Abstract
Background:
The associations between neuropsychiatric symptoms (NPSs) and Alzheimer’s disease (AD) have been well-studied, yet gaps remain.
Objective:
We aimed to examine the associations of four subsyndromes (hyperactivity, psychosis, affective symptoms, and apathy) of NPSs with cognition, neurodegeneration, and AD pathologies.
Methods:
Totally 1,040 non-demented elderly (48.07% males) from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) were included. We assessed the relationships between NPSs and AD neuropathologies, cognition, neurodegeneration, and clinical correlates in cross-sectional and longitudinal via multiple linear regression, linear mixed effects, and Cox proportional hazard models. Causal mediation analyses were conducted to explore the mediation effects of AD pathologies on cognition and neurodegeneration.
Results:
We found that individuals with hyperactivity, psychosis, affective symptoms, or apathy displayed a poorer cognitive status, a lower CSF amyloid-β (Aβ) level and a higher risk of clinical conversion (
Conclusions:
NPSs could be strongly associated with AD. The influences of NPSs on cognitive impairments, neurodegeneration might be partially associated with Aβ.
Keywords
INTRODUCTION
Alzheimer’s disease (AD) is characterized by abnormal amyloid deposition, tau phosphorylation and neurodegeneration in pathology, cognitive deficits, and neuropsychiatric symptoms (NPSs) in clinic. In the period of advanced AD (cognitive impairment), it is almost impossible to achieve a satisfactory outcome with medicines and other interventions [1]. Therefore, access to early diagnosis and treatment of AD is especially meaningful. NPSs manifest primarily as emotional, perceptual, and behavioral disturbances associated with neurodegenerative disorders, and the Neuropsychiatric Inventory (NPI) was used to assess the NPSs of the subjects. NPSs appear early in dementia and are linked to an increased risk of AD [2, 3], indicating that NPSs may be promising predictive targets.
Previous studies had demonstrated that the syndrome of apathy was correlated with accelerated cognitive decline and elevated cerebral amyloid-β (Aβ) burden [4]. They also found that Aβ pathology mediated the influences of apathy on cognitive decline and quality of life [4]. However, there is currently little in-depth research in this field or continued exploration of whether this mechanism can be confirmed in other NPSs. Studies also found that hippocampal volume (HV) was smaller in patients with late-life depression relative to healthy comparison subjects [5]. Therefore, whether these effects on brain structures atrophy are also mediated by AD core pathologies needs to be further explored. Here, in non-demented elderly, we aimed to 1) investigate the relationships of NPSs with AD CSF biomarkers, cognition, neurodegeneration, and whether relationships above could be further verified through multi-year follow-up; 2) explore whether AD core pathologies are associated with the effects of NPSs on cognition and neurodegeneration; and 3) explore whether the emergence of NPSs could predict the progression of AD.
METHODS
Participants
The ADNI database (http://adni.loni.usc.edu) provided the data required in this study. It was a multisite databank launched in 2003 and aimed at determining the relationships among clinical, cognitive, imaging, genetic, and biochemical biomarkers across the entire spectrum of AD. This project was authorized by the Institutional Review Board and informed consent was signed in accordance with the Declaration of Helsinki. People absorbed into the database were older adults aged 55 to 90. Their pathological biomarkers, cognitive trajectories, and neurodegeneration were assessed periodically to track disease progression.
In the study, a total of 1,040 non-demented elderly with NPI data and without a history of NPSs or related medication use records were enrolled at baseline. Beforehand, 582 entries were excluded. Of those 582 subjects, 402 subjects diagnosed with dementia, 28 subjects with missing diagnosis, and 152 subjects had a history of NPSs or related medication use records at baseline. We respectively obtained NPI data, AD CSF biomarkers information, 18F-florbetapir-positron emission tomography (AV45-PET) data, global cognition, cognitive domains data, and MR imaging information such as brain structures at baseline and follow-up. They were followed up at least 1 year, ranging from 1 to 8 years (Fig. 1A).
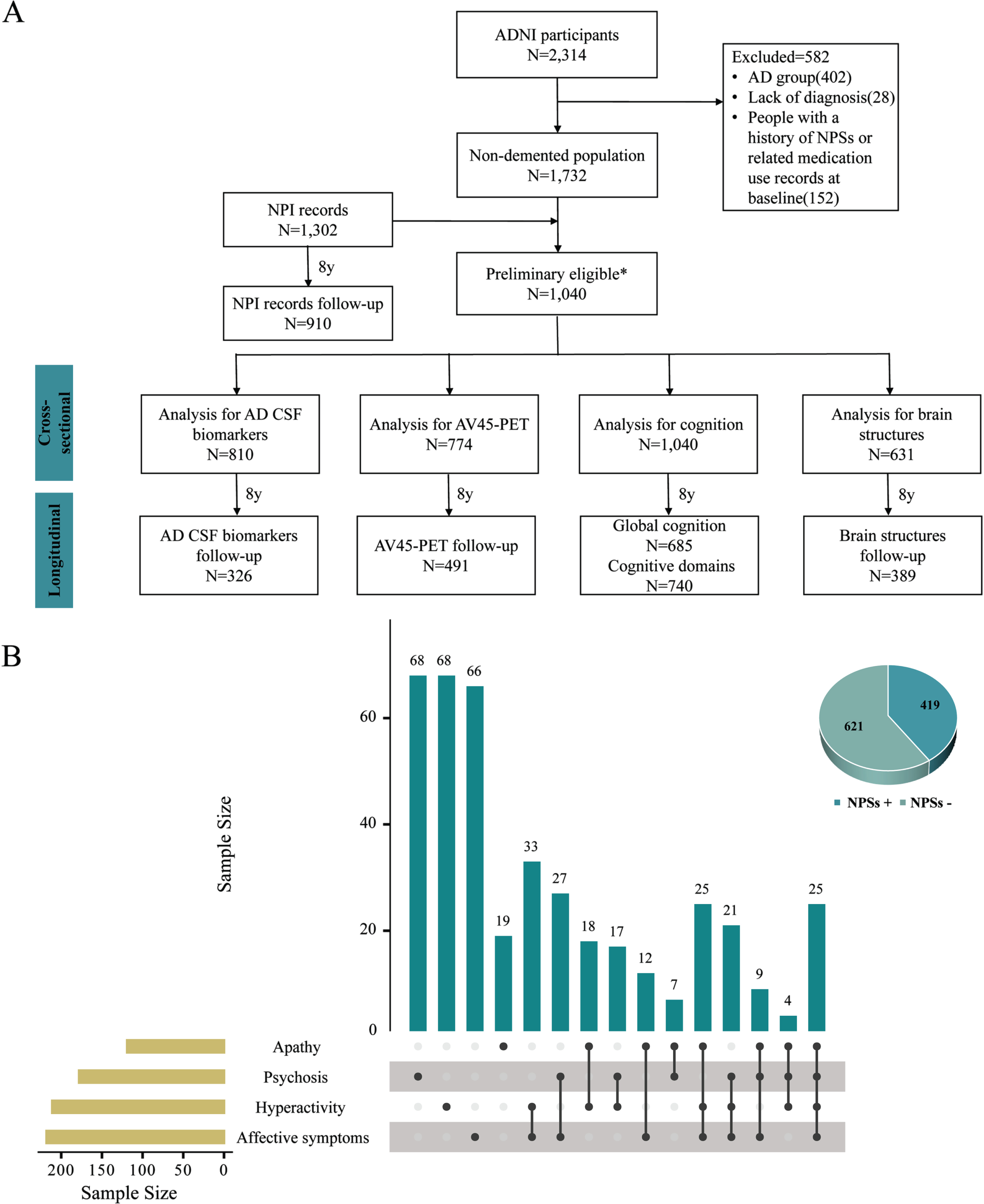
Flow diagram of studied population selection. Data of 2,314 ADNI participants were downloaded. We excluded those who were with a diagnosis of dementia, a lack of diagnosis, and had a history of NPSs or related medication use records at baseline. Ultimately, 1,040 non-demented individuals with NPI data and without a history of NPSs or related medication use records at baseline were eligible for the inclusion criteria of the present study. 810 participants had AD CSF biomarkers information, 774 participants had AV45-PET data, 1,040 participants had cognition information, and 631 participants had brain structures information. After 8 years of follow-up, 910 participants had NPI records, and 326, 491, 740, 389 individuals were respectively included in the longitudinal analysis (A). In the total population, 621 participants were NPSs- and 419 participants were NPSs+. The distribution of NPSs + population with any one subsyndromes of NPSs or more was shown in B. *Non-demented population with NPI records and without a history of NPSs or related medication use records at baseline. ADNI, Alzheimer’s Disease Neuroimaging Initiative; NPSs, neuropsychiatric symptoms; NPI, Neuropsychiatric Inventory; AD, Alzheimer’s disease; CSF, cerebrospinal fluid; AV45-PET, 18F-florbetapir-positron emission tomography.
NPI
NPI was used to measure and quantify NPSs. It included 12 domains which could be divided into four subsyndromes: hyperactivity (euphoria, aggression, irritability, disinhibition and aberrant motor behaviors), psychosis (hallucination, delusion and night-time behavior disturbances), affective symptoms (anxiety and depression), and apathy (apathy, appetite and eating abnormalities) [6]. It included presence, severity, frequency, and caregiver distress [7]. First, frequency was multiplied by severity to calculate the score for each domain, and then scores for each domain were added together to calculate a gross score.
CSF measurements of AD core biomarkers
Lumbar puncture was conducted through L3-L5 intervertebral space, after which CSF was collected, centrifuged, subpackaged, and stocked in polypropylene tubes in a refrigerator at –80°C. The ADNI used the fully automated and highly standardized Roche Elecsys immunoassay to assess AD biomarkers, including total tau (t-tau), phosphorylated tau (p-tau) (181P), and Aβ1 - 42. A single lot of reagents for each of the three measured biomarkers were used (provided in UPENNBIOMK9.csv file available at http://adni.loni.usc.edu) [8]. Levels below 976.6 pg/ml (Aβ42) or above 21.8 pg/ml (P-tau181P) and 245 pg/ml (T-tau) are categorized as positive (A+, T+, N+, respectively), otherwise are classified as negative (A-, T-, N-, respectively) [8]. CSF biomarkers were assessed in 810 subjects at baseline and 326 subjects during follow-up.
APOE genotype
Early morning fasting blood samples were taken, and DNA was extracted from them using the QIAamp DNA Blood Mini Kit (Qiagen, Hilden, Germany). Polymerase chain reaction amplification was performed with the assistance of forward primers 5’-ACGGCTGTCCAAGGAGCTG-3’ (rs429358) and 5’-CTCCGCGATGCCGATGAC-3’ (rs7412). The extracted DNA was genotyped for apolipoprotein E4 (
AV45 PET
The images acquisition process and related data were documented in detail on the ADNI website (http://adni.loni.usc.edu/methods/documents/). In brief, AV45 SUVRs calculated by averaging across four cortical regions (frontal, anterior/posterior cingulate, lateral parietal, lateral temporal) were used for brain amyloid burden, and then divided by the whole cerebellum as reference region. The acquisition of Aβ PET images was in four frames of 5 min each, 50–70 min p.i. for 18F-florbetapir and 90–110 min p.i. for 18F-florbetaben.
MRI structures
Brain structures imaging were collected by using a 1.5-T magnetic resonance imaging (MRI) system with T1-weighted MRI scans using a sagittal volumetric magnetization-prepared rapid acquisition gradient-echo sequence. The software program (https://surfer.nmr.mgh.harvard.edu/) was used to quantify cortical thickness and subcortical volumes. The volume of the ventricles, hippocampus, entorhinal, and mid temporal was extracted as the regions of interest. The atrophy of medial temporal lobe structures, such as hippocampus and entorhinal cortex (EC), preceded the onset of dementia [9–11].
Cognitive assessment
Multiple scales were applied to assess generic cognitive functions and specific cognitive functions, including the global cognition by Mini-Mental State Examination (MMSE), the cognitive section by Alzheimer’s Disease Assessment Scale (ADAS), the Clinical Dementia Rating Sum of Boxes (CDRSB), and the life quality by Functional Activities Questionnaire (FAQ). In addition, neuropsychological assessments were conducted on three cognitive domains (memory [MEM], executive function [EF], and language [LAN]). To identify items that could be viewed as indicators of the three domains, the neuropsychological batteries were used to evaluate these domains [12, 13]. All assessments were carried out at baseline and at annual follow-up.
Statistical analysis
Based on the scoring rules of NPI, we calculated the total score of each subsyndrome separately. We prescribed that scores in any one of the domains of one subsyndrome > 0, the subsyndrome was defined as positive (+), and all domains of one subsyndrome = 0, the subsyndrome was defined as negative (–). Similarly, it was defined as NPSs + if scores in anyone of 12 domains > 0, and it was defined as NPSs- if all domains were 0. Eventually, 621 participants were NPSs- and 419 participants were NPSs+. The distribution of NPSs + population with any one subsyndromes of NPSs or more was shown in Fig. 1B. Data were displayed as mean (standard deviation, SD) or number (percentage, %). To test the difference between two groups, we used Mann-Whitney u test for continuous variables and
Multiple linear regression and interaction analysis
First, we assessed the associations of NPSs’ four subsyndromes (each of the subsyndromes was respectively added to the model as independent variables) with AD CSF pathologies, cognition (cognitive domains and global cognition), and neurodegeneration (brain structures and AV45-PET) (dependent variables) using multiple linear regression (MLR) models. Furthermore, we included an analysis of the interaction between NPSs’ four subsyndromes and
Causal mediation analyses
Next, we performed a mediation analysis with 10,000 bootstrap iterations to explore whether AD core pathologies were associated with the correlations between NPSs and cognitive, neurodegeneration. The linear regression model was fitted according to the method put forward by Kenny and Baron [14] with reference to the detailed method in previous studies [15]. Mediation analysis must meet the following conditions: 1) NPSs were correlated with AD core pathological biomarkers significantly; 2) NPSs were correlated with cognitive indicators or neurodegeneration significantly; 3) AD core pathological biomarkers had a remarkable correlation with cognitive indicators or neurodegeneration; 4) the correlation of NPSs with cognitive indicators and neurodegeneration was attenuated after adding AD core pathological biomarkers (mediators) to the model. Gender, age, education degree, and
Linear mixed effects
Then, linear mixed effects (LME) models were designed for tracking the follow-up correlations between NPSs’ four subsyndromes and AD CSF pathologies, cognition, neurodegeneration. The LME models were employed because they could handle unbalanced and censored data as well as a continuous variable for time [16]. The random intercepts and slopes for time and an unstructured covariance matrix for the random effects were included into the LME models, and the interaction between time (continuous) and the independent variable (hyperactivity+ versus hyperactivity–, psychosis+ versus psychosis–, affective symptoms+ versus affective symptoms–, apathy+ versus apathy–) was regarded as a forecast. The covariates included age, gender, educational level,
Cox proportional hazard models
Finally, the relationships between hyperactivity, psychosis, affective symptoms, apathy, and clinical progression were tested by calculating cumulative incidence using the Kaplan-Meier method. Clinical progression was defined as: diagnosed as AD dementia for baseline CN or MCI individuals (with the end of study time or dropout as censor points). The Cox proportional hazards model was used to assess the hazard ratios (HR) with 95% confidence interval (CI). Additionally, the multivariate cox proportional hazard model adjusted for gender, age, education degree, and
The R software (version 4.1.0), Jamovi 1.6.3, and IBM SPSS Statistics 26 were used for statistical analyses and the preparation of figures. Two-tailed
RESULTS
Participants characteristics
Table 1 summarized the baseline characteristics of participants in ADNI. Totally 1,040 non-demented subjects were enrolled, who were in their late midlife (aged 71.76±6.85), with secondary education level (mean 16.55 years) and undamaged cognition (mean MMSE score = 28.55). Men made up of 48.07% and an
Population characteristics at baseline
Values are mean±standard deviation (SD), or n (% of the group),
Baseline associations of NPSs with cognition, AD CSF biomarkers, and neurodegeneration
After controlling for covariates at baseline, we found that individuals with hyperactivity, psychosis, affective symptoms or apathy displayed worse global cognition, MEM, EF, LAN (e.g., hyperactivity, psychosis, affective symptoms, apathy:
Amyloid pathology might be associated with the relationships between NPSs and cognitive impairments, neurodegeneration
All above findings indicated that NPSs’ four subsyndromes were not only independent risk factors for cognitive decline and HV shrinkage, but also relevant to amyloid pathology. Mediating outcomes indicated that Aβ might be associated with the relationships between hyperactivity, psychosis, affective symptoms, and apathy, with cognitive decline (Fig. 2 and Supplementary Figure 2). In the total samples, hyperactivity, psychosis, affective symptoms, and apathy were remarkably correlated with higher level of CSF Aβ respectively in the first equation. In the second equation, they displayed a remarkable correlation with worse global cognition, worse MEM, EF, and LAN (
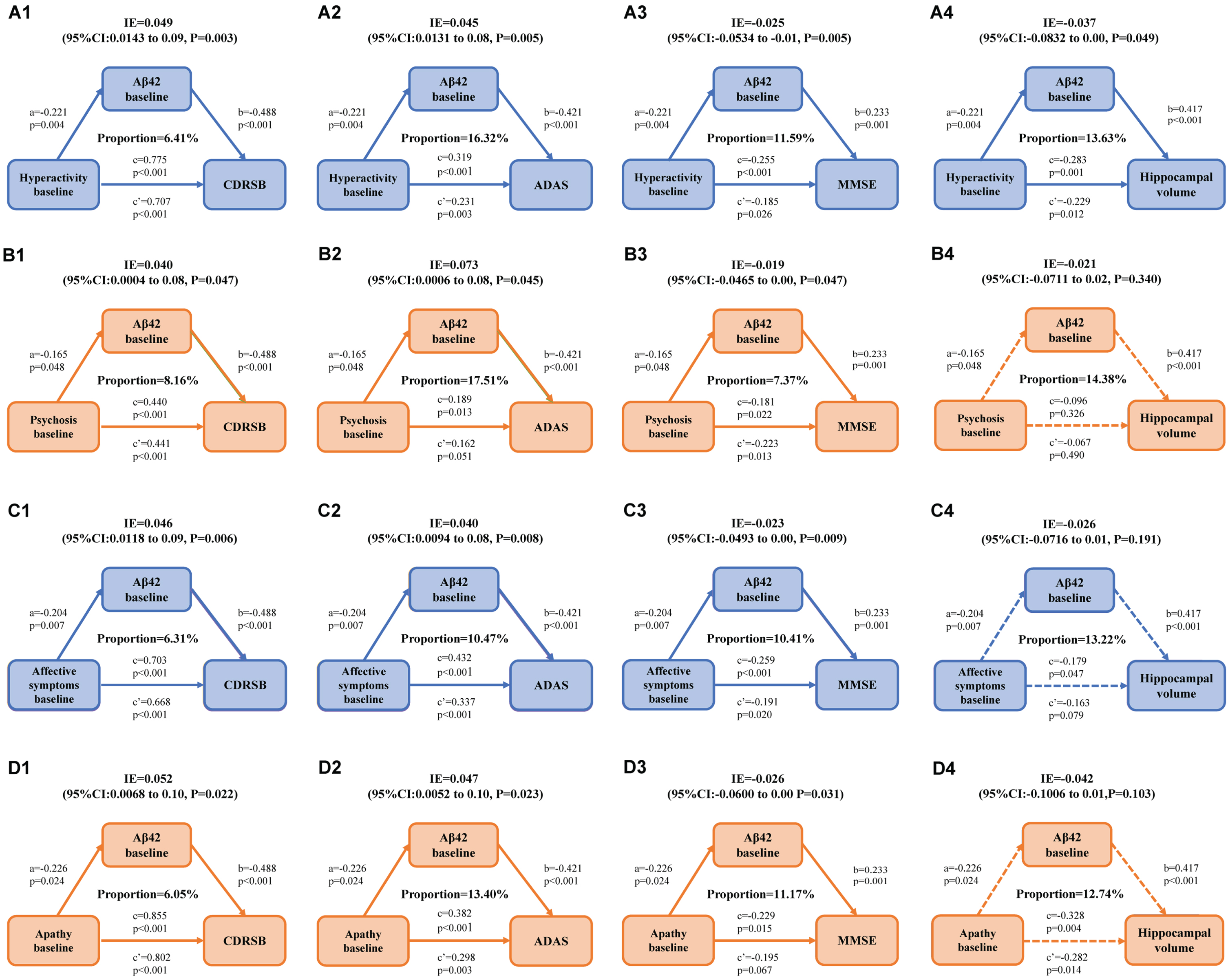
Mediation analyses with CDRSB, ADAS, and MMSE as global cognition outcomes and HV as brain structure outcomes in non-demented elderly. Amyloid-β might be associated with the relationships between hyperactivity, psychosis, affective symptoms, apathy, and global cognition measured by CDRSB (A1, B1, C1, D1), ADAS (A2, B2, C2, D2), and MMSE (A3, B3, C3, D3). Amyloid-β might be associated with the relationships between hyperactivity and HV (A4), but psychosis affective symptoms or apathy were not (B4, C4, D4). a is the effect of independent variable on mediators; b is the effect of mediators on dependent variables after controlling the influence of independent variables; c is the total effect of independent variables on dependent variables; c’ is the direct effect of independent variables on dependent variables after controlling the influence of mediators; IE is the indirect effect of independent variables on dependent variables, in this intermediary model,
Longitudinal associations of NPSs with cognition, AD CSF biomarkers, and neurodegeneration
In this longitudinal study, after controlling for covariates, we found no definite indication that NPSs were connected with a rapid decline in global cognition (
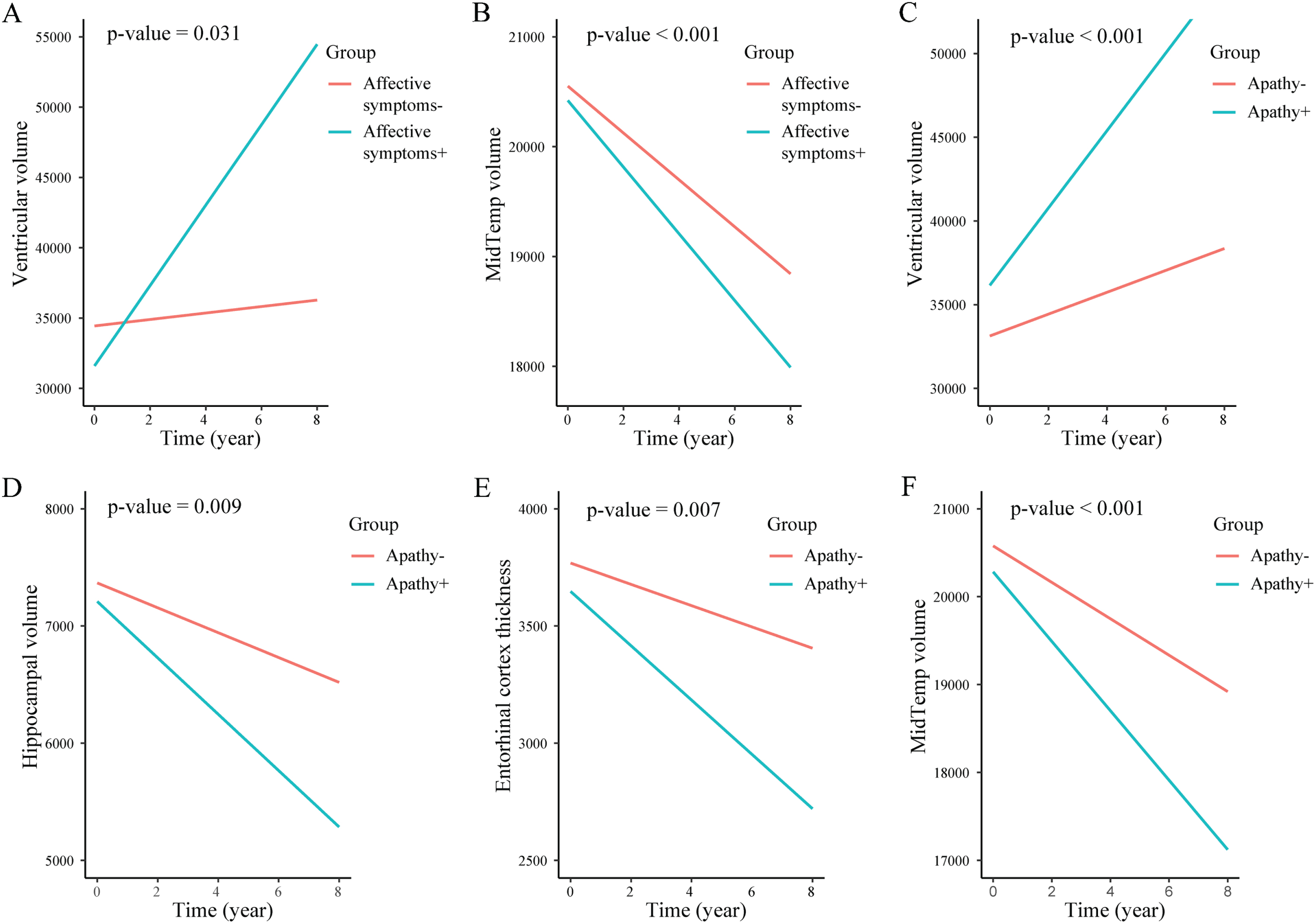
Longitudinal changes of brain structures volume according to NPSs status. Longitudinal changes of ventricular volume and middle temporal region volume in non-demented affective symptoms individuals (A, B). Longitudinal changes of ventricular volume, hippocampal volume, entorhinal cortex volume and middle temporal region volume in non-demented apathy individuals (C, D, E, F). NPSs, neuropsychiatric symptoms; MidTemp, middle temporal region volume.
NPSs predict the risk of AD
The survival curves of four subsyndromes of NPSs and clinical progression were calculated in non-demented participants and were exhibited in Fig. 4. Individuals with hyperactivity, psychosis, affective symptoms, or apathy groups showed higher conversion risks of AD dementia (Supplementary Table 4) in clinical progression, compared with the negative groups. Individuals with hyperactivity (+) presented a progression rate to AD dementia of 21.99%, while hyperactivity (–) individuals presented 8.43% (HR = 2.89, 95% CI = 1.93–4.34,
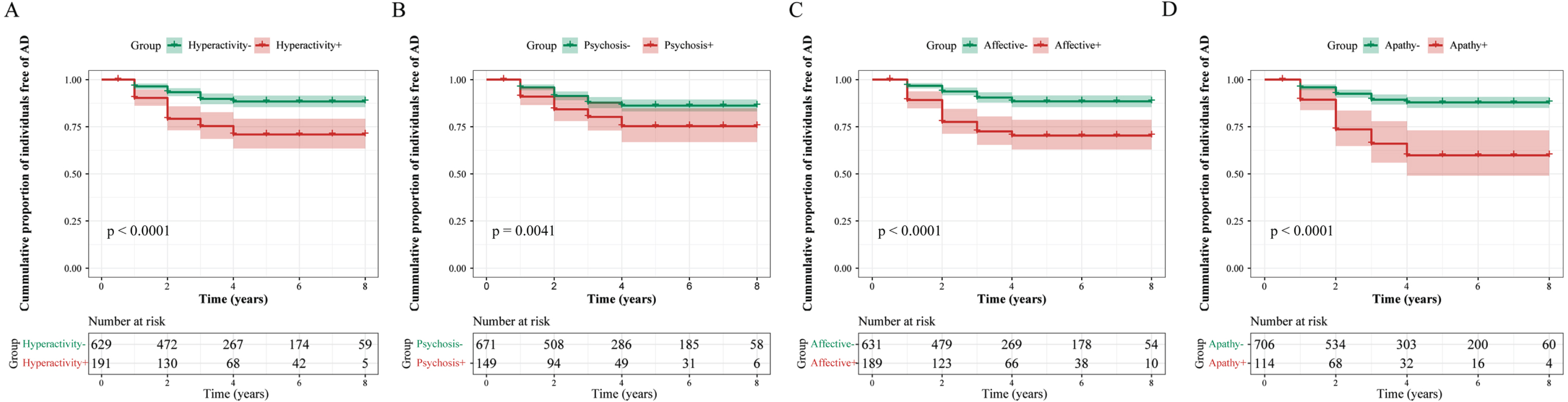
Longitudinal relationships between NPSs and AD risk. Compared with normal participants, those with hyperactivity (A), psychosis (B), affective symptoms (C), and apathy (D) respectively had 2.89 times, 1.92 times, 3.16 times, and 3.71 times risk of developing AD dementia. NPSs, neuropsychiatric symptoms; AD, Alzheimer’s disease.
DISCUSSION
The current research indicated that among elderly without dementia: 1) NPSs’ four subsyndromes were probably significantly associated with the faster cognitive decline, lower CSF Aβ levels, and the atrophy of HV. 2) The relationships between NPSs’ four subsyndromes and cognitive decline, neurodegeneration might be associated with amyloid pathology. 3) NPSs’ four subsyndromes could significantly elevate the risk of AD. Findings above reinforced the close associations of NPSs with amyloid pathology, cognition, and neurodegeneration. These results supported that NPSs could provide essential information for early detection and prevention.
This study contributed multiple aspects of evidence to the relationships of NPSs with AD pathologies and the increased risk of conversion to AD in the crowd of non-demented elderly. Our results are consistent with mainstream studies but seem more comprehensive. Reviewing previous studies, a cross-sectional study in a mixed population found that decreased CSF Aβ levels and increased t-tau and p-tau levels were related to anxiety. Decreased CSF Aβ levels and smaller HV were related to apathy [17–20]. Nevertheless, AD biomarkers were not associated with depression, agitation, irritability, and sleep disturbances [17]. In addition, two cross-sectional studies in patients with MCI [21] and AD [22] showed a relationship between apathy and cortical Aβ deposition. In comparison, a sizable scale study using a combined sample of older adults with CN, MCI, and AD did not find such a relationship [23]. Although follow-up studies detected a positive correlation between lower CSF Aβ level and greater probability of apathy [24], apathy was correlated with Aβ burden [25], and higher Aβ burden at baseline predicted the development of depression-anxiety symptoms [26]. However, no further association was found between NPSs and CSF Aβ levels and Aβ burden in our study with at least one follow-up, ranging from 1 to 8 years. We think it might be explained by differences in study design such as database selection, sample size, population selection, follow-up years, or differences in the measurement of both biomarkers and NPSs.
In the aspect of cognition, researchers had proposed that apathy, anxiety, and depression were regarded as predictors of cognitive decline in individuals without dementia in some earlier studies [25, 28]. Consequently, the improvement of cognition should be a treatment strategy for AD patients dealing with NPSs [29]. Several reports have also shown that the poorer individual’s cognitive status, the more frequent and severe the NPSs will be [30, 31]. In contrast, our study included both broader cognitive aspects (global cognition and cognitive domains) and more comprehensive NPSs (12 types of NPSs which were grouped into four subsyndromes). The findings underscored the link between NPSs and cognition, suggesting that the four subsyndromes of NPSs constituted risk markers for more rapid cognitive decline and conversion to AD. However, to date, only a few studies have explored the effects of NPSs on longitudinal cognition. It remained significant in our 8-year longitudinal study that the relationships between the four subsyndromes of NPSs and functional decline in specific cognitive domains, which including memory, executive and language function. This significant correlation had not been further confirmed in global cognition yet. We considered that the association between NPSs and cognitive performance diminished with longer follow-up, suggesting that NPSs were primarily a precursor to cognitive decline rather than a causal risk factor. The selective withdrawal of people with NPSs might also diminish the long-term relevance.
The hippocampal formation is a key brain structure for learning, memory [32, 33] and stress regulation [34] and is implicated in many neuropsychiatric disorders. The atrophy of HV, especially when combined with the atrophy of EC and temporal neocortical volume [10], are a common morphological abnormality in AD, which has been shown to be an early marker for the presence of AD [35, 36]. Previous and our studies further validated the relationship between baseline NPSs and HV in non-demented individuals [37, 38]. Hypersecretion of glucocorticoids may be a valid explanation [38].
Compared with normal participants, individuals with NPSs were found to have a higher risk of conversion to AD over a maximum of 8 years cohort study. As a prognostic indicator of dementia, NPSs warrant more focus in the elderly. Although many studies have targeted prognostic biomarkers with more advanced and accurate MRI and CSF analyses, the evaluation of NPS still needs to be considered due to its advantages of being less invasive, less costly, and easier to be scaled up [39–41].
Our study further confirmed the predictive relationship between baseline NPSs and amyloid pathologic progression in non-demented elderly. Previous study has shown that the severity of apathy leads to cognitive impairment through the mediating effect of cerebral Aβ burden through a 2-year follow-up [4]. We also confirmed it. Besides, we found the same results for the other three subsyndromes, namely Aβ was associated with the relationships between hyperactivity, psychosis, affective symptoms, apathy, and the impairment of cognition. This is the first comprehensive look at the possible relationship between NPSs and the impairment of cognition through amyloid pathology. We believe that it fills a blank in the existing studies and points the way for future research.
The mechanisms by which NPSs regulating amyloid pathology and amyloid pathology mediating the cognitive impairment as well as neurodegeneration remain obscure. A few of investigators believed that the core amyloid plaques disrupted the dopamine transporter, leading to the impairment of motivation [42]. Previous research had hypothesized that “mood disorders” [43] such as depression can increase neuroinflammatory cytokines and disrupting brain-derived neurotrophic factors by affecting the gamma-aminobutyric acid system in the altered hypothalamic-pituitary-adrenal axis [44]. Meanwhile, another study showed the activation of indoleamine-2,3-dyoxigenase (IDO) (a rate-limiting enzyme in the catabolism of tryptophan along the kynurerine pathway), was a causative factor of Aβ-associated inflammation in AD [45]. Emerging evidence suggested that the β-site amyloid precursor protein cleaving enzyme (BACE1) might affect processes underlying broader brain mechanisms and functions, affecting emotional fields, decision-making, circadian activity, and others [46–49]. BACE1, the limiting velocity enzyme for Aβ production in AD [50], was one of the prospective therapeutic targets against cognitive decline and amyloid pathology [50, 51]. The roles of these pathways or mechanisms in mediating the effects of NPSs on amyloid pathology merit further investigation as they are potential targets for early intervention in AD.
There are some limitations to our study: 1) The NPI was used as the primary NPSs measure and was measured indirectly through relatives or caregivers. The provider’s familiarity with the patients and the levels of the expectation might have an impact on the diagnosis and thus might reduce the translatability of the results to the individual levels. 2) There might be missing data during the follow-up, such as the unavoidable factors withdrawal and death. LME was used as the most appropriate analysis method to minimize this problem. It was still possible to not reject the null hypothesis as a result of the inadequate power.
Conclusion
To sum up, this study indicated that NPSs’ four subsyndromes (hyperactivity, psychosis, affective symptoms, apathy) were all early manifestations of cognitive decline and precede imaging findings. Amyloid pathology was not only contributed from NPSs, but also a key mediator of NPSs effects on cognitive decline as well as neurodegeneration, and could increase the risk of AD. These findings in favor of the hypothesis that NPSs reflect an early manifestation of preclinical AD and could be used to help identify high-risk populations suitable for early disease prevention.
Footnotes
ACKNOWLEDGMENTS
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012).
ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
FUNDING
This study was supported by grants from the National Natural Science Foundation of China (81971032, 82271475), Taishan Scholars Program of Shandong Province (tsqn20161078), and Medical Science Research Guidance Plan of Qingdao (2021-WJZD001).
CONFLICT OF INTEREST
Lan Tan is an Editorial Board Member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer-review.
All other authors have no conflict of interest to report.
DATA AVAILABILITY
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
