Abstract
Background:
Sleep disturbances are considered a hallmark of dementia, and strong evidence supports the association between alterations in sleep parameters and cognitive decline in patients with mild cognitive impairment and Alzheimer’s disease (AD).
Objective:
This systematic review aims to summarize the existing evidence on the longitudinal association between sleep parameters and cognitive decline, with the goal of identifying potential sleep biomarkers of AD-related neurodegeneration.
Methods:
Literature search was conducted in PubMed, Web of Science, and Scopus databases from inception to 28 March 2023. Longitudinal studies investigating the association between baseline objectively-measured sleep parameters and cognitive decline were assessed for eligibility.
Results:
Seventeen studies were included in the qualitative synthesis. Sleep fragmentation, reduced sleep efficiency, reduced REM sleep, increased light sleep, and sleep-disordered breathing were identified as predictors of cognitive decline. Sleep duration exhibited a U-shaped relation with subsequent neurodegeneration. Additionally, several sleep microstructural parameters were associated with cognitive decline, although inconsistencies were observed across studies.
Conclusions:
These findings suggest that sleep alterations hold promise as early biomarker of cognitive decline, but the current evidence is limited due to substantial methodological heterogeneity among studies. Further research is necessary to identify the most reliable sleep parameters for predicting cognitive impairment and AD, and to investigate interventions targeting sleep that can assist clinicians in the early recognition and treatment of cognitive decline. Standardized procedures for longitudinal studies evaluating sleep and cognition should be developed and the use of continuous sleep monitoring techniques, such as actigraphy or EEG headband, might be encouraged.
Keywords
INTRODUCTION
Cognitive decline and dementia are primary and growing public health challenges, with rapidly increasing prevalence and high social burden [1, 2]. Alzheimer’s disease (AD) is the most common type of dementia, accounting for 60–80% of all cases [3, 4]. Growing attention is focused on the recognition of the early stages of the disease such as subjective cognitive decline (SCD) and mild cognitive impairment (MCI), which are widespread conditions with a high risk of conversion to dementia [5–7]. Given the complexity and the difficult management of dementia’s clinical manifestations, early diagnosis, preventive strategies, and biomarkers identification are currently widely acknowledged as research priorities [1, 3]. Therefore, the early recognition of the disease might help in predicting its course for possibly stopping or reverting the cascade of events leading to cognitive impairment, also considering the upcoming era of disease-modifying treatment strategies [8–10].
Indeed, recent research lines focused on the early identification of specific biomarkers of cognitive decline in adults and elderly, with biofluid and neuroimaging emerging as the most promising biomarkers for predicting dementia onset and conversion from preclinical stages to AD [11–17]. However, alongside these established markers, there has been a parallel exploration of accessible and feasible, non-specific biomarkers. This exploration encompasses a wide array of readily available clinical data and parameters, such as blood pressure [18], heart rate [19], diet [20], physical activity [21], and sleep. In particular, an expanding body of literature has focused on sleep alteration as a candidate biomarker for evaluating the risk for dementia, and the associations between cognitive decline and several sleep variables have been investigated, including sleep duration, alterations in sleep macro- and micro-architecture, specific sleep disorders (i.e., insomnia and sleep disordered breathing - SDB), and circadian sleep-wake rhythm desynchronization [22]. Consistently, the association between sleep and cognitive functioning in older adults has been widely shown [23–25], and emerging evidence suggests that sleep alteration may play a relevant role in the development of AD. Sleep disorders are common among SCD, MCI, and AD patients [26–30], and individuals in a preclinical AD stage showing comorbid sleep disorders such as SDB might experience worse longitudinal cognitive deterioration with a possibly more rapid conversion to dementia [31–34].
Amyloid-β (Aβ) is the primary marker of AD, and the alteration of cerebrospinal fluid (CSF) levels of Aβ isoforms or the cerebral deposition of Aβ plaques have been both considered the hallmark of the disease from the very early stages [35, 36]. Therefore, the literature investigating the risk for AD has focused on evaluating the interference of different factors on CSF levels and brain deposition of Aβ. Among these factors, it has been demonstrated that sleep impairment and disorders (such as sleep deprivation, insomnia, and SDB) are related to Aβ cerebral levels and hence possibly increase the risk for AD [37–41]. Indeed, sleep disruption contributes to Aβ accumulation in the brain, and thus may be largely implied in the complex pathophysiology of AD [42–44]. Consistently, associations between Aβ burden and sleep complaints [45], short sleep duration [46], and SDB [47] have been reported among cognitively unimpaired elderly individuals, further highlighting the importance of searching for sleep-related biomarkers that can predict the onset of AD and dementia.
Several reviews highlighted alterations in the aforementioned objectively-measured sleep parameters such as sleep macrostructure (i.e., sleep duration, sleep continuity, and conventional sleep stages), sleep microstructure (e.g., spindles and K-complexes, EEG power spectra, and cyclic alternating pattern [48]), and SDB in subjects with MCI or AD in comparison with elderly controls [49–52]. Nonetheless, these studies solely investigated the results of cross-sectional research, and thus do not provide conclusive evidence concerning the feasibility of the reported sleep alteration as early biomarkers of dementia. Despite large cohort studies and recent meta-analyses showed significant associations between subjectively-evaluated sleep quality and sleep problems and subsequent cognitive decline [53–56], further evidence relying on objective measures is needed to corroborate the consequential nature of these relations. Therefore, this systematic review aims to summarize the findings of longitudinal research investigating whether objective sleep parameters and circadian sleep-wake rhythms measured by polygraphic, polysomnographic, or actigraphic recordings of sleep may predict dementia and AD-related neurodegeneration in older adults.
MATERIALS AND METHODS
Search strategy
This review was conducted following the preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement [57]. A comprehensive search was performed to identify relevant scientific articles on PubMed, Web of Science, and Scopus databases from their inception to 28 March 2023, using the following search query in titles and abstracts: (“Alzheimer’s disease” OR “mild cognitive impairment” OR “cognitive decline” OR “subjective cognitive impairment”) AND (PSG OR polysomnography OR polygraphy OR EEG OR electroencephalography OR electroencephalogram OR actigraphy) AND (“sleep duration” OR “total sleep time” OR “sleep architecture” OR “sleep macrostructure” OR “sleep microstructure” OR “spindle*” OR “slow wave sleep” OR “slow wave activity” OR “NREM” OR “REM” OR “sleep-wake rhythm”). The search results were imported into the Zotero reference manager, and duplicates records were automatically excluded. Two reviewers (MC and CL) manually screened the remaining records according to the eligibility criteria reported below. Relevant reviews and the references lists of screened full-text articles were also examined to identify potentially omitted articles. No language restrictions were applied, and non-English articles were screened using automatic translation tools.
Eligibility criteria
To be eligible for inclusion, studies had to meet the following criteria: 1) longitudinal research design; 2) older (mean age above 60 years for the whole sample) cognitively intact, SCD, or MCI participants at baseline, with no clinical diagnosis of dementia or AD; 3) objective sleep assessment with polysomnography (PSG) and/or actigraphy or headbands with frontopolar EEG signals (polygraphy) performed at least at baseline; 4) at least one relevant outcome concerning cognitive decline obtained at follow-up, including diagnosis of MCI or dementia, longitudinal changes in cognitive test scores, or longitudinal changes in AD-related biomarkers, such as Aβ; 5) explicit exploration of the association between baseline sleep parameters and longitudinal cognitive decline. Studies were excluded if: 1) they were case reports, case series, cross-sectional studies, reviews, abstracts, or editorials; 2) the full-text was inaccessible; 3) they exclusively investigated forms of dementia other than AD; or 4) the sample primarily included patients with Parkinson’s disease or sleep parasomnias related to synucleinopathies, such as REM sleep behavior disorder [58].
The study selection was independently conducted by two authors (MC and CL). Any disagreements on inclusion were resolved by consensus or by a third reviewer’s (MF) arbitration.
Data extraction
Two reviewers (MC and CL) extracted data from the full-text articles using a pre-defined codesheet. For each article, the following records were extrapolated: first author and publication year, country, sample characteristics (size, mean age, female proportion, and cohort name when available), follow-up (number of visits and mean/median time), sleep assessment methods, sleep parameters evaluated, outcomes, covariates, and main findings.
Assessment of study quality
The evaluation of study quality and assessment of risk of bias were conducted collaboratively by two reviewers (MC and CL) using the Newcastle-Ottawa scale for nonrandomized observational studies (cohort studies version) [59]. Each included record underwent scrutiny across nine items encompassing three domains (selection, comparability, and outcome) employing the recommended star rating system, with a maximum achievable score of nine stars. For the purpose of this review, obtained scores were classified into three distinct categories: low quality (0–3 stars), medium quality (4–6 stars), and high quality (7–9 stars).
RESULTS
Search results and quality assessment
The study selection process is illustrated in Fig. 1, which was generated using the PRISMA flow diagram dedicated tool [60]. The literature search of the three databases initially identified 933 records. After removing 537 duplicates, 396 records were screened by title and abstract, resulting in the exclusion of 369 records. Twenty-six full-text articles were assessed for eligibility (one full text was not retrieved), and 11 of them were excluded for various reasons (see Fig. 1 for details). Five full-text articles identified from a search on the references lists of the screened records and other relevant reviews from the literature were considered, and two of them were subsequently included. Table 1 provides a comprehensive list of records excluded after full-text screening along with the reasons for their exclusion.
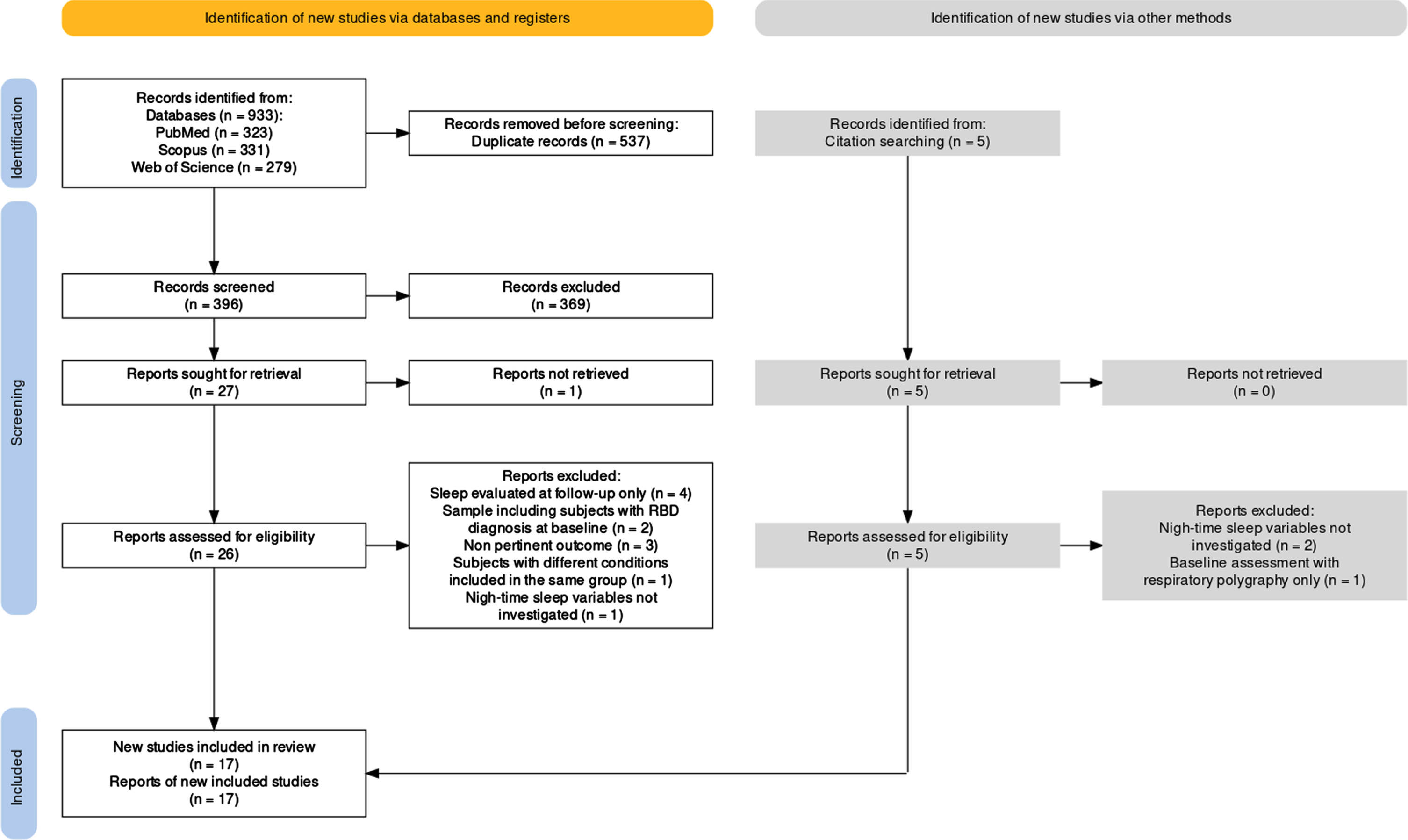
Flow diagram of the study selection process. RBD, rapid eye movement sleep behavior disorder.
List of studies excluded after full-text screening with reasons
SCD, subjective cognitive decline; MCI, mild cognitive impairment; RBD, rapid eye movement sleep behavior disorder; PSG, polysomnography; SDB, sleep-disordered breathing; AD, Alzheimer’s disease.
Ultimately, a total of 17 records were included in the qualitative synthesis. Overall, the study quality was medium-to-high, with a mean Newcastle-Ottawa scale score of 6.3 out of 9 for the included studies. Nine studies were of high quality, and eight received a medium-quality score, with none falling into the low-quality category. Primary concerns included sample size, selection bias, control for confounders, adequacy of follow-up cohorts, and follow-up duration. Quality ratings for each study are reported in Table 2.
Quality assessment of included studies conducted with the Newcastle-Ottawa scale for cohort studies
The maximum number of stars is four for Selection, two for Comparability, and three for Outcome.
Table 3 presents the characteristics and main findings of the studies included in this review. Among the 17 studies, 13 (76%) were conducted in the US, one was conducted in Italy, one in the Netherlands, one in India, and one in South Korea. Most of the US studies were part of large cohort studies, with four studies relying on samples drawn from the Study of Osteoporotic Fractures cohort, three studies from the Osteoporotic Fractures in Men Study cohort, and two studies from the Atherosclerosis Risk in Communities Study cohort. Twelve studies evaluated sleep using PSG, while six studies used actigraphy (from one to seven consecutive nights), with one study combining actigraphy with continuous frontopolar EEG signals monitoring (Sleep Profiler headband) for six nights. Regarding sleep parameters, 15 studies investigated at least one parameter of sleep macrostructure at baseline, nine studies investigated indices of SDB at baseline, and four studies considered sleep microstructure at baseline.
Characteristics of the 17 included studies
3MS, Modified Mini Mental State Examination; AD, Alzheimer’s disease; AHI, apnea-hypopnea index; APOE, apolipoprotein E; Aβ, amyloid-β; BMI, body mass index; CAP, cyclic alternating pattern; CDR, Clinical Dementia Rating; CSF, cerebrospinal fluid; CVD, cardiovascular disease; DSST, digit symbol substitution test; DWR, delayed word recall; EEG, electroencephalography; ESS, Epworth Sleepiness Scale; FU, follow up; IADL, instrumental activities of daily activities; LWEP, long wake episodes; MCI, mild cognitive impairment; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; NREM, non-REM; ODI, oxygen desaturation index; OSA, obstructive sleep apnea; PACC, Preclinical Alzheimer Cognitive Composite; PET, positron emission tomography; PLMI, periodic leg movement index; PSG, polysomnography; PSQI, Pittsburgh Sleep Quality Index; REM, rapid eye movement; SDB, sleep-disordered breathing; SE, sleep efficiency; SL, sleep latency; SWA, slow wave activity; TIA, transient ischemic attack; Trails B, Trail Making Test - Part B; TST, total sleep time; WASO, wake after sleep onset; WFT, word fluency test; y, year. aOnly longitudinal findings concerning the MCI group were reviewed.
Sleep macrostructure and cognitive decline
Sleep quality and cognitive decline
Objective sleep duration (total sleep time, TST) and sleep efficiency (SE, a sleep parameter obtained by the ratio between TST and time in bed) were considered as main measures of sleep quality in the included studies. Among the studies evaluating dementia or MCI onset at follow-up as the primary outcome, Carnicelli et al. [77] (n = 19 older adults with MCI) did not find significant differences in baseline PSG-evaluated TST between MCI patients converting to AD and MCI patients not converting to AD at the two-year follow-up, and Djonlagic and colleagues [79] (n = 170 older women) did not observe differences in TST and SE between women diagnosed with MCI or dementia (n = 85) and cognitively stable women (n = 85) at the five-year follow-up. Both the studies by Suh et al. [88] (n = 235 older adults) and by Yaffe et al. [91] (n = 298 older women) did not find any relationship between TST or SE and the prospective risk of cognitive decline, and nor did the study by Pase et al. [86], although the authors reported a trend for higher risk associated with short sleep duration in 321 older adults in their long term investigation with a mean follow-up of 12 years.
Using actigraphy to measure TST and SE over three nights, Diem and collaborators [78] did not observe any relationship between TST and diagnosis of MCI or dementia at follow-up in 1245 older women, whereas lower SE was found to be associated with higher risk of cognitive decline in their study. Lysen et al. [83] reported similar findings, highlighting an association between higher SE (as well as later lights out time, but not TST) and a reduced risk of both all-cause dementia and AD over a long time interval (mean follow-up of 9.5 years) in 1322 cognitively intact older adults.
Studies examining longitudinal changes in cognitive performance as main outcome revealed a different pattern of results. Blackwell et al. [75] found that lower actigraphy-measured SE (but not changes in TST) was associated with a longitudinal decline in general cognitive functioning as measured with the Modified Mini-Mental State Examination (MMSE) and with increased odds of clinically significant decline in executive function as measured with Trail Making Test Part B (TMT-B) in their sample of 2822 older men. Similarly, in the actigraphy-based study by McSorley et al. [84], lower percentage of sleep (an actigraphic estimation of SE) and sleeping less than six hours were both associated with increased risk of cognitive decline at the five-year follow up in 555 older adults, as indicated by a significant decrease in Montreal Cognitive Assessment scores. In a well-conceived study using actigraphy and continuous single-electrode EEG monitoring (Sleep Profiler headband) to model non-linear associations between sleep and cognitive performance in a cohort of 100 elderly subjects, Lucey et al. [80] observed an inverse U-shape relationship between TST and longitudinal trends in scores of a neuropsychological tests battery over five assessment visits, with subjects with low (<4.5 h) or high (>6.5 h) TST showing a worse cognitive performance than subjects with medium sleep duration. This association was not replicated for SE. Conversely, Menon et al. [85] did not find any associations between PSG-evaluated TST and SE and longitudinal trends in general cognitive functioning and verbal memory using conventional linear models in 37 participants with amnestic MCI.
Finally, Winer and collaborators [92] found that lower SE (but not TST) at baseline predicted higher cerebral Aβ deposition in subsequent PET scans at the four-year follow-up in the only included study investigating the relationship between PSG-measured sleep parameters and longitudinal changes in AD-related biomarkers in 32 healthy elderly.
Sleep impairment, sleep fragmentation, and cognitive decline
Several macrostructural indices of sleep fragmentation were considered in the included studies, such as sleep latency (SL), wake after sleep onset (WASO), arousal index (number of arousals per hour), number of awakenings, and number of long wake episodes. Among the studies evaluating MCI or dementia onset at follow-up as the outcome, Carnicelli et al. [77], Djonlagic et al. [79], Suh et al. [88], and Yaffe et al. [91] did not find any significant associations between baseline polysomnographic measures of sleep fragmentation and subsequent cognitive decline. However, Pase et al. [86] reported that higher PSG-measured WASO (but not SL) was associated with a higher risk of all-cause dementia at follow-up, whereas Diem and colleagues [78] found that actigraphy-measured longer SL (but not WASO) predicted subsequent diagnosis of MCI or dementia. Consistently, Lysen et al. [83] observed that longer SL was associated with a higher risk of developing all-cause dementia and AD at follow-up, and that longer WASO was solely associated with higher AD risk (although these associations were possibly limited to APOE ɛ4-negative participants in their sample).
Concerning longitudinal changes in cognitive performance, the actigraphic study by Blackwell et al. [75] reported that higher WASO and a higher number of long wake episodes were both associated with clinically significant decline in executive function at follow-up, and that having eight or more long wake episodes was also associated with significant decline in general cognitive functioning. Similarly, McSorley et al. [84] found that both actigraphy-measured WASO and sleep fragmentation (an actigraphy estimation given by the sum of the percentage of recorded epochs with any movement and the percentage of immobile periods of less than one minute) were associated with a significant decline in cognitive functioning in cognitively intact older adults. Conversely, Lutsey et al. [81] (n = 966 older adults) and Menon et al. [85] reported no significant associations between PSG-measured WASO and arousal index and longitudinal change in cognitive test scores.
Finally, the PSG study by Winer et al. [92] found no relationship between WASO and cerebral Aβ accumulation at follow-up.
Sleep stages and cognitive decline
Out of eight studies included in this review reporting data about sleep stages, five [79, 92] used Rechtschaffen and Kales (R&K) sleep scoring criteria [93], whereas the remaining three [77, 87] used the American Academy of Sleep Medicine (AASM) scoring manual [94]. However, in the PSG studies using R&K criteria, Non-REM (NREM) sleep phase 3 and phase 4 were reported jointly as slow wave sleep (SWS), which will be considered equivalent to AASM stage N3 from here on. In all these studies, time (absolute or percentage of TST) spent in each sleep stage was considered along with additional parameters including REM latency, number of REM periods. A single study [88] also considered specific parameters related to NREM/REM cycles.
Regarding the studies investigating MCI or AD diagnosis at follow-up as the main outcome, Carnicelli et al. [77] did not find any differences in baseline sleep stages between MCI patients converting to AD and MCI patients not converting to AD, and neither did Djonlagic and collaborators [79] when comparing older women developing MCI or dementia at follow-up with cognitively stable counterparts. In contrast, Pase et al. [86] observed that a lower percentage of REM sleep was associated with increased risk of both all-cause dementia and AD dementia (although the relation with AD incidence was found solely for APOE ɛ4 carriers in adjusted analyses), and that higher REM latency was also associated with higher risk of dementia. Lastly, Suh et al. [88] found no associations between sleep stages and dementia at follow-up, but they observed that shorter mean duration of NREM/REM cycling and shorter mean time spent in NREM and REM sleep per cycle were all associated with increased risk of developing MCI or dementia.
Among the studies considering longitudinal change in cognitive performance as the primary outcome, Menon et al. [85] did not find any relation between sleep stages and longitudinal trends of general cognitive functioning and verbal memory. Conversely, Song et al. [87] found that participants spending more time in stage NREM 1 (N1) showed a greater decrease in executive and general cognitive functioning scores (as measured with the TMT-B and the Modified MMSE, respectively) at follow-up in comparison to those with the shortest amount of time in N1 in a sample of 2,601 cognitively intact older men; additionally, participants with the least amount of REM sleep exhibited more pronounced worsening in general cognitive functioning compared with those with higher REM sleep duration. In the same study, higher % N1 also predicted clinically significant decline in executive function (as measured with TMT-B). Additionally, in their study employing non-linear statistical methods, Lucey and collaborators [80] observed that both low and high amounts of time spent in NREM sleep (N2 and N3 stages only) and REM sleep were associated with worse cognitive performance over time (in an inverse-U relationship, with cognitive performance remaining stable over time within the middle range of time in NREM and REM sleep), although these findings should be considered with caution given that sleep stages were evaluated solely on the basis of single-channel EEG recording in this study.
Pertaining to the longitudinal evaluation of AD-related biomarkers, Winer et al. [92] did not find any associations between sleep stages and cerebral Aβ accumulation at follow-up in their study.
Sleep microstructure and cognitive decline
Of the four studies that evaluated microstructural sleep parameters, one [77] examined cyclic alternating pattern (CAP), and three [79, 92] used EEG spectral power analysis across conventional frequency bands. In the two studies that evaluated dementia onset as the primary outcome, Carnicelli et al. [77] found lower total CAP rate (CAP time over total NREM sleep time), lower CAP rate in stage N1, and lower CAP rate in stage N2 (i.e., decreased sleep instability) in MCI patients who later converted to AD, compared to MCI patients who did not convert to AD (with no significant differences in other evaluated CAP parameters). Since CAP rate represents a marker of sleep instability, with high values corresponding to lower sleep quality [48, 95], these results point out that patients with preclinical AD may show reduced sleep instability, somewhat in contradiction with the previously reported physiological increase of CAP rate in aging [96]. On the other hand, Djonlagic and colleagues [79] observed higher alpha and theta power in NREM sleep and higher alpha and sigma power in REM sleep in women who developed MCI or AD at follow-up, compared to women remaining cognitively intact. Baseline alpha power and theta power significantly predicted subsequent cognitive decline, and higher slow oscillations (0.5–1 Hz), slow wave activity (SWA, 0.75–4.5 Hz), and theta frequencies were associated with decline in Short MMSE (measuring general cognitive functioning), whereas there were no relations between quantitative sleep EEG parameters and decline in TMT-B (measuring executive functions).
Lucey and colleagues [80] found significant non-linear associations between longitudinal changes in cognitive functioning and NREM SWA, with worse cognitive performance over time at low and high values of both <1 Hz and 1–4.5 Hz NREM SWA. However, it should be noted that SWA spectral power across these two bands was the only microstructural parameter evaluated in this study, and that EEG data were obtained from a single electrode continuous recording over the study period.
Lastly, Winer et al. [92] found that a lower proportion of SWA between 0.6 and 1 Hz measured at frontal electrodes during baseline PSG was associated with increased cerebral Aβ accumulation in their sample. Furthermore, negative associations were observed between EEG power of frequency bands between 0.6 Hz and 1 Hz and brain Aβ deposition at follow-up, whereas power at frequencies from 1 Hz to 6 Hz was positively associated with cerebral Aβ accumulation in this study. No relations were found between NREM sleep spectral power at higher frequencies (6–40 Hz), total SWA (0.8–4.6 Hz), or REM sleep spectral power at any frequencies (0.6–40 Hz) and the rate of Aβ change.
Circadian sleep-wake rhythm dysregulation and cognitive decline
Although six studies included in this analysis used actigraphy (from one to seven consecutive nights) to monitor participants, only Tranah et al. [89] analyzed specific actigraphic parameters of the circadian sleep-wake cycle in 1,282 healthy older women from the Study of Osteoporotic Fractures cohort. In particular, they considered activity rhythm parameters including amplitude (i.e., the strength of the rhythm measured as the peak to nadir difference), mesor (the mean level of activity), robustness of the circadian activity rhythm, and acrophase (i.e., the time of day when peak activity is reached) and found that women in the lowest quartiles of amplitude and rhythm robustness were more likely to develop dementia or MCI at the five-year follow-up. An increased risk of dementia or MCI was also found to be related to a delayed time of peak activity.
Consistently, Lysen et al. [83] reported an association between later lights out time and a reduced risk of both all-cause dementia and AD.
SDB and cognitive decline
Of the nine studies included in this review that evaluated indices of SDB, two [80, 87] did not address the association between SDB and cognitive decline. Several indices of SDB were considered, including the apnea-hypopnea index (AHI, i.e., the number of apnea and hypopnea events per hour of sleep), the oxygen desaturation index (ODI, i.e., the number of desaturation episodes per hour of sleep), the percentage of sleep time with oxygen saturation below 90%, and the percentage of sleep time spent in apnea or hypopnea.
Among the studies evaluating the relationship between SDB and the prospective risk of developing MCI or dementia, Djonlagic and colleagues [79] did not find any significant difference in baseline measures of SDB between women who developed MCI or dementia at follow-up and those who did not, and Pase et al. [86] observed no association between AHI and dementia risk in their sample. Lutsey et al. [82] reported that severe sleep apnea (i.e., AHI ≥30) was associated with increased risk of MCI or all-cause dementia over a long time interval (median follow-up of 14.9 years) in their large cohort of older adults (n = 1,667) when compared to subjects with no significant sleep apnea events, but this effect was attenuated when the model was adjusted for comorbidities and behavioral risk factors. No association was found between high percentage of sleep time with oxygen saturation <90% and dementia risk in this study. On the other hand, Yaffe and colleagues [91] observed that presence of SDB (i.e., AHI ≥15) and two indices of hypoxia (ODI ≥15 and percentage of sleep time spent in apnea or hypopnea >7%) were associated with an increased risk of developing MCI or dementia in their sample of older women drawn from the Study of Osteoporotic Fractures cohort.
When longitudinal changes in cognitive performance were investigated, Blackwell et al. [76] did not find any significant associations between SDB parameters and clinically significant decline in executive and general cognitive functioning in a sample of 2636 older men, although higher ODI and higher sleep time with oxygen saturation <90% at baseline predicted a greater decline in Modified MMSE between baseline and follow-up visits. In the study conducted by Lutsey et al. [81], no associations were observed between OSA severity and changes in cognitive tests assessing verbal memory, verbal fluency, attention and visuo-perceptual functions over up to 15 years of follow-up. Finally, Menon et al. [85] found no associations between OSA severity and longitudinal trends in general cognitive functioning and verbal memory over three years.
DISCUSSION
This systematic review explored the longitudinal relationship between objectively-measured sleep parameters and cognitive decline with the aim of evaluating the clinical potential of using objective sleep parameters as early biomarkers of neurodegeneration.
Overall, the studies included in the qualitative synthesis reported heterogeneous findings, with both clear patterns and inconsistencies.
Pertaining to sleep duration and sleep quality, poor sleep efficiency was consistently associated with cognitive decline (broadly defined as dementia onset and worsening in cognitive performance) and even with Aβ accumulation; similar but less sharp results were reported for sleep duration, with short or long total sleep time seemingly predicting more pronounced cognitive decline. Indeed, according to the reviewed evidence, a medium amount of sleep (∼7 hours, i.e., the amount of sleep for healthy adults recommended by the AAMS [97]) might be a protective factor against cognitive decline in comparison with either shorter or longer sleep. On the other hand, measures of sleep fragmentation such as wake after sleep onset, sleep latency, long wake episodes, and nigh-time activity were also linked with higher dementia risk and higher decline in cognitive test scores over time. Interestingly, for both sleep quality (i.e., sleep efficiency and total sleep time) and sleep fragmentation, these findings were mostly observed in studies using actigraphy rather than PSG. In fact, actigraphy permits to monitor more nights of sleep and thus can broaden the significance of the results. However, the sleep-wake cycle parameters were seldom analyzed despite the actigraphic recording permits the quantification of the circadian rhythm activity parameters [98].
Regarding the findings on sleep macrostructure obtained with PSG, sleep stages’ duration, reduced REM sleep, increased light sleep (stage N1), lower mean duration of NREM/REM cycles, and low and high amounts of both REM and NREM sleep were all found to be associated with cognitive decline over time, although these relations were not consistent across the reviewed studies. Among microstructural sleep parameters, lower cyclic alternating pattern rate, higher alpha and theta power in NREM sleep, and higher alpha and sigma power in REM sleep were associated with cognitive decline, but again with several inconsistencies across studies. Most studies did not find any relationship between slow wave sleep and cognitive decline, but in the work of Djonlajic et al. [79] higher slow oscillations (0.5–1 Hz range) and higher slow wave activity (1–4.5 Hz range) predicted a more pronounced decline in cognitive test scores in older women. Conversely, another study [80] pointed out that both low and high amounts of slow oscillations and slow wave activity might predict worse cognitive performance over time, and a lower proportion of slow oscillations was also found to predict higher Aβ accumulation in cognitively unimpaired subjects [92], consistently with the purported role of slow wave activity in glymphatic clearance [99].
Lastly, the evidence on SDB was conflicting: some studies reported associations between baseline apnea severity and oxygen desaturation and subsequent cognitive decline, while others failed to detect such relation.
As a whole, these findings mostly converge with those reported in previous studies [49–51, 55], and reinforce the evidence supporting the pathogenetic role of sleep disruption in neurodegeneration [33, 43], highlighting the suitability of sleep biomarkers for predicting cognitive decline.
However, it should be pointed out that the evidence base for these claims is still limited. Indeed, the studies included in this review were highly heterogeneous in research designs and employed methodologies, with a high variability in sample sizes (from 19 up to 2,822 participants), follow-up times (from one year up to 15 years), type and duration of sleep evaluations, and reported outcomes. Contradictory findings on the same sleep parameters were observed in studies with large samples, whereas results regarding sleep microstructure were mostly obtained in small samples. Such heterogeneity and shortcomings limit the generalizability of the reported associations and prevented us from conducting a meta-analysis on the reviewed studies. In order to pursue high-quality and meaningful clinical evidence, the establishment of consensus guidelines on feasible sleep measures and adequate follow-up intervals to detect specific relations would be highly warranted.
Moreover, future studies should further address some critical points. In particular, the relation between cyclic alternating pattern and cognitive decline was only investigated in a small study with unexpected results indicating that a higher sleep stability is associated with longitudinal cognitive decline in patients with MCI [77]. Since this promising microstructural sleep feature showed a clear relationship with development and aging [96], additional research on its possible role as a biomarker for neurodegeneration is needed. Furthermore, no studies considered sleep spindles characteristics and the coupling between spindles and slow wave oscillations as possible predictors of cognitive decline despite cross-sectional evidence highlighted that sleep spindles might be altered in both MCI and AD [49, 51] and that the age-related impairment in slow wave-spindle coupling could threaten memory consolidation [100]. On the other hand, the association between SDB and cognitive decline was not fully supported by the studies included in this review despite robust evidence endorsing the implication of sleep apnea and hypoxia in the mechanisms leading to neurodegeneration and dementia [26, 101]. Forthcoming studies might explore this complex relation in more selected samples (e.g., middle-aged adults) with adequate research designs, possibly evaluating relevant mediators and moderators. Finally, more research exploring the association between sleep parameters and well-established biomarkers of cognitive decline such as Aβ in both cognitively unimpaired individuals and in subjects with SCD is needed to better understand the reliability and sensitivity of sleep-related biomarkers and their clinical feasibility.
Recent efforts towards the integration of data from large cohort studies such as the Sleep and Dementia Consortium [102] might represent a viable solution to overcome these shortcomings and provide more reliable and robust evidence. Considering the importance of building such large databases, the use of practical, non-invasive instruments to measure sleep-wake rhythms and to assess sleep disorders should be taken into account and included in the screening for cognitive decline and dementia. Notably, the most consistent findings in this review were those obtained within actigraphic studies linking reduced sleep efficiency and greater sleep fragmentation to the risk of subsequent cognitive decline. Actigraphy has been acknowledged as a central assessment tool in clinical sleep medicine [98, 103], and actigraphy-based estimates of sleep parameters were shown to be reliable and comparable to those obtained with PSG [104–106]. Furthermore, actigraphic monitoring provides ecologically valid measures with continuous recording over multiple days and nights and hence might be particularly effective for studying sleep, activity patterns, and circadian rhythms in older people and patients with dementia, for whom adherence to PSG instructions might be more difficult [26, 107]. Indeed, the role of circadian rhythms in neurodegenerative diseases deserves consideration [108, 109], and two studies included in this review showed an association between delayed circadian patterns and subsequent dementia [83, 89]. Although other recent works have started using actigraphy to investigate parameters of circadian activity patterns predicting cognitive impairment [110, 111], future research might combine actigraphic recordings with continuous EEG monitoring performed with wearable devices, as shown in the study by Lucey et al. [80], in order to comprehensively evaluate the association between sleep-wake patterns and cognitive decline.
In any case, these lines of evidence support the link between disruption of objectively-measured sleep parameters and risk of subsequent cognitive impairment, and encourage the development of preventive interventions targeting sleep quality. Beyond direct treatment of sleep disorders co-occurring with AD [112], both low intensity and structured non-pharmacological strategies to enhance sleep such as psychoeducation, sleep hygiene, behavioral interventions, cognitive-behavioral treatment, and slow wave activity enhancement [113–118] might represent a valuable asset in primary and secondary prevention of cognitive decline in the elderly population. Moreover, continuous positive airway pressure is considered the gold standard treatment for SDB and has the potential to completely restore sleep and protect against the detrimental effects of this sleep disorder on brain health [119–121]. Given the suitability of these interventions to be broadly delivered in a stepped-care fashion [122, 123] and their potential to improve sleep quality [124–126], these approaches could yield a cost-effective reduction in the risk of dementia and related neurodegenerative conditions. Their effectiveness for this purpose should be investigated in controlled studies, which might evaluate cognitive status as proximal and long-term outcome of sleep enhancement interventions and treatments.
In conclusion, the beneficial effects of sleep have been widely discussed and the recent insights into the glymphatic system’s dynamics further reinforce the importance of ensuring sleep quality and continuity for preventing cognitive decline and neurodegeneration [43, 128]. Sleep fragmentation emerges as a plausible biomarker for early identification of individuals at risk for cognitive decline. Nevertheless, further research is needed to thoroughly characterize sleep fragmentation and to establish a viable marker for its detection, with actigraphy being a potential avenue due to its versatility in monitoring sleep and circadian rhythms over extended periods.
Footnotes
ACKNOWLEDGMENTS
The authors thank the Italian scientific society SIN-dem, Dr. Biancamaria Guarnieri, Dr. Michelangelo Maestri, and Prof. Enrica Bonanni for their support in shaping the research strategies focused on assessing the interplay between sleep disorders and dementia, and for the organization of the webinar titled “Ruolo del sonno nella neurodegenerazione della Malattia di Alzheimer”, which provided the opportunity to delve into the relevant literature pertaining to the scope of this review.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
Matteo Carpi, Mariana Fernandes, and Nicola Biagio Mercuri have no conflict of interest to report.
Claudio Liguori is an Associate Editor of this journal but was not involved in the peer-review process nor had access to any information regarding its peer-review.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article. The corresponding author can be contacted for further inquiries.
