Abstract
Background:
Bilateral oophorectomy (BO) confers immediate estradiol loss. We examined prevalence and predictors of Alzheimer's disease (AD) in women with early BO comparing their odds ratios of AD to those of women with spontaneous menopause (SM).
Methods:
A cohort from UK Biobank (n = 34,603) included women aged 60 + at baseline with and without AD who had early BO or SM. AD was determined based on AD related ICD-10 or ICD-9 code. We used logistic regression to model the association of menopause type with AD. Model predictors included age, education, age at menopause, hormone therapy (HT), APOE4, body mass index (BMI), cancer history, and smoking history.
Results:
Those with early BO had four times the odds of developing AD (OR = 4.12, 95% CI [2.02, 8.44]) compared to those with SM. APOE4 (OR = 4.29, 95% CI [2.43, 7.56]), and older age (OR = 1.16, 95% CI [1.05, 1.28]) were associated with increased odds of AD in the BO group. Greater years of education were associated with reduced odds of AD for both BO (OR = 0.91, 95% CI [0.85, 0.98]), and SM (OR = 0.95, 95% CI [0.90, 0.99]), while ever use of HT was associated with decreased odds of AD only for the BO group (OR = 0.43, 95% CI [0.23, 0.82]).
Conclusions:
Women with early BO, particularly with an APOE4 allele, are at high risk of AD. Women with early BO who use HT and those with increased education have lower odds of developing AD.
Introduction
Alzheimer's disease (AD) is projected to affect 12.7 million individuals 65 and older by 2050. 1 Women comprise two-thirds of individuals with AD. 2 Thus, understanding the specific AD-related risk and resilience factors in women is pivotal to decreasing the number of persons developing AD.1,3 One key sex-related factor is the loss of 17β-estradiol (E2) during menopause.4–10 A few studies using the UK biobank (a prospective population-based database of more than 500,000 individuals with extensive phenotypic and genetic data) have analyzed risk for all cause dementia in women, but the results regarding menopause are mixed.11–16 Notably, none of these studies have focused solely on AD which is the type of dementia prevalent in women.
Thus, there is need for further research to understand how AD risk varies by menopause type. 17 Spontaneous menopause (SM) occurs at approximately 51 years of age; it is the most studied with respect to women's cognitive changes with aging. From the UK biobank, studies report that SM is associated with decreased performance on prospective memory tasks, and decreased parahippocampal and hippocampal volume. 11 Menopause can also result from surgery; women with early bilateral oophorectomy (early BO; removal of both ovaries prior to the average age of SM) experience a complete and abrupt loss of endogenous E2 production, unlike women in SM in which E2 production wanes gradually with advancing age.17,18 Early BO has been linked to higher risk of all cause of death and close to two times the risk of developing late life AD.9,19 Indeed, the association between early BO and increased risk of cognitive impairment and/or dementia is now well-established.9,10,20,21 This risk decreases linearly with older age at surgery and in those given hormone therapy (HT) until age 50. 9 As compared to women with SM, women with early BO show reductions in different memory dimensions (e.g., episodic, semantic, working memory) up to 20 years post-surgery. 6 Women who had BO surgery prior to age 49 show steeper cognitive decline over a period of up to 18 years; at autopsy, there is increased AD pathology 22 as well as decline in parahippocampal-entorhinal cortical thickness. 23 Notably, women with early BO show accelerated rates of multimorbidity for several chronic conditions, including cardiovascular and pulmonary disease. 24
However, literature from the UK Biobank related to dementia risk in women with early BO is mixed. For instance, two studies looking at lifetime exposure to estrogens reported that only hysterectomy without BO increases dementia risk.12,13 In this respect, there may have been cases of ovarian failure since even ovarian sparing hysterectomy might lead to changes in blood supply to the ovaries. 25 Other studies have reported that increased exposure to estrogens is associated with lower AD risk, 14 and earlier age at menopause with increased AD risk. 15 Although both studies included history of surgical menopause, neither study differentiated between the different types (e.g., hysterectomy without BO versus BO). Another UK Biobank study 16 did find that BO was linked to 10% higher risk for dementia than for women with SM which is in agreement with other cohorts, typically showing an increased dementia risk for women with BO9,10,20,21; however, this study found no main effects related to genetic risk score, cardiometabolic factors, or HT. Indeed, results about the benefit of HT are mixed in UK Biobank studies with some finding no benefit 12 and others reporting a positive association of HT with cognitive performance, 13 larger medial temporal lobe volumes, 11 neuroprotective effects, 14 and decreased risk of AD but only for women who had early SM. 16 Thus, with the exception of one study, the mixed results of the UK Biobank studies are surprising given that other cognitive and brain studies identify decrements in the early months and years post-BO in verbal tests and memory performance, including associative, spatial, working and episodic memory.26,27 There is also volume loss in the dentate gyrus/CA2/CA3 hippocampal subfield. 28 However, E2-based HT ameliorates some of these changes.26,28,29
In order to clarify the range of possible risk and resilience proxies for AD in women from the UK Biobank, we accessed the cohort to examine whether BO prior to SM (early BO) would increase the odds of having AD. We hypothesized that 1) women with early BO would have greater odds of AD than women with SM; 2) women with early BO and APOE4 would be at greater odds than those without APOE4; and 3) women with early BO who had ever taken HT would be at reduced odds of AD compared to those who had never taken HT. Our approach was to directly compare women with early BO without AD (BO), women with early BO and AD (BO-AD), women with SM without AD (SM), and women with SM and AD (SM-AD) to determine risk and resilience factors for these different menopauses.
Methods
Participants
Data were obtained from UK Biobank which comprises demographic, medical, genetic, and lifestyle variables. 30 The sample was restricted to women aged 60 and older who either had BO at age 49 or younger (4403 of whom 47 developed AD) or SM at age 51 or older (30,200 of whom 61 developed AD). We included only women who had BO prior to age 49 to make sure we captured effects related to BO and not SM, i.e., bilateral oophorectomy before SM. Regarding SM, it has been reported that the common average age at SM is 51 years, 31 and thus we included in our SM group women who reported SM at 51 or older. Menopause type was determined by the questionnaire in their “female-specific factors” category. Separate self-report questions were used to assess BO and SM and the ages at which menopause occurred. Use of HT was collected as a dichotomous measure (never use vs ever use). Diagnosis for those who developed AD was determined with at least one AD-related ICD-10 or ICD-9 code. 32
All human subjects provided informed consent to participate in the UK Biobank (Figure 1).
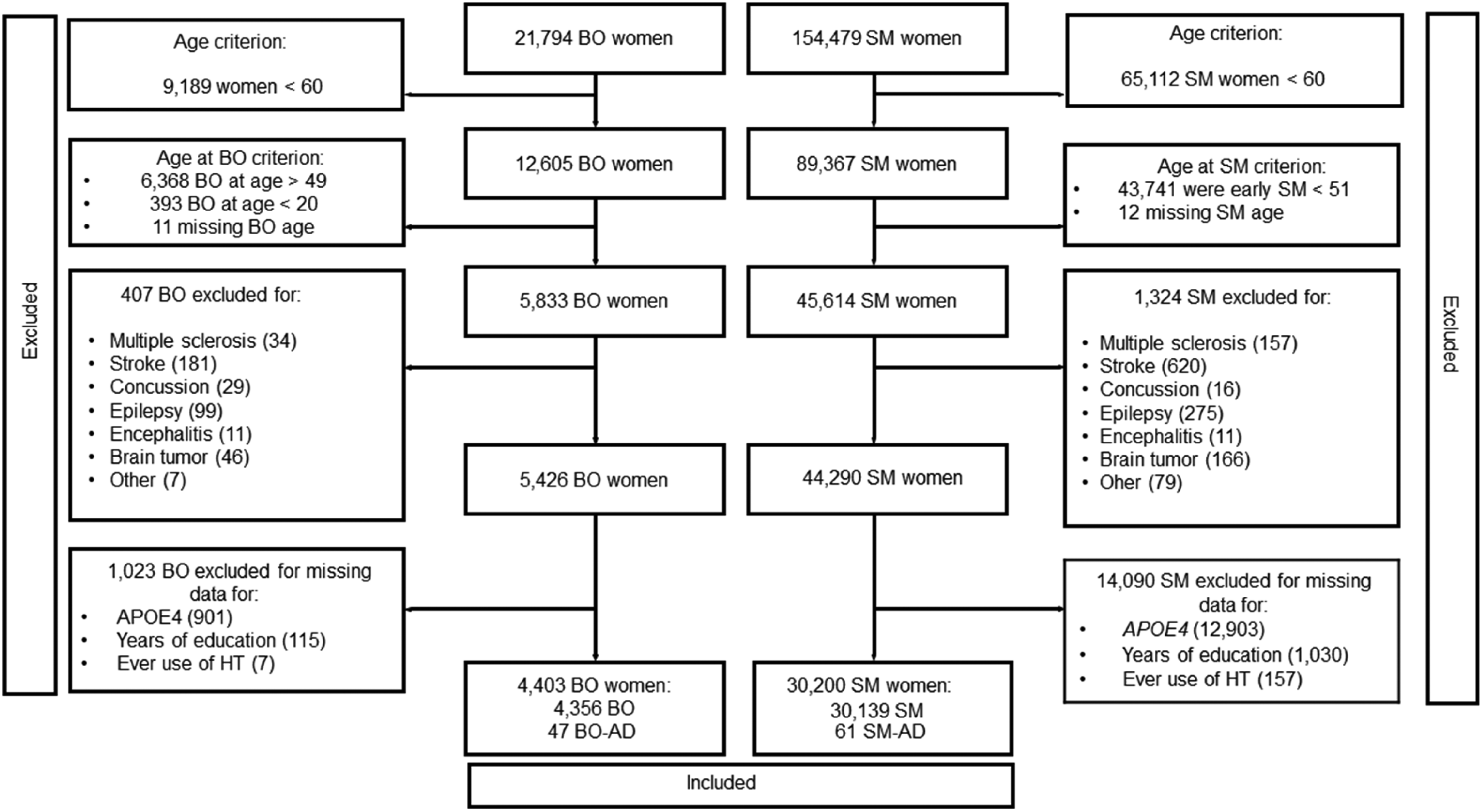
Flow diagram of participant selection
Measures
For APOE genotype, UK Biobank used Applied Biosystems UK BiLEVE Axiom array by Affymetrix and Applied Biosystems UK Biobank Axiom Array which share 95% common content 33 to determine single nucleotide polymorphism (SNP) data for rs429358 and rs7412. 34 The APOE ε4 carriers had either one ε4 allele (ε3/ε4, ε4/ε4) or two ε4 alleles, we coded APOE genotype as a dichotomous variable (APOE4 status = yes/no). Participants with an E2/E4 genotype were included in the no E4 group because the E2 allele is thought to counteract the deleterious effects of E4 on AD risk. 35 Years of education at baseline were defined using International Standard Classification of Education categories 36 : No qualifications = 7 years; Certificate of Secondary Education/O level/General Certificate of Secondary Education, or equivalent = 10 years; A level/AS level or equivalent = 13 years; other professional qualification = 15 years; National Vocational Qualification, Higher National Diploma or equivalent = 19 years; college or university degree = 20 years. To calculate body mass index (BMI) at baseline, weight was divided by the square of height (Kg/m2). Weight and estimated percentage fat were measured with the Tanita BC418ma bioimpedance device (Tanita, Tokyo, Japan). Diabetes, hypertension, depression, and anxiety diagnoses were determined using ICD-10 codes as dichotomous measures (yes/no). Cancer history was determined by UK biobank cancer register based on ICD-10 records and treated as a dichotomous variable. Detailed information about UK Biobank data and protocols is available. 37
Statistical analyses
All analyses were conducted in R 4.1.2. 38 Bivariate analysis (ANOVA or χ2 tests) were used to compare the following groups: women with early BO without AD or any other dementia (BO), women with early BO and AD (BO-AD), women with SM without AD (SM), and women with SM and AD (SM-AD). Characteristics differing between groups were entered into Firth's bias-reduced logistic regression analyses 39 as full model predictors. We used Firth's regression to account for the relatively rarer cases of AD participants compared to non-AD participants in this cohort. Exponentiation was then used on the regression coefficients to calculate odds ratios. To determine the combined and stratified effects of model predictors on odds of AD, analyses were conducted for combined menopause types (BO and SM) as well as for menopause groups separately. Model predictors included: menopause type (combined model only), APOE4, ever use of HT, age, years of education, age at menopause, BMI, cancer history, and smoking history. A post-hoc sensitivity analysis was also conducted to clarify whether the observed findings were primarily due to BO status as opposed to early menopause. This analysis included all respondents with SM > 40 and those with BO <49.
Results
Participant characteristics
A total of 34,603 participants were included: BO (n = 4356), BO-AD (n = 47), SM (n = 30,139), SM-AD (n = 61). Overall, participants were 63.8 years old on average (SD = 2.79) at baseline, had 13.0 years of education (SD = 5.11), and 25.4% had an APOE4 allele. Less than 5 participants reported diabetes or hypertension in the SM-AD group, and fewer than 2% of the women in each group had a diagnosis of depression or of anxiety. Thus, these variables were removed from further analysis.
ANOVAs revealed that there was a statistically significant difference among the groups in age F (3, 34599) = 7.28, p < 0.001; education F (3, 34599) = 41.46, p < 0.001; age at menopause F (3, 34599) = 20318, p < 0.001; and BMI F (3, 34599) = 33.24, p < 0.001. Tukey's HSD test adjusting for multiple comparisons found that the mean value of age was significantly different between women with BO and women with BO-AD (p < 0.001, 95% C.I. = 0.68, 2.78); and between women with BO-AD and women in SM (p = 0.002, 95% C.I. = −2.70, −0.61). Regarding education, Tukey's test showed a significant difference between the following groups: BO-AD and BO (p = 0.01, 95% C.I. = −4.16, −0.32); SM and BO (p < 0.001, 95% C.I. = 0.63, 1.05); BO-AD and SM (p = 0.001, 95% C.I. = 1.18, 5). Tukey's test for age at menopause, showed a significant difference between the following groups: BO and BO-AD (p < 0.001, 95% C.I. = −2.76, −0.69); SM and BO (p < 0.001, 95% C.I. = 10.79, 11.02); SM-AD and BO (p < 0.001, 95% C.I. = 10.17, 11.19); SM and BO-AD (p < 0.001, 95% C.I. = 11.60; 13.66); SM-AD and BO-AD (p < 0.001, 95% C.I. = 11.45, 14.18). Regarding BMI, Tukey's test indicated a significant difference between SM and BO (p < 0.001, 95% C.I. = −0.98, −0.57), as well as a trends towards significance between SM-AD and BO ((p = 0.069, 95% C.I. = −3.14, −1.53).
χ2 tests showed a significant difference in HT χ2(3, N = 34.603) = 2377.2, p < 0.001; APOE4 χ2(3, N = 34.603) = 38.73, p < 0.001; and cancer history χ2(3, N = 34.603) = 48.04, p < 0.001. There were no significant differences in smoking history χ2(3) = 3.19, p = 0.36. Participants with BO were more likely to have ever taken HT; and those with BO who developed AD were more likely to be APOE4 carriers and have cancer diagnoses (Table 1).
Participant characteristics.
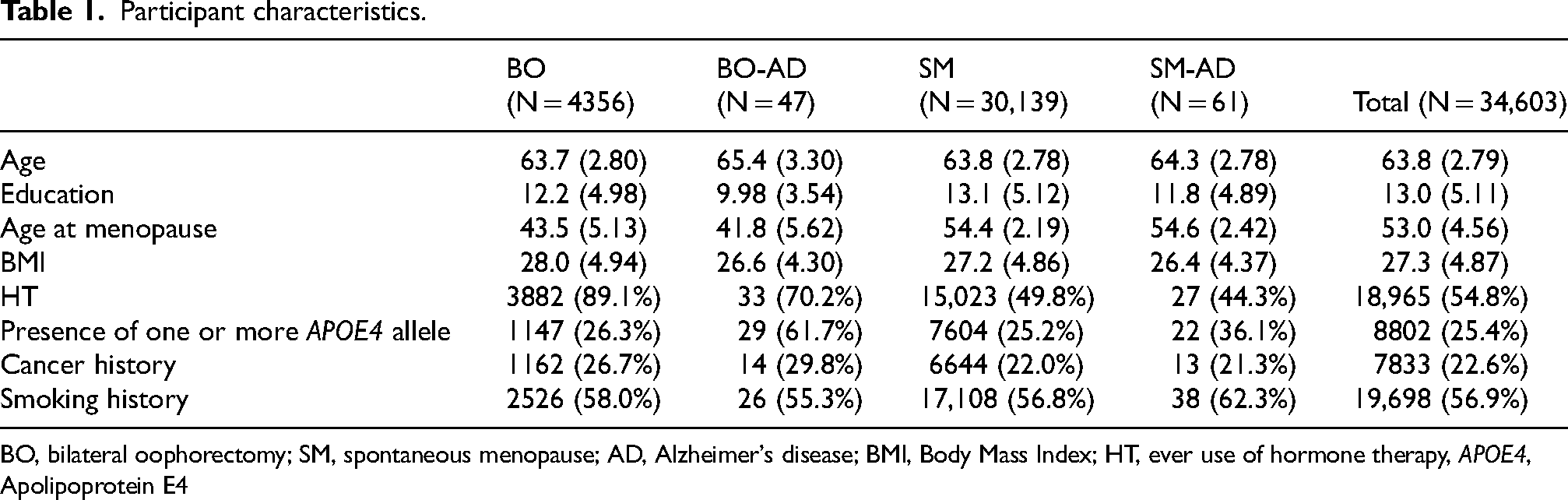
BO, bilateral oophorectomy; SM, spontaneous menopause; AD, Alzheimer's disease; BMI, Body Mass Index; HT, ever use of hormone therapy, APOE4, Apolipoprotein E4
Combined model
The combined menopause type model showed an association of BO with increased odds of AD (OR = 4.12, 95% CI [2.02, 8.44], p = <0.001). Additionally, carrying an APOE4 allele (OR = 2.55, 95% CI [1.76, 3.69], p = <0.001), and older age (OR = 1.11 per year, 95% CI [1.05, 1.19], p = 0.001) were associated with higher odds of AD, while ever use of HT (OR = 0.59, 95% CI [0.39,0.89], p = 0.014), every unit higher BMI (OR = 0.94, 95% CI [0.90,0.98], p = 0.005), and each additional year of education (OR = 0.93, 95% CI [0.90, 0.97], p = 0.001) were associated with decreased odds of AD (Table 2; Figure 2A).
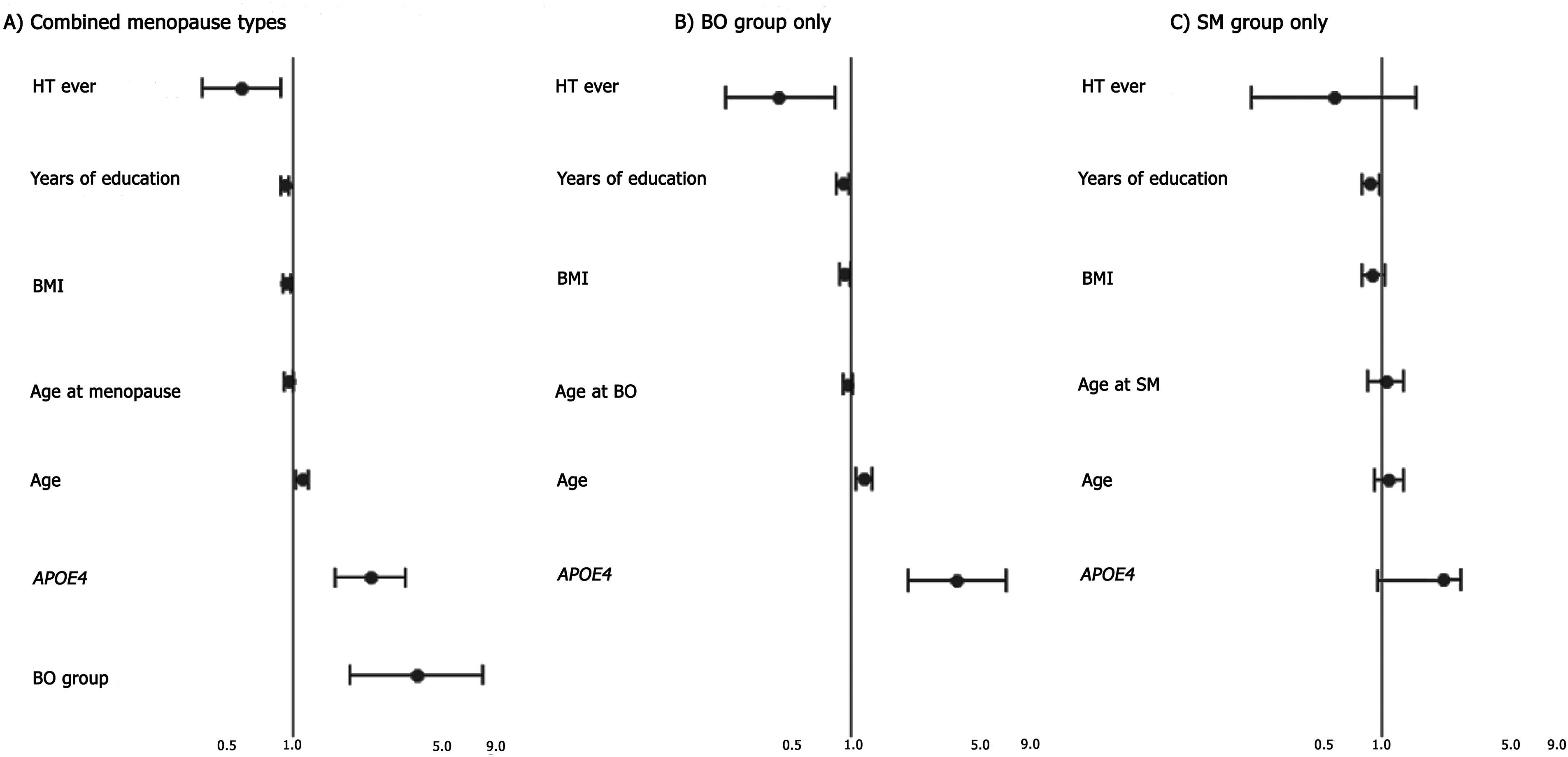
Key risk and resilience factors for each model. (A) Odds ratios for the combined menopause model. (B) Odds ratios for the model including only women with BO. (C) Odds ratios for the model including only women with SM.
Odds of Alzheimer's disease by menopause type.
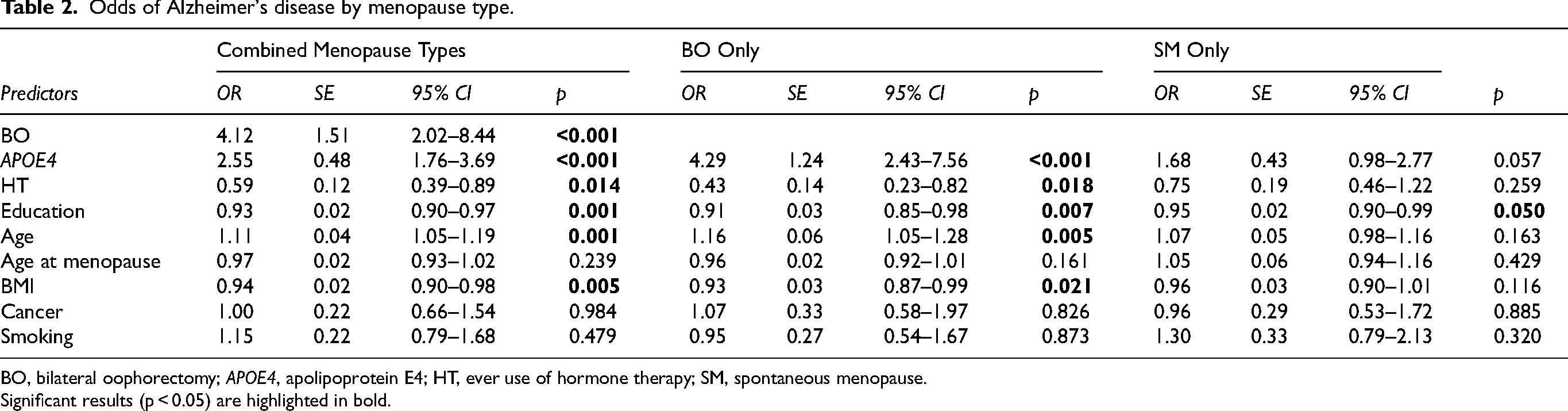
BO, bilateral oophorectomy; APOE4, apolipoprotein E4; HT, ever use of hormone therapy; SM, spontaneous menopause.
Significant results (p < 0.05) are highlighted in bold.
Stratified models
The stratified model for BO group only (Table 2, Figure 2B) showed APOE4 allele (OR = 4.29, 95% CI [2.43, 7.56], p = <0.001), and older age (OR = 1.16, 95% CI [1.05, 1.28], p = 0.005) were associated with a higher odds of AD. Ever use of HT (OR = 0.43, 95% CI [0.23, 0.82], p = 0.01), every unit higher BMI (OR = 0.93, 95% [0.87–0.99], p = 0.02) and each additional year of education (OR = 0.91, 95% CI [0.85, 0.98], p = 0.007) were associated with decreased odds of AD (Table 2, Figure 2B). In the sample restricted to women with SM, years of education might be related with lesser odds of AD (OR = 0.95, 95% CI [0.90, 0.99], p = 0.05) - table 2, Figure 2C. All other factors included in the model for women with SM failed to reach statistical significance.
Sensitivity analysis
Results from the sensitivity analysis which considered all women with SM > 40 and those with BO <49 showed similar results to the previous analysis. Overall, BO (OR = 5.25, 95% CI [3.34–8.25], p = <0.001), carrying an APOE4 allele (OR = 2.64, 95% CI [1.98–3.53], p = <0.001), and older age (OR = 1.12, 95% CI [1.07–1.18], p = <0.001) were associated with increased odds of AD. Higher years of education mitigated these effects as it was associated with reduced odds of AD (OR = 0.96, 95% CI [0.93–0.99], p = 0.008). Importantly, ‘age at menopause’ was not significant (OR = 0.99, 95% CI [0.96–1.02], p = 0.50), which indicates that the risk of AD for the BO group is associated to the removal of ovaries (BO) and not to early age at menopause.
Discussion
Using the UK Biobank, a large population-based cohort, we identified five possible risk and resilience factors for women with early life BO: APOE4, HT, education, age, and BMI. Some of these factors are in agreement with what others have found using the entire UK Biobank cohort, and others, are different. The differences might arise because previous studies were of the whole female cohort, while ours included only the subset of early BO and SM in order to compare odds of AD in these groups alone. Also, previous studies have analyzed all cause of dementia while our study focused only on AD. Thus, our study is the first from the UK Biobank to show odds of AD specifically related to early BO in comparison with SM.
Previous studies looking at reproductive factors and cognitive decline have had sources of variability,40,41 one of which importantly is a concatenation of different menopause types 17 or dementia types.42,43 Perhaps, it is due to this variability that studies using the UK Biobank have themselves reported mixed results with respect to menopause type and increased risk for AD, with some showing no effect of early BO 13 and others, an effect for hysterectomy without BO and only in women with low socioeconomic status. 12 Since early BO leads to an abrupt loss of E2, it well might have different long-term effects than SM.17,24
Increased odds of AD: risk factors
We found that early BO is associated with a fourfold increased odds of AD compared to SM. This is high compared to previous studies finding a 1.5 times increase in the odds of AD compared to controls. 9 This difference may be due to including the APOE4 allele in the model. E2 loss may interact in complex ways with genetic and metabolic risk factors.10,44 In animal models, estradiol's neurotrophic effects are lost in the presence of the genetic risk factor for late-onset AD, apolipoprotein ε4 (APOE4 45 ). A recent meta-analysis concluded that the presence of an APOE4 allele is generally associated with approximately two- to threefold risk of AD in women. 46 However, in our BO group, the presence of the APOE4 allele indicated a fourfold increased odds of AD compared to women in SM (OR = 4.12, 95% CI [2.02, 8.44]). This OR estimate may be larger compared to previous studies on AD risk because previous studies have not analyzed data focused solely on women with early BO- for this population, carrying an APOE4 allele may be more deleterious. Given the interaction of E2 with APOE4, 47 the detrimental effects of E2 loss may combine with those of APOE4 to further increase AD risk, placing women with early BO and APOE4 in a state of ‘double jeopardy’.
Age at menopause was not associated with odds of AD in any of our models. This may be due to the small number of respondents who developed AD which limits the power of the analysis. However, we were interested in analyzing age at menopause in all women, and then separated by menopause type because younger age at BO has been linked to cognitive decline, 22 and AD risk.9,20 Also, some of the previous studies using the UK Biobank reported age at menopause to be related to AD risk, but without differentiating menopause type.14,15 Note that in some of these studies, dementia risk was found when BO was performed before the age of 40 years but also after 55 years. The latter finding is in contrast to multiple other studies.9,10,19,48 This U-shape relationship reported in 2 studies is complex; one needs to consider deeply whether or not it is actually related to early surgical menopause. For instance, if BO was performed after 55 years old for some women, the effect might be related to SM before BO. Thus, we wanted to explore these relationships using the same cohort, but the results were not significant in our models. We also conducted a supplementary analysis to disentangle the role played by BO status as opposed to early menopause in the association with AD. This sensitivity analysis included all respondents with SM > 40 and those with BO <49. As shown in Supplementary Table 1, ‘age at menopause’ alone was not significant in the combined model (OR = 0.99, 95% [0.96–1.02], p = 0.50), while BO was significant and the odds ratio was also higher than in the previous analysis [(OR = 5.25, 95% [3.34–8.25], p = <0.001)]. These findings indicate that it is BO rather than ‘age at menopause’ which is driving the association with AD. Future studies using other cohorts could investigate this question further.
Decreased odds of AD: factors of resilience
Ever taking HT was associated with less than half the odds of AD in women with early BO, which underscores the importance of HT for women with BO in mitigating the odds of AD. While our study is in accordance with the study by Lindseth et al. 13 in a cohort of 221,124 women from the UK Biobank showing the benefits of HT to mitigate cognitive decline, it is in contrast with those studies using the same cohort which did not find an effect for HT related to dementia.12,15 Again, this may be because their cohort and comparator groups were different than ours. Previous work has demonstrated a beneficial effect of estradiol replacement for short-term cognitive improvements and in mitigating later-life AD risk. 9 As well, smaller cohort studies have shown that without estradiol therapy, there are cognitive declines shortly after BO compared to preoperative baselines 27 and at an average of five years post-oophorectomy compared to age-matched controls. 26 E2-based HT can ameliorate these changes. 49 Future research should investigate the effect of length of time between BO or SM and the onset of HT treatment on HT efficacy as well as the relative benefits of type, route of administration, and duration of HT for the different menopause types.
We also observed that, in both menopause types, more education was associated with decreased odds of AD. In mixed sex studies controlling for sex, education has been suggested as a proxy for cognitive reserve—the brain's capacity for resilience throughout healthy or pathological aging, including AD.50–52 Indeed, higher education has been linked to an older age at onset of menopause, 53 and this seems to be the case in our cohort of women in which the average age of SM is 2 years older than the typical average of 51. Note that UK biobank participants have higher education and socioeconomic position than the average person their age in Britain. 54 However, there is little research directly examining the role of education on cognitive reserve specifically for women with early E2 loss. In this respect, only one study using the UK Biobank 12 reported lower socioeconomic status (SES) linked with dementia risk but only in women who had hysterectomy. Our study is the first to suggest that education is beneficial for all menopause types. It may be especially beneficial for women with early BO. The interactions between education as a resilience factor in the face of E2 loss may be an important aspect of cognitive reserve in women and a critical area for further study.42,55
We also found a modest positive association of higher BMI with decreased odds of AD in both the combined model, and women with BO. In this respect, the literature shows mixed results with some studies reporting high BMI or obesity associated with increased dementia risk,56–59 and others reporting obesity as a protective factor against dementia. 60 Our study is also the first to report this association in the UK Biobank cohort. It is possible that higher BMI at an older age is associated with a decreased AD risk because adipose tissue makes estrone 61 which, in the absence of E2, may help maintain cognitive function. 62
Taken together, our findings support and extend recent findings from the UK Biobank cohort which reported an association between women's reproductive lifespan and cognitive impairment, 13 as well as an increased odds of dementia in women with BO. 16 Our results are also in accordance with reports using other large data bases.9,10,20 Importantly, they also provide further evidence for greater odds of AD in women with BO compared to their SM counterparts and identify a particular risk for women with early BO and APOE4, underscoring the differences between two types of menopauses, early BO and SM. They also indicate that women with BO may benefit from early monitoring and prevention efforts, especially if they also carry an APOE4 allele. Further, we found that ever use of HT specifically associates with lower odds of AD for women with BO, and that education decreases the odds of AD for both menopause types. This suggests that education and ever use of HT should be considered as resilience proxies in women.
Strengths and limitations
One strength of this study is that the participants are from a large, prospective, population-based database that collected a wide variety of demographic, medical, genetic, and lifestyle data. This provided a large number of women with early BO and allowed examination of some modifiable and unmodifiable risk factors for AD. A limitation, however, is that when we specified the groups—women with early BO and SM who developed AD—the design was unbalanced. This may have led to wide CIs especially with respect to the risk of APOE4. However, to counteract the unbalanced design, we used the Firth method which has proved to be efficient with these type of issues. 39 Another limitation is that the current cohort had a relatively low percentage of women with BO at 38 or earlier. Considering the strongest relationship between BO and AD risk has been observed in this group,22,48 it is possible that our findings may be underestimating BO's deleterious effects. Moreover, HT use in our study was self-reported. In general, evidence on the use of HT as a mitigating factor against cognitive decline and AD has been mixed. Specifically, there is a discrepancy between the findings of randomized clinical trials63,64 and observational studies using self-reported HT use.65,66 Our results regarding HT are in line with previous observational studies using self-reported HT and a previous UK Biobank study using the same measure. 11 It would have been useful to include information about the time window for participant's HT and this is an important factor that future studies should consider. As was previously mentioned, investigation of type, duration and time to onset of HT use would benefit future research. Future studies could also benefit from accounting for immunological changes or psychological stress, which would help obtain a broader perspective on the effects of BO in aging.
It is also important to note that participants in the UK Biobank are less likely than the general population to partake in adverse health behaviors, have fewer health conditions, and are less likely to live in socioeconomically depressed areas—evidence of a ‘healthy volunteer’ selection bias that may reduce generalizability. 54 This was seen in our cohort in which fewer than 2% of the participants were diagnosed with depression or anxiety, and thus we could not analyze the association among depression, anxiety and AD. Similarly, hypertension and diabetes were more likely in the early BO group, but still very few participants were diagnosed with hypertension or diabetes. These typical AD comorbidities were even less likely in the SM-AD group (less than 5 participants). It is notable that in otherwise relatively healthy women, early BO is associated with a substantial increased odds of developing AD.
Conclusion
We identified the odds of AD in women with early BO. Early BO itself greatly increased the odds of AD. In addition, for women with early BO, carrying one or more APOE4 allele was associated significantly with increased odds of AD. Ever use of HT, higher BMI and greater education were associated with lesser odds. Events conferring early E2 loss are not always avoidable and are, in fact, recommended in some cases (e.g., as prophylaxis against cancer risk in BRCA 1/2 carriers). 67 Thus, it is critical to understand how early E2 loss may affect cognition and AD risk and what factors may confer resilience. Our study identifies women with the combination of early BO and APOE4 as likely a particularly at-risk group and demonstrates the importance of ever use of HT for lowering that risk. Further, the decrease of estradiol during SM is a universal event for all women who reach older age with ovaries intact, and our study points to the role of education in lowering cognitive decline and odds of AD for women with SM.7,68 Broadly, this work underscores the importance of continued research into the effects of estrogens on cognition and odds of AD for all women and contributes to a greater understanding of sex differences in AD.
Supplemental Material
sj-docx-1-alz-10.3233_JAD-240646 - Supplemental material for Associated risk and resilience factors of Alzheimer’s disease in women with early bilateral oophorectomy: Data from the UK Biobank
Supplemental material, sj-docx-1-alz-10.3233_JAD-240646 for Associated risk and resilience factors of Alzheimer’s disease in women with early bilateral oophorectomy: Data from the UK Biobank by Noelia Calvo, G Peggy McFall, Shreeyaa Ramana, Michelle Galper, Esme Fuller-Thomson, Roger A Dixon and Gillian Einstein in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
This research uses data provided by patients and collected by the NHS as part of their care and support (application reference 57934). The authors would like to thank Dr Rebekah Reuben for contributing with preliminary analysis for this manuscript.
Author contributions
Noelia Calvo (Data curation; Formal analysis; Investigation; Methodology; Writing – original draft; Writing – review & editing); G Peggy McFall (Conceptualization; Methodology; Supervision; Writing – review & editing); Shreeyaa Ramana (Data curation; Writing – review & editing); Michelle Galper (Data curation; Writing – review & editing); Esme Fuller-Thomson (Conceptualization; Methodology; Supervision; Writing – review & editing); Roger Dixon (Conceptualization; Methodology; Supervision; Writing – review & editing); Einstein Gillian (Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; Writing – review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wilfred and Joyce Posluns Chair in Women's Brain Health and Aging from the Posluns Family Foundation, Women's Brain Health Initiative, Canadian Institutes of Health Research (CIHR), Ontario Brain Institute, Center for Aging + Brain Health Innovation, and Alzheimer Society of Canada to GE [grant WJP-150643 and WJD-180960], the Canadian Consortium on Neurodegeneration in Aging (CCNA) Phase II to GE (grant CCNA 049-04; CIHR reference number: CNA 163902), and the Jacqueline Ford Gender and Health Fund to GE. RAD acknowledges support from the CCNA through a funding partnership between Alberta Innovates (#G2020000063) and CIHR (#163902).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Supplemental material
Supplemental material for this article is available online.
