Abstract
Background:
Portable digital health technologies (DHTs) could help evaluate non-cognitive symptoms, but evidence to support their use in patients with dementia with Lewy bodies (DLB) is uncertain.
Objective:
1) To describe portable or wearable DHTs used to obtain digital biomarkers in patients with DLB, 2) to assess the digital biomarkers’ ability to evaluate non-cognitive symptoms, and 3) to assess the feasibility of applying digital biomarkers in patients with DLB.
Methods:
We systematically searched databases MEDLINE, Embase, and Web of Science from inception through February 28, 2023. Studies assessing digital biomarkers obtained by portable or wearable DHTs and related to non-cognitive symptoms were eligible if including patients with DLB. The quality of studies was assessed using a modified check list based on the NIH Quality assessment tool for Observational Cohort and Cross-sectional Studies. A narrative synthesis of data was carried out.
Results:
We screened 4,295 records and included 20 studies. Seventeen different DHTs were identified for assessment of most non-cognitive symptoms related to DLB. No thorough validation of digital biomarkers for measurement of non-cognitive symptoms in DLB was reported. Studies did not report on aspects of feasibility in a systematic way.
Conclusions:
Knowledge about feasibility and validity of individual digital biomarkers remains extremely limited. Study heterogeneity is a barrier for establishing a broad evidence base for application of digital biomarkers in DLB. Researchers should conform to recommended standards for systematic evaluation of digital biomarkers.
Keywords
INTRODUCTION
Dementia with Lewy bodies (DLB) is second only to Alzheimer’s disease (AD) in terms of the prevalence of neurodegenerative causes of dementia [1], with the first diagnostic criteria being proposed in 1996 [2, 3]. Aside from cognitive impairment, patients with DLB often suffer from various non-cognitive symptoms such as visual hallucinations, REM sleep behavior disorder (RBD), and parkinsonism [4] either preceding the diagnosis of dementia or emerging at later stages of the disease course [5, 6]. While cognitive symptoms are defining for dementia, non-cognitive symptoms are of equal importance for the specific diagnosis of DLB [4], and they significantly impact patients’ quality of life [7, 8]. Moreover, some non-cognitive symptoms may be manageable such as parkinsonism and autonomic dysfunction (see Table 1). However, it is difficult to evaluate the presence and severity of non-cognitive symptoms in patients with DLB as cognitive deficits may compromise reliable symptom description. Also, high interobserver variability for assessment scales of non-cognitive symptoms such as motor examination using the UPDRS (Unified Parkinson’s Disease Rating Scale) part III has been reported [9]. Thus, there is a need for objective and precise tools to improve the ability to detect non-cognitive symptoms and to assess their severity. At the same time, growing demands on the efficiency of health care systems require implementation of low-cost, data-driven tools in the clinical assessment and decision-making to ensure maximal resource and capacity utilization and quality of future patient care [10].
Non-cognitive symptoms in Dementia with Lewy bodies
Digital biomarkers, which are biomarkers collected from digital health technologies (DHTs, i.e., “A system that uses computing platforms, connectivity, software, and sensors for healthcare and related uses”) [11], show potential in closing these gaps. Indeed, the research field of digital biomarkers is rapidly expanding with an increasing number of available DHTs measuring biological and physiological data [12]. This may be done in an unobtrusive manner such as with smartwatches, which can monitor numerous parameters including limb movement, heart rate, and whereabouts with automatic upload and analysis of data. Portable, or wearable DHTs could become valuable tools due to their convenience, however, evidence to support their use in the assessment of patients with DLB is uncertain. Indeed, application of DHTs in a population with dementia may pose a challenge, thus evaluation of feasibility is of special importance, when considering new digital biomarkers for future clinical practice.
Several use-cases for DHTs in the management of patients with DLB could be envisioned, e.g., as diagnostic and prognostic biomarkers, the latter missing almost entirely from the current biomarker landscape. Moreover, accurate measures of symptoms to track treatment effects and even as trial outcomes are also highly relevant applications.
This review seeks to identify and synthesize current knowledge on digital biomarkers applied for the assessment of non-cognitive symptoms in patients with DLB. Specifically, the objectives were: 1) to identify wearable or portable DHTs used to obtain digital biomarkers in patients with DLB, 2) to assess the ability of these biomarkers to monitor and evaluate non-cognitive symptoms in DLB, and 3) to assess the feasibility of applying digital biomarkers in patients with DLB. To our knowledge, no systematic review exists on this topic.
METHODS
A study protocol for this review was uploaded to the PROSPERO database (ID: CRD42023400196). Reporting on this systematic review was conducted according to the PRISMA 2020 Statement [13].
Eligibility criteria
Original observational or interventional studies published in peer-reviewed international journals were considered eligible if they met all inclusion criteria and none of the exclusion criteria:
Inclusion criteria
Reporting on adult humans (age > 17 years) meeting diagnostic criteria for DLB (McKeith, 3rd and 4th edition [4, 14]). Reporting on digital biomarkers (according to reported definition [11]), which assessed non-cognitive symptoms in DLB and were obtained by wearable or portable DHTs (as evaluated by the reviewers). Full text available in English language.
Exclusion criteria
Studies including < 5 patients with DLB. Studies only reporting on patients with dementia of mixed types (e.g., DLB and vascular dementia) or Parkinson’s disease dementia (PDD). Studies reporting on digital biomarkers primarily reflecting cognitive features of symptoms (as evaluated by the reviewers), e.g., number of spoken words per minute during cognitive tests.
Information sources and search strategy
We performed searches in MEDLINE and Embase using the Ovid-platform, and in Web of Science from inception to February 28, 2023. References cited in the included studies were also examined for eligibility. Search strategies and results are available in the Supplementary Material.
Selection process
Screening for eligibility was executed by two independent reviewers (NS, MG) using the web based systematic review tool Covidence®. A senior researcher (KF) was consulted in cases where consensus could not be reached between NS and MG. Papers with duplicate data but additional relevant data (e.g., data from different comparators) were included and designated accordingly. Otherwise, the paper based on the most complete dataset was included in the review.
Data collection process
Data was independently extracted by two reviewers (NS, MG) using a modified data extraction sheet in Microsoft Excel®. Any discrepancies upon comparison were resolved by the two reviewers. KF was consulted in cases of no consensus between NS and MG.
Data items
The following data items were extracted from the included reports: a) study design and purpose, b) eligibility criteria for included participants, c) number of participants, d) age and sex of participants, e) diagnosis of participants, f) cognitive scores of participants (e.g., Mini-Mental State Examination (MMSE) scores, or The Montreal Cognitive Assessment (MoCA) scores), g) symptoms assessed, h) DHT hardware and methods including period of measurement for obtainment of digital biomarker, i) DHT software, j) digital biomarker metrics, k) statistical analysis methods, l) reported values of the digital biomarkers measured, m) comparator, n) comparisons with validated symptom assessment (e.g., validated clinical rating scales or ancillary investigations), o) data loss/quality, p) safety assessments (e.g., adverse events), q) methods of assessment of feasibility (e.g., questionnaires, or drop-out), r) funding, s) reported conflicts of interests for study authors.
When data collected from patients with DLB were not reported for these participants specifically, biomarker values and comparisons with validated symptom assessment were not extracted, however, remaining data items were still extracted to identify relevant DHTs and to assess the feasibility of applying digital biomarkers in DLB. Biomarker values for comparators were extracted as a further measure of the ability of the biomarker to measure non-cognitive symptoms in patients with DLB.
Assessments of study quality
Two reviewers (NS, MG) carried out independent, qualitative assessments of the methodology of included studies using a modified check list (see Supplementary Material) adopting seven criteria from the National Institute of Health (NIH) Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies [15]. Not all criteria were adopted as they were to be applied on studies of heterogenous design, and we did not investigate any exposures. Any discrepancies upon comparison were resolved by the two reviewers, and KF was consulted in cases of disagreement. Studies without extracted biomarker values for patients with DLB were not assessed.
Data synthesis
A narrative data synthesis was carried out. No assessment of meta-bias or strength of the body of evidence was made in this review.
RESULTS
The study selection process is illustrated in the PRISMA flow-chart in Fig. 1. We identified 4295 unique citations through database searches. After screening of title and abstract, 67 papers were retrieved for full text assessment of eligibility, resulting in 20 included studies. Of note, one study [16] did meet inclusion criteria but was excluded due to the results already being included in the study by Kazui et al. [17].
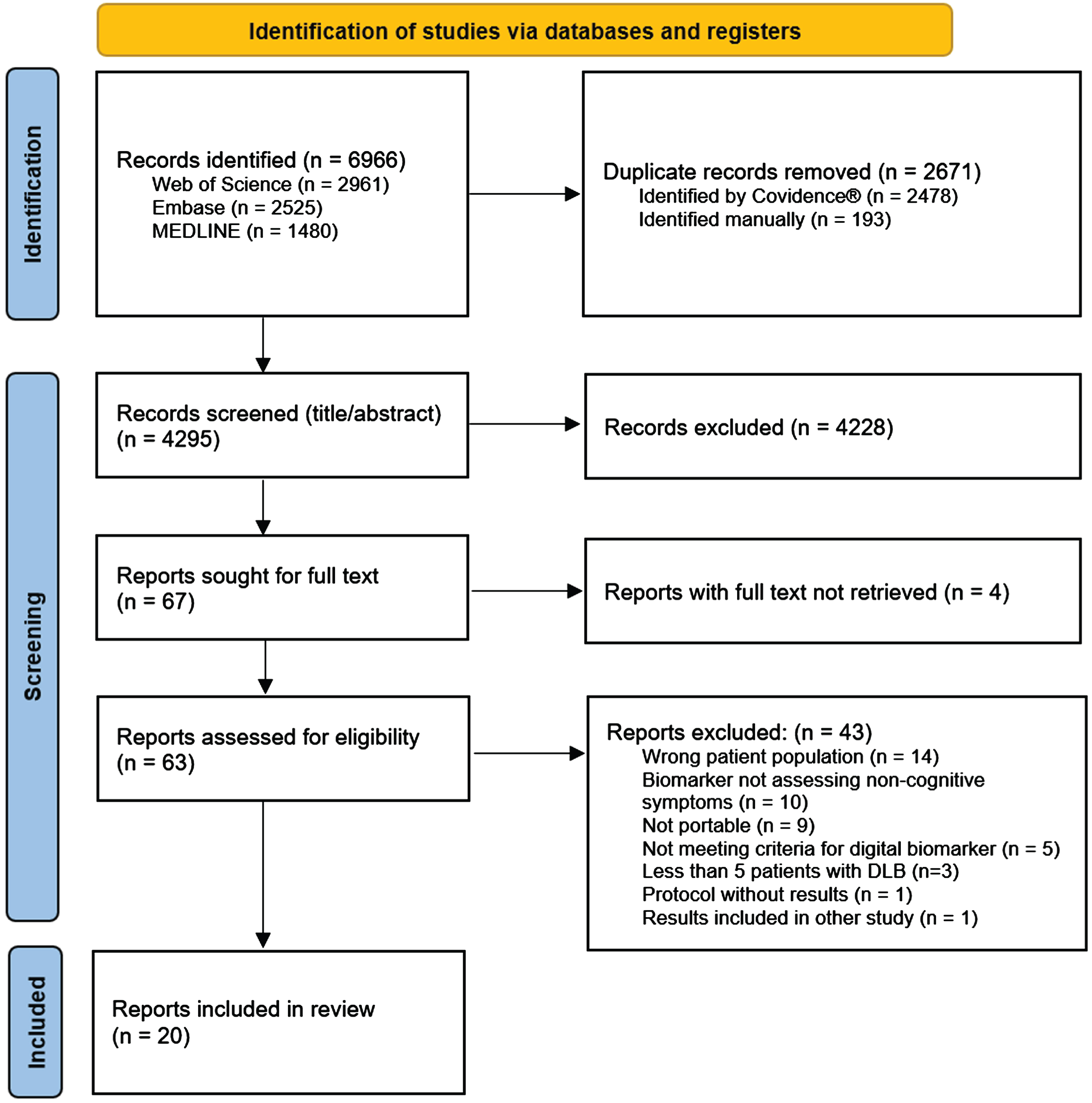
PRISMA flow diagram of the screening process.
Study presentation
Table 2 gives the characteristics of the included studies. The DHTs applied in the included studies assessed the following groups of symptoms: fine motor skills (n = 4), sleep (n = 4), gait and balance (n = 3), dysautonomia (n = 3), neuropsychiatric symptoms (n = 2), physical activity and rest (n = 2), speech (n = 2), color vision impairment (n = 1), and hypomimia (n = 1). Four studies investigated interventions (pharmacological [17, 18], or non-pharmacological [19, 20]) where DHTs were used to assess outcomes, whereas the remaining studies had observational, cross-sectional designs. Four studies [18, 22] did not provide any subgroup analysis of DLB patients but were still included to identify relevant DHTs and assess the feasibility of applying digital biomarkers in DLB. Most studies were carried out in an outpatient setting, whereas one study investigated the application of DHTs in a hospital setting [20], and one study investigated patients in a long-term care facility [19].
Characteristics of included studies
DLB, dementia with Lewy bodies; MCI, mild cognitive impairment; AD, Alzheimer’s disease; FTD, frontotemporal dementia; PDD, Parkinson’s disease dementia; BPSD, behavioral and psychological symptoms of dementia; VaD, vascular dementia; SMI, subjective memory impairment; PD, Parkinson’s disease; CVD, cardiovascular disease; Pro-DLB, MCI with REM sleep behavior disorder or parkinsonism but not meeting criteria for PD. *Record appears more than once in table. Several studies provided results on more than one digital biomarker and may occur under different symptom grouping accordingly. #Reporting on participants who are also included in other records by same lead author.
The number of patients with DLB in the included studies ranged from five to 62 with a median of 23. Additionally, one study [18] reported on measurements of 238 patients with Lewy body dementias (DLB or PDD). For participants with DLB, the mean and median age ranged from 66 to 80 years and mean and median MMSE score ranged from 16 to 26.5 reflecting a broad range of disease severity. In five studies, participants with DLB had a mean MMSE score below 22 [17, 23–26], whereas in 10 studies a mean score of 22 or greater was reported [27–36]. Comparisons with healthy controls was reported in 10 studies [17, 33–36], and/or with patients with AD in 12 studies [21, 31–36].
Digital health technologies
The various DHTs applied in the included studies are illustrated in Fig. 2, and their application is described in Table 2. Fifteen studies [17, 30–36] reported sufficient details on DHT hardware for identification of the specific DHT, and 15 studies [17–19, 32–36] reported on applied software, i.e., described algorithms or named computer programs used for data processing. A total of 17 different DHTs were applied in the included studies, but only one DHT (AX3 Axivity) was reported across multiple studies [18, 32–35]. An inertial measurement unit (IMU) was the most widely used type of DHT in the included studies with eight papers reporting biomarkers derived from IMUs [17–19, 32–35]. An IMU, such as an actigraph, measures movement using accelerometry, and was applied in studies, which measured characteristics of balance, gait, agitation, and sleep utilizing varying hardware, software, and assessment paradigms. Indeed, most types of DHTs were reported to give rise to several digital biomarkers and have the potential to assess multiple symptoms.
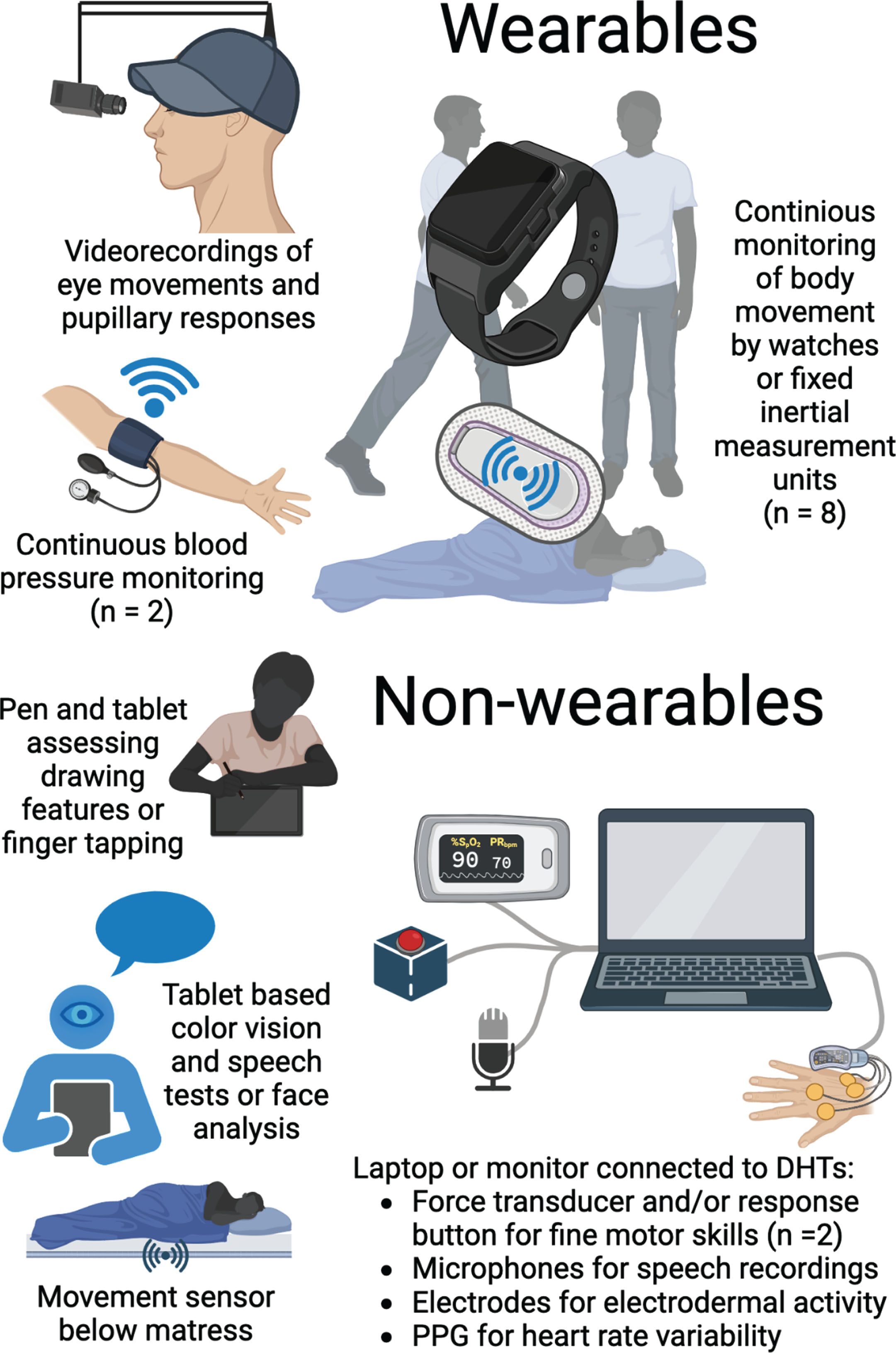
Graphical representation of applied DHTs. A graphical representation of the DHTs applied for symptom assessment in the included studies. An illustration might represent multiple DHTs if various assessments are described in the adjacent text. Some types of DHTs were applied across multiple studies as illustrated by the numbers in parentheses. DHT, digital health technology; PPG, photoplethysmograph. Created with BioRender.com.
Digital biomarkers and corresponding symptoms
A full list of the investigated digital biomarkers derived from DHTs is given in Table 2. We divided symptoms assessed into nine different types as illustrated by the following examples: Biomarkers such as those representing characteristics of drawing (e.g., pressure applied by a digital pen) and other small movements (e.g., frequency of finger or alternating hand tapping) were grouped under fine motor skills. Sleep was assessed by means of e.g., sleep duration and efficiency. Biomarkers related to autonomic function (e.g., change of mean blood pressure from day to night) were grouped under dysautonomia, whereas biomarkers grouped under neuropsychiatric symptoms group include those reported by the authors as measures of e.g., agitation or pareidolias. Biomarkers reported as characteristics of gait (e.g., asymmetry of step time, or step length) or balance (e.g., acceleration in mediolateral direction) were similar in nature and grouped together accordingly. Biomarkers representing movement on a more general level (e.g., average acceleration per day, steps per day, and bout length variability) were grouped under physical activity and rest. Speech was assessed through, e.g., fundamental frequency range, and phoneme rate, while biomarkers grouped under color vision impairment was reported as such by the authors. Hypomimia was assessed using the likelihoods of different facial emotions expressed including neutral.
Discriminatory power of digital biomarkers
Seven studies [17, 33–36] comparing DLB to controls found group differences on at least one biomarker (Table 3). Assessments of fine motor skills, sleep, and gait or balance resulted in the highest number of different digital biomarker values between these two groups. In seven studies comparing DLB with AD [24, 36], group differences in biomarker values were reported. These were mainly from assessments of fine motor skills, gait, dysautonomia, and color vision impairment (Table 3). In studies comparing DLB with both controls and AD, more biomarkers differed between DLB and controls than between DLB and AD, and DLB differed more significantly from controls than AD [27, 34–36] on the same biomarkers. Of note, four digital biomarkers differed between DLB and PDD (in assessment of gait, balance, and physical activity/rest) [32, 35], one between DLB and Parkinson’s disease (PD, dysautonomia) [25], and one between DLB and frontotemporal dementia (fine motor skills) [28].
Main findings from included studies
Main results include only significant results if not otherwise stated. Due to space limitations, insignificant results are not always presented, but all investigated biomarkers are mentioned in Table 2. For comparisons of digital biomarkers with validated symptom assessments, only results specific to the DLB-group were reported. DLB, dementia with Lewy bodies; AD, Alzheimer’s disease; FTD, frontotemporal dementia; PDD, Parkinson’s disease dementia; UPDRS, Unified Parkinson’s disease rating scale; UHDRS, Unified Huntington’s disease rating scale; PD, Parkinson’s disease. *Record appears more than once in table. Several studies provided results on more than one digital biomarker and may occur under different symptom grouping accordingly. #Reporting on participants who are also included in other records by same lead author.
Comparisons with validated symptom assessment methods
Eight studies [17, 35] compared results from digital biomarkers in patients with DLB to validated symptom assessment methods (Table 3). One study [33] found that digital biomarkers assessing physical activity showed moderately strong, positive correlations with self-reported ability to maintain balance as measured with the Activities-specific Balance Confidence (ABC) scale. In the same study, weak correlations were found between digital biomarkers assessing physical activity and motor symptoms according to clinical evaluation using the UPDRS-III. One digital biomarker assessing sleep was significantly correlated to delusional behavior as assessed with the Neuropsychiatric Inventory (NPI) [17]. In contrast, Kazui et al., who reported these findings, also reported scores from assessments of sleep disturbances using the NPI to be reduced after taking donepezil, but changes of the digital biomarkers which was applied in the trial were not significantly associated to these changes. Of interest, Kragh et al. reported that the variability of certain digital biomarkers assessing fine motor skills (e.g., frequency of finger tapping, and frequency of alternating pronation/supination of hand) were lower than the variability of results from corresponding clinical assessments [28]. Consequently, the authors suggest that digital biomarkers could be more sensitive to small changes that would go undetected by regular clinical examination.
Aspects of feasibility
Data on feasibility of DHTs in the studied patient population was sparse and not collected systematically. Most studies reported pooled results on the whole study population, whereby specific data on aspects of feasibility in patients with DLB could not be extracted (Table 3). In general, data was obtained from assessments of most of the included participants in almost all studies.
A single study, by Negami et al., explicitly reported on adverse events, and participants in this study did not experience adverse events in relation to measurement of symptoms of dysautonomia using automated blood pressure monitoring [24]. We found no studies purposely reporting on participant experience by e.g., questionnaires. However, five studies made mention of user related issues regarding usability of DHTs applied for continuous monitoring (i.e., discomfort [33], unwillingness to wear the DHT [34], difficulties wearing the DHT [19], or participants removing the DHT [17, 20]) but did not provide additional details. Aside from these user related issues, difficulties in the obtainment of useful data were mainly reported as related to technical problems (e.g., device malfunctioning [20], data upload issues [33, 34]), or poor data quality. Specifically, two studies raised concerns about the reliability of DHTs concerning data quality [19, 33], while the remaining did not report on this matter at all. One study [33] quantified the amount of data evaluated as being of inadequate quality, but details from data quality assessments were not reported in any studies. Regarding the two randomized controlled trials using DHTs to assess the effects of interventions, no drop-out were reported to be related to the DHT in one study [19], and reasons for drop-out or non-compliance were not reported on in the other study [18].
Two studies considered feasibility of the applied DHT to be limited based on the reported amount of data loss due to user related issues or low-quality data. Moyle et al. [19] reported almost half of participants not wearing the device for at least 10 of the 12 planned hours, which the authors required for inclusion in the analysis, and Fukuda et al. [20] report unreliable data from the DHT in addition to the DHT not being able to monitor participants due to their behavior. Of note, Wang et al. [18] reported a drop from 80% to 60% of participants adhering to daily finger tapping test completions during the 16-week study period but the authors did not consider this indicative of limited feasibility of applying the digital biomarker for assessment of the effect of the pharmacological intervention.
Results from assessments of study quality
The results of the study methodology assessments are presented in Fig. 3. Four studies were not assessed since no biomarker values for patients with DLB were extracted [18, 22]. In general, studies performed well on providing a clearly stated research question/objective, applying uniform eligibility criteria for all participants, and implementing clearly defined outcomes. In contrast, most studies failed to provide sample size justifications, blind outcome assessors from participant diagnosis, and account for key potential confounders in the statistical analyses. Half of the studies clearly specified the study population.
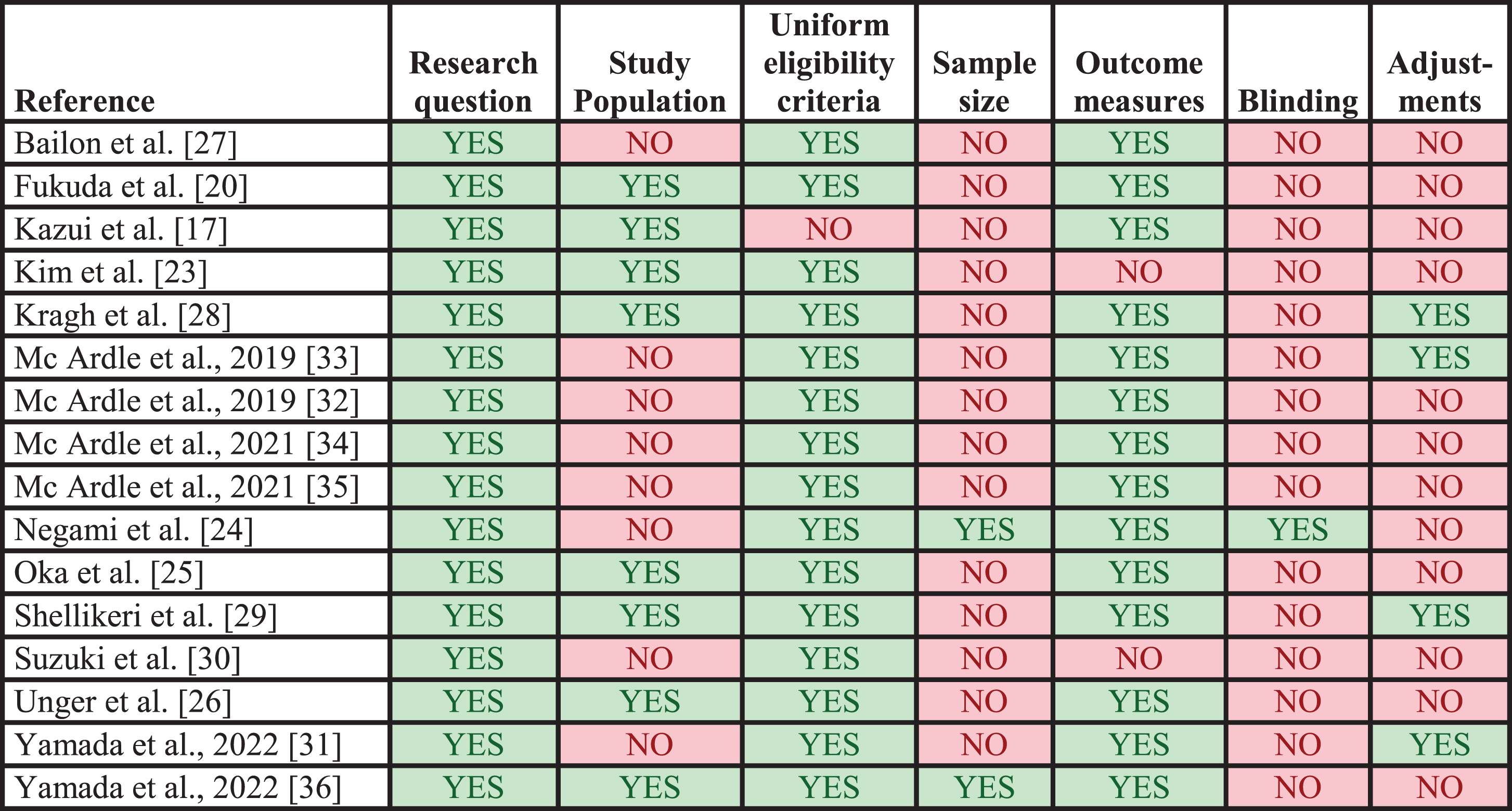
Quality assessments of included studies. Results from application of the modified check list for assessment of study methodology. Each column represents a criterion from the check list. Studies marked with YES did meet the criterion, whereas studies marked with NO did not. The applied check list is available in the Supplementary Material.
DISCUSSION
We conducted a systematic review on digital biomarkers for the assessment of non-cognitive symptoms in patients with DLB. A wide variety of different DHTs were used across studies mostly assessing motor symptoms. Digital biomarkers assessing physical activity and fine motor skills showed reasonable comparability when compared with other methods for assessment of similar symptoms in patients with DLB, but such comparisons were seldom reported on. Issues related to feasibility were due to technical problems or participants not being compliant to continuous monitoring. However, feasibility was not reported on in a systematic way, and not in patients with DLB, specifically.
So far, data on digital biomarkers related to non-cognitive symptoms in DLB is limited. Reviews show that research in this area extends further in AD [37, 38] and especially in PD [39–43], which however share some symptoms with DLB [44, 45]. In AD and PD, comparable (and sometimes identical) DHTs have been used for assessments of similar symptoms as those in the included studies, e.g., IMUs for assessment of gait [46, 47], sleep [48, 49] and activity [50, 51], and dysautonomia assessed using photoplethysmography [52] or blood pressure monitors [53]. Our findings align with those in AD and PD. Specifically, a vast assortment of DHTs and paradigms have been used, which is a barrier to establish a broad evidence base for specific DHTs that show promise and in terms of comparing DHTs which has been done in relatively few studies [54–57]. We identified no studies directly comparing portable or wearable DHTs in DLB, but one study [32] showed that a wearable IMU used for assessment of gait was superior to an instrumented walkway in terms of providing significant biomarker differences between DLB and other diagnostic groups. Although two studies [17, 20] reported on identical outcomes (i.e., sleep duration and sleep efficiency) in DLB, populations and settings of the studies differed noticeably (i.e., admission in a dementia care unit versus home-based monitoring) thus limiting the value of comparing the DHTs applied. Future research in digital biomarkers in DLB might benefit from the efforts that have been made to compare different DHTs by using a numbered scale reflecting their level of readiness for clinical use in PD [58], although we did not examine thisaspect.
Most common types of symptoms in DLB were investigated, however considering core clinical features of DLB, we did not identify DHTs assessing fluctuations or REM sleep behavior disorder although application of IMUs in measurements of patients with PD have shown high specificity for RBD [59]. Moreover, certain symptoms considered as supportive clinical features of DLB such as constipation or hyposmia [4] were not assessed by any of the included studies. Neither have such symptoms been assessed in patients with PD or AD [37, 40]. Existing wearable DHTs and digital biomarkers might however be used such as a smart belt for assessment of gastrointestinal symptoms [39], and a vocal biomarker for detection of hyposmia [60].
In general, most studies did not report comparisons of digital biomarkers with other methods of symptom assessments in patients with DLB specifically, or they reported comparisons that were not evidently suitable for evaluation of the digital biomarkers’ ability to measure symptoms (e.g., correlations between parkinsonian symptoms and decreased fall in nighttime blood pressure [23], and NPI delusions score and features of sleep [17]). In this regard, evidence to proper evaluate digital biomarkers’ ability to capture non-cognitive symptoms in DLB is lacking. This illustrates a need for a more standardized approach in the validation of digital biomarkers for symptom assessment in DLB. Indeed, the lack of standardization has been raised as a general concern regarding development of novel digital biomarkers, and a checklist to ensure proper validation have been provided for the design of future clinical trials [61]. Ideally, new biomarkers should be validated against current gold standards for symptom assessment.
As all but two studies were cross-sectional, limited evidence is available on the longitudinal application of the investigated digital biomarkers in DLB (i.e., sensitivity to symptom progression) advising the need for prospective study designs in future assessment of the most promising DHT’s. It has been suggested that test-retest repeatability and sensitivity to symptom progression is a valuable and important approach for validating biomarkers [40], and future studies could incorporate these aspects to elucidate this further.
Uncertainty remains as to whether most of the applied DHTs can be considered feasible for use in patients with DLB as data are not reported for key aspects such as adverse events, user experiences, and data quality assessments. Feasibility of applying digital biomarkers in PD [42, 43] or cognitive impairment [62] have been reviewed with similar findings. Of interest, our findings on amounts of data lost due to human factors using wearables matched those of similar studies in patients with PD and were lower than in studies on stroke-patients [43]. This suggests that DHTs could be applied in assessments of patients with DLB on the same terms as in the mentioned patients without dementia, which might make portable and wearable DHTs preferable to demanding methods for obtainment of biomarkers in patient with dementia.
The relevance of specific information on feasibility remains as minor issues such as discomfort or subjective preferences in relation to device usage is of importance to patients, and such information might help in ensuring data collection. Indeed, it has been possible to identify barriers for using DHTs by people with dementia and addressing these barriers have resulted in higher patient satisfaction and a reduction in data lost due to human factors as reported in [43, 63]. One approach to ensure systematic data collection of aspects of feasibility is to include patient reported outcome measures (PROMs) specifically on the use of DHTs for symptom assessment. Such PROMs have been assessed in studies including patients with PD and AD using questionnaires and interviews [51, 64], and similar assessments could be applied in future studies including patients with DLB.
None of the identified studies specifically aimed to investigate DHTs in early versus late disease stages. Between studies, there were differences on mean cognitive performance but there were no tendencies to be detected in terms of which DHTs were applied nor for measures of diagnostic accuracy. However, there were indications of reduced feasibility in studies reporting on populations with more severe behavioral disturbances. It is likely that different DHTs are relevant in early compared to late disease stages, but more studies are needed to further elucidate this.
We recognize that the present review is not without limitations. For instance, there is a possibility of publication bias, since studies on DHTs resulting in adverse events or without significant, positive outcomes might not have been submitted, an aspect we did not investigate by means of meta-analysis, which was not pre-planned. Another limitation is that we included studies not reporting results for patients with DLB explicitly, which might have skewed our findings. Finally, studies not published in English language were not considered due to lack of resources, which might limit retrieval of relevant studies. Despite these shortcomings, this review offers a useful summary of the field and provides important recommendations for future research to attend to. Strengths of this review included a well-planned, rigorous approach and our broad search strategy encompassing several databases, ensuring adequate capture of relevant literature.
Research on digital biomarkers from wearable or portable DHTs in patients with DLB is still in its very infancy in the assessment of the non-cognitive symptoms. Most non-cognitive symptoms have been assessed, but the field is very diverse, without standardized protocols, and thus no broad evidence base exists for individual, digital biomarkers. Currently, no fully validated methods for assessments of non-cognitive symptoms in DLB by wearable or portable DHTs exist and means of feasibility were not formally assessed in the included studies. Thus, there is a need for more knowledge about feasibility and validity of symptom assessment before conclusions can be made concerning the use of digital biomarkers in DLB. Future studies should focus on closing these gaps before digital biomarkers may enter routine clinical practice, ideally by systematically applying outcomes to evaluate feasibility and validity. Our findings support proposed recommendations for future research in wearable DHTs emphasizing the importance of standardization of evaluation of digital biomarkers [42].
AUTHOR CONTRIBUTIONS
Nikolai Sulkjær Sjælland (Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Project administration; Visualization; Writing – original draft); Mathias Holsey Gramkow (Conceptualization; Investigation; Methodology; Writing – review & editing); Steen Gregers Hasselbalch (Conceptualization; Methodology; Writing – review & editing); Kristian Steen Frederiksen (Conceptualization; Methodology; Supervision; Writing – review & editing).
Footnotes
ACKNOWLEDGMENTS
We thank information specialist Trine Kæstel at Rigshospitalet Medical Library for her help with developing search strings.
FUNDING
The study was supported by a grant from the Lundbeck Foundation.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
