Abstract
Background:
Type 2 diabetes mellitus (T2DM) affects ∼25% of Veterans, a prevalence rate double that of the general population. T2DM is associated with greater dementia risk and has been shown to exacerbate the impact of Alzheimer’s disease (AD) risk factors on declines in daily functioning; however, there are few studies that investigate these patterns in older Veterans.
Objective:
This study sought to determine whether T2DM moderates the association between amyloid-β (Aβ) positron emission tomography (PET) and 1-year change in everyday functioning in older Veterans.
Methods:
One-hundred-ninety-eight predominately male Vietnam-Era Veterans without dementia from the Department of Defense-Alzheimer’s Disease Neuroimaging Initiative (DoD-ADNI) with (n = 74) and without (n = 124) T2DM completed Aβ PET imaging and everyday functioning measures, including the Clinical Dementia Rating–Sum of Boxes (CDR-SB) and Everyday Cognition (ECog). Linear mixed effects models tested the moderating role of T2DM on the association between Aβ PET and 1-year change in everyday functioning.
Results:
The 3-way T2DM×Aβ PET×time interaction was significant for CDR-SB (p < 0.001) as well as the Memory (p = 0.007) and Language (p = 0.011) subscales from the ECog. Greater amyloid burden was associated with greater increases in functional difficulties, but only in Veterans with T2DM.
Conclusions:
Higher Aβ was only associated with declines in everyday functioning over 1 year in Veterans with T2DM. Given that people with T2DM are more likely to have co-occurring cerebrovascular disease, the combination of multiple neuropathologies may result in faster declines. Future studies should examine how diabetes duration, severity, and medications impact these associations.
INTRODUCTION
With the rapid increase in the older adult population of the US [1], both type 2 diabetes mellitus (T2DM) and Alzheimer’s disease (AD) are significant public health concerns. While over 35 million US adults have diabetes (14.7% of the US adult population) [1], the prevalence of T2DM is disproportionately higher among Veterans, with roughly 25% of Veterans diagnosed with T2DM [2, 3]. In addition to increasing risk of multiple medical conditions including heart disease, chronic kidney disease, nerve damage, and retinopathy, T2DM also increases the risk of cognitive impairment [4–7]. People with T2DM are at greater risk for developing amnestic mild cognitive impairment (MCI), dementia due to vascular and AD neuropathologies, and changes in everyday functioning [4, 8]. Thus, T2DM and AD may interact in unique ways to accelerate declines in cognitive and everyday functioning.
AD currently impacts 6.7 million (10.7%) of the US older adult population and this estimate is expected to nearly double (12.7 million) by 2050 [9]. Military-related exposures such as vascular risks secondary to toxin exposures, posttraumatic stress disorder (PTSD), and traumatic brain injury (TBI) all increase risk of dementia, including AD [10–12]. AD is characterized by the presence of amyloid-β (Aβ) plaque and neurofibrillary tau tangle pathologies and an insidious, progressive course of cognitive and functional decline [13]. Although there are some mixed results regarding the direct causal link between Aβ and cognitive functioning [14, 15], there is consistent evidence that higher Aβ levels as measured in vivo using cerebrospinal fluid (CSF) or positron emission tomography (PET) are associated with worse cognitive and functional outcomes [16–19].
While investigations into the association between T2DM and Aβ have yielded somewhat mixed results [20–22], a recent population-based study demonstrated that participants with T2DM had higher rates of amyloid burden on PET imaging [22]. One possible mechanism for this pattern may be that T2DM results in damage to the blood-brain barrier as well as cerebral hypoperfusion, which initiates a cascade that promotes AD pathology (e.g., Aβ and tau) and, in turn, accelerates neurodegeneration [23, 24]. Further, several studies have now demonstrated interactive or synergistic effects of T2DM and AD risk factors such as an MCI classification, genetic susceptibility, or elevated tau pathology on worse cognitive and functional outcomes [25–27], greater cortical volume loss and reduced glucose metabolism [28], and more severe AD neuropathology at autopsy [29].
The current study uses data from the Department of Defense-Alzheimer’s Disease Neuroimaging Initiative (DoD-ADNI), which provides a unique opportunity to examine AD biomarkers in Vietnam-era Veterans. It is critical to examine T2DM and AD biomarkers within Veteran-only cohorts since there are comorbidities and exposures unique to Veterans that may contribute to both diabetes (e.g., Agent Orange [30]) and dementia (e.g., PTSD, TBI) and put Veterans at particularly high risk of cognitive and functional declines. The current study aims to examine to what extent T2DM moderates the association between Aβ PET and 1-year change in everyday functioning in older Veterans. We anticipated that T2DM and higher Aβ would act synergistically to predict faster rates of functional decline.
MATERIALS AND METHODS
Participants
Data used in this study were obtained from the publicly available Brain Aging in Vietnam War Veterans/DoD-ADNI database (http://adni.loni.usc.edu). The study is directed by principal investigator Dr. Michael Weiner of the San Francisco VA Medical Center and University of California, San Francisco. The overarching goals of the DoD-ADNI study are to characterize the long-term neural and behavioral consequences of TBI and/or PTSD. The main aims and methods of DoD-ADNI as well as up-to-date information can be found at http://www.adni-info.org. This research was approved by the institutional review boards of all participating sites within ADNI and written informed consent was obtained for all study participants.
The analytic sample included 198 Vietnam-Era Veterans without dementia at baseline. Participants were included if they had variables of interest including diabetes status, amyloid PET, and everyday functioning measures as well as important covariate data to adjust for including relevant demographics, psychiatric symptoms, TBI history, apolipoprotein E (APOE) ɛ4 carrier status, and general ischemia risk. At the 1-year follow-up, 160 participants had longitudinal data.
T2DM status
T2DM status was first determined based on self-report of diabetes diagnosis (yes or no). Then, we examined the self-reported medical history details and excluded participants with type 1 diabetes. Participants who reported having prediabetes were coded in the no T2DM group since laboratory values were not available.
Amyloid PET
Florbetapir (AV45) PET was used to measure amyloid burden. The details of data acquisition and processing of ADNI florbetapir PET data are available at http://adni.loni.usc.edu. A summary standardized uptake value ratio (SUVR) was calculated by dividing the mean uptake across AD-vulnerable cortical regions (frontal, anterior/posterior cingulate, lateral parietal, and lateral temporal cortices) by whole cerebellar (white and gray matter) uptake. Greater retention of florbetapir is reflective of a greater cortical Aβ load. Aβ PET was log-transformed to improve the normality of the model residuals. While Aβ was used continuously in the models, to describe the sample and for the figures, Aβ positivity was defined using the cutoff of > 1.11 [17]. The baseline study visit was generally close to the amyloid PET visit (mean difference = 7.52 days, median difference = 1 day, range = 0–128 days).
Everyday functioning
The Clinical Dementia Rating–Sum of Boxes (CDR-SB) and Everyday Cognition measures (ECog) were used to measure change in everyday functioning over 1-year. The CDR is a semi-structured interview that assesses the degree of everyday impairment across domains of Memory, Orientation, Judgment & Problem Solving, Community Affairs, Home & Hobbies, and Personal Care. The CDR-SB provides a greater range of scores compared to the CDR global score and ranges from 0–18, with higher scores indicating worse functional status [31, 32]. The ECog measure is a 39-item questionnaire in which the participants rate their ability to perform everyday tasks relative to 10 years ago on a scale of 1 (“better or no change”) through 4 (“consistently much worse”) [33]. A score of 9 (“don’t know”) was coded as missing. The everyday tasks are divided into cognitive domains that are involved with the task, including memory, language, visuospatial, planning, organization, and divided attention. The ECog total score is based on the mean of all the items and the ECog domain subscales were calculated by taking the mean of the items that go into that specific domain. Higher scores represent more subjective everyday cognitive and functional difficulties.
Additional measures
Hachinski Ischemia Scale total score was used to measure ischemia risk and includes items assessing history of hypertension, stroke, atherosclerosis, focal neurologic signs, and more. Given the limited range of Hachinski scores in DoD-ADNI (all participants had scores < 6) and the very few participants with a score of 3, 4 or 5 (n = 6), the Hachinski score was categorized as 0 = score of 0, 1 = score of 1, and 2 = score of 2 + . There were a small number of participants who identified as a race other than white (n = 29), and therefore race had to be coded as white or non-white (American Indian/Alaska Native, Asian, Black/African American, more than one race, or Unknown) for inclusion as a covariate. Ethnicity was coded as Hispanic or non-Hispanic. APOE ɛ4 genotype (non-carrier versus carrier) was included as a measure of genetic susceptibility to AD. Current PTSD symptom severity was measured using the Clinician-Administered PTSD Scale (CAPS) and depressive symptom severity was measured using the Geriatric Depression Scale (GDS). Since the GDS includes an item about memory problems that is similar to subjective memory concern items that are also asked on the ECog, that memory concern item on the GDS was excluded in the total GDS score used as a covariate. TBI severity was based on Veteran Affairs (VA)/DoD criteria 2021 Clinical Practice Guidelines [34]. Each TBI was coded and severity was based on the most severe TBI sustained in their lifetime. An injury was classified as mild if the participant had a loss of consciousness (LOC) of≤30 min, or alteration of consciousness (AOC) or post-traumatic amnesia (PTA) up to 24 h. The moderate and severe TBI groups were combined since the information for PTA > 1 day was not available. Thus, the moderate-to-severe TBI criteria included: LOC > 30 min, AOC > 24 h, or PTA > 1 day.
Statistical analyses
Independent samples t-tests and chi-squared tests were used to examine demographic and clinical characteristics by T2DM status. Linear mixed effects models using Full Information Maximum Likelihood examined whether T2DM moderated the association between Aβ PET SUVR and 1-year change in everyday functioning measured using the CDR-SB and ECog. Model 1 was minimally-adjusted and included age, education, and Hachinski score as covariates. Next, Model 2 was fully-adjusted for measures that either differed between Veterans with and without T2DM or could potentially impact Aβ levels or the everyday functioning outcomes. Covariates for Model 2 included age, education, Hachinski score, race/ethnicity, APOE genotype, PTSD and depressive symptom severity, and traumatic brain injury history. Random intercept was included in all models. Given the possibility of PET scanner effects across sites, we examined the models with and without adjustment for site. Since the inclusion of site did not impact the effects of interest, we did not include it in the final models in order to minimize model parameters.
RESULTS
Participant characteristics for the whole sample and split by T2DM status are shown in Table 1. At baseline, there were significant differences between T2DM–and T2DM+ groups on Hachinski score with T2DM+ having higher vascular/ischemia risk. There were no statistical differences between T2DM groups on baseline age, years of education, sex, race/ethnicity, APOE ɛ4 carriers, Aβ PET SUVR levels or positivity rates, depressive or PTSD symptoms, TBI history, global cognition, or neuropsychological measures of memory (Rey Auditory Verbal Learning Test), language (Boston Naming Test, category fluency), and executive functioning (Trails B). There was a trend for slower performance for processing speed (Trails A) by the T2DM+ group. Related to baseline everyday functioning, the T2DM+ group endorsed significantly more difficulties on the ECog Planning subscale relative to the T2DM- group, and there was a pattern of more difficulties for the T2DM+ group on the ECog Memory and Organization subscale as well as the CDR-SB, but these effects did not reach statistical significance. There were no group differences in the proportion of participants with a CDR global score of 0 versus 0.5 or on the ECog total score as well as the ECog Language, Visuospatial, and Divided Attention subscales. Within participants in the T2DM+ group, the most common medications were metformin (77.0% were taking metformin) and insulin (23.0% were taking insulin).
Sociodemographic and clinical characteristics by diabetes group
APOE, apolipoprotein E; GDS, Geriatric Depression Scale; CAPS, Clinician-Administered PTSD Scale; TBI, traumatic brain injury; MMSE, Mini-Mental State Exam; AVLT, Rey Auditory Verbal Learning Test; BNT, Boston Naming Test (30-item); CDR, Clinical Dementia Rating; ECog, Everyday Cognition; CDR was missing data from 8 participants (n = 190).
Linear mixed effects models were used to examine the two-way interactions of of T2DM×time and Aβ PET×time on change in CDR-SB or ECog prior to including the three-way T2DM×Aβ PET×time interaction in the models. T2DM was not independently associated with change in CDR-SB or ECog (ps > 0.05) in the minimally-adjusted models (Model 1) or the fully-adjusted models (Model 2). Aβ PET was associated with faster increases in functional difficulties on the CDR-SB in minimally- and fully-adjusted models (Model 1: β= 0.126, 95% CI [0.010, 0.234], p = 0.033; Model 2: β= 0.122, 95% CI [0.006, 0.238], p = 0.039), but not on the ECog for either models.
Next, we examined the moderating impact of T2DM on the association between Aβ PET and change in everyday functioning (see Table 2 for full results). For CDR-SB, the three-way T2DM×Aβ PET×time interaction was significant (β= 0.223, 95% CI [0.115, 0.331], p < 0.001) for Model 1, such that greater amyloid burden was associated with faster increases in functional difficulties over 1 year, but only in Veterans with T2DM (Fig. 1). This three-way interaction was unchanged when additional covariates were added to Model 2 (β= 0.220, 95% CI [0.112, 0.328], p < 0.001). A similar pattern was found for the 1-year change in ECog for both Models 1 (β= 0.093, 95% CI [–0.004, 0.190], p = 0.061) and 2 (β= 0.093, 95% CI [–0.003, 0.190], p = 0.058), but the 3-way interaction did not reach statistical significance.
Estimates for minimally-adjusted (Model 1) and fully-adjusted (Model 2) models examining T2DM as a moderator of the association between amyloid PET and 1-year change in everyday functioning
GDS, Geriatric Depression Scale; CAPS, Clinician-Administered PTSD Scale; TBI, traumatic brain injury; APOE, apolipoprotein E; T2DM, type 2 diabetes.
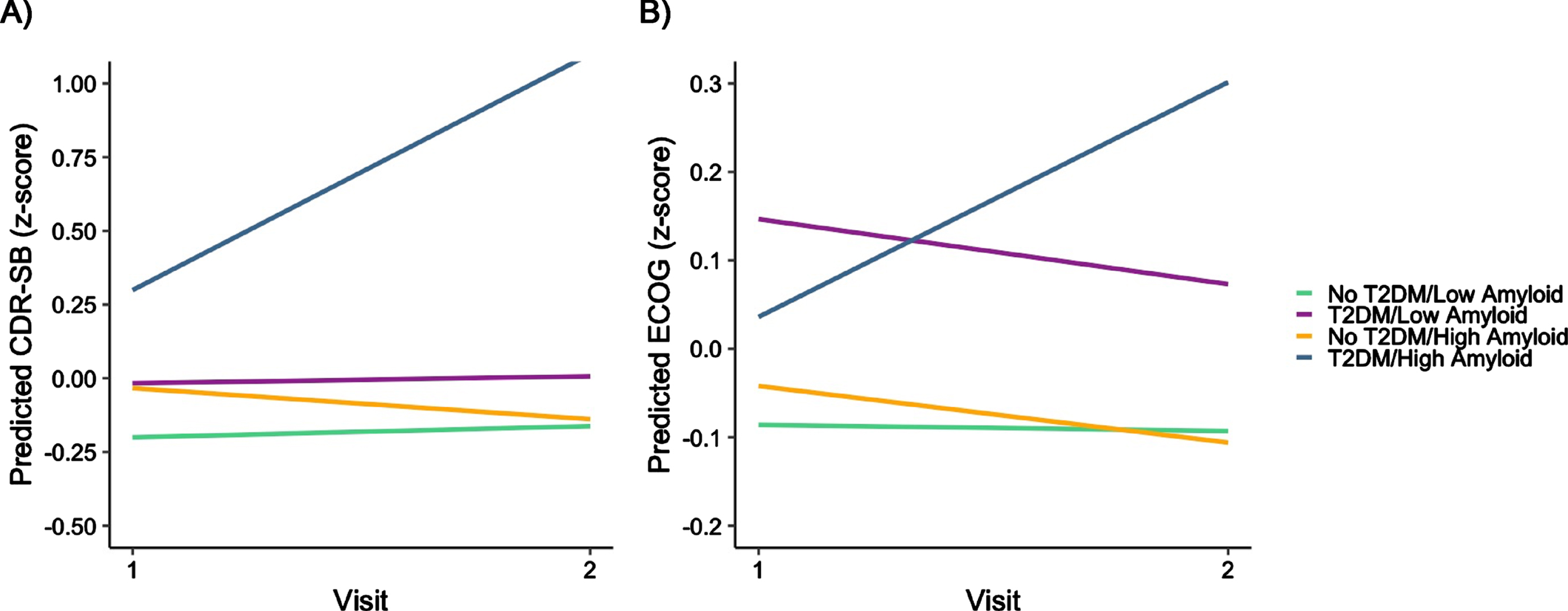
One-year change in CDR-SB and ECog scores by T2DM and Amyloid status. Amyloid was a continuous variable in the model, but for graphing purposes amyloid PET was dichotomized at High/Low based on previous cutoffs [17]. High Amyloid, SUVR > 1.11; Low Amyloid, SUVR < 1.11.
To learn more about the types of everyday functioning changes participants were endorsing, we examined the 3-way interaction predicting change in domain-specific subscores of the ECog. T2DM only moderated the association between Aβ PET and change on the ECog Memory (Model 1: β= –0.144, 95% CI [0.041, 0.246], p = 0.006; Model 2: β= 0.142, 95% CI [0.040, 0.244], p = 0.007) and Language (Model 1: β= 0.139, 95% CI [0.033, 0.246], p = 0.011; Model 2: β= 0.139, 95% CI [0.033, 0.245], p = 0.011) subscores, but not Visuospatial, Planning, Organization, or Divided Attention subscores.
DISCUSSION
The current study found that T2DM impacted the association between Aβ burden and rate of decline in everyday functioning in older Veterans without dementia. Specifically, in participants with T2DM, greater Aβ burden was associated with a faster increase in functional difficulties as measured by the CDR-SB and on the memory and language subscales of the ECog. These changes in functioning are largely consistent with changes observed due to AD, as the CDR is associated with AD pathology [31, 35] and memory and language are often the first cognitive domains to be impacted in typical AD [36].
Prior work has demonstrated that both Aβ and T2DM independently predict decline in everyday functioning in older adults without dementia [37–41]. Within our study, however, there was no effect of T2DM on change in everyday functioning, and Aβ PET was only associated with change in CDR-SB, but not ECog. These findings may be due to the short follow-up period, as we would not expect substantial change in everyday functioning over 1 year in older adults without dementia. These findings could also be due to differences specific to a Veteran sample (e.g., in which multiple other factors that could impact functioning such as psychiatric symptoms and unique exposures and health factors more prevalent in Veterans such as TBI). While we adjusted for many of these factors in our models, it is possible that these Veteran-relevant factors have complex and interacting relationships that could not be fully accounted for in the model as individual covariates and thus could complicate subjective ratings of everyday functioning.
The findings of accelerated declines in Veterans with multiple risk factors such as higher Aβ accumulation plus T2DM are particularly notable given the high rates of older Veterans with T2DM. Prior work in cognitively unimpaired older adults from the Alzheimer’s Disease Neuroimaging Initiative who were mostly non-Veterans showed that T2DM interacted with multiple AD risk factors including APOE genotype, CSF p-tau, CSF total tau, and subtle cognitive changes, to accelerated decline in everyday functioning over 4 years [27]. Notably, this prior study did not find an interactive effect with CSF Aβ; however, studies show that PET may be a more robust marker of Aβ than CSF [42–44], which may partially explain the difference in results. Further, it is likely that the participants with higher amyloid burden also have higher tau levels in the brain. Given that tau tends to be more strongly associated with cognition than amyloid [15], it is possible that tau pathology is a key contributor to the current results. Additionally, there may be factors more prevalent in older Veterans that put them at greater risk for earlier amyloid accumulation (e.g., greater vascular burden, toxin exposure).
The results of the study are important for older Veterans as the prevalence of diabetes in the Veteran population is already high and likely to increase in coming years with the aging of both Vietnam-Era Veterans as well as Veterans who participated in the Iraq and Afghanistan conflicts. Given these high rates of diabetes and exposures that increase risk of dementia, including AD, it highlights the need for careful monitoring of cognitive and functional changes in middle-aged and older Veterans with T2DM. Further, while this study adjusted for co-occurring psychiatric (e.g., PTSD, depressive symptoms) and health factors (TBI, general vascular burden) that could also contribute to changes in functioning, more work is needed to understand the unique and interactive nature of these factors with both T2DM and higher Aβ pathology that put one at greater risk for dementia.
There are several possible reasons for the interactive effect of Aβ PET and T2DM to accelerate functional decline in this study. It is possible that individuals with T2DM patients are more likely to have co-occurring cerebrovascular disease and the combination of multiple pathologies (i.e., AD plus vascular) results in faster declines. The mechanisms for this pattern may be that greater vascular disease results in damage to the blood-brain barrier as well as greater cerebral hypoperfusion, which may exacerbate the negative effects of AD pathology [23, 45]. Specifically, cerebral hypoperfusion has been found in people with T2DM [46] and cerebral blood flow has also been shown to interact with Aβ to impact cognitive functioning [47]. A separate speculation for the pattern of findings could be due to the subjective nature of the everyday functioning measures in combination with the complex medication management and health monitoring sometimes required in T2DM. For example, it is possible that people with T2DM, due to engaging in these complex medication management (to ensure correct doses, often a combination of pills and/or insulin) and monitoring (e.g., remembering to check blood sugar; consistent awareness of what they are eating, etc.) may notice subtle cognitive changes associated with AD, which make managing their diabetes more difficult at an earlier stage than someone with similar pathology but who does not have the daily cognitive and functional demands that it often takes to manage T2DM. Finally, while we may have speculated that the T2DM+ group simply has more advanced AD pathology than the T2DM- group and therefore are progressing faster, we show in Table 1 that neither the continuous Aβ PET SUVR nor the proportions of Aβ positivity differ between T2DM groups at baseline.
Amyloid PET is the gold standard of in vivo measurement of Aβ pathology. However, this is also an expensive tool that is also not often accessible for individuals who are not affiliated with a larger medical center. In coming years, plasma biomarkers such as p-tau217 may offer a more accessible and less invasive and costly alternative to PET imaging given strong associations with Aβ PET and future cognitive declines [48]. As these plasma biomarkers become more widely available as a clinical tool, a potential use of these plasma measures may be for close monitoring in individuals with T2DM in order to improve early detection of AD-related cognitive and functional declines.
The current study is one of the first to look at intersecting vascular and AD risk in older Veterans. Additionally, the DoD-ADNI database has a number of different advantages, including multiple AD biomarkers, psychiatric measures, multiple measures of everyday functioning to increase the likelihood of reliable findings, and longitudinal data. While more years of longitudinal follow up data would be beneficial, results demonstrated accelerated decline in everyday function in Veterans with T2DM and higher Aβ burden on two different measures, even over the short 1-year follow up period. Despite the strengths of the data, the current study is limited by the lack of representation of women Veterans and non-representative proportions of race/ethnicity among Veterans as well as the over-sampling of participants with a history of TBI and PTSD given the initial goals of DoD-ADNI. Additionally, more detailed data such as diabetes severity (e.g., HbA1c) and duration of diabetes would provide helpful information to characterize the sample and conduct additional sensitivity analyses. We did have access to their current diabetes medications and included the proportion who are on metformin and insulin (the two most used medications) to help characterize the sample. Given more recent evidence that diabetes medications such as metformin or DPP-4 inhibitors may alter AD-related declines [49–52], future research looking at how specific diabetes drug types may impact these trajectories is needed in older Veterans.
Taken together, this study offers initial evidence that T2DM moderates the association between Aβ and rate of everyday functioning decline across a one-year period. The findings demonstrated that greater Aβ burden relates to greater decline in everyday functioning over 1-year, but only in Veterans with T2DM. These results offer new insights into the impact of T2DM on functioning in Veterans already at risk for AD and suggest close monitoring of cognitive and functional changes in Veterans with T2DM.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
This work was supported by the U.S. Department of Veterans Affairs Clinical Sciences Research and Development Service (1IK2CX001865 to KRT, 1I01CX001842 to KJB). Authors also receive salary support from the National Institutes of Health/National Institute on Aging grants (R03 AG070435 to KRT, R01 AG063782 to KJB) and the Alzheimer’s Association (AARG-22-723000 to KRT).
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; BioClinica, Inc.; Biogen Idec Inc.; Bristol-Myers Squibb Company; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; GE Healthcare; Innogenetics, N.V.; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Medpace, Inc.; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Synarc Inc.; and Takeda Pharmaceutical Company. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Disease Cooperative Study at the University of California, San Diego. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California. This research was also supported by NIH grants P30 AG010129 and K01 AG030514.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Disease Cooperative Study at the University of California, San Diego. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California. This research was also supported by NIH grants P30 AG010129 and K01 AG030514.
CONFLICT OF INTEREST
Drs. Thomas and Bangen are Editorial Board Members of this journal but were not involved in the peer-review process nor had access to any information regarding its peer-review. Other authors report no conflicts of interest.
