Abstract
Coronary artery disease is a prevalent ischemic disease that results in insufficient blood supply to the heart muscle due to narrowing or occlusion of the coronary arteries. Various reperfusion strategies, including pharmacological thrombolysis and percutaneous coronary intervention, have been developed to enhance blood flow restoration. However, these interventions can lead to myocardial ischemia/reperfusion injury (MI/RI), which can cause unpredictable complications. Recent research has highlighted a compelling association between MI/RI and cognitive function, revealing pathophysiological mechanisms that may explain altered brain cognition. Manifestations in the brain following MI/RI exhibit pathological features resembling those observed in Alzheimer’s disease (AD), implying a potential link between MI/RI and the development of AD. The pro-inflammatory state following MI/RI may induce neuroinflammation via systemic inflammation, while impaired cardiac function can result in cerebral under-perfusion. This review delves into the role of extracellular vesicles in transporting deleterious substances from the heart to the brain during conditions of MI/RI, potentially contributing to impaired cognition. Addressing the cognitive consequence of MI/RI, the review also emphasizes potential neuroprotective interventions and pharmacological treatments within the MI/RI model. In conclusion, the review underscores the significant impact of MI/RI on cognitive function, summarizes potential mechanisms of cardio-cerebral communication in the context of MI/RI, and offers ideas and insights for the prevention and treatment of cognitive dysfunction following MI/RI.
Keywords
INTRODUCTION
Cardiovascular disease is a significant global health concern associated with high mortality and disability rates [1]. Reperfusion strategies have been widely implemented to restore blood flow following a cardiovascular event, improving patient outcomes [2]. However, this approach can lead to myocardial ischemia/reperfusion injury (MI/RI), causing further damage to the heart tissue [3, 4]. At the same time, cognitive impairment and dementia are prevalent among cardiovascular disease patients [5, 6], particularly after cardiac surgery, with a prevalence ranging from 9% to 54% [7]. Post-operative neurocognitive deficit poses significant clinical challenges due to their association with increased mortality and limited treatment options [8]. Additionally, these deficits contribute to reduced long-term quality of life and increased economic burden [9]. Growing evidence suggests that I/R in peripheral organs, including the liver [10–12], kidneys [13, 14], intestines [15, 16], and limbs [17], is a risk factor for cognitive impairment. Cardiac I/R has been specifically identified as an important contributor to cognitive dysfunction following cardiac surgery [18]. Animal studies have revealed that cognitive decline associated with cardiac I/R is linked to various brain pathological manifestations, such as blood-brain barrier (BBB) breakdown, increased oxidative stress and neuroinflammation, mitochondrial dysfunction, amyloid-β (Aβ) accumulation, neuronal apoptosis, and dendritic spine loss [19].
Considering the distinct relationship between the heart and the brain, it is intriguing how the brain undergoes pathological changes in response to MI/RI. This review aims to compile and discuss potential mechanisms underlying cognitive impairment following MI/RI, as well as provide insights into treatment and protective strategies against cognitive decline in this context.
SEARCH STRATEGY
We performed a search of all literature available in PubMed and Google scholar. The literature search was performed without any language or study publication year restrictions, and the following key words were used: (“myocardial ischemia reperfusion injury” OR “MI/RI” OR “cardiac ischemia reperfusion” OR “cardiac I/R” OR “Cardiac Surgery” OR “percutaneous coronary intervention” OR “PCI” OR “percutaneous coronary transluminal angioplasty” OR “coronary artery bypass grafting” OR “CABG” OR “coronary artery revascularization”) AND (“cognition” OR “cognitive dysfunction” OR “cognitive decline” OR “cognitive impairment” OR “dementia” OR “perioperative neurocognitive dysfunction” OR “PND” OR “post operative cognitive dysfunction” OR “POCD”). We evaluated the abstracts of all articles and screened them for relevance to the purpose of this review. We also searched the reference lists of the articles identified through this search strategy and selected those considered relevant. Ultimately, we screened 22 clinical articles and three preclinical articles that met our research objectives. The characteristics of these studies are shown in Tables 1 and 2.
Study Characteristics of MI/RI-Related Postoperative Cardiac Cognitive Dysfunction
Behavioral assessment of MI/RI-related cognitive impairment
CLINICAL CLUES SUGGEST THAT MI/RI MAY BE THE CAUSE OF COGNITIVE IMPAIRMENT
In contemporary medical practice, a significant proportion of cardiac-related procedures involves intervention aimed at reconstructing coronary blood flow, including percutaneous coronary intervention (PCI), percutaneous coronary transluminal angioplast, and coronary artery bypass grafting (CABG) [20, 21]. Despite the inevitable occurrence of varying degrees of MI/RI during these interventions, it is noteworthy that this phenomenon is seldom regarded as a potential risk factor for cognitive changes following cardiac surgery or procedures from a clinical standpoint. This may be attributed to the challenge of clinically distinguishing, monitoring, or predicting MI/RI. However, it is crucial to emphasize that MI/RI should not be discounted as a contributing factor to cognitive dysfunction post-cardiac surgery or procedures.
A large cohort study involving 3,105 participants revealed comparable levels of memory loss following both cardiac surgery and cardiac catheterization [22]. Moreover, a recent high-quality retrospective study indicated that, under average population characteristics, the anticipated probability of dementia five years post-revascularization stood at 10.5% for the CABG group and 9.6% for the PCI group, with no statistically significant difference [23]. In addition, evidence from a systematic review proposed that the type of reconstructive surgery (PCI and CABG) is not significantly linked to variations in cognitive performance deterioration [24]. It is widely recognized that PCI, as an alternative coronary revascularization method to CABG, mitigates exposures to several medical factors believed to contribute to cognitive decline after CABG, such as extracorporeal circulation pumps, general anesthesia, and surgical trauma. Consequently, this evidence underscores the importance of considering factors like MI/RI that manifest following both PCI and CABG in relation to cognitive decline post-cardiac surgery. Additionally, the presence of collateral circulation has the potential to attenuate MI/RI, and existing evidence suggests that collateral circulation in coronary arteries diminishes the risk of postoperative cognitive impairment [25]. Remarkably, the vast majority of the patient population (85%) who exclusively underwent coronary angiography displayed no postoperative cognitive dysfunction [26]. Collectively, this body of evidence strongly implies that MI/RI may play a pivotal role in postoperative cognitive decline. Detailed characteristics of MI/RI-related postoperative cardiac cognitive dysfunction studies are listed in Table 1.
Indirect clinical evidence suggests a potential association between MI/RI and cognitive dysfunction. In clinical scenarios, MI/RI can manifest as reperfusion arrhythmias, myocardial failure, low cardiac output, and perioperative myocardial infarction [27]. For instance, a study reported that approximately 9% of patients with acute ST-segment elevation myocardial infarction develop atrial fibrillation during or immediately after PCI [28], potentially induced by MI/RI. A well-established causal relationship between atrial fibrillation and cognitive dysfunction has been documented [29–31]. Furthermore, the low cardiac output or myocardial infarction resulting from MI/RI leads to inadequate cerebral perfusion, mirroring the pathophysiological features of cognitive impairment observed in heart failure cases [32]. In addition, systemic inflammation is activated after MI/RI, releasing substantial amounts of inflammatory factors into the bloodstream [33]. Also, the role of postoperative serum or plasma cytokines, particularly interleukin 6 (IL-6), in postoperative cognitive dysfunction has been extensively validated in patients [34, 35].
Despite the compelling evidence, drawing a definitive causal link between MI/RI and cognitive impairment from available clinical data proves challenging. This difficulty likely arises because MI/RI cannot be isolated as a singular factor in clinical settings. Nonetheless, avenue for investigation persist. Focusing on new MI/RI-related cardiovascular events at specific time points (e.g., during and after cardiac reperfusion surgery) and exploring their correlation with long-term cognitive status could prove valuable. Additionally, patients with acute coronary syndromes undergoing acute PCI are more prone to exhibit higher levels of depression and fatigue, along with poorer concentration and attention 0–5 days after PCI compared to patients undergoing elective PCI [36]. This implies the importance of considering population characteristics, suggesting that the impact on postoperative cognitive outcomes after coronary revascularization may differ for acute and chronic coronary syndromes. Furthermore, urgent attention is needed for exploring markers or measures of MI/RI in the clinic settings. This exploration is crucial not only to understand MI/RI-related complications but also to delve beyond cognitive function.
COGNITIVE-RELATED BRAIN PATHOLOGY AND BRAIN REGIONS FOLLOWING MI/RI
Significant pathological changes occur in the brain after MI/RI, and these pathological manifestations are critical for understanding the role of the condition in cognitive dysfunction. One prominent pathological feature after cardiac I/R is the disruption of the BBB, which allows the entry of numerous inflammatory mediators into the brain [19]. This disruption is accompanied by a decrease in the number of CD 11b/CD 45++low cells and an increase in the number of CD 11b/CD 45++++high cells, along with morphological changes indicative of microglia proliferation and hyperactivation [55]. Additionally, reactive astrogliosis leads to a cascade of neuroinflammation amplification [56]. Another potential mechanism of brain damage during cardiac I/R is increased oxidative stress brought on by fission and malfunction of brain mitochondria [57]. Consequently, these pathological events lead to neuronal apoptosis and dendritic spine damage, predominantly in the hippocampal region [56, 58]. Interestingly, cardiac I/R-induced pathological events also exhibited features associated with Alzheimer’s disease (AD) in the brain, such as Aβ formation and aggregation, as well as tau hyperphosphorylation [58]. In summary, the pathological events induced by cardiac I/R shown in Fig. 1 appear to contribute to the development of cognitive dysfunction collectively.
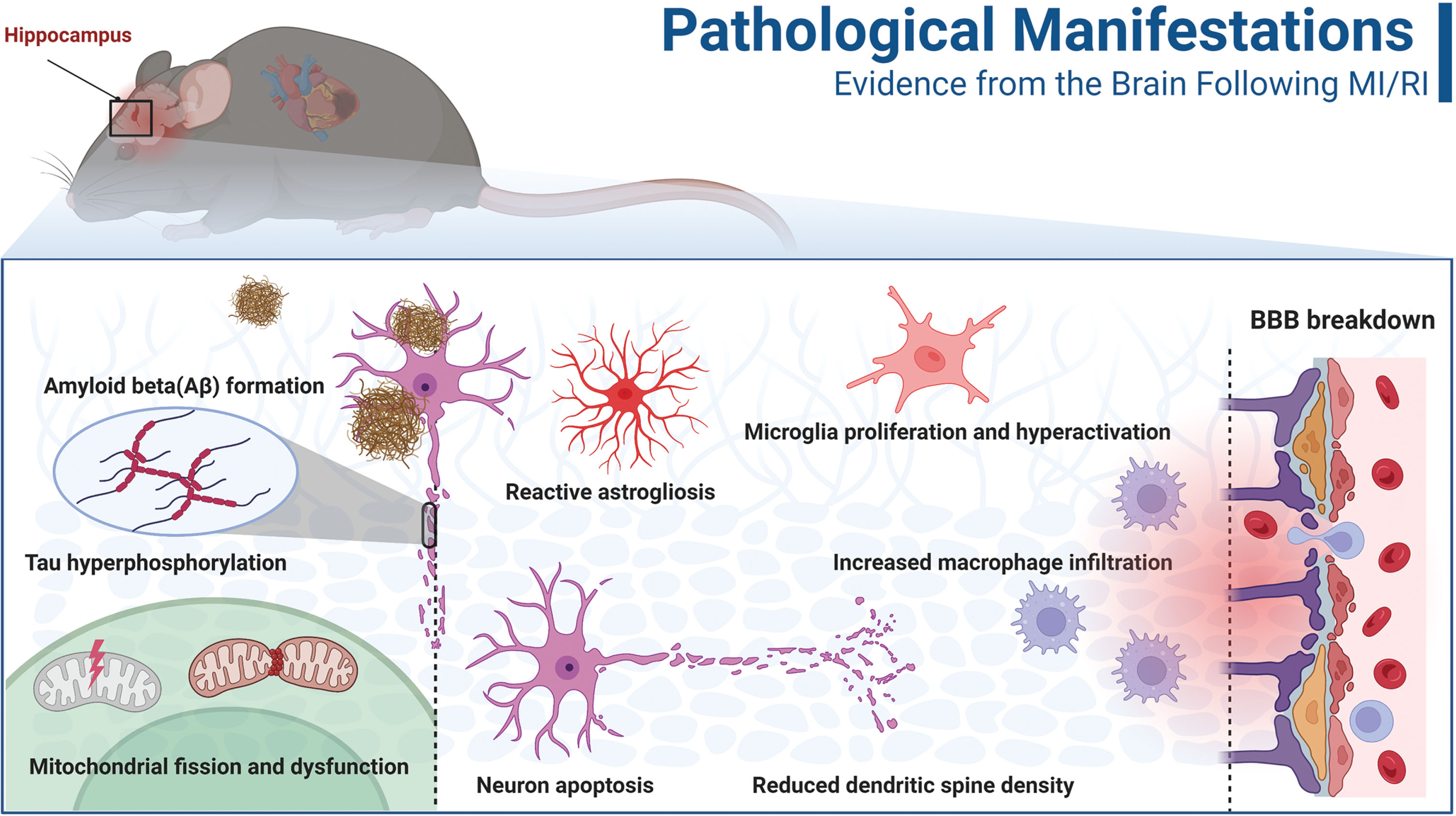
The hippocampus is one of the most extensively studied brain regions involved in cognitive decline after MI/RI. As mentioned earlier, most brain pathologies associated with cognition and memory are found in the hippocampus [18]. Furthermore, the paraventricular nucleus of the hypothalamus and prefrontal cortex have also been implicated [59]. Although the hypothalamus is primarily known for controlling physiological processes such as appetite, body temperature, sexual behaviors, and hormone release, it is interconnected with the hippocampus, amygdala, and prefrontal cortex. It may serve as an interface for various cognitive functions [60]. Moreover, a study utilizing proton magnetic resonance spectroscopy revealed alternation in metabolites in the hippocampus, striatum, thalamus, and temporal cortex following MI/RI [61]. Interestingly, the relevance of these brain regions to the formation and control of various cognitive functions is receiving increasing attention [62–64]. Overall, there is limited current research on cognition-related brain regions after MI/RI, and significant efforts are warranted in the future.
MANIFESTATIONS OF COGNITIVE IMPAIRMENT DUE TO MI/RI IN ANIMALS
Rats exhibit degradation of spatial learning and memory after cardiac I/R [59]. In a study by Evonuk and colleagues, cognitive function in mice was assessed through behavioral measures two months after cardiac I/R. The results revealed that cardiac I/R mice performed worse in contextual fear conditioning and object location memory tasks compared to sham-operated mice [18]. Similarly, rats subjected to cardiac I/R also demonstrated poorer short-term cognitive performance than sham rats in the eight-arm maze test [65]. Our unpublished data also support a significant decline in cognitive function in mice following cardiac I/R, with this decline occurring in both the short-term (7 days) and long-term (a month). In conclusion, despite insufficient attention from most investigators, converging evidence indicates that MI/RI is indeed a crucial factor contributing to cognitive dysfunction after cardiac surgery. A summary of the cognitive behavioral assessment findings in animal studies following MI/RI is presented in Table 2.
POTENTIAL MECHANISMS OF MI/RI-INDUCED COGNITIVE DYSFUNCTION
To understand the mechanisms underlying cognitive impairment induced by MI/RI is to identify the specific pathological changes and processes that occur in the body following MI/RI, ultimately contributing to or directly causing cognitive deficits. Indeed, MI/RI-induced cognitive dysfunction is an outcome of a complex series of pathological processes at the molecular, cellular, tissue, and even systemic levels. Based on the existing literature, we categorize the potential mechanisms of cognitive decline in the brain following MI/RI into three domains.
The role of circulating inflammatory molecules or cells in cognitive dysfunction after MI/RI
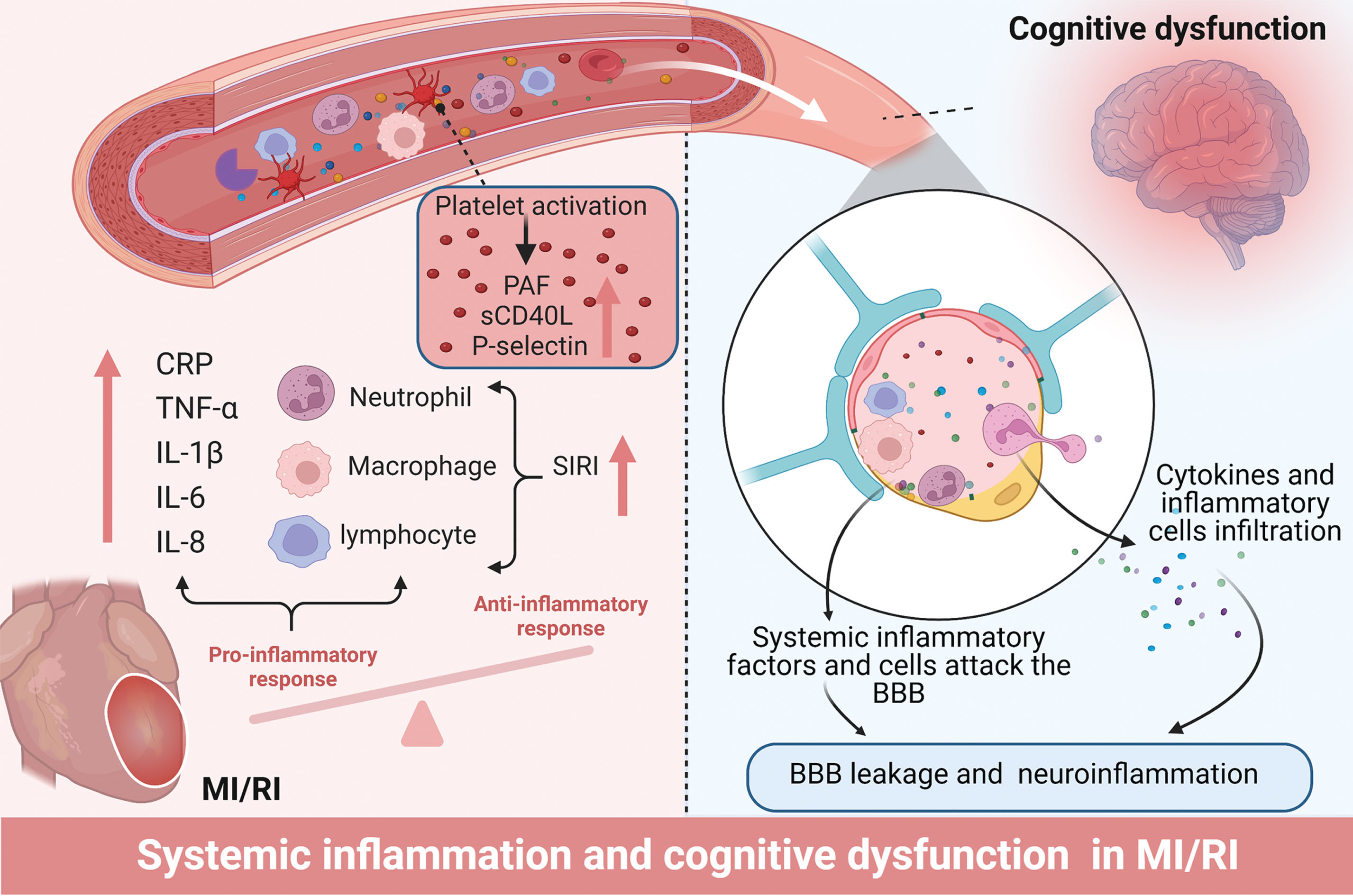
It is widely recognized that activating inflammation, specifically sterile inflammation, contributes to the progress of MI/RI [3]. Surprisingly, in the context of the I/R model, inflammatory mediators can act as implicated agents in the emergence of systemic complications [66]. The reliable marker of systemic inflammation, C-reactive protein (CRP)/high sensitivity C-reactive protein (hsCRP), has been shown as an indicator of MI/RI [67]. Clinical data indicate that CRP levels remain elevated after PCI, peaking at three days and persisting up to 7–14 days post-procedure [68, 69]. Notably, the relative variation of hsCRP (ΔhsCRP) from pre- to 24-h post-PCI has shown superior predictive value for future major adverse cardiac events compared to pre- or post-procedure hsCRP levels alone [70]. Importantly, elevated levels of CRP due to MI/RI play a significant role in predicting short- and long-term cardiac complications. Given the strong association between CRP and cognitive dysfunction, this predictive advantage of ΔhsCRP may also be reflected in cognitive decline after cardiac I/R [71, 72]. Several studies have reported noteworthy links between CRP and postoperative neurocognitive dysfunction (PND) after off-pump CABG and heart valve replacement [73, 74]. Moreover, in animal models, interleukin-6 (IL-6), interleukin-8 (IL-8), and tumor necrosis factor-alpha (TNF-α) levels in the blood were significantly elevated after myocardial I/R [75, 76]. Similar findings were observed in patients, with serum TNF-α and IL-6 levels substantially increased after PCI compared to baseline, particularly TNF-α, which continued to rise 14 days after PCI in acute myocardial infarction cases [69]. Deterioration in the systemic condition can profoundly impact the brain, with significant consequences. A meta-analysis and systematic review of 170 studies on AD and mild cognitive impairment (MCI) highlighted the influence of peripheral inflammation on cognitive function and that the elevated levels of circulating TNF-α have been associated with postoperative cognitive dysfunction [77]. Increased TNF-α and IL-6 have been linked to cognitive decline after cardiac surgery [78] and CABG [79], and the cytokines in the cerebrospinal fluid may serve as predictors of impaired cognition.
The systemic inflammation response index (SIRI), a novel micro-inflammatory marker based on peripheral blood neutrophil, monocyte, and lymphocyte counts, has recently gained attention. Like ΔhsCRP, this composite inflammatory index is an independent risk factor following PCI and reflects cellular-level inflammation responses [80, 81]. Furthermore, SIRI is correlated with cognitive function, as older individuals with higher SIRI levels are at a heightened risk of developing MCI [82]. Strikingly, there appears to be a logical relationship in cardiac revascularization and vascularization procedures. In patients undergoing PCI post-acute ST-segment elevation myocardial infarction (STEMI), SIRI levels have been independently associated with the no-reflow phenomenon (NRP) [83], which is a clinical characteristic manifestation of MI/RI [84]. Therefore, SIRI may serve as a representation of the degree of MI/RI in patients following PCI. Furthermore, the systemic inflammatory response syndrome score is a robust and independent risk factor for of developing PND in patients after CABG [85]. Hence, SIRI may provide additional prognostic information for the development of cognitive dysfunction after MI/RI. Additionally, the neutrophil-lymphocyte ratio (NLR), a readily available marker of systemic inflammation, has shown involvement in the pathogenesis of AD, PND, and MCI in the elderly [86–88]. However, its role in cognitive impairment after MI/RI remains unexplored. Notably, these systemic inflammation markers are inexpensive, readily available, and widely employed in clinical practice. Large-scale, well-controlled clinical studies on the relationship between these inflammatory indicators and cognitive function after cardiac I/R would be valuable in providing predictive biomarkers for impaired cognitive decline.
A growing body of evidence highlights the crucial role of platelets in inflammatory and immune responses. After cardiac I/R, platelets undergo activation via various modes, such as the release of platelet-activating factor (PAF), soluble CD40 ligand (sCD40 L), and P-selectin [89, 90]. Platelet activation is a common occurrence following PCI, and despite employing multiple antiplatelet and anticoagulant drugs, a significant and prolonged elevation of sCD40 L in plasma is observed [68]. Moreover, plasma calprotectin, associated with platelet activation, may serve as an early predictive biomarker of NRP in patients with acute coronary syndromes [91]. Platelets play a crucial role in neuroinflammation [92], and studies have shown a strong association between the degree of platelet activation and decreased cognitive function in AD [93]. However, one study reported that neuroprotective PAF antagonists (lexipafant) did not differentially reduce the level of cognitive impairment following CABG [94]. The authors suggest that this finding may be attributed to the lower-than-expected incidence of cognitive impairment in their experiment, which was insufficient to detect a protective effect of lexipafant [94]. Overall, evidence regarding the role of platelet activation in cognitive function after MI/RI is still preliminary and requires further validation.
How systemic inflammation contributes to impaired cognitive performance after MI/RI is another interesting topic. For these circulating elevated inflammatory molecules and inflammatory cells to exert adverse effects on the brain, they must transverse the BBB. Unfortunately, the BBB sustains significant damage after MI/RI. Data indicated impaired BBB integrity in 50% of post-operative cardiac patients [95]. Animal evidence also confirms a decrease in the expression levels of the BBB protein Claudin5 following cardiac I/R [19, 96]. Current evidence suggests that MI/RI induces the release of pro-inflammatory mediators from coronary endothelial cells and various inflammatory cells into the bloodstream, activating the immune response [27]. These heightened peripheral blood factors subsequently assault the compromised BBB alongside inflammatory cells [97]. On the one hand, microglia and astrocytes become activated in response to these inflammatory factors, further promoting the development of neuroinflammation. On the other hand, these blood factors and inflammatory cells can induce brain remodeling by modulating endothelial cells and neuronal signaling, resulting in the secretion of pro-inflammatory factors, oxidative stress, and the aggregation of AD-related proteins [98–100]. Moreover, inflammatory factors can directly adversely affect neuronal survival and neuronal signaling, leading to neuronal death and synaptic dysfunction [101]. The underlying mechanisms of systemic inflammation induced by MI/RI and the subsequent development of cognitive dysfunction are depicted in Fig. 2.
Production of local cardiac pathological mediators and heart-to-brain transport after MI/RI
Is there a mechanism other than direct invasion of the brain by soluble blood factors and peripheral immune cells that can cause cognitive changes in the brain following MI/RI? Given the considerable distance between the heart and brain, it is necessary to consider the potential for heart-brain crosstalk. Extracellular vesicles (EVs) have emerged as novel messengers facilitating information exchange between organs, including the heart [102].
EVs refer to cell-released membrane organelles such as exosomes, microvesicles, and microparticles [103]. In this review, we refer to all types of EVs collectively. These EVs are synthesized by cells and released into circulation, allowing them to reach various organs, including the brain. By transporting diverse contents, such as proteins, lipids, DNA, mRNAs, microRNAs (miRNAs), and circular RNAs (circRNAs), EVs exert significant biological effects on recipient cells. The concept that EVs mediate communication between the heart and brain is not novel [104, 105]. Evidence shows that EVs from peripheral organs can carry harmful contents into the brain via circulation and contribute to the development of cognitive dysfunction [106]. Similar evidence is emerging for brain injury and cognitive impairment after peripheral organ I/R [107, 108]. Encouragingly, EVs have been implicated in pathophysiological processes after MI/RI [109, 110].
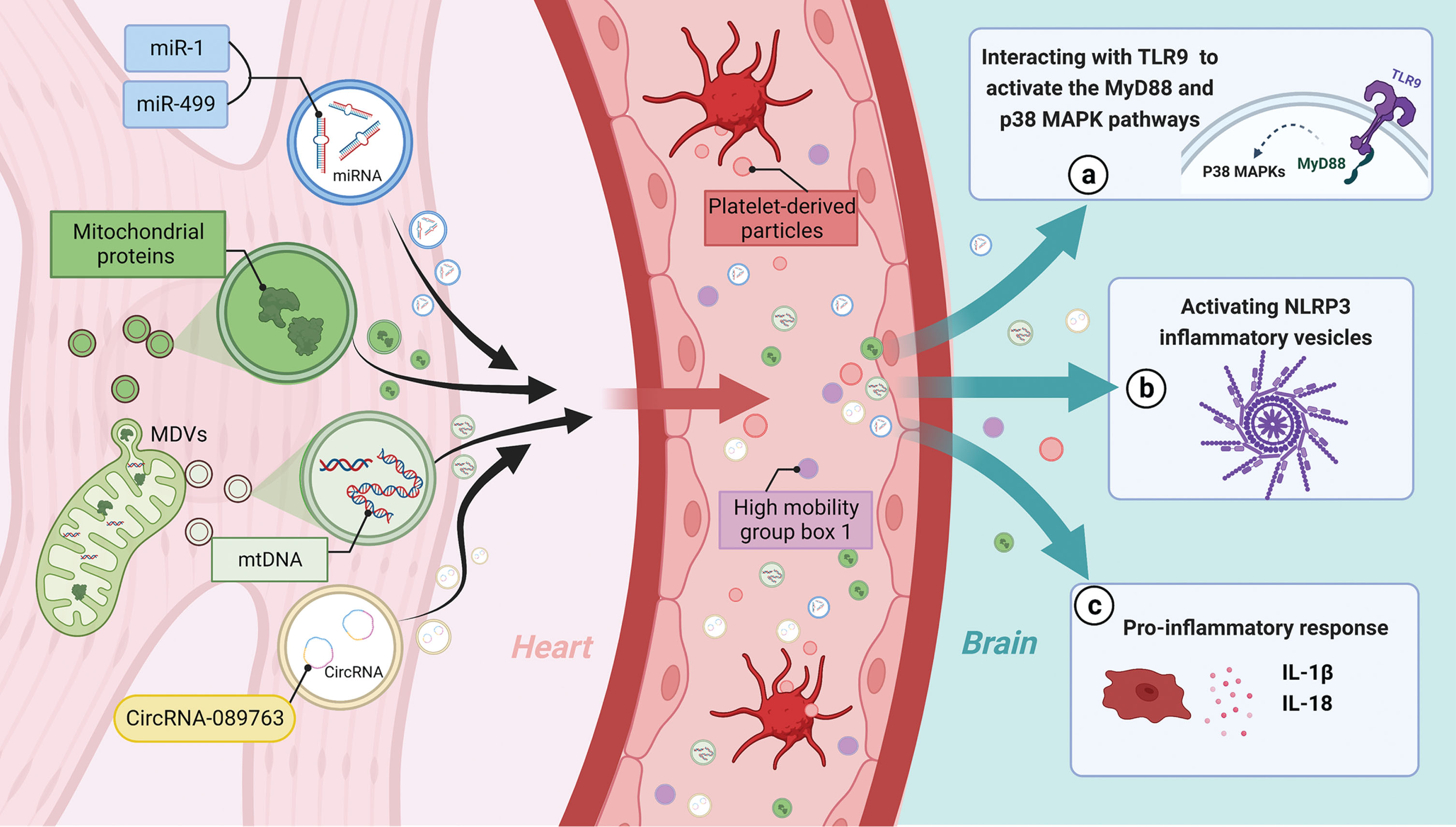
Among the EVs’ contents, miRNAs have been extensively studied. Research by Deddens and Gidlöf et al. demonstrated increased release of cardiac-derived EVs after cardiac I/R in an animal model [111, 112], as evidenced by plasma levels of muscle-specific extracellular miRNAs (miR-1, miR-133b, miR-208b, and miR-499) were found to increase up to 750-fold [112]. Surprisingly, the increased release of EVs carrying miR-1, miR-208b, and miR-499 also occurred in plasma samples collected from patients before and after PCI, particularly after reperfusion [113]. Similar findings include increased plasma EVs concentrations in patients undergoing CABG and increased expression of miR-1, miR-24, miR-133a, and miR-133b in EVs [114]. These findings consistently suggest increased cardiac secretion and release of circulating EVs containing miR-1, miR-208b, miR-133a, and miR-133b, miR-499 post-MI/RI. Can these EVs potentially enter the brain and lead to impaired cognitive function after MI/RI? The answer appears to be affirmative. Mice with cardiac overexpression of microRNA-1 experience cognitive impairment [115]. Inhibition of miR-1 in hippocampal EVs significantly attenuates infarct-induced neuronal microtubule damage [116]. Additionally, miR-1 from cardiac EVs has been shown to attenuate infarct-induced synaptic vesicle exocytosis in the hippocampus by regulating the target gene synaptosomal-associated protein 25 (SNAP-25) [117]. Furthermore, miR-499 was significantly increased among the miRNAs carried by EVs after MI/R. Notably, miR-499-5p is implicated in neurocognitive dysfunction in central nervous system disorders by causing dendritic disruption and dysregulation of calcium homeostasis [118]. Apart from miRNAs, circRNAs within EVs may also contribute to this theory. CircRNA-089763 was remarkably increased in plasma EVs of patients with cardiac I/R occurrence (post-CABG), and the functional annotation analysis and target gene prediction suggested a significant enrichment in GO keywords and pathways associated with PND [119].
MI/RI-induced EVs not only exacerbate local inflammation in the heart but also trigger systemic inflammation in distant organs [120]. This raises the question of how EVs may facilitate inflammation spreading to the central nervous system (CNS). Oxidative phosphorylation function in mitochondria is impaired after MI/RI, leading to increased formation of free radicals/reactive oxygen species [121]. A recent study reveals that under graded oxidative stress in vitro, mitochondria isolated from the myocardium actively generate vesicles known as mitochondria-derived vesicles, which carry many mitochondrial proteins demonstrated by a proteomic analysis [122]. Interestingly, several of these mitochondrial proteins are also present in circulating EV isolated from the blood of patients with diseases associated with oxidative stress disorders [123]. Furthermore, mitochondrial DNA (mtDNA) in cardiac cells is not protected from oxidative stress damage during MI/RI, and mitochondria-derived vesicles can act as intermediaries, mediating the transfer of mtDNA into EVs [124]. Once cardiac cells release mtDNA into circulation during MI/RI, it can remain protected within EVs and travel through bloodstream to mediate the inflammatory response in the target organ [125]. Circulating mtDNA has been shown to activate TLR9 (toll-like receptor 9), promoting inflammation through activation of the myeloid differentiation primary response 88 (MyD88) and p38 MAPK (mitogen-activated protein kinase) pathways [126, 127]. Additionally, mtDNA activates NLRP3 inflammasomes, triggering the production of IL-1β and IL-18 [128, 129]. The mtDNA in EVs can potentially activate neuroinflammation in neurodegenerative diseases [125]. Interestingly, it was also demonstrated that injection of mtDNA into the hippocampus of mice induces neuroinflammation [130]. These finding also provides directions for future research. It has also been shown that MI/RI-induced EVs contribute to the dissemination of inflammation, as evidenced by the significant elevation of high mobility group box 1 (HMGB1), an injury-associated molecular pattern, in plasma after cardiac I/R [131]. These increased HMGB1 levels in reperfused coronary arteries mainly originate from platelet-derived particles and are predominant in patients with MI/RI injury presenting as no flow phenomenon [132].
These aforementioned pieces of evidence support the possibility that MI/RI triggers the production of EVs, which can transport harmful mediators (miRNAs, circRNAs, inflammatory proteins, cytokines, and mitochondrial components) through circulation into the brain, thereby activating neuroinflammation and resulting in neurocognitive impairment (Fig. 3). However, despite the burgeoning interest in EVs, there is currently insufficient data to evaluate the brain effects of EVs released into the circulation after MI/RI.
Indirect mechanisms of cognitive dysfunction due to altered cardiac function after MI/RI
One well-known consequence of MI/RI is the impairment of cardiac function. This can manifest as arrhythmias, heart failure, and myocardial infarction. A direct consequence of these manifestations is a decrease in cardiac output, which may lead to varying degrees of cerebral hypoperfusion. A study showed that MI/RI rats exhibited cardiac insufficiency and reduced cerebral blood flow in the accumbens core, left caudate putamen, hippocampus, left hypothalamus, olfactory, superior colliculus, right midbrain, ventral tegmental area, inferior colliculus and left thalamus whole [133]. The hypothesis that heart failure exacerbates cerebral blood flow (CBF) and contributes to cognitive decline is supported by existing literature [134]. However, it has been argued that lower CBF does not fully explain cognitive impairment in patients with hemodynamic dysfunction along the cardio-cerebral axis [135]. Nevertheless, the study conducted by the authors established a cross-sectional association between CBF and cognitive function in patients with heart failure, but not a longitudinal analysis. In patients undergoing CABG, postoperative changes in CBF are commonly associated with PND [136, 137]. On the other hand, there is inconsistent evidence suggesting that a significant reduction in CBF after CABG does not correlate directly with neuropsychological test results [138]. In fact, patients with PND showed an average decrease of approximately 7% in regional CBF (rCBF), while patients without PND exhibited an increase of about 2% in rCBF. These results did not reach statistical significance, possibly due to the small sample size and the large standard deviation. Additionally, ischemic heart disease is strongly linked to cognitive status after cardiac surgery [139], a factor not addressed in this study. In addition, inadequate cerebral perfusion leads to decreased cerebral oxygen saturation, which can be noninvasively measured using near-infrared spectroscopy. Studies suggest that long-term declines in regional cerebral oxygen saturation (rSO2) predict cognitive decline [140]. Though there are no long-term monitoring data on brain oxygen saturation after MI/RI, indirect evidence supports this theory. During CABG, intraoperative declines in brain oxygen saturation significantly correlate with early and late cognitive decline in patients [141, 142]. In addition, a study monitoring oxygen saturation after cardiac surgery found that decreased postoperative brain saturation was significantly associated with postoperative delirium [143]. Taken together, low cerebral perfusion or cerebral oxygen saturation is a risk factor for cognitive dysfunction resulting from impaired cardiac function. However, whether MI/RI specifically drives this phenomenon remains largely unknown and requires further investigation. Notably, it is essential to highlight that the cognitive outcome for patients undergoing On-Pump CABG is worse than that of those for patients undergoing Off-Pump CABG [144]. The acknowledged adverse effect of cardiopulmonary bypass (CBP) on cognition is partly attributed to reduced CBF [137]. This suggests that cognitive dysfunction after On-Pump CABG may be more related to CBP-induced cerebral hypoperfusion. Additionally, atrial fibrillation occurring after MI/RI may lead to micro-clots formation and occlude small cerebral vessels, potentially exacerbating cognitive dysfunction [145]. Although anesthesia also has the potential to influence cognitive function in cardiac surgery, it is not further discussed in our review, as our main focus is determining the role of MI/RI in postoperative cardiac cognitive dysfunction and elucidating potential mechanisms [146]. It is essential to emphasize that the mechanisms described above are indirect pathways through which MI/RI may contribute to cognitive dysfunction, and they are not specific to MI/RI. Future research is needed to establish causal evidence for these indirect mechanisms.
PREVENTION STRATEGIES FOR COGNITIVE DYSFUNCTION AFTER MI/RI
Research focused on preventing the cognitive changes associated with MI/RI is primarily driven by efforts to circumvention MI/RI. Although the precise mechanisms underlying MI/RI-related cognitive impairment remain incompletely understood, there are methods to prevent MI/RI partially and thereby address the fundamental cause of mental issues associated with it. Ischemic preconditioning (IPC), which involves subjecting the heart to brief ischemic stress, is the most effective intrinsic preventive measure against MI/R. IPC reduces subsequent MI/RI and mitigates the range of extracardiac injuries resulting from MI/RI [147]. However, the translational relevance of IPC is limited. Therefore, remote ischemic preconditioning (RIPC) has gained more interest, and it is a protective strategy focused on the circulation between ischemia and reperfusion within a specific vascular region (e.g., the arm) [148–150]. Heusch et al. comprehensively summarized the cardiac impact of RIPC in prior elective I/R studies (PCI and CABG). They provided compelling evidence supporting the beneficial effects of RIPC in mitigating MI/RI, particularly when applied to the upper limb [151]. Thus, RIPC holds promise as an approach to alleviate MI/RI-induced cognitive dysfunction.
Encouragingly, studies have demonstrated RIPC improved performance on behavioral tests in rats subjected to brain I/R and enhanced cognitive control in healthy adults [152, 153]. Investigation on the role of RIPC in MI/RI-induced cognitive dysfunction has yielded mixed results. One study reported that RIPC prevented postoperative decline in cognitive function over the short term in patients undergoing cardiac surgery, the majority of whom underwent surgery for CABG [154]. Three other studies observed a tendency of RIPC to attenuate MI/RI-induced neurocognitive outcomes, as indicated by reduced serum markers of brain damage and a non-significant decrease in incidence [155–157]. Contrastingly, two studies found no impact of RIPC on neurocognition [158, 159]. Notably, one of these studies did not specify the composition and proportion of cardiac surgery type performed on the patient, and surgery under propofol anesthesia was involved [159]. It is noteworthy that the occurrence and severity of MI/RI vary among different types of cardiac procedure, predominantly PCI and CABG, and multiple factors, including age, hypertension, beta-blocker, sex, diabetes, heart failure, and propofol, may influence the clinical utilization and effectiveness of RIPC [160]. Furthermore, propofol has been shown to diminish the protective effects of RIPC [161, 162]. In another study, cognitive assessment was conducted using a six-category test, and cognitive morbidity was used as an indicator [158]. Although the results suggested a lower cognitive decline in five out of the six categories among participants in the RIPC group compared to the control group, the overall prevalence rate of cognitive impairment was not significantly different or even higher in the RIPC group [158]. The methodology for calculating cognitive prevalence was not clearly defined in the study, and the interpretation of the results may be due to consideration of poor performance on any one of the six tests as cognitive impairment. Consequently, more individuals in the RIPC group have a lower level of impairment, while fewer people in the control group have a higher level of impairment. It remains uncertain whether the assessment of morbidity in this context provides an objective evaluation of the patient overall cognitive profile. The issue of confounding by procedure type was also present in the two studies that reported trends but lacked statistically significant differences, where valve placement was performed in 75% and 45% of the respective patients’ cohorts [156, 157].
The aforementioned unpromising results raise concern due to the heterogeneity of the patient population and potential bias introduced by variations in surgery type and anesthesia. The clinical translation of RIPC is also influenced by the number of cycles and the duration of I/R. Although most human studies on CABG utilize 3–4 cycles of I/R (5 min/5 min), the most common approach in PCI is three cycles of I/R (3 min/3 min). Nonetheless, 3–4 cycles of I/R (5 min/5 min) yield greater effectiveness in preventing MI/RI. It is disappointing that no studies to date have specifically examined the effect of RIPC on cognitive function after PCI, although such research is encouraged, given the lower confounding factors compared to CABG and the relevance to MI/RI. Furthermore, most of the studies primarily focus on short-term cognitive impairment, whereas investigating the long-term cognitive protective effects of RIPC is an important direction for future research.
In conclusion, the favorable cognitive outcomes observed in these studies enhance the credibility of RIPC as a preventive measure against MI/RI-induced cognitive decline. However, larger, standardized multicenter studies are still required to determine the extent to which RIPC translates into clinical benefit. It is important to emphasize that blood pressure control, glucose control, cerebral perfusion and cerebral oxygen saturation management, temperature management, use of some preoperative medications, preoperative cognitive training, avoidance of some anesthetics as well as CPB, etc. have the potential to prevent cognitive impairment after cardiac surgery [144, 163–168] However, the subject of our review is the relationship between MI/RI and cognitive dysfunction after cardiac surgery, thus highlighting the neuroprotection of ischemic preconditioning in MI/RI. However, we are in no way dismissing the role of other strategies in MI/RI-related cognitive deficits; moreover, the evidence for other strategies has been detailed in previous reviews [146, 169].
TREATMENTS OF COGNITIVE IMPAIRMENT DUE TO MI/RI
The post-ischemic treatment corresponding to ischemic preconditioning is ischemic postconditioning (I-postC), involving multiple brief reperfusion/ischemic treatments before sustained reperfusion following ischemia. Studies have shown the beneficial effect of I-postC on brain injury after MI/RI, partly through inducing glycogen synthase kinase-3 beta (GSK-3β) autophosphorylation [136]. However, similar to IPC, the clinical application of I-postC is not promising. Animal experiments have suggested alternative effective interventions for MI/RI-induced brain cognition, including electroacupuncture [65]. Extensive researchers have also explored the pharmacological treatment of brain injury after MI/RI, using inhibitors such as p-donepezil (a well-known inhibitor of acetylcholinesterase), and proprotein convertase subtilisin/kexin type 9 inhibitor, as well as various cell death inhibitors [19, 58] (Table 3). Neuroprotective and cytoprotective peptides like humanin and the pharmacological mitochondrial dynamics regulator mitochondrial fusion promoter (M1) have demonstrated the ability to reverse brain pathology caused by MI/RI [57, 96]. Additionally, evidence supports the effectiveness of metformin and sevoflurane, among other clinical agents [170–172]. Notably, gas anesthetics (desflurane, isoflurane, and sevoflurane) have been associated with better postoperative cognitive performance in CABG patients compared to propofol, suggesting a preference for gas anesthetics in CABG patients at risk for MI/RI impairment [172–174]. In summary, the current strategies for the treatment of MI/RI-induced brain injury are still in the early stage, calling for future attention and research in this area.
Treatment of brain injury and cognitive dysfunction following MI/RI
CLINICAL IMPLICATION
Previous studies have indicated that patients who undergo CABG may experience PND, as evidenced by biomarker changes similar to those seen in AD [176]. Additionally, mouse models have shown AD-like manifestations in brain pathology after MI/RI, suggesting a potential connection between MI/RI and AD. However, the etiology of postoperative cognitive decline in current clinical studies still does not take M/RI into account, so it is difficult to conclude the causal relationship between MI/RI and cognitive impairment from the limited clinical evidence available. Consequently, it is crucial to investigate the impact of MI/RI on cognitive dysfunction after cardiac surgery. Recognition of this role may promote the exploration of safe and non-invasive methods like IPC or RIPC for preventing and protecting against cognitive impairment after cardiac surgery. Additionally, studying the mechanisms by which MI/RI leads to cognitive impairment is essential. One promising avenue is the use of EVs that contain a significant amount of non-coding RNAs and serve as specific biomarkers for MI/RI-related cognitive decline [177]. Furthermore, EVs can potentially be both pathogenic and therapeutic mediators in cardiovascular diseases [178], and EVs inhibitors hold promise for various clinical applications [179]. In addition, actively improving cardiac function and reducing inflammation can help mitigate or prevent cognitive decline associated with MI/RI. In conclusion, this opens up new avenues for identifying and intervening in MI/RI-related cognitive impairment in clinical settings, providing valuable insights and directions.
SUMMARY AND OUTLOOK
MI/RI is common in CAD patients undergoing cardiac procedures, but the underlying mechanism linking MI/RI to brain pathologies remains unclear. Besides systemic inflammation, cardiac insufficiency following MI/RI is recognized as a factor contributing to cerebral under-perfusion and cognitive impairment. Based on recent clues, we hypothesize that releasing harmful molecules after MI/RI may induce brain pathology through circulation, either directly or within encapsulated EVs. Moreover, various pharmacologic treatments and interventions have shown promise in reducing brain pathology and improving cognition in animal models of MI/RI. Despite substantial research demonstrating the impact of MI/RI on cognitive impairment, studies in this area, particularly in the clinical setting, have been limited. Currently, clinical practice primarily focuses on myocardial injury factors such as creatine kinase-MB and cardiac troponin I, neglecting the significance of MI/RI for cognitive impairment. Thus, future studies should prioritize the development of markers for predicting MI/RI in clinical settings to provide direct evidence of its role in cognitive impairment. Mechanistically, there are gaps in the understanding of cognitive decline after MI/RI, as research has primarily focused on the initial event (MI/RI) and subsequent brain pathology without establishing a clear connection between them. Therefore, comprehensive investigations are needed to establish a cohesive pathogenesis narrative. In conclusion, the available evidence is preliminary, and mechanisms, preventive and therapeutic measures, as well as additional interventions, requires exploration and validation in in both clinical and basic aspects.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
This work was supported by the National Natural Science Foundation of China (No.82171185 and No.U23A20422 to Dr. Chan Chen); Central Guidance on Local Science and Technology Development Fund of Sichuan Province (No.23ZYZYTS0135 to Dr. Chan Chen); The Post-Doctor Research Project, West China Hospital, Sichuan University (No. 2021HXBH061 to Dr. Erya Chen); The Youth Project of Natural Science Foundation of Sichuan Provincial Department of Science and Technology (No. 2023NSFSC1563 to Dr. Erya Chen); The China Postdoctoral Science Foundation (No.2022M722254 to Dr. Erya Chen).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
