anthocyanins from LRf reduce body weight in animal models by altering the gut microbiota, suggesting their anti-obesity effects [73, 74].
Black goji berries are gaining increasing attention as an underutilized source of health-promoting bioactive compounds with potential functional food and cosmetic applications. Although the cultivation of L. ruthenicum is widespread outside Asia, there is very little literature on the phytochemical composition and bioactive potential of LRf from Europe. The chromatographic analysis confirmed that hydroxycinnamic acid derivatives and acylated anthocyanins are the predominant polyphenols. In addition to antioxidant properties, the results confirmed antienzyme inhibitory effects on α-amylase, α-glucosidase, acetylcholinesterase, and tyrosinase, as well as on 15-lipoxygenase. Also, LRe had growth-promoting effects on several probiotics, with a marked impact on the growth of yeast Saccharomyces boulardii. This study supports further research of LRf as sources of ingredients for developing innovative health-promoting products.
Footnotes
Acknowledgments
The authors thank “Ljuba i sinovi” from Niš for donating black goji berry samples.
Funding
This work was supported by the Ministry of Science, Technological Development and Innovation, Republic of Serbia, through a Grant Agreement with the University of Belgrade-Faculty of Pharmacy [grant number 451-03-47/2023-01/200161].
Conflict of interest
The authors have no conflict of interest to report.
Author contributions
Conceptualization, T.I., and B.V.; Methodology, T.I., N.K., S.S., M.M., G.Z., and D.B.; Formal analysis and investigation, T.I., N.K., S.S., G.Z., and D.B.; Writing—original draft preparation, T.I.; Writing—review and editing B.V.; Resources, B.V.; supervision, B.V. All authors have read and agreed to the final version of the manuscript.
