Abstract
Background:
Alzheimer’s disease (AD) is the most common neurodegenerative disease. Unfortunately, efficient and affordable treatments are still lacking for this neurodegenerative disorder, it is therefore urgent to identify new pharmacological targets. Astrocytes are playing a crucial role in the tuning of synaptic transmission and several studies have pointed out severe astrocyte reactivity in AD. Reactive astrocytes show altered physiology and function, suggesting they could have a role in the early pathophysiology of AD.
Objective:
We aimed to characterize early synaptic impairments in the AppNL-F knock-in mouse model of AD, especially to understand the contribution of astrocytes to early brain dysfunctions.
Methods:
The AppNL-F mouse model carries two disease-causing mutations inserted in the amyloid precursor protein gene. This strain does not start to develop amyloid-β plaques until 9 months of age. Thanks to electrophysiology, we investigated synaptic function, at both neuronal and astrocytic levels, in 6-month-old animals and correlate the synaptic activity with emotional behavior.
Results:
Electrophysiological recordings in the hippocampus revealed an overall synaptic mistuning at a pre-plaque stage of the pathology, associated to an intact social memory but a stronger depressive-like behavior. Astrocytes displayed a reactive-like morphology and a higher tonic GABA current compared to control mice. Interestingly, we here show that the synaptic impairments in hippocampal slices are partially corrected by a pre-treatment with the monoamine oxidase B blocker deprenyl or the fast-acting antidepressant ketamine (5 mg/kg).
Conclusions:
We propose that reactive astrocytes can induce synaptic mistuning early in AD, before plaques deposition, and that these changes are associated with emotional symptoms.
INTRODUCTION
Alzheimer’s disease (AD) is a progressive age-related neurodegenerative disorder accounting for 75% of dementia cases worldwide [1]. Patients show significant memory loss [2] together with multiple secondary symptoms such as social isolation [3] and depressive symptoms [4]. In recent years, significant improvements have been done in clinical care of patients and recent anti-amyloid-β (Aβ) immunotherapies have shown promising results in early phases of the pathology [5]. However, the underlaying mechanisms of this pathology are still unknown. Intracellular tau neurofibrillary tangles [6] and Aβ aggregation [7] are the main molecular hallmarks of the pathology and have been given considerable attention in the attempt to understand the disorder. Extensive cellular rearrangements occur around Aβ plaques, including synaptic degeneration and astrocyte reactivity. In addition, Aβ can take other aggregated forms, including oligomers that are known to deteriorate neuronal activity [8] and affect astrocytic secretome [9], and has been suggested to have an even stronger adverse effect than Aβ plaques on neuronal and astrocytic function. Thus, Aβ oligomers could induce neurobiological changes before plaque deposition.
Early symptoms of AD reach beyond memory loss, including sleep disturbance, psychosis, and social isolation [10]. Depressive symptoms are also common in the early stages and depression is a strong risk factor for AD. Whether depression is causative, or a prodromal indication is still a question of discussion [11]. Interestingly, antidepressant treatment such as ketamine or monoamine oxidase inhibitors has been shown to reduce cognitive deficits as well as pathology in AD [12, 13].
Recent imaging work suggests reduced synaptic density already at early stages of AD [14, 15] whereas clinical data show increased functional connectivity and synchronization of neuronal activity early in the disease [16]. These results appear contradictory, and we still need to better understand the functional changes of synaptic transmission in early AD. In this context, a deficit in neuronal activity set-point such as an imbalance between firing and neuronal plasticity, has been proposed to underlay early development of the pathology [17]. Interestingly, preclinical studies further emphasize the development of early synaptic deficits in AD, with demonstration of synaptic impairments at the pre-plaque stage in an App knock-in mouse model [18, 19] and impaired synaptic plasticity in a hypercholesterolemia mouse model of AD [20].
Reactive astrocytes around plaques are very well described [21] and can be a consequence of Aβ accumulation [22]. More recently, astrocytic reactivity has been demonstrated at an early stage in the progression of AD [23, 24] and increased level of the glial fibrillary acidic protein (GFAP) has been detected in the cerebrospinal fluid of patients early in the disease [25]. Together with the fact that healthy astrocytes are important for Aβ clearance, thus reactive astrocytes could facilitate Aβ accumulation [26], these findings have led to the proposal of an astrocytic origin of the disease [27]. Early astrocyte reactivity in AD is compatible with synaptic changes due to the close interaction between astrocytic and synaptic function. Reactive astrocytes affect synapses through several mechanisms [28]. Among other things, reactive astrocytes have been shown to express monoamine oxidase type B (MAO-B) [29] resulting in de novo synthesis of GABA and an increased tonic inhibition of adjacent neurons [30].
Many animal models have been engineered in order to understand the mechanisms underlying AD, yet none of them recapitulate all the symptoms of the pathology [31]. One common feature in older models is an overproduction of the amyloid precursor protein, resulting in an important Aβ accumulation at a young age. The AppNL-F mouse line is a late onset knock-in model of AD that is useful to investigate early stages of the pathology. This model carries two disease causing mutations (the Swedish mutation, NL and the Beyreuther/Iberian mutation, F) in the endogenous App gene [32] and hence free from App overexpression. The AppNL-F mouse does not develop pathological plaques until 9 months of age [33]. While no tau pathology has been unveiled in this model [34], except increased phosphorylation of some sites [35], many molecular changes such as autophagy disturbances [36] and mitochondria dysfunctions [37] were reported both in vivo and in vitro [38]. Interestingly, early changes in the synapse structure and function has been identified in the cortex of 6-month-old AppNL-F animals [19, 39].
In the present study we used behavioral, electrophysiological and molecular readouts to understand early changes in this AppNL-F knock-in mouse model of AD. Our analysis revealed reduced synaptic activity and impaired synaptic plasticity associated to reactive-like astrocytes. These neuronal and astrocytic dysfunctions correlate with increased depressive-like behavior and could be partially corrected by treatment with either the MAO-B inhibitor deprenyl or the fast-acting antidepressant ketamine.
MATERIAL AND METHODS
Animals
AppNL-F mice, model of AD, were bred locally at Uppsala University. These mice carry the Swedish (KM670/671NL) and the Beyreuther/Iberian (I716F) mutations in the App gene. Age-matched C57Bl6 mice were bought from Charles River (Germany). Both strains were housed in the same conditions, on a 12 : 12 light-dark cycle and with access to food and water ad libitum. All experiments were conducted in 6-month-old males, where increased insoluble Aβ was detected but where Aβ-plaques were absent (Supplementary Figure 1).
The study was conduct in accordance with the Declaration of Helsinki and was approved by the Swedish board of animal use (Jordbruksverket permit #5.2.18–03389/2020) and accepted by the ethics committee at Uppsala University (application DOUU 2020-022).
Drugs
Tetrodotoxin (TTX; Tocris 4368-28-9) was diluted in artificial cerebrospinal fluid (aCSF) and used in patch clamp experiments to measure mini excitatory post-synaptic currents (mEPSC), at a concentration of 1 μM.
NBQX (Tocris 1044), DL-AP5 (Tocris 3693), and Picrotoxin (PTX; Tocris 1128) were diluted in aCSF and used in patch clamp experiments to measure tonic GABA current, at a concentration of 10 μM, 50 μM, and 100 μM respectively.
(R)-(-)-Deprenyl hydrochloride (deprenyl; Tocris 1095) was diluted in aCSF and was used in the patch clamp and the field recording experiments, at a concentration of 100 μM.
Ketamine (Ketaminol® vet., 100 mg/kg, Intervet) was diluted in vehicle solution (natriumchloride, NaCl 0.9%) and delivered intraperitoneally (i.p.; 5 mg/kg) 24 h prior to experiments.
Behavioral analysis
Animals were habituated to the experimental room 20 min prior to experiments. The luminosity of the room was set on 30 lux and the temperature at 21°C±1°C. The tests were performed in the following order, with at least three days between each test.
Open field test
The animals were free to explore a spatial-cue-free square arena (50 cm×50 cm) for 10 min. The central zone and peripheral zone were defined in the Ethovision software (Noldus tech, The Netherlands). The area of the central zone was defined as 50% of the area of the arena. The total travelled distance, the time spent in the central area, as well as the number of entries in the central zone, were automatically analyzed by the software.
Five-trials social memory test
The mice were habituated to an empty square arena (50 cm×50 cm) for 10 min, followed by 5 min exploration of the same arena with an empty removable cage (8 cm×8 cm×9 cm). An unknown mouse (same genotype, same fur color, same age, and same gender as the test mouse) was placed for 5 min in the removable cage for four consecutive trials, separated by 10 min. One hour after the fourth trial, a new unknown mouse was placed in the cage for 5 min. Social interactions were manually scored.
Olfactory habituation/dishabituation test
The mice were isolated in individual standard cages in which they performed the whole experiment and habituated for at least 2 min. Cotton swabs soaked with non-social odors (melon and banana) or social odors (one from their homepage and one from a non-familiar cage) were presented to the animal for 2 min. Each odor was presented three times.
Elevated plus maze
The maze was composed of two open arms, two closed arms, linked together at a central platform and positioned 50 cm above ground. Mice were placed on the central platform facing one open arm and were let to explore for 5 min. The number of entries and the time spent in the open arms were manually scored. An animal was considered “in the arm” when the four paws crossed the virtual line between the central platform and the considered arm.
Forced swim test
The mice were placed in a cylinder (30 cm high, φ 18 cm) filled with 25±1°C water to a height of 17 cm where no part of the animal could touch the bottom. The mice were left in the cylinder for 6 min, and the time spend immobile was quantified during the last 4 min. Latency to first immobility and the total time of immobility were manually scored. An animal was considered as immobile when it was floating and none of the paws nor the tail was moving.
Emotionality z-score
Z-normalization was used in this study as a complementary measurement for emotionality-related behavior, obtain from different paradigms [40]. Raw behavioral data (parameters of interest) were normalized to the control group using the following equation z = (X-μ)/σ with X being the individual value for the considered parameter, μ being the mean of the control group and σ being the standard deviation of the control group. Each parameter of interest gave a parameter z-score (zSC_param), adjusted so that an increased score reflects an increased emotionality. For each test, all parameter scores were averaged into a test score (zSC_test), and eventually averaged to give an individual emotionality z-score.
More details about the exact list of parameters of interest and the mathematical method are available in Supplementary Tables 1 and 2.
Electrophysiology
Brain slicing
For the preparation of acute slices, mice were anesthetized with isofluorane and decapitated soon after the disappearance of corneal reflexes. 300 μm thick horizontal sections were prepared using a Leica VT1200 vibrating microtome (Leica Microsystems, Nussloch, Germany) in dissection solution containing 250 mM sucrose, 2.5 μM KCl, 1.4 mM NaH2PO4, 26 mM NaHCO3, 10 mM glucose, 1 mM CaCl2 and 4 mM MgCl2, and bubbled with carbogen gas (5% CO2, 95% O2). Hippocampi were dissected and slices were placed in a recovery chamber filled with aCSF containing (in mM): 130 NaCl, 3.5 KCl, 1.25 NaH2PO4, 24 NaHCO3, 10 glucose, 2 CaCl2 and 1.3 MgCl2 and bubbled with carbogen gas.
Patch clamp recording
After a recovery period of at least 2 h, slices were transferred in a submerged recording chamber with a perfusion rate of 2–3 mL per min with standard aCSF, tempered at 32±1°C and bubbled with carbogen gas. Borosilicate glass pipettes with a tip resistance of 4–5 MΩ were used for patching neurons. The glass pipettes were filled with a solution containing (in mM) 110 K-gluconate, 10 KCl, 4 Mg-ATP, 10 Na2-phosphocreatine, 0.3 Na-GTP, 10 4-(2-hydroxyethyl)piperzine-1-ethanesulfonic acid (HEPES) and 0.2 ethylene glycol tetraacetic acid (EGTA) (pH 7.2–7.4; 270–290 mOsm). Glutamatergic neurons were identified by shape and patched at the border between stratum pyramidale and stratum radiatum in area CA1 of the hippocampus. Spontaneous excitatory post-synaptic currents (sEPSC) were recorded in absence of any drugs, whereas mEPSC were recorded in the presence of 1 μM TTX. To block the monoamine oxidase B (MAO-B), deprenyl (100 μM) was added in the recovery chamber and all the recording were done under tonic deprenyl treatment.
The access resistance was monitored throughout the recordings and data were included only for stable values (<30% variation). The signal was acquired using an Ag/AgCl electrode connected to a Multiclamp 700B amplifier, digitized with Digidata 1440A and handled with the Clampex software (v. 10.0; Molecular Devices). Traces were analyzed using the Easy electrophysiology software (v. 2.4). The event detection was based on a template fitting method. Probe recordings (three recordings of 1 min each) from C57Bl6 wild type acute hippocampus slices were used to generate and refine a template from representative events corresponding to an average of ten to twenty detected EPSC. Event detection in AppNL-F and C57Bl6 controls acute slices was then run on a semi-automatic method: each event detected fits the previously generated template. A minimum amplitude threshold of 10 pA was applied: all events that were detected between 0 pA and 10 pA were discarded. Each recording was analyzed on a period of 1 min.
GABA currents [41, 42] were recorded in the presence of NBQX (10 μM) and DL-AP5 (50 μM). After recording a stable baseline, picrotoxin (PTX; 100 μM) was added to block GABAA receptors. Tonic GABA current was calculated as the difference in mean holding current for 1 min after achieving a stable shift in baseline with PTX compared to before PTX.
Field recording
After a recovery period of at least 2 h, slices were transferred in a submerged recording chamber with a perfusion rate of 2–3 ml per minute with either standard aCSF or deprenyl (100 μM)-containing aCSF, tempered at 32±1°C and bubbled with carbogen gas. An extracellular borosilicate recording pipette filled with aCSF was placed in the stratum radiatum of area CA1 and field excitatory post-synaptic potentials (fEPSP) were evoked by electrical stimulation of the Schaffer collaterals using a bipolar concentric electrode (FHC Inc., Bowdoin, ME), connected to an isolated stimulator (Digitimer Ltd., Welwyn Garden City, UK). Recordings were performed with the stimulus intensity to elicit 30–40% of the maximal response and individual synaptic responses were evoked at 0.05 Hz (every 20 s). The acquired signal was amplified and filtered at 2 kHz (low-pass filter) using an extracellular amplifier (EXT-02F, NPI Electronic, Tamm, Germany). Data were collected and analyzed using a Digidata 1440A, Axoscope and Clampfit softwares (Molecular Devices, San Jose, CA). Responses were quantified by determining the slope of the linear rising phase of the fEPSP (from 10% to 70% of the peak amplitude). The response was normalized to the average baseline measured in the last 10 min prior to the start of the experiment.
To study long term synaptic plasticity, we used ten theta-bursts separated with 200 ms. Each theta-burst is four pulses at 100 Hz, a stimulation protocol tested not to be enough to trigger long term potentiation (LTP) in slices from C57Bl6 mice.
Immunohistochemistry
Perfusion and brain extraction
Mice were deeply anesthetized with a mixture of ketamine (100 mg/kg) and xylazine (16 mg/kg) i.p. In absence of corneal reflexes, mice were transcardially perfused with NaCl 0.9% followed by a 4% PFA solution. The brain was extracted and post-fixed in a 4% PFA solution for 48 h, washed in PBS for 24 h and placed in a 30% sucrose + 1% natrium azide solution until further use.
Slicing and staining
Brains were cut in a cryostat at 140 μm thick sections (for astrocytic morphology analysis) and 40 μm (for inflammation markers analysis). For GFAP staining, free-floating slices were treated with a peroxide solution (10% methanol and 10% H2O2 in PBS) for 20 min followed by antigen retrieval solution (Tris 10 mM EDTA, 10% Tween-20) at 80°C. After blocking steps with normal donkey serum, slices were incubated with primary glial fibrillary acidic protein (GFAP, Synaptic system 173–004; 1 : 500) and primary connexin 43 (Cx43, Invitrogen 71-0700; 1 : 500) for 72 h (for the morphology analysis); 40 μm thick slices were incubated with vimentin primary antibody (Merk, AB5733) for 48 h. Slices were incubated with the appropriate secondary antibodies for 2 h, DAPI (1 : 5000) was added to counterstain nuclei. Slices were then mounted on non-coated microscope glass coverslips using the fluoromount mounting medium (Invitrogen, 00-4958-02). Two z-stacks (0.25 μm step size) per slice (two slices per animal), were obtained from the CA1 region of the hippocampus using a confocal microscope (Zeiss LSM700) with a 63× oil objective.
Image analysis
For morphological analysis, the z-stacks were deconvoluted using the Hyugens software (Scientific Volume Imaging) and noise-saturated pixels were removed. GFAP/DAPI images were segmented thanks to a pixel classification protocol using the Ilastik software [43]. Using Image J (NIH), centroids of the nuclei were determined, DAPI/GFAP images were merged and each astrocyte were manually separated from the astrocytic syncytium thanks to the Cx43 staining giving the precise location of each connexon. Every astrocyte touching at least one border of the z-stack was automatically removed in order to exclude truncated astrocytes. The center of the sholl analysis was defined by the centroid of the nuclei. Branch counting started at a radius of 7 μm and ended at 45 μm from the center, with a step size of 2 μm. Subsequent data (i.e., sholl analysis and volume occupied by the cell) were automatically generated thanks to the Image J software using a custom macro. Detailed workflow is available in the Supplementary Figure 2.
For the vimentin signal analysis, maximal intensity projections images where thresholded and cells expressing vimentin were identified and analyzed automatically in the ImageJ software. A size filter was applied to exclude vessels. The total vimentin staining in all detected regions of interest was summed for each image.
Statistical analysis
IBM SPSS Statistics (v. 28 IBM corp) was used to perform the statistical analysis. For each measured variable, a Shapiro-Wilk test for normality and Levene’s test for homogeneity of variances were conducted. If none of them was significant (indicating a normal distribution and homogeneous variances), a student t-test was conducted. If one or the other was significant, a Mann-Whitney test was conducted. Because of the experimental design, and the presence of multiple variables measured at once, a two-way ANOVA followed, if applicable, by a Bonferroni correction was used to analyze the ELISA (see Supplementary Material) and the astrocytic morphology experiments. A one-way ANOVA followed, if applicable, by a Bonferroni correction was used for the five-trials social memory test and the olfactory habituation/dishabituation test. For all the tests, 0.05 was set as the significance threshold. All results are shown as mean±S.E.M. and the individual values are displayed on each graph. The number of animals used, the number of slices and the number of recorded cells is described in each figure caption.
RESULTS
Astrocytes display reactive-like phenotype in 6-month-old AppNL-F mice
Morphologically, reactive astrocytes are characterized by a swollen soma and increased branching. Morphology analysis of GFAP stained astrocytes in brain sections from 6-month-old mice revealed a reactive-like morphology in AppNL-F mice compared to C57Bl6 mice (Fig. 1A–C). The morphological complexity was quantified as an increased number of branches, counted every 2 μm from the soma (Two-way ANOVA with distance and genotype as the main factors. Distance: F(8,230) = 98.167, p < 0.001; Genotype: F(1,40) = 26.845; p < 0.001; Interaction: F(8,230) = 19.927, p < 0.001). The difference was particularly prominent between 7 and 25 μm from the soma (Fig. 1B). Moreover, the volume occupied by each astrocyte was bigger in AppNL-F mice compared to C57Bl6 mice (C57Bl6 : 2396±486.1 μm3 versus AppNL-F: 10133±1568 μm3, p < 0.01; Fig. 1C). The number of astrocytes (GFAP-positive cells) was not significantly different in AppNL-F compared to C57Bl6 controls (Supplementary Figure 2D) when estimated as the number of astrocytes per image. Astrocytic reactivity is not limited to morphological changes, and we further analyzed the commonly used astrocytic reactivity marker vimentin. The analysis in the hippocampus revealed a significant increased vimentin signal in AppNL-F mice compared to C57Bl6 controls (46218±17227 a.u. versus 222674±46862 a.u, p < 0.001; Fig. 1D, E).
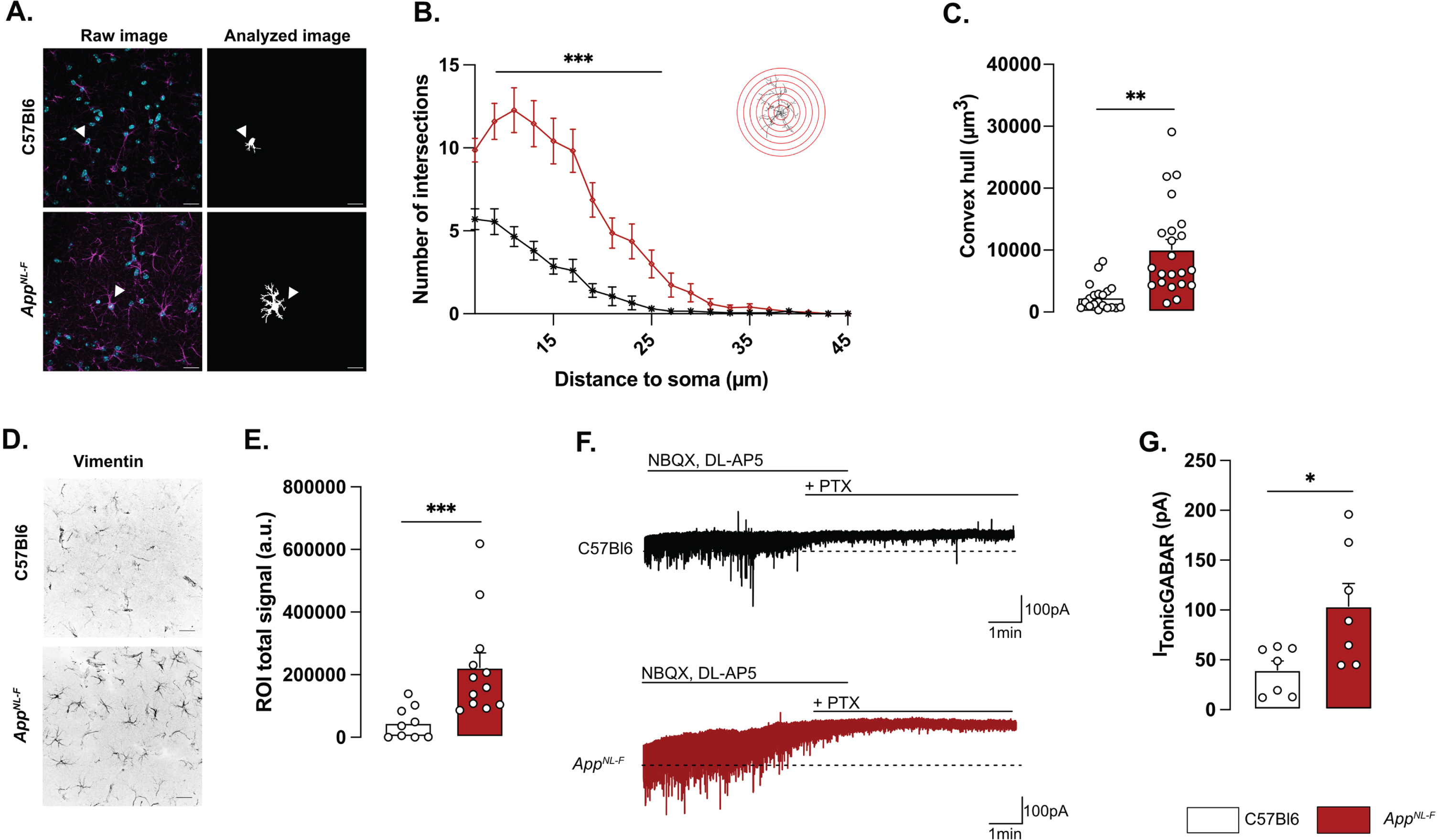
Reactive astrocytes have been shown to synthesize and release GABA [30] and we thus recorded tonic GABA currents in pyramidal neurons in area CA1 (Fig. 1F). Indeed, tonic GABA currents were significantly higher in AppNL-F mice compared to C57Bl6 (103.8±22.63 pA versus 39.83±9.057 pA, p < 0.05; Fig. 1G).
Impaired synaptic transmission and plasticity in AppNL-F mice
Whole-cell patch clamp recordings from pyramidal neurons of area CA1 showed that resting membrane potential was significantly depolarized in AppNL-F mice compared to C57Bl6 mice (Fig. 2A; –51.74±1.32 mV versus –56.18±1.38 mV; p < 0.05). Spontaneous synaptic activity in active hippocampal network (sEPSC) was not different between the two strains in neither frequency amplitude, decay or rise time of sEPSC (Fig. 3A-D). However, when action potentials were blocked with the sodium channel blocker TTX (1 μM) to record spontaneous release of individual synaptic vesicles (mEPSC), a reduced frequency of the mEPSC (Fig. 2B) was observed in AppNL-F mice compared to C57Bl6 mice (0.27±0.04 Hz versus 0.56±0.08 Hz; p < 0.01), whereas the amplitude of mEPSCs remained unchanged (C57Bl6: –21.71±1.48 pA, AppNL-F: –19.65±1.43 pA, Fig. 2C). There were no differences in the decay or rise time of mEPSC (Supplementary Figure 3E, F respectively).
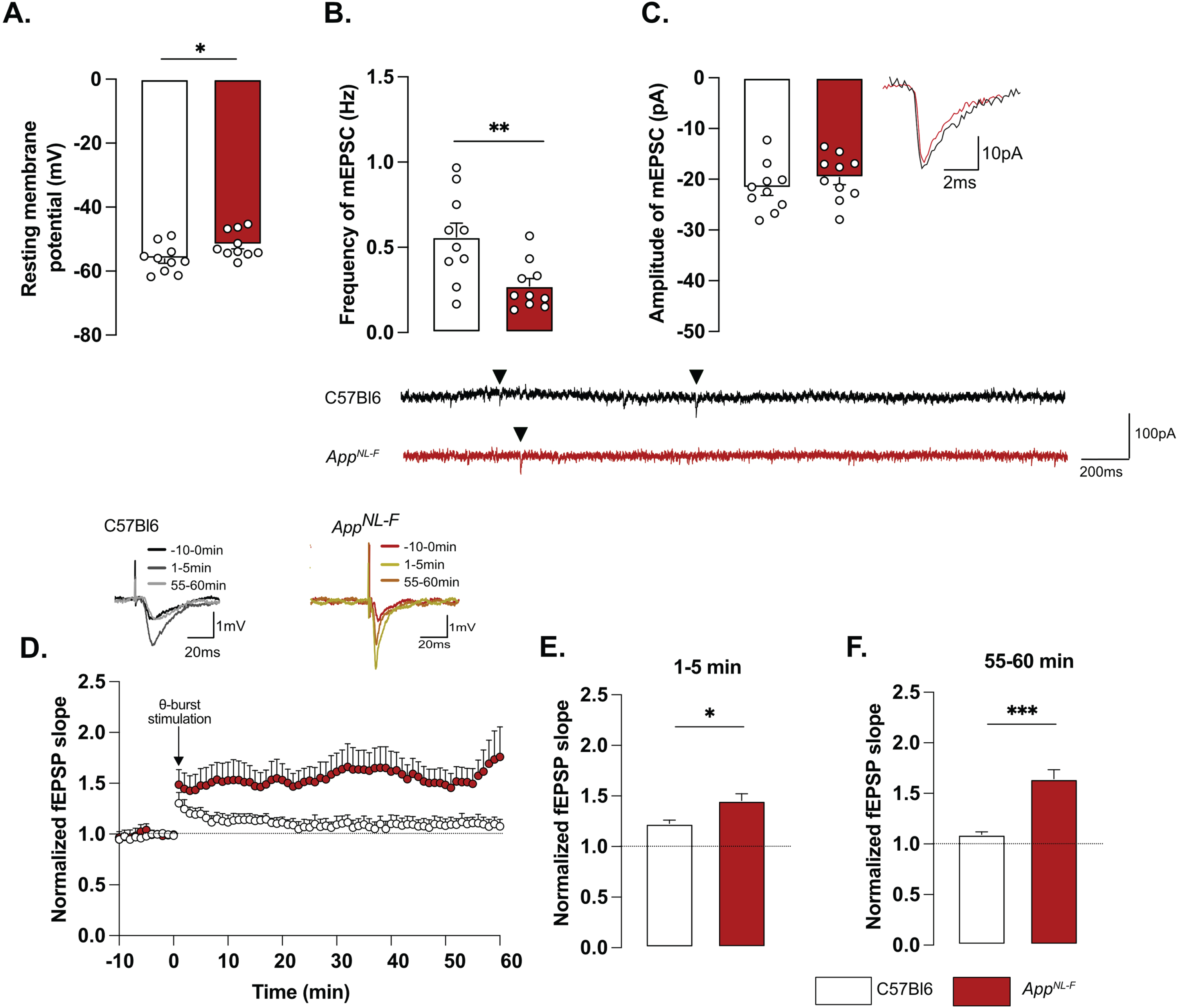
Changes in basic synaptic transmission can entail changes in synaptic plasticity, previously described as synaptic mistuning [44]. To investigate the state of plasticity in AppNL-F, the possibility to undergo long-term potentiation (LTP) was examined, using a subthreshold stimulation protocol that did not evoke LTP in C57Bl6 mice. Ten theta-bursts stimulation of Schaffer collaterals induced an immediate potentiation of synaptic strength, as measured as the increased slope of the evoked fEPSP, in both genotypes (p < 0.001; Fig. 2D, E). However, 1 h after stimulation, the fEPSP had progressively returned to baseline in control animals, whereas the magnitude of the fEPSP was still significantly increased in AppNL-F mice (p < 0.001 different from the baseline; Fig. 2D, F). Thus, already at 6 months, AppNL-F mice have changes in synaptic function, detected as reduced frequency of synaptic events and a reduced threshold for potentiation.
MAO-B blocking restores synaptic transmission and plasticity in AppNL-F mice
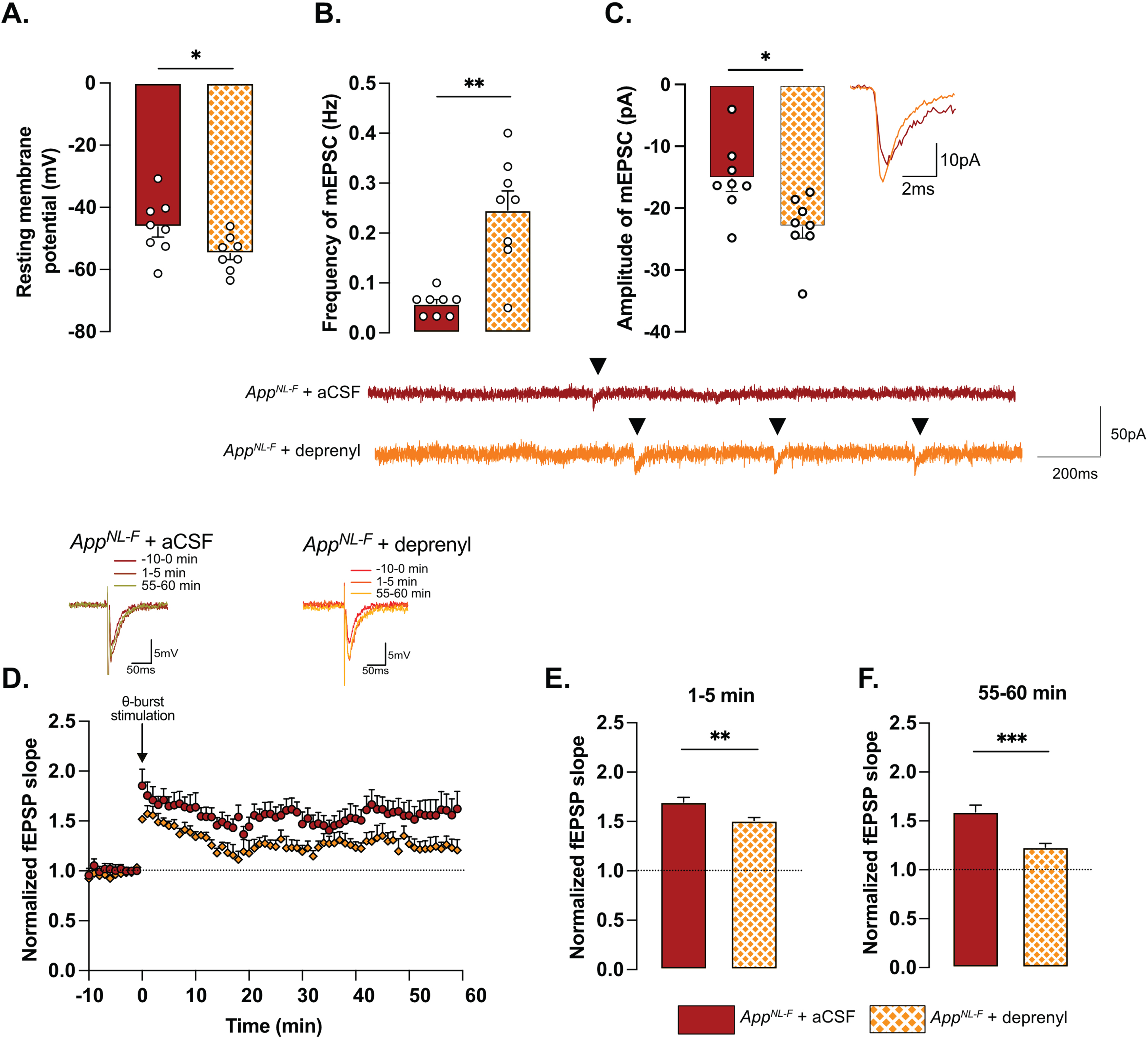
To examine the relationship between tonic GABA and synaptic function, the astrocytic GABA-synthesizing enzyme MAO-B was blocked by pre-incubating the slices with deprenyl (100 μM). Blocking MAO-B caused an increase in the resting membrane potential in deprenyl treated slices from AppNL-F mice compared to non-treated slices (–54.84±1.994 mV versus –46.30±3.251 mV, p < 0.05, Fig. 3A). Moreover, deprenyl increased the frequency of mEPSC events from 0.05±0.008 Hz, in non-treated slices to 0.26±0.03 Hz in deprenyl treated slices (p < 0.01, Fig. 3B) and the amplitude from –15.23±2.10 pA to –23.06±1.79 pA (p < 0.05, Fig. 3C). For spontaneously evoked synaptic events, deprenyl treatment significantly increased the amplitude of sEPSC but not the frequency (Supplementary Figure 3G, H). Deprenyl did not affect rise time nor decay time of either sEPSC or mEPSC (Supplementary Figure 3I-L).
Pre-treatment with deprenyl also had an effect on synaptic plasticity. Unexpectedly, synaptic potentiation was reduced in slices from AppNL-F mice that had been pretreated with deprenyl compared to non-treated slices (Fig. 3D). Deprenyl reduced fEPSP magnitude both at 1–5 min (AppNL-F + aCSF: 1.70±0.05, AppNL-F + deprenyl: 1.51±0.03) and 55–60 min (AppNL-F + aCSF: 1.519±0.07, AppNL-F + deprenyl: 1.23±0.04) after the same sub-threshold stimulation as used above (Fig. 3E, F).
Taken together, our results show that already at a pre-plaque stage AppNL-F mice have reactive-like astrocytes. We further suggest that GABA release from astrocytes contribute to synaptic impairment since in the hippocampus, MAO-B is predominantly expressed by reactive astrocytes [45]. A neuronal origin of tonic GABA is however not explicitly ruled out.
AppNL-F mice show a lack of motivation and a mild depressive-like behavior
Deficits in plasticity is typically linked to memory problems. However, despite deficits in plasticity at 6 months, AppNL-F mice have no spatial memory impairment at this age [46]. Other forms of memory have not been explored, prompting us to investigate the possible effect on social memory. In the five-trials social memory test the social interactions with an unknown animal in four consecutive trials is used to evaluate social memory whereas the interaction with a new animal on the fifth trial is used to assess the social novelty preference and motivation [47, 48] (Fig. 4A). A similar decrease in social interactions was observed over the four first trials in both AppNL-F and C57Bl6 animals (Fig. 4B), suggesting no social memory impairments at 6 months of age. However, on the fifth trial the social interactions with a new animal were increased in control mice (8.93±2.98 s on trial 4 versus 28.41±2.98 s on trial 5; p < 0.05) whereas no change was observed in the fifth trial compared to the fourth in AppNL-F mice (10.74±1.95 s on trial 4 versus 12.92±4.19 s on trial 5). The latency to first interaction was similar in both groups during the first trial, (C57Bl6 : 26.32±13.31 s, AppNL-F: 11.69±6.903 s); however, it was significantly increased in AppNL-F mice compared to C57Bl6 mice on the fifth trial (132.01±41.24 s versus 9.73±5.13 s; p < 0.01; Fig. 4C). Together, these results show an intact social memory in AppNL-F mice at 6 months, and suggest a slight reduced preference for social novelty, evident at the fifth trial. To rule out potential olfactory dysfunctions, which could interfere with the social memory tests, we performed an olfactory habituation/dishabituation test [49] (Fig. 4D). Four odors were tested and no significant differences between odor recognition was observed between C57Bl6 and AppNL-F mice. Both strains spent more times interacting with social odors, especially from an unknown cage, showing the absence of olfactory problem as well as a capacity of AppNL-F mice to discriminate between social and non-social odors. Furthermore, since motor coordination could be a confounding factor in behavior tests, motor coordination was evaluated on the rotarod. No difference between C57Bl6 and AppNL-F mice was observed in the performance in the rotarod (Supplementary Figure 4).
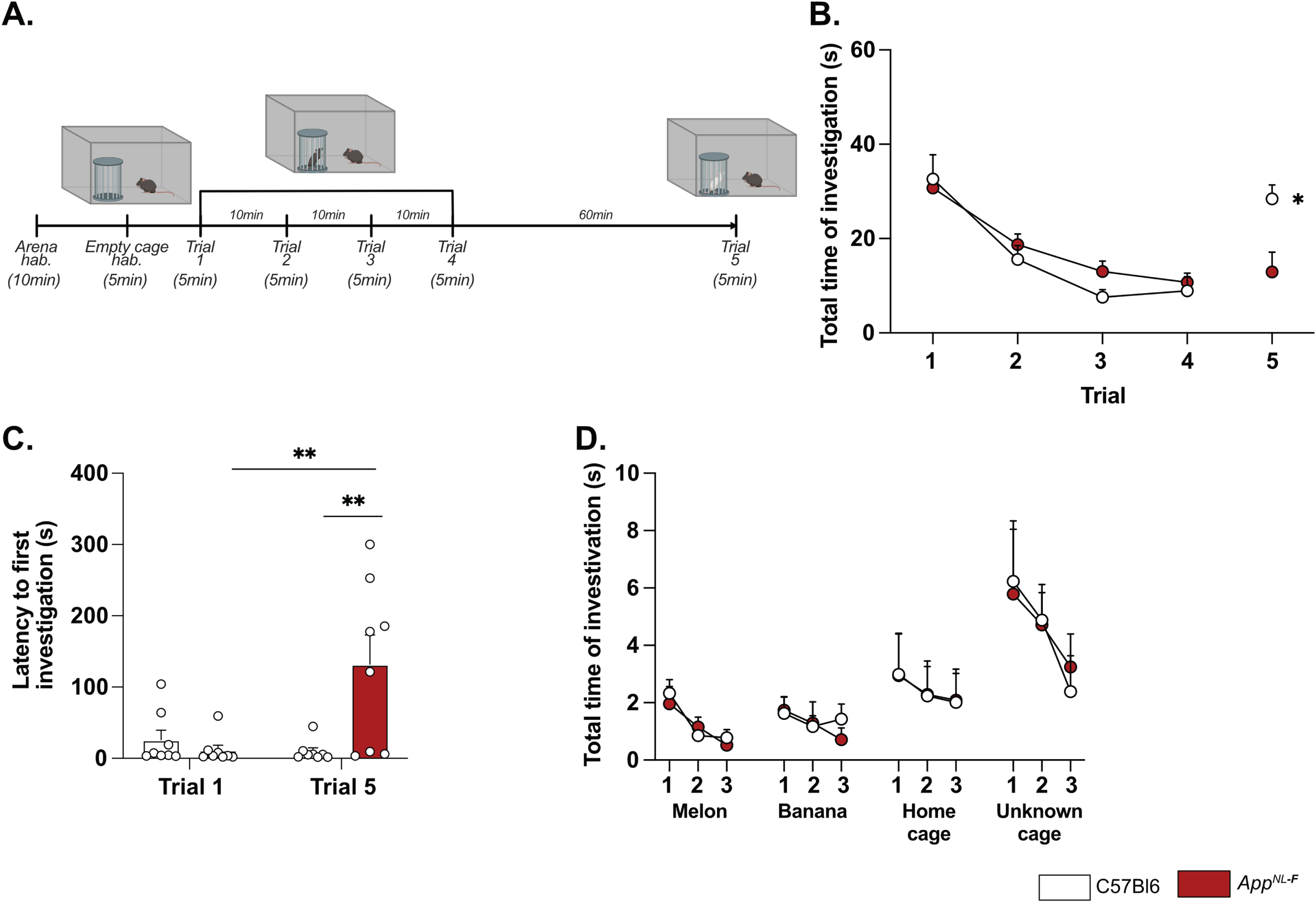
A reduced preference for social novelty in 6-month-old AppNL-F mice is consistent with clinical studies on patients with AD [50] and prompted further research of emotional symptoms. In the open field test AppNL-F animals had a significant reduction of the travelled distance compared to C57Bl6 (3543±61.1 cm versus 5663±421.1 cm; p < 0.001; Fig. 5A). The number of entries in the central area was significantly lower in the AppNL-F mice compared to C57Bl6 mice. (6.50±1 entries versus 30.75±4 entries; p < 0.01; Fig. 5B) whereas the time spent in the central zone was not significantly different (C57Bl6 : 81.16±6.23 s, AppNL-F: 82.30±7.22 s; Fig. 5C). In the elevated plus maze the number of entries in the open arms (anxiogenic area) was significantly reduced in AppNL-F mice compared to C57Bl6 (3.25±1 entries versus 6.12±1 entries; p < 0.05; Fig. 5D) whereas the total time spent in this area was not different between the strains (C57Bl6 : 38.38±5.68 s, AppNL-F: 26.63±5.76 s; Fig. 5E).
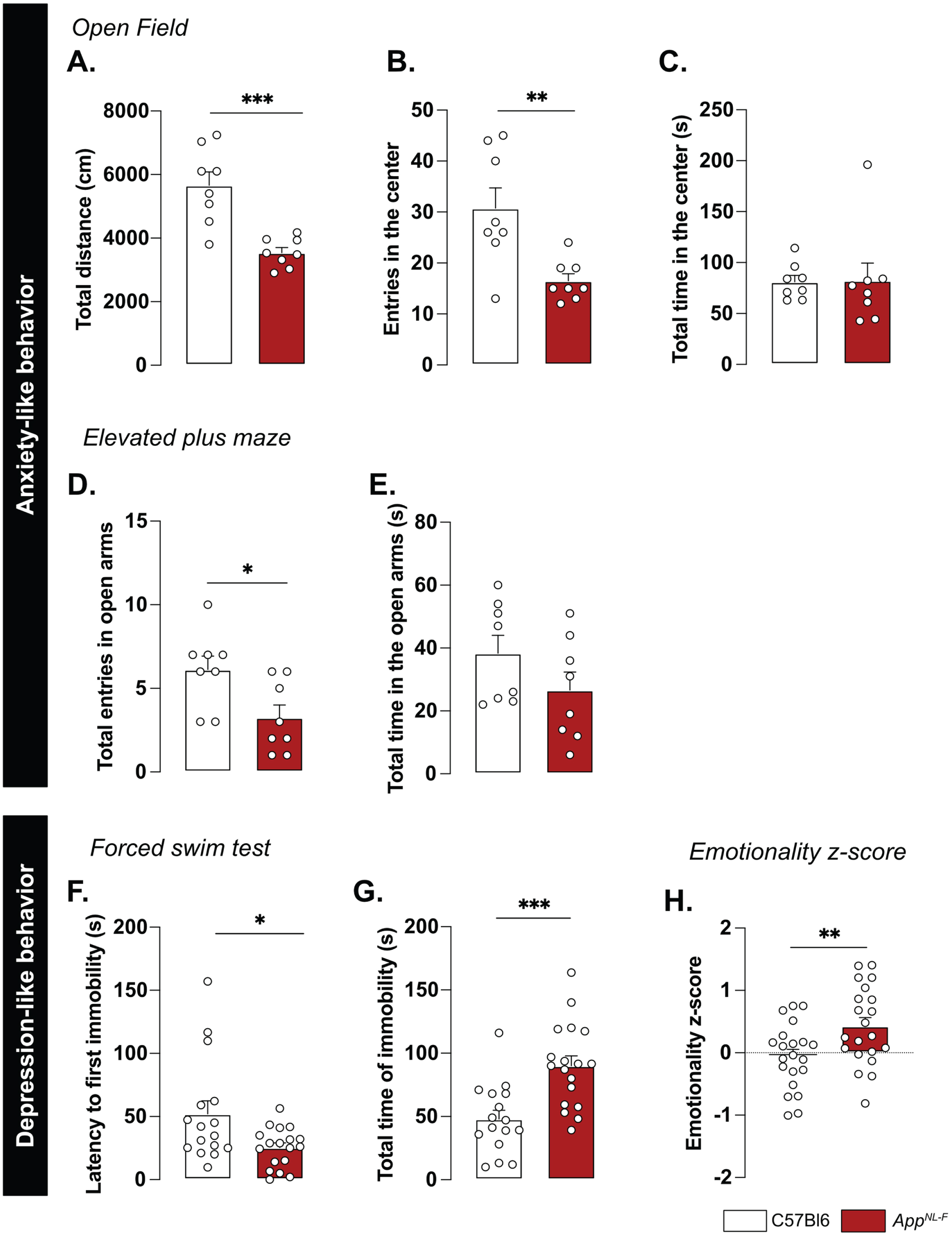
We then explored the depression-like behavior in three different paradigms. In the forced swim test, the immobility was used as an indicator of helplessness. The latency to the first immobility was significantly reduced in AppNL-F mice (C57Bl6 : 52.05±0.30 s, AppNL-F: 25.43±3.70 s, p < 0.05; Fig. 5F) whereas the total time of immobility was significantly increased (C57Bl6 : 48.07±6.89 s, AppNL-F: 90.07±7.79 s; p < 0.001; Fig. 5G). In the splash test (see Supplementary Material), the lack of grooming reflects a lack of self-care. No significant differences in the grooming behavior were observed in the AppNL-F mice compared to C57Bl6 (Supplementary Figure 4C-E). The sucrose preference test (supplementary methods), where a lack of preference for sugar compared to water is considered to reflect anhedonia, revealed no difference between the two strains (Supplementary Figure 4F).
These behavioral data suggest a slight anxiety- and depression-like behavior and this was quantified as an emotionality z-score [40]. We used several parameters per test to establish a z-score for each test (ZSc_test; Supplementary Table 1) and each ZSc_test was then combined into an emotionality z-score (Fig. 5H). The C57Bl6 mice were used as the control group and have an average z-score of zero (–0.05±0.11). The AppNL-F mice had a significantly increased z-score (0.42±0.13, p < 0.01) confirming depressive-like behavior in the 6-month-old AppNL-F mice.
A single dose of ketamine (5 g/kg) partially corrects depressive-like behavior in AppNL-F mice
A low dose of ketamine has antidepressant effects, and can also revert dysfunction in synaptic tuning [44]. Thus, an i.p. injection of either ketamine (5 mg/kg) or vehicle solution (NaCl 0.9%) was administered to 6-month-old AppNL-F mice and behavior was tested 24 h after injection. In the five-trials social memory test, ketamine did not affect social memory as shown by a similar decrease of social interaction over four consecutive trials, in both treatment conditions (Fig. 6A). As in our previous experiments, the social interaction measured in the fifth trial was not significantly different from the fourth trial in the vehicle treated animals (trial 4 : 18.90±4.66% versus trial 5 : 38.20±5.91%, Fig. 6B). Ketamine did not have a significant effect on the social interaction on the fifth trial (trial 4 : 22.085±4.97% versus trial 5 : 40.12±9.48%).
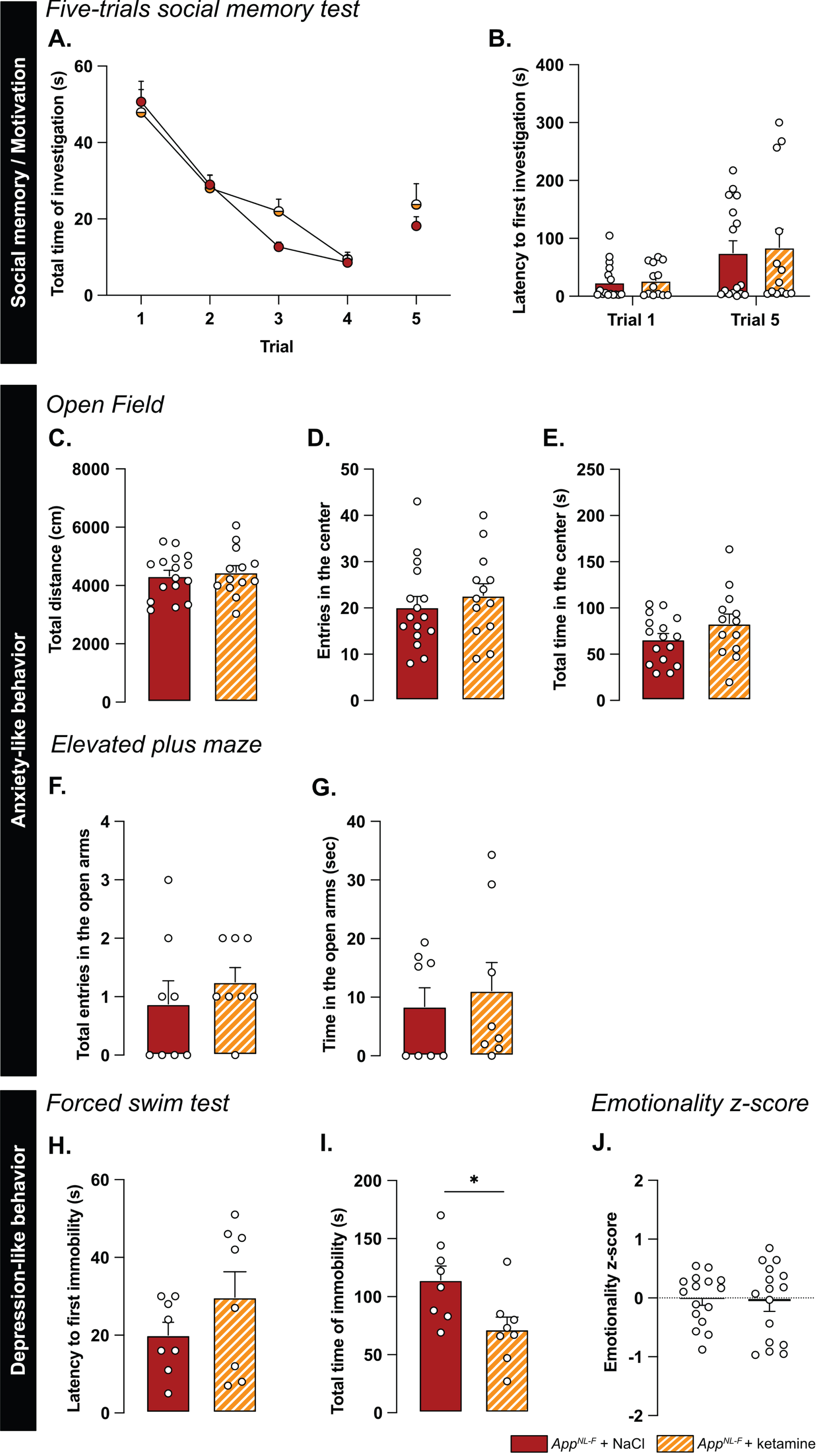
We further explored the antidepressant action of a single injection of 5 mg/kg ketamine. In the open field, none of the parameters total distance travelled (NaCl: 4331±190.5 cm, ketamine: 4454±229.3 cm; Fig. 6C), the number of entries in the center (NaCl: 20.19±2.30 entries, ketamine: 22.69±2.55 entries; Fig. 6D) and total time spent in that zone (NaCl: 66.11±6.35 s, ketamine: 83.13±10.32 s; Fig. 6E) were changed by the ketamine treatment. Likewise, ketamine had no effect on the number of entries in the open arms in the elevated plus maze (NaCl: 0.87±0.40 entries, ketamine: 1.25±0.25 entries; Fig. 6F) nor the total time spent in that zone (NaCl: 8.41±3.2 s, ketamine: 1.12±4.79 s; Fig. 6G).
In the forced swim test, no changes were observed in the latency to first immobility (NaCl: 20±3.32 s, ketamine: 29.75±6.56 s; Fig. 6H). However, there was a reduced total time of immobility in ketamine treated mice compared to NaCl treated (NaCl: 114.4±2.02 s, ketamine: 71.88±10.61, p < 0.05; Fig. 6I). Similarly, in the splash test, a reduced latency to first grooming (NaCl: 32.52±3.71 s, ketamine: 16.30±2.56 s, p < 0.01; Supplementary Figure 4G), increased frequency of grooming (NaCl: 7±0.27, ketamine: 9.13±0.69, p < 0.05; Supplementary Figure 4H) and an increased total time of grooming (NaCl: 145.7±6.01 s, ketamine: 188.8±3.56 s, p < 0.05; Supplementary Figure 4I) were observed after ketamine treatment.
The behavioral data was integrated into the emotional z-score (Fig. 6J) to assess the overall effect of ketamine on the depressive-like behavior of AppNL-F. Animals who received the vehicle solution were used as the control group with an average emotionality z-score of zero. No statistical difference was shown in the z-score between the two different groups. Thus, ketamine did not reverse all emotional symptoms in the AppNL-F mice. However, in line with the well-known antidepressant effect of ketamine [51], we show an effect of ketamine in specific test for depressive-like behavior in these mice.
Ketamine (5 mg/kg) does not affect astrocyte morphology but partially restores synaptic dysfunctions and reduces astrocytic tonic inhibition
Astrocyte morphology was evaluated 24 h after ketamine (5 mg/kg) injection. The sholl analysis (Fig. 7A, B) did not unveil differences between NaCl and ketamine treated animals (Two-way ANOVA with distance and treatment as the main factors. Distance: F(2.244,630.4)=536.8, p < 0.001; Genotype: F(1,181)=0.5445; p = 0.461; Interaction: F(19,5339)=0.2808, p = 0.999). No differences were detected in the convex hull analysis (Fig. 7C) showing that a single injection of ketamine did not alter astrocytic morphology when given 24 h prior to analysis.
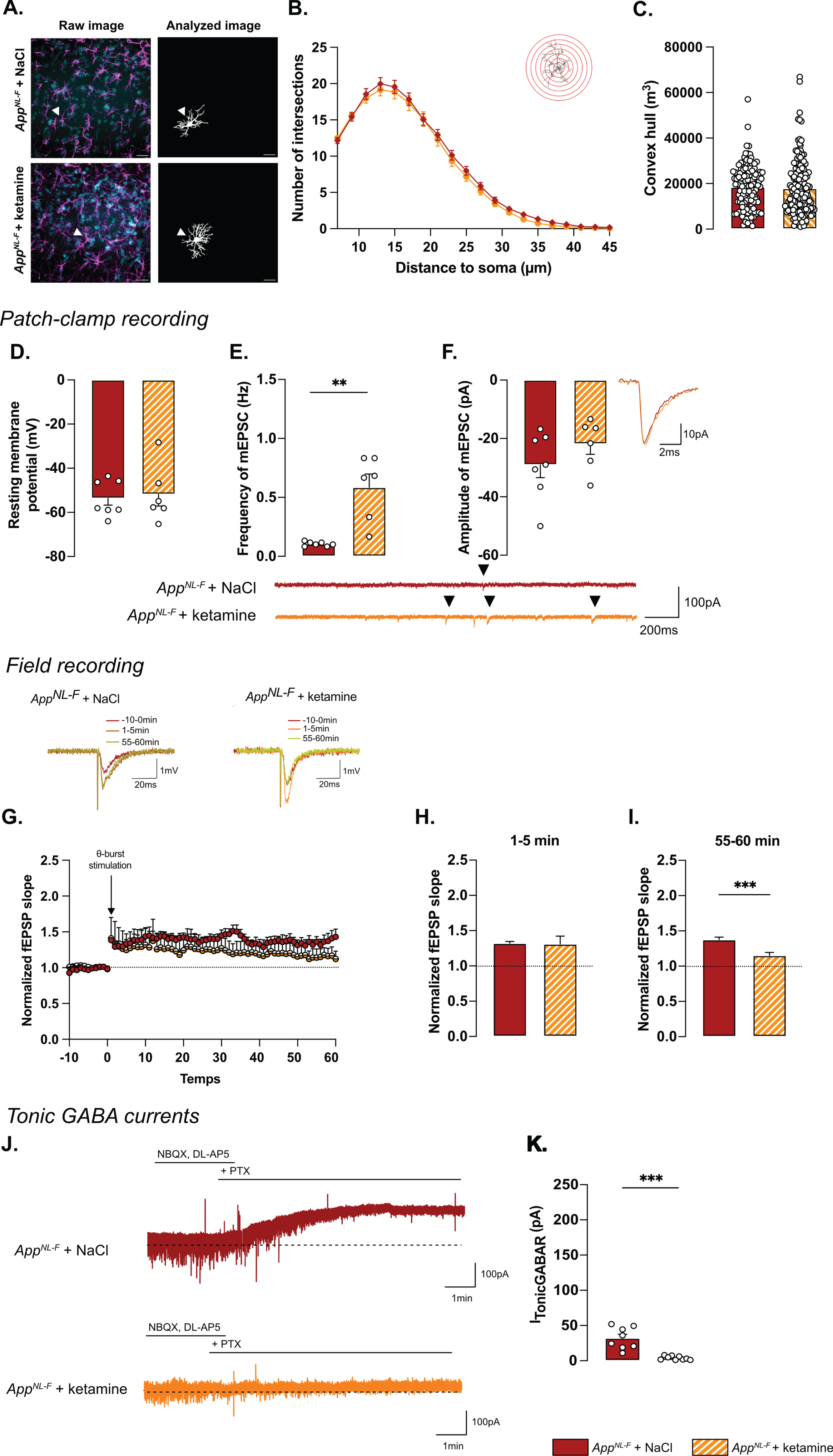
Regarding electrophysiological activity, no difference was observed on resting membrane potential (NaCl: –53.4±3.08 mV, ketamine: –51.8±5.29 mV; Fig. 7D) or any sEPSC parameters (Supplementary Figure 3M-P). However, ketamine induced a significant increase in frequency of mEPSC (NaCl: 0.10±0.001 Hz, ketamine: 0.59±0.11 Hz, p < 0.01; Fig. 7E) while the amplitude (NaCl: –29.03±4.40 pA, ketamine: –21.89±3.52 pA; Fig. 7F), decay time (Supplementary Figure 3Q) and rise time (Supplementary Figure 3R) were not affected.
Synaptic plasticity deficits in 6-month-old AppNL-F mice were restored by ketamine: a subthreshold theta-burst stimulation gave an initial potentiation of the fEPSP in both conditions (NaCl: 1.32±0.02, ketamine: 1.31±.11; Fig. 7G, H). Fifty-five minutes after stimulation, the magnitude of the long-term potentiation was significantly smaller in AppNL-F + ketamine compared to AppNL-F + NaCl (Fig. 7G-I; p < 0.001). Taken together, these electrophysiological recordings confirm the ability of ketamine to retune misregulated synaptic transmission and plasticity.
Since we hypothesize that the synaptic mistuning in AppNL-F mice is mediated through astrocytes, we tested whether ketamine would affect the astrocytic tonic inhibition (Fig. 7J). Tonic GABA currents were recorded in hippocampal slices from AppNL-F mice that had been treated with ketamine 24 h earlier. Tonic GABA currents were significantly decreased in ketamine treated AppNL-F mice compared to saline treated group (3.94±0.9 pA versus 32.11±5.457 pA, p < 0.001; Fig. 7K), suggesting a role for astrocytes in the antidepressant effect of ketamine.
DISCUSSION
As the final product of the dysregulated amyloid cascade, Aβ plaques have traditionally been studied as one of the main cause of AD pathology [52], although we know that both behavioral and pathophysiological changes occur before plaques can be detected [53]. In a late onset mouse model of AD (AppNL-F), at 6 months of age, limited occurrence of plaques has been reported [33] and we did not detect any Aβ plaques in the hippocampus in 6-month-old AppNL-F mice. However, an increase in non-soluble oligomers of Aβ was detected. Most importantly, at 6 months of age, AppNL-F mice have reactive-like astrocytes, increased tonic GABA, and synaptic misfunction. This is in good agreement with the hypothesis that Aβ oligomers are inducing several toxic events in early AD [54]. It is noteworthy that only male animals have been used in the present study. A recent study describes similarities and differences in behavior in male and female AppNL-F mice [46], however more work in this regard is needed. In particular, the effect of ketamine on female AppNL-F mice should be investigated since it has been shown that female wildtype mice are more sensitive to ketamine [55].
A reduced threshold for long-term potentiation in AppNL-F mice is a surprising finding. Memory impairments and dementia disorders have typically been associated with a decrease in synaptic potentiation; previous work in the AppNL-F mice at 6 months of age show no effect on LTP after a strong tetanic stimulation [56]. However, synaptic potentiation is not linearly correlated with memory, rather synaptic strength is constantly adjusted based on activity patterns in the neuronal network [57]. To learn and adapt to the changing environment, synaptic transmission needs to be plastic, while keeping the overall activity within optimal range. In fact, it has been proposed that cognitive deficits in early AD is not about reduced plasticity, but an imbalance at the network level [58], which is compatible with a reduced threshold for potentiation. Our results in the AppNL-F mice are consistent with previous work, where authors have been using a low or even sub-threshold stimulation protocol in other model with memory impairment [20]. The increase in synaptic potentiation, as well as the lack of change in evoked synaptic response, in 6-month-old AppNL-F mice has also been described by others [19], who attribute the effect to a decreased turnover of presynaptic proteins.
In this work we reveal that blocking MAO-B with deprenyl changes synaptic transmission in AppNL-F mice in several ways. As expected, deprenyl pre-treatment increases the amplitude of synaptic events, both when evoked by spontaneous action potentials (sEPSC) and when arising from non-evoked release of individual vesicles (mEPSC). This effect is easily explained by an increase in membrane resistance due to the decreased GABA tone. In agreement, the frequency of mEPSC is increased in deprenyl-treated slices, however the frequency of sEPSC is not. Interestingly, the threshold for LTP is increased by blocking MAO-B and not reduced as one would expect with a reduced GABA tone. Thus, blocking GABA synthesis in astrocytes in AppNL-F mice entails more and other changes than a direct increase in membrane resistance. It would be interesting for future studies to explore the mechanism underlying the apparent discrepancy between increased astrocytic tonic GABA and the reduced threshold for LTP. Previous work in our lab has shown that synaptic activity and plasticity are strongly interacting in a well-tuned relationship [44]. Thus, the increased synaptic activity induced by deprenyl could tune synaptic plasticity to specifically reverse synaptic deficits in the AppNL-F mice.
The synthesis and release of GABA from reactive astrocytes are well described phenomena and were first shown in a mouse model overexpressing App and with a severe plaque load [59]. Here we describe that astrocytes are reactive already in 6-month-old AppNL-F mice through a change in morphology as well as increased vimentin expression. As a consequence, and in line with previous research [41], tonic GABA is increased already before plaque formation in the AppNL-F mice model of AD. Reactive astrocytes [28] have been described as the starting point of several brain dysfunctions and proposed as an interesting therapeutic target in various brain disorders [60], and more recently in neurodegeneration and aging [61]. We do not directly rule out a neuronal origin of GABA that can be a consequence of excessive inhibitory activity and activation of extra synaptic GABAA receptors [62, 63]. However, the astrocytic origin of GABA is strongly supported by results showing that the main effect of blocking MAO-B in the hippocampus is a reduction in astrocytic GABA [42, 45].
Reactive astrocytes, as well as synaptic impairments, are linked to depressive-like behavior [64]. AppNL-F mice do indeed display depressive like behaviors in some tests already at 6 months. There is no test that directly translates to clinical depression, partly due to the fact that clinical depression can be describes as a syndrome, with different clinical presentations [65]. To get an overall vision of the emotional state of the AppNL-F mice, we compiled our behavioral data into an emotionality z-score, relevant to study depression-like behaviors assessed on different scales [66]. Since the reduced mobility could be another confounding factor in several of the tests used to assess depression-like behavior, data obtained in the open field and elevated plus maze were normalized with the travelled distance. When all the affective behavioral test are integrated in a z-score there is a significant difference in AppNL-F compared to C57Bl6 mice. This is in good agreement with early depressive symptoms observed in AD patients [10] as well as results from other animals models of the disease [67]. Moreover, we confirm the lack of memory impairment at this age using the five-trials social memory test. In the last session of this test, the AppNL-F mice do not show increased time of interaction with the novel individual, as does the C57Bl6 mice. Having ruled out that the difference is due to olfactory problems or motor coordination problems, we suggest that this lack of interaction is due to a reduced social novelty interest, an indicator of an apathy-like behavior, which is consistent with the depressive like phenotype. However, another explanation could be that AppNL-F mice have deficits in social recognition consistent with recent observation in early diagnose patients [68]. We here use as the “presenting animal”, mice with the same fur color, same age, same gender as the tested animal. Thus, AppNL-F mice might present early pattern separation deficits, as they cannot discriminate two very close situations (i.e., identify the mice as a new individual), in line with deficits observed in early AD [69]. Further work will need to be done to clarify this.
Ketamine was identified as a fast-acting antidepressant drug in the early 2000 s [70] and has been suggested for treatment also in depression associated to AD [71, 72]. We investigated the therapeutic effect of a low dose of ketamine (5 mg/kg) delivered i.p., 24 h prior to experiments. In line with the literature [73], we successfully show the effect of this drug in the forced swim test and the splash test. There was no significant effect in the anxiety related tests and the overall emotionality z-score was not significantly improved. Moreover, the lack of exploration of the novel mice at the fifth trial in the social exploration test was not affected by ketamine.
The partial effect of ketamine on behavior contrasts with the significant effect of ketamine on the synaptic dysfunction in the AppNL-F mice, where mEPSC frequency is increased and the threshold for LTP induction is restored. In other models, especially when LTP is induced with a strong stimulation, ketamine has been shown to enhance LTP [74, 75]. Nevertheless, the reduction of LTP inducing threshold by ketamine is in good agreement with previous work from our lab where we show that ketamine affects synaptic transmission and plasticity in a state-dependent manner, to stabilize the synaptic tuning [44]. Therefore, caution should be exercised when comparing the impact of ketamine on synaptic function across various animal groups or treatment methods.
Thus, a single dose of ketamine partially corrects early synaptic dysfunctions, in the AppNL-F mice, but do not reverse behavioral deficits. However, ketamine does have an antidepressant effect in several of the tested paradigms. In this respect it is interesting to note that deprenyl, that also reverts synaptic dysfunctions in the AppNL-F mice is also used clinically as an anti-depressant drug [76], this effect has typically been ascribed to its effect on monoamine synthesis rather than the effect on astrocytes.
Increasing synchronous neuronal activity in the gamma frequency has been shown to slow-down disease progress and improve cognitive performance in AD [77] (but see also [78]). Moreover, recent work suggest that neuronal activity can indeed affect Aβ accumulation and plaque deposition [79]. Restoring synaptic transmission and plasticity may thus have a prophylactic effect on AD rather than an acute effect. It has been shown that depression is a risk factor for AD and interestingly antidepressant drugs seem to prevent the development if the disease [80] although the long term effect of ketamine in this regard has not been evaluated and whether this effect is mediated by retuning of synaptic function remains to be shown. We can however speculate that restoring synaptic tuning could also be a biological mechanism underlying the beneficial effect of a mental and physical active life-style [81]. Thus, it would be interesting to follow-up the long-term effect of chronic ketamine treatment, with its beneficial effect on synaptic tuning on the progression of AD.
Ketamine’s mechanism of action is still debated. Interestingly, we see that ketamine and deprenyl have the same effects on synaptic transmission and plasticity in AppNL-F mice. Furthermore, we show that ketamine reduces tonic GABA, thus one possibility is that ketamine acts on reactive astrocytes to restabilize synaptic tuning, through changes in gliotransmission. This is in good agreement with recent results showing that ketamine affects astrocytic function [82], as well as astrocytic GABA metabolism [83]. Somewhat to our surprise, whilst morphology and function are generally considered to go hand in hand, ketamine did not affect astrocyte morphology after 24 h. This is consistent with the fact that GFAP expression is not reduced after ketamine treatment [84] and underscores the need of better understanding of what defines a reactive astrocyte [28].
Research on AD treatment has focused mainly on Aβ reducing treatments [85] or the cholinergic system [86]. Here we show that targeting the glutamatergic system can correct early synaptic impairment as well as early depression-like behaviors observed in an AD model. How an early intervention restoring synaptic function affects the progression of AD remains to be shown.
AUTHOR CONTRIBUTIONS
Benjamin Portal (Formal analysis; Funding acquisition; Investigation; Methodology; Resources; Validation; Writing – original draft; Writing – review & editing); Moa Södergren (Investigation; Writing – original draft); Teo Parés i Borrell (Investigation; Writing – original draft); Romain Giraud (Investigation; Writing – original draft); Nicole Metzendorf (Formal analysis; Investigation; Writing – original draft); Greta Hultqvist (Supervision; Writing – original draft; Writing – review & editing); Per Nilsson (Conceptualization; Writing – original draft; Writing – review & editing); Maria Lindskog (Conceptualization; Formal analysis; Funding acquisition; Investigation; Supervision; Validation; Writing – original draft; Writing – review & editing).
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank the BioVis imaging facility, the Uppsala University Behavioral Facility (UUBF) and the Animal Behavior Core Facility (ABCF) at Karolinska Institutet for their expertise and their help on setting up the different experiments. The authors would like to thank Takaomi Saido and Takashi Saito at RIKEN Center for Brain Science (Japan) for providing the AppNL-F mice.
Cartoons were created with BioRender.com.
FUNDING
This work was supported by the Swedish Alzheimer’s Foundation (to Maria Lindskog), Wenner-Gren Stiftelsen (to Maria Lindskog), O.E Edla Johansson Vetenskapliga stiftelsen (to Benjamin Portal) and Gun och Bertil Stohnes stiftelsen (to Benjamin Portal).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
All the data generated and analyzed in this study are included in the main manuscript. The custom macro used in the morphological analysis and the detailed statistics are available on reasonable request to the corresponding author.
