Abstract
Background:
Mild behavioral impairment (MBI) and loneliness are associated with cognitive decline and an increased risk of dementia.
Objective:
Our aim was to examine the validity of the Japanese version of the MBI checklist (MBI-C) and investigate the relationship between loneliness and MBI.
Methods:
The participants in this cross-sectional study included 5 cognitively normal persons and 75 persons with mild cognitive impairment. MBI-C and the revised University of California at Los Angeles loneliness scale (LS) were used to assess MBI and loneliness, respectively. Diagnostic performance of MBI-C was examined using receiver operating characteristic analysis. The relationship between MBI-C and LS was examined using multiple linear regression in 67 subjects who were assessed with both scales, with MBI-C total or domain score as the dependent variable and LS as the independent variable, adjusted for age, gender, living situation, presence of visual and hearing impairment, and Mini-Mental State Examination score.
Results:
Per the Youden index, in this mostly MCI sample, the optimal MBI-C cut-off score was 5.5 with sensitivity 0.917 and specificity 0.949. In multiple linear regression analysis, LS score was detected as a significant predictor of MBI-C total scores, and MBI-C decreased motivation, affective dysregulation, and abnormal thought and perception scores.
Conclusions:
The caregiver-rated Japanese MBI-C has excellent diagnostic performance. Loneliness is associated with a greater MBI burden, especially in the decreased motivation, affective dysregulation, and abnormal thought and perception domains. Interventions for loneliness in older people may have the potential to improve MBI.
Keywords
INTRODUCTION
Mild behavioral impairment (MBI) is characterized by the emergence of significant and persistent neuropsychiatric symptoms at ≥50 years of age [1]. While MBI is distinct from dementia, it can be identified in individuals with cognitive normality, subjective cognitive decline, and mild cognitive impairment (MCI) [1]. Importantly, while the symptoms of MBI are not a result of psychiatric illnesses, such illnesses can be complicated by MBI [2]. MBI has been proposed as a risk factor for dementia and may be an early marker of dementia in some people [1]. Indeed, MBI is associated with cognitive decline and incident dementia [3–7]. Neuroimaging studies support MBI as a prodromal marker due to associations with regions affected early in the disease course [8]. MBI comprises five domains of decreased motivation (apathy), affective dysregulation (mood/anxiety symptoms), impulse dyscontrol (agitation, impulsivity), social inappropriateness (impaired social cognition), and abnormal perception or thought content (psychotic symptoms). Affective dysregulation and impulse dyscontrol are most common among these domains [9]. While each MBI domain is associated with a significantly greater risk of dementia, this risk is highest for abnormal perception or thought [10, 11], intermediate for decreased motivation [12], and lowest for affective dysregulation [13].
Loneliness has also been examined as a risk factor for dementia, related to cognitive impairment [14, 15] and greater risk [16–18]. A neuroimaging study suggested that loneliness in older people may be linked to atrophy of the left hippocampus, left entorhinal cortex, and bilateral thalamus [19], while loneliness in cognitively normal older people has been associated with cortical amyloid burden [20, 21] and tau burden in the right entorhinal cortex [21]. Loneliness in older people is also associated with psychiatric symptoms, including depressive symptoms [22–28], anxiety [24], hallucinations [26, 29], and delusions [26].
Previous studies have shown associations of MBI with activities of daily living [30], frailty [31, 32], diabetes mellitus [33], low vitamin D [34], high serum triglyceride [34], and hearing impairment [35, 36]. However, the relationship of MBI with loneliness has not been examined. Since both MBI and loneliness are linked to dementia and loneliness is associated with psychiatric symptoms, loneliness may be related to MBI. The aim of this study was to examine the validity of a Japanese version of the MBI checklist (MBI-C) and investigate the potential link between MBI and loneliness using MBI-C.
METHODS
Participants
This cross-sectional study was conducted at the Center for Diagnosis of Dementia at Kyoto Prefectural University of Medicine. The inclusion criteria were: 1) age 60 and above; and 2) no psychiatric diseases, organic brain diseases, or dementia. Eighty participants were enrolled after providing informed consent, including 5 cognitively normal persons and 75 persons with MCI. The participants included 51 females and had a mean age±standard deviation (SD) of 78.5±6.3 years. MCI was diagnosed using the Petersen criteria [37]. Cognitively normal people were defined as those who visited the Center for Diagnosis of Dementia because of SCD or a caregiver’s concern about the patient’s cognitive decline, but did not meet criteria for MCI or dementia. MBI was diagnosed in accordance with the International Society to Advance Alzheimer’s Research and Treatment (ISTAART) research diagnostic criteria [1]. The diagnoses of MBI, MCI, and cognitively normal were made without knowing the MBI-C scores. These diagnoses were based on clinical assessments that took into account all available data, including medical interviews, neurological examinations, neuropsychological evaluations, blood tests, and neuroimaging studies. The study was approved by the ethics committee of Kyoto Prefectural University of Medicine (ERB-C-842-5).
Assessment
Information was collected for age, gender, and living situation (alone or not alone). As in our previous studies [26, 38], visual or hearing impairments were retrospectively based on descriptions in medical charts, which characterized them as diminished visual or hearing abilities observed during clinical examinations and in daily life. The MBI-C (http://www.MBItest.org) [39] was used to assess MBI. This scale uses 34 items to assess decreased motivation (6 items), affective dysregulation (6 items), impulse dyscontrol (12 items), social inappropriateness (5 items), and abnormal thoughts and perception (5 items). Each item is scored as 0 (absent), 1 (mild), 2 (moderate), and 3 (severe). Therefore, the MBI-C total score ranges from 0 to 102.
The English version of the MBI-C was first translated into Japanese, and then we followed a standard back-translation procedure to ascertain the semantic equivalence of the Japanese version and the original English version [40]. The initial version of the Japanese MBI-C was tested in five participants in the study, after which minor modifications were made to create the final version. These five participants were not included in the final analyses, which were performed in 75 participants (Fig. 1), incorporating caregiver MBI-C ratings.
The revised University of California at Los Angeles loneliness scale (LS) [41, 42] was used to assess loneliness. This scale includes 20 items, with a score of 1 to 4 for each item. Thus, the total score ranges from 20 to 80, and higher scores represent more severe loneliness. Participants self-rated this scale. The Neuropsychiatric Inventory-12 item version (NPI-12) [43, 44] was used as a standard assessment of neuropsychiatric symptoms and was completed by the caregiver. The Mini-Mental State Examination (MMSE) [45] was used to evaluate cognitive impairment.
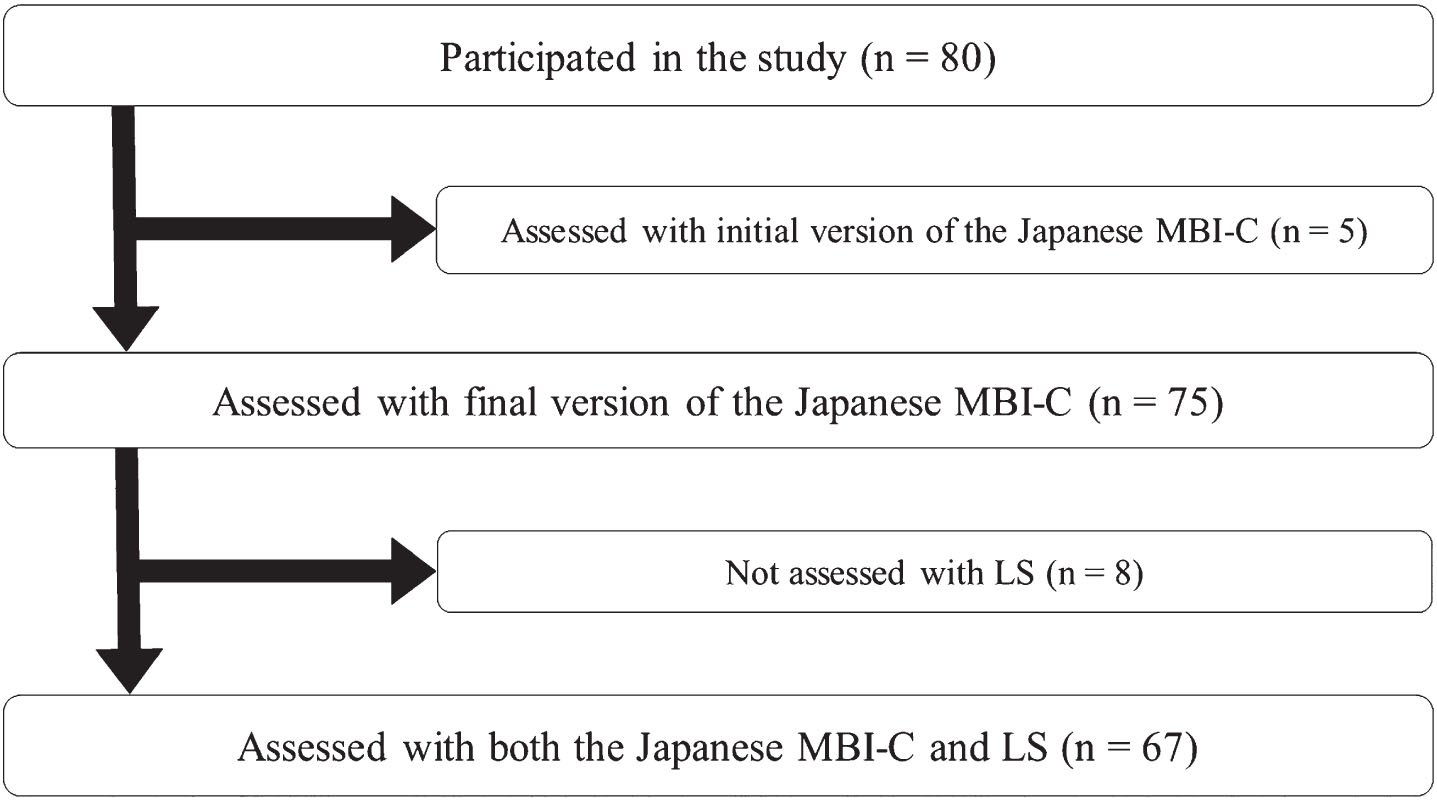
Flow of the study participants. LS, Loneliness Scale; MBI-C, Mild Behavioral Impairment Checklist.
Sample size
The sample size for the correlation analysis was calculated using G*Power 3.1 (Heinrich-Heine-Universitat Dusseldorf, Dusseldorf, Germany) [46, 47] with the following conditions: effect size, 0.3; α, 0.05; 1-β, 0.8. Consequently, 67 participants were needed, and recruitment was stopped when the number of participants with LS scores reached 67. Assessments other than LS were completed in 75 participants (Fig. 1).
Statistical analysis
The validity and reliability of the Japanese version of the MBI-C were first examined in 75 participants. Concurrent validity of the MBI-C was assessed based on the correlation of MBI-C and NPI-12 scores using Spearman correlation analysis. Internal consistency was examined with Cronbach’s alpha coefficient. The diagnostic performance of the MBI-C was evaluated using receiver operating characteristic (ROC) analysis.
The relationship between MBI-C and LS was then examined using Spearman correlation and multiple linear regression in 67 participants. In multiple linear regression analysis, the MBI-C total or domain score was used as the dependent variable and the LS score as the independent variable, adjusted for age, gender, living situation (alone or not alone), presence of visual and hearing impairment, and MMSE score. A normal distribution of residuals was confirmed using a percentage probability (P-P) plot and Durbin-Watson ratio. Statistical analyses were performed using SPSS 29 (IBM Corp., Armonk, NY, USA), with the significance threshold set at a p value of 0.05.
RESULTS
Characteristics of participants
The characteristics of the participants are shown in Table 1. The prevalence of MBI diagnosed by ISTAART criteria was 48.0% in this sample of participants, who were mainly female (64.0%) and about one-third (29.3%) lived alone, and with a cognitive diagnosis of MCI (93.3%). The prevalence of visual impairment was 9.3% and that of hearing impairment was 17.3%.
Characteristics of participants (n = 75)
Data are presented as a number or a mean±standard deviation (SD). LS, Loneliness Scale; MBI-C, Mild Behavioral Impairment Checklist; MMSE, Mini-Mental State Examination; NPI, Neuropsychiatric Inventory.
Validation of the MBI-C
MBI-C total scores were significantly correlated with NPI-12 total scores (ρ= 0.749, p < 0.001). Cronbach’s coefficient alpha was 0.868. The area under the curve (AUC) was 0.970 (95% confidence interval: 0.937–1.004, p < 0.001) (Fig. 2). With the previously published MBI-C cut-off score of 6.5 for MCI, the sensitivity was 0.833 and specificity was 0.949. However, per the Youden index, the optimal MBI-C cut-off score was 5.5 with an AUC of 0.866, sensitivity 0.917, and specificity 0.949.
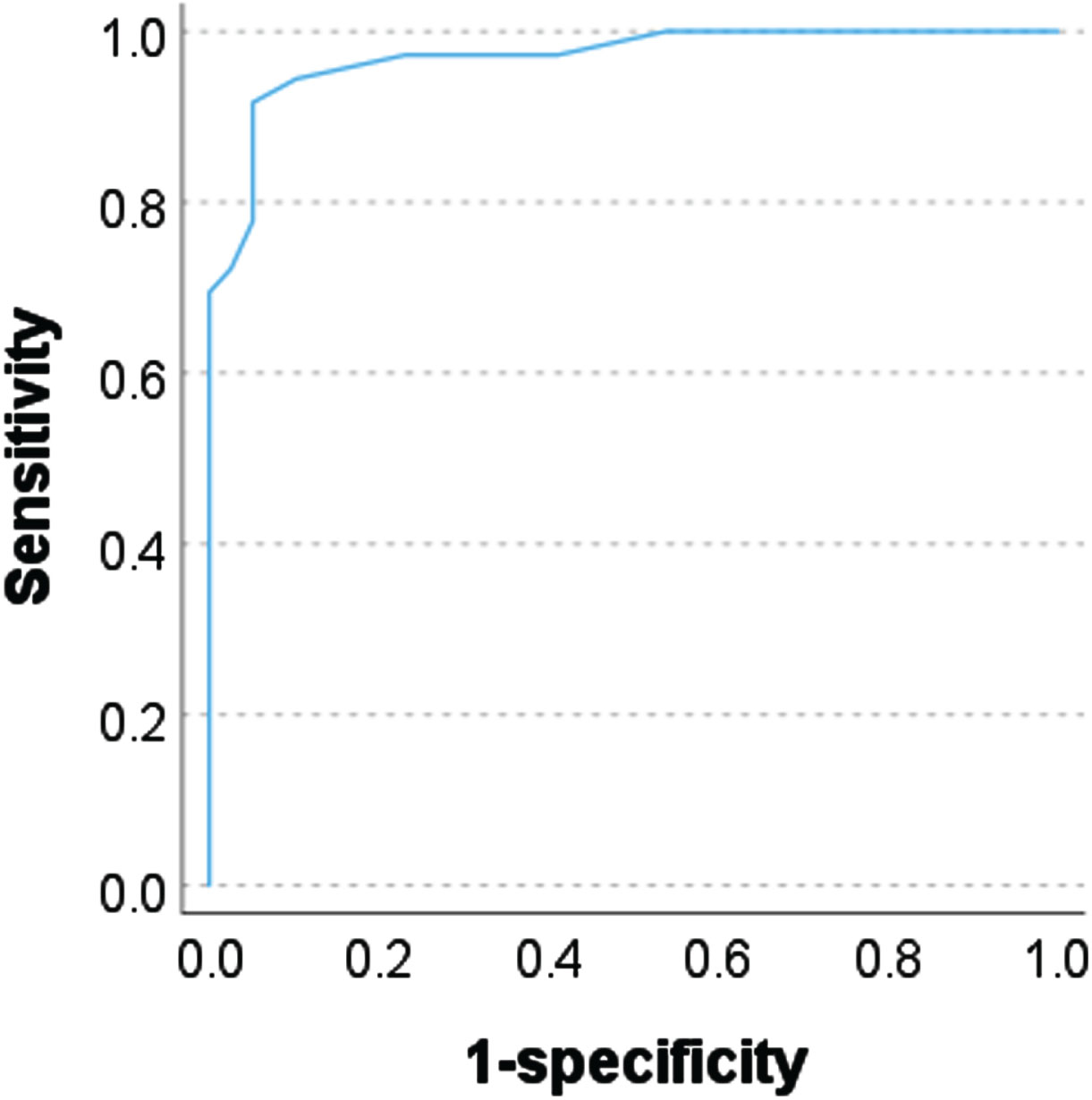
Receiver operating characteristic analysis using the Mild Behavioral Impairment Checklist.
Correlation analyses between loneliness and MBI-C
LS scores were significantly correlated with MBI-C total scores (ρ= 0.366, p = 0.002), and scores for decreased motivation (ρ= 0.292, p = 0.017), affective dysregulation (ρ= 0.307, p = 0.011), impulse dyscontrol (ρ= 0.332, p = 0.006), and abnormal thought and perception (ρ= 0.264, p = 0.031), but not with those for social inappropriateness (ρ= 0.065, p = 0.602).
Multiple linear regression analysis
In multiple linear regression analysis, LS score was identified as a significant predictor of MBI-C total score (adjusted R2 = 0.323, F = 5.508, p < 0.001) (Table 2). Female gender and living alone were also significant variables (Table 2). When each domain score was entered as a dependent variable in multiple linear regression analysis, the LS score was a significant predictor of scores for MBI-C decreased motivation (adjusted R2 = 0.274, F = 4.567, p < 0.001), affective dysregulation (adjusted R2 = 0.312, F = 5.282, p < 0.001), and abnormal thought and perception (adjusted R2 = 0.389, F = 7.000, p < 0.001) (Table 3), but not for scores for MBI-C impulse dyscontrol (adjusted R2 = 0.073, F = 1.742, p = 0.117) and social inappropriateness (adjusted R2 = 0.009, F = 1.088, p = 0.383). There were also significant relationships of MMSE score and age with MBI-C decreased motivation score, and of female gender and living alone with MBI-C affective dysregulation score. Visual impairment, living alone, female gender, and age were also significant predictors of the MBI-C abnormal thought and perception score (Table 3). P-P plots in analyses of MBI-C total, decreased motivation, affective dysregulation, and abnormal thought and perception scores showed that the distribution of residuals was approximately normal. The Durbin-Watson ratios in these analyses were nearly 2, which indicates non-autocorrelation between residuals (1.974, 2.286, 2.061, 1.995, respectively).
Results of multiple regression analysis for MBI-C total scores in persons assessed with the MBI-C and LS (n = 67)
CI, confidence interval; LS, Loneliness Scale; MBI-C, Mild Behavioral Impairment Checklist; MMSE, Mini-Mental State Examination; VIF, variance inflation factor.
Results of multiple regression analyses for MBI-C domain scores
CI, confidence interval; LS, Loneliness Scale; MBI, mild behavioral impairment; MMSE, Mini-Mental State Examination; VIF, variance inflation factor.
DISCUSSION
This study demonstrated the association of loneliness with MBI, and especially in the MBI decreased motivation, affective dysregulation, and abnormal thought and perception domains. Visual impairment was linked to abnormal thought and perception, and cognitive impairment was involved in decreased motivation. Age was also related to decreased motivation and abnormal thought and perception, while female gender and living alone were associated with affective dysregulation and abnormal thought and perception. Thus, although various factors contributed to MBI, loneliness may be among the most important due to the consistency of its effect across analyses.
In this study, loneliness was most involved with MBI-C total scores, and female gender and living alone were also linked to these scores. Loneliness, rather than social isolation, is related to depressive symptoms [48] and dementia onset [16], and these findings indicate the importance of loneliness in MBI. Loneliness is associated with various psychiatric symptoms [22–29], and a relationship between loneliness and depressive symptoms is most commonly reported [22–28]. Associations of loneliness with anxiety [24], hallucinations [26, 29], and delusions [26] have also been found. These findings are consistent with the relationship of loneliness with MBI found in the current study, and especially with the associations with decreased motivation, affective dysregulation, and abnormal thought and perception.
Visual impairment was the strongest predictor of MBI abnormal thought and perception in this study. Visual and hearing impairment have been suggested to not be linked to MBI [49], but other studies have shown involvement of hearing impairment in MBI [35, 36]. There is also a relationship between visual impairment and psychosis in older people [29, 38], and thus, it is possible that visual impairment is involved in abnormal thought and perception in MBI. Assessment of retinal structures using optical coherence tomography suggested that the association of visual impairment with psychosis in older people was due to central nerve system dysfunction, rather than a causal association [50]. Visual impairment may also be a symptom of Alzheimer’s disease [51], although a meta-analysis framed visual impairment as a risk factor for dementia [52]. A recent study reported that the presence of both amyloid and putative Lewy body pathologies may be associated with MBI abnormal thought and perception [53]. Therefore, both MBI abnormal thought and perception, and visual impairment may be early symptoms of dementia. Further studies of the causal relationships between visual impairment and MBI are needed.
In this study, female gender was linked to MBI affective dysregulation and abnormal thought and perception. This result is consistent with previous reports of gender differences in MBI showing that affective dysregulation is more common in females [54], while decreased motivation [32, 55], impulse dyscontrol [54, 55], and social inappropriateness [54] are more common in males. Late-onset psychosis has also been found to be more common in female than male outpatients [38] and community samples [56], but other studies have not found prevalence of MBI psychosis to differ between genders [11]. However, despite our findings that affective dysregulation and abnormal thought and perception are more common in females, the link of loneliness to mild to moderate depression has a stronger association in males [28]. This previous finding is inconsistent with our results showing that affective dysregulation is related to loneliness and female gender, and a further investigation is warranted.
A systematic review showed the effectiveness of intervention for loneliness and social isolation in community-dwelling older people, although the evidence level was low [57]. It has been estimated that removing loneliness could prevent 11–18% cases of depression in people aged 50 years and above [27]. Therefore, it is plausible that intervention for loneliness could improve MBI. Whether MBI symptoms are risk factors or prodromal symptoms of dementia, reducing loneliness might prevent or reduce cognitive decline and improve quality of life. These findings suggest that interventions studies on loneliness are worth exploring.
Several validations of the MBI-C have been published [58], with translations into about 30 languages, including Italian [59], Chinese [60–62], Mandarin [63], Persian [64], French [65], German [66], Czech [67], and Spanish [68]. In differentiating between the presence and absence of MBI, cut-off scores of 6.5 for MCI [69] and 8.5 for SCD [70] have been most commonly used. In one study using a cut-off of 6.5, 50% of people with MCI had MBI [64]. In our study, we demonstrated that the Japanese version of the MBI-C has satisfactory concurrent validity and reliability. The prevalence of MBI in this study was 48.0%, which is consistent with previous studies in MCI showing prevalence of 14.2–85.3% [5, 72]. In our sample, a cut-off of 5.5 optimized sensitivity and specificity to obtain the best discriminative performance of the MBI-C, but sensitivity and specificity were still relatively high at the 6.5 cut-off [69]. Our findings are consistent with a recent validation in Brain Health Registry participants, in which cut-offs as low as >5 discriminated MBI+ from MBI- for poorer memory and executive function [73], although in largely cognitively normal participants. Similarly, an MBI-C cut-off of >5 identified a subgroup from mixed cognitively normal and MCI participants with fMRI changes in the default mode network and salience network in early Alzheimer’s disease [74]. The value of the 5.5 cut-off warrants further examination. Studies have also shown that the prevalence of MBI endorsed by informants was higher than that self-reported by subjects, sometimes with weak correlations [75]. We did not assess these differences, but self-informant differences require exploration in future studies.
There are some limitations in this study. First, the number of participants was relatively small, although the sample size was calculated to be sufficient. Moreover, it was not possible to examine construct validity using exploratory factor analysis due to the small sample size. Second, the prevalence of the MBI social inappropriateness and abnormal thought and perception domains was low, with low mean scores for these domains. Third, visual and hearing impairments were assessed using descriptions on medical charts only. Fourth, the cross-sectional study design cannot determine causality, and the MBI affective dysregulation domain might overlap with loneliness. Fifth, the split-half reliability and repeatability reliability of the Japanese version of MBI-C were not examined. Further studies are needed to overcome these limitations.
Conclusion
Loneliness may exacerbate MBI, and especially the decreased motivation, affective dysregulation, and abnormal thought and perception domains. Interventions for loneliness in older people may improve MBI and a further intervention study is warranted.
CREDIT AUTHOR STATEMENT
Teruyuki Matsuoka (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Writing – original draft); Zahinoor Ismail (Conceptualization; Methodology; Supervision; Writing – review & editing); Ayu Imai (Conceptualization; Investigation; Writing – review & editing); Keisuke Shibata (Conceptualization; Investigation; Writing – review & editing); Kaeko Nakamura (Conceptualization; Investigation; Writing – review & editing); Yukihide Nishimura (Conceptualization; Investigation; Writing – review & editing); Ellen Rubinstein (Conceptualization; Methodology; Writing – review & editing); Hiroyuki Uchida (Conceptualization; Methodology; Supervision; Writing – review & editing); Masaru Mimura (Conceptualization; Methodology; Supervision; Writing – review & editing); Jin Narumoto (Conceptualization; Investigation; Methodology; Supervision; Writing – review & editing).
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
This work was supported by JSPS Grant-in-Aid for Science Research (C) (21K07505).
CONFLICT OF INTEREST
Teruyuki Matsuoka has received speaker’s fees from Takeda, Eisai, Otsuka, MSD, and Sumitomo Pharma; and grants from AbbVie (2023), Otsuka (2023), and Boehringer Ingelheim (2023). Teruyuki Matsuoka is an Editorial Board Member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
Zahinoor Ismail has received advisory board fees from Eisai, Lundbeck, Otsuka, and Roche, and research support from the Canadian Institutes of Health Research Institute of Aging (BCA2633).
Hiroyuki Uchida has received advisory board fees from Lundbeck, Sumitomo Pharma, Takeda Pharmaceutical Company, and Boehringer Ingelheim Japan; speaker’s fees from Eisai, Janssen, Lundbeck, Meiji Seika Pharma, Otsuka, and Sumitomo Pharma; and grants from Daiichi Sankyo (2021), Eisai (2021–2022), Mochida (2020–2022), Otsuka (2020–2022), and Sumitomo Pharma (2020–2022).
Masaru Mimura has received speaker’s honoraria from Biogen Japan, Byer Pharmaceutical, Daiichi Sankyo, Dainippon-Sumitomo Pharma, Demant Japan, Eisai, Eli Lilly, Fuji Film RI Pharma, Hisamitsu Pharmaceutical, H.U. Frontier, Janssen Pharmaceutical, Mochida Pharmaceutical, MSD, Mylan EPD, Nippon Chemipher, Novartis Pharma, Ono Yakuhin, Otsuka Pharmaceutical, Pfizer, Shionogi, Takeda Yakuhin, Teijin Pharma, and Viatris (2020–2022). Dr. Mimura has also received grants from Daiichi Sankyo, Eisai, Fronteo, Shionogi, Takeda, Tanabe Mitsubishi and Tsumura outside the submitted work (2020–2022).
Jin Narumoto has received advisory board fees from Ono, Sumitomo Mitsui Card Company, Unicharm Corporation, Sompo Japan Insurance, Sumitomo Forestry, and Money Forward; speaker’s fees from Eisai, Janssen, Lundbeck, Pfizer, Meiji Seika Pharma, Otsuka, Eli Lilly Japan, Shionogi Pharma, Mylan EPD, Mochida Pharmaceutical, EA Pharma, Viatris, Towa, Yoshitomi Pharmaceutical Company, MSD and Sumitomo Pharma; and grants from Eisai (2020–2022), Otsuka (2020–2022), Shionogi Pharma (2020–2022) and Sumitomo Pharma (2020–2022).
All authors have no conflict of interest to report.
DATA AVAILABILITY
The research data cannot be shared due to patient privacy concerns.
