Abstract
Background:
There is evidence that aerobic exercise is beneficial for brain health, but these effects are variable between individuals and the underlying mechanisms that modulate these benefits remain unclear.
Objective:
We sought to characterize the acute physiological response of bioenergetic and neurotrophic blood biomarkers to exercise in cognitively healthy older adults, as well as relationships with brain blood flow.
Methods:
We measured exercise-induced changes in lactate, which has been linked to brain blood flow, as well brain-derived neurotrophic factor (BDNF), a neurotrophin related to brain health. We further quantified changes in brain blood flow using arterial spin labeling.
Results:
As expected, lactate and BDNF both changed with time post exercise. Intriguingly, there was a negative relationship between lactate response (area under the curve) and brain blood flow measured acutely following exercise. Finally, the BDNF response tracked strongly with change in platelet activation, providing evidence that platelet activation is an important mechanism for trophic-related exercise responses.
Conclusions:
Lactate and BDNF respond acutely to exercise, and the lactate response tracks with changes in brain blood flow. Further investigation into how these factors relate to brain health-related outcomes in exercise trials is warranted.
Keywords
INTRODUCTION
Recent meta-analyses have shown that exercise and physical activity are beneficial to the brain and may reduce risk for cognitive decline and dementia [1, 2]. However, clinical trials in cognitively healthy older adults and those with Alzheimer’s disease (AD) have shown the benefits of exercise programs vary amongst individuals [3, 4]. This individual variability in brain benefit may be due in part to differences in the physiological responses to each bout of acute exercise, including changes in circulating biomarkers. These biomarkers include bioenergetic substrates like glucose and lactate, as well as neurotrophic molecules, such as brain-derived neurotrophic factor (BDNF). However, the immediate response of these biomarkers to an acute exercise bout in older adults and their relationships to brain-relevant neurovascular and cognitive outcomes remains unclear.
Circulating biomarkers likely play an important role in coupling the peripheral and central responses to exercise. For instance, exercise is associated with increases in circulating lactate. The kinetics of lactate entry into the brain indicate that the blood-brain barrier is about half as permeable to lactate as glucose, but that intracellular uptake of lactate is greater than that of glucose [5]. During exercise, there is a switch from net release to net uptake of lactate by the brain [6], when lactate has been shown to account for over a third of brain energy use [7]. However, lactate’s role stems beyond that of an energy substrate—the lactate receptor GPR81/HCAR1 exists on brain vascular endothelial cells [8], and others have shown that lactate can augment cerebral blood flow [9]. Importantly, shear stress is a known factor that contributes to platelet activation [10] and acute BDNF release [11]. Although there is much to be learned about mechanisms, blood flow increases throughout the vascular system with exercise. We postulate that shear stress from increasing blood flow and resultant platelet activation may contribute to increases in BDNF following acute exercise. This increase in BDNF would occur wherever platelets were activated, and is relevant to brain health because peripherally released BDNF can readily cross the blood-brain barrier via a high capacity transport system [12]. In the brain, BDNF has been shown to play important roles in neurodevelopment and neuroprotection [13]. Increasing our understanding of the dynamic relationship between circulating lactate, changes in brain blood flow, and neurotrophic responses is critically important. This is especially true in older adults, where vascular health, physical activity, and cardiorespiratory fitness begin to decline more rapidly and may be more variable between participants.
Here, we present data to characterize the impact of acute exercise on lactate and BDNF, as well as the relationship between lactate and brain blood flow. We further describe changes in platelet activation as a potential mechanism of BDNF release. The goal of this characterization was to shed light on important factors that may explain differences in responses between individuals in exercise trials, which may underlie differences in benefit. We hypothesized that a bioenergetic stimulus (acute exercise) would stimulate the response of blood biomarkers such lactate and neurotrophins, while also modulating brain blood flow.
METHODS
We performed an ancillary analysis of data from 61 cognitively healthy individuals (60 + years old) who had undergone a single study visit to characterize the blood biomarker response to acute moderate intensity exercise, as well as the relationship between biomarker response and brain blood flow using magnetic resonance imaging (MRI). The protocol was approved by the Institutional Review Board at the University of Kansas Medical Center (#142822).
Fasting blood collection
Upon arrival to the study visit, whole blood was collected into vacutainer tubes containing acid citrate dextrose, aliquoted into cryotubes and stored at –80°C for apolipoprotein E4 (APOE4) genotyping as previously described [14]. Additional blood was collected into tubes containing EDTA as an anti-coagulant and processed to collect plasma and platelet poor plasma. Tubes were inverted 8× to mix and blood was centrifuged at 1500 × min at 4°C. Plasma was aliquoted into fresh tubes and frozen until further analysis. Additional plasma aliquots immediately underwent a subsequent centrifugation at 1700 × g for 15 min at 4°C to generate a platelet pellet. Supernatant was carefully collected and frozen in fresh tubes as platelet poor plasma.
Exercise-stimulated blood collection
All participants engaged in a single, discrete, bout of moderate intensity exercise (45–55% heart rate reserve): a 5-min warm-up, followed by 15 min of exercise, and a 3-min cool down. The full exercise protocol has been published previously [15]. Briefly, individuals reported to the Hoglund Brain Imaging Center, where they first completed approximately 20 min of cognitive testing using the NIH toolbox prior to entering the MRI suite. Vitals were obtained, and the resting MRI scan was completed. Individuals were then escorted to the room where cognitive testing occurred for resting phlebotomy and exercise. Following blood collection, age-predicted heart rate maximum was calculated to determine HR range. Each participant mounted a cycle ergometer and began a 5-min warm-up where resistance was gradually increased to achieve the HRR range (45–55% HRR) by minutes 4–5. We used the Karvonen formula to calculate HRR. Participants were asked to maintain a cadence of 60–70 rpm while HR was checked each minute and watts adjusted to maintain HR range. After 15 min, resistance was reduced to 10 watts and participants pedaled at their own pace for 3 min to cool down prior to obtaining the “immediate post” blood draw prior to the MRI scan (average 32.3 min (sd 10.2) following the start of exercise). An additional “delayed post” blood draw was performed an average of 68.8 min (sd 18.2) following the start of exercise. Both the immediate and delayed post draws were processed in an identical manner as the baseline blood draw. Blood pressure measurements (Welch Allyn ProBP3400) were taken prior to each blood draw while the participant was seated. Pulse pressure was computed as the difference between the systolic and diastolic values.
Blood biomarkers
Time course responses of BDNF (R&D Systems) and Platelet Factor 4 (PF4; Abcam), were measured using ELISA in platelet poor EDTA plasma. Glucose and lactate were measured in traditionally processed EDTA plasma (YSI 2500 Glucose and Lactate Analyzer). We further characterized baseline plasma biomarkers related to brain health, including amyloid-β 42, amyloid-β 40, neurofilament light (NfL), and glial fibrillary acidic protein (GFAP) in EDTA plasma using commercially available assays (Neuro 3 Plex A and Neuro 2 Plex B) on a Simoa HD-X (Quanterix).
Neuroimaging procedures
Neuroimaging data were collected with a 3-Tesla whole-body scanner (Siemens Skyra, Erlangen, Germany) fitted with a 20-channel head and neck receiver coil. The MRI session was split into two parts: pre-exercise and post-exercise. Prior to exercise, two 3D turbo gradient spin echo (TGSE) pseudo-continuous arterial spin labeling (pCASL) sequences were collected as previously described [14]. These sessions lasted were performed back-to-back and lasted approximately 6 min each for a total of 12 min. A magnetization prepared rapid gradient echo (MPRAGE) structural scan was collected following the pCASL sequences. Immediately following exercise, individuals underwent an additional 4 consecutive pCASL sequences lasting approximately 6 min each for a total of 24 min. Identical parameters were used for all pCASL sequences, which were designed to include the entire brain and optimized for older adults (repetition time/echo time (TR/TE)=4300/22.42 ms, flip angle = 120°, field of view (FOV)=300 × 300 mm2, matrix = 96 × 96 voxels, voxel in-plane resolution = 3.125 × 3.125 mm2, slice thickness = 2.5 mm, 48 axial slices, 20 volumes). Parameters for the MPRAGE were as follows, TR/TE = 2300/2.95 ms, inversion time (TI) = 900 ms, flip angle = 9°, FOV = 253×270 mm, matrix = 240×256 voxels, voxel in-plane resolution = 1.05×1.05 mm2, slice thickness = 1.2 mm, 176 sagittal slices, in-plane acceleration factor = 2, acquisition time = 5 : 09. We calculated mean cerebral blood flow post exercise as well as area under the curve (AUC) which spanned the resting and exercise conditions, which was the sum of the mean global cerebral blood flow over the entire duration of acquisition (mL*100 g tissue-1). AUC methods have been described previously [14]. Briefly, we used Loft Lab packages for quantification of cerebral blood flow (loft-lab.org). Statistical Parametric Mapping CAT12 was used to generate regions of interests for gray matter. For each of the 6 separate sequences (2 pre-exercise and 4 post-exercise), both labeled and control pCASL images were motion corrected prior to calculating cerebral blood flow, which produced a timeseries of 9 subtraction images for each sequence (56 cerebral blood flow estimates total).
Statistical analyses
Descriptive measures of the overall cohort, including means and standard deviations for continuous variables and frequencies for categorical measures were generated. For biomarker endpoints measured at the 3 timepoints in the overall group (baseline, immediate post-exercise, and delayed post-exercise), we performed linear mixed-effects analysis using R v4.1.3 and the lme4 package [16]. we used linear mixed models, with age, sex, and APOE4 as covariates. For assessment of biomarker responses as continuous measures, we calculated an area under the curve for each blood biomarker of interest. p-values for effects of interest were obtained by likelihood ratio tests of the full model against a reduced model without the effect of interest.
RESULTS
Participant characteristics for the entire sample are given in Table 1. Overall, this group of cognitively healthy older adults were normoglycemic with elevated blood pressure. On average, markers of neurodegeneration were normal given the age of the cohort [17]. In the overall cohort, linear mixed modeling performed with age, sex, and APOE4 genotype as covariates revealed a significant effect of time post exercise on lactate (p < 0.001, Fig. 1A) and BDNF (p < 0.001, Fig. 1B). Lactate peaked immediately post exercise, while BDNF elevations were greatest at the delayed post-exercise timepoint. There was also a significant effect of time post exercise on glucose (p = 0.001, Fig. 1C).
Participant characteristics
Values are mean (sd) unless categorical, which are given as n (%). APOE, apolipoprotein E; mmHg, millimeters of mercury; bpm, beats per minute; NfL, neurofilament light; GFAP, glial fibrillary acidic protein.
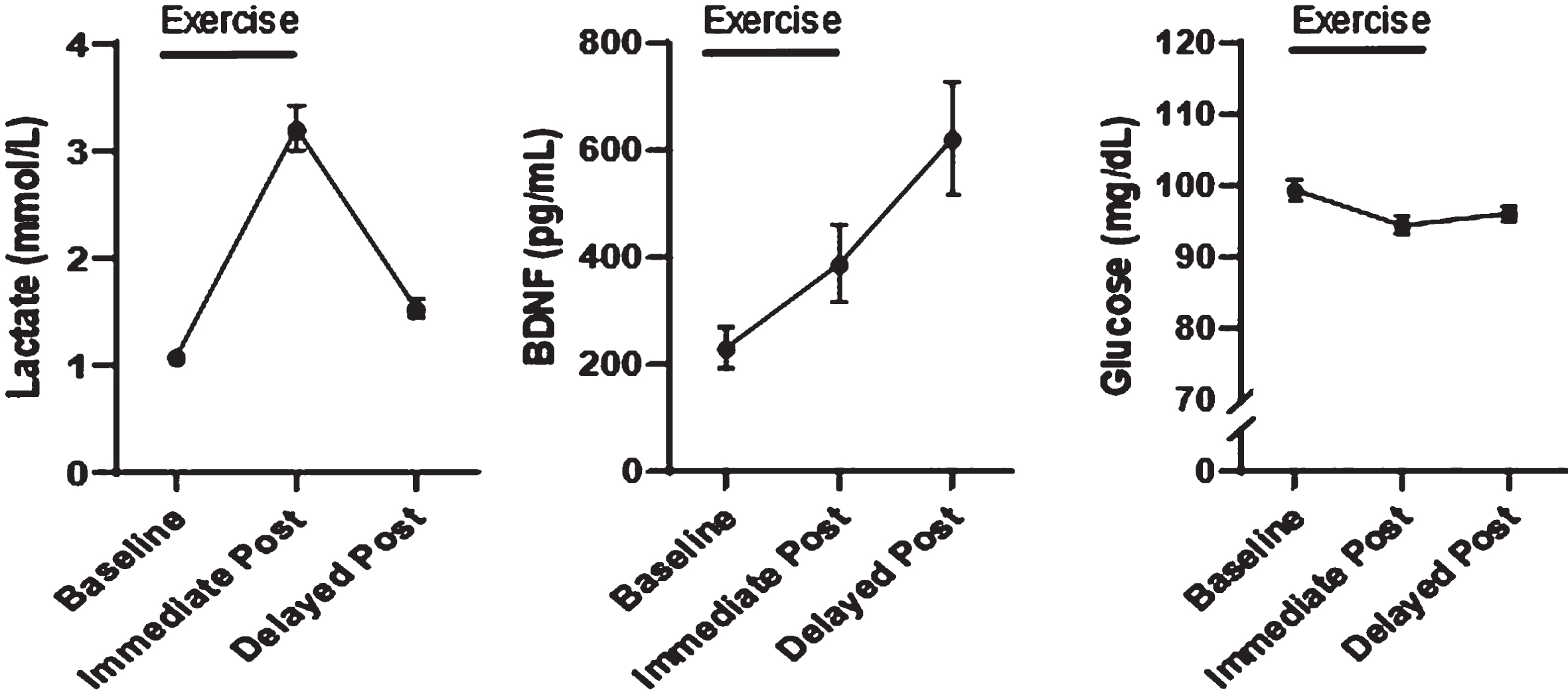
Metabolic and neurotrophic responses to acute exercise in older adults. A) Lactate, BDNF, and glucose change with time post exercise. Lactate peaks immediately post exercise (A), while BDNF continues to rise with time (B). Glucose levels drop post exercise (C). Values are plotted as means (SE).
Given that lactate is known to affect vascular outcomes through various mechanisms, we evaluated the relationship between exercise-stimulated lactate response (lactate AUC) and brain blood flow measured post-exercise. Lactate AUC was strongly and negatively associated with post exercise whole-brain blood flow (r2 = 0.523, p < 0.001; Fig. 2A). These relationships were consistent between brain regions (Fig. 2B-D2; precuneus r2 = 0.540, p < 0.001, Superior Parietal, r2 = 0.512, p < 0.001, Middle Frontal Cortex r2 = 0.498, p < 0.001). These relationships with Lactate AUC also held when white mater was examined (r2 = 0.353, p < 0.001; Supplementary Figure 1). Given that our lactate AUC contained resting values (0 timepoint) we also calculated a brain blood flow AUC value which incorporated values obtained in the resting state, prior to exercise, as well as post-exercise values. When we assessed the relationship between the brain blood flow AUC value and lactate AUC, we observed a similar relationship (r2 = 0.373, p < 0.001; Supplementary Figure 2). No relationship between BDNF AUC or Glucose AUC and brain blood flow post exercise was observed (data not shown).
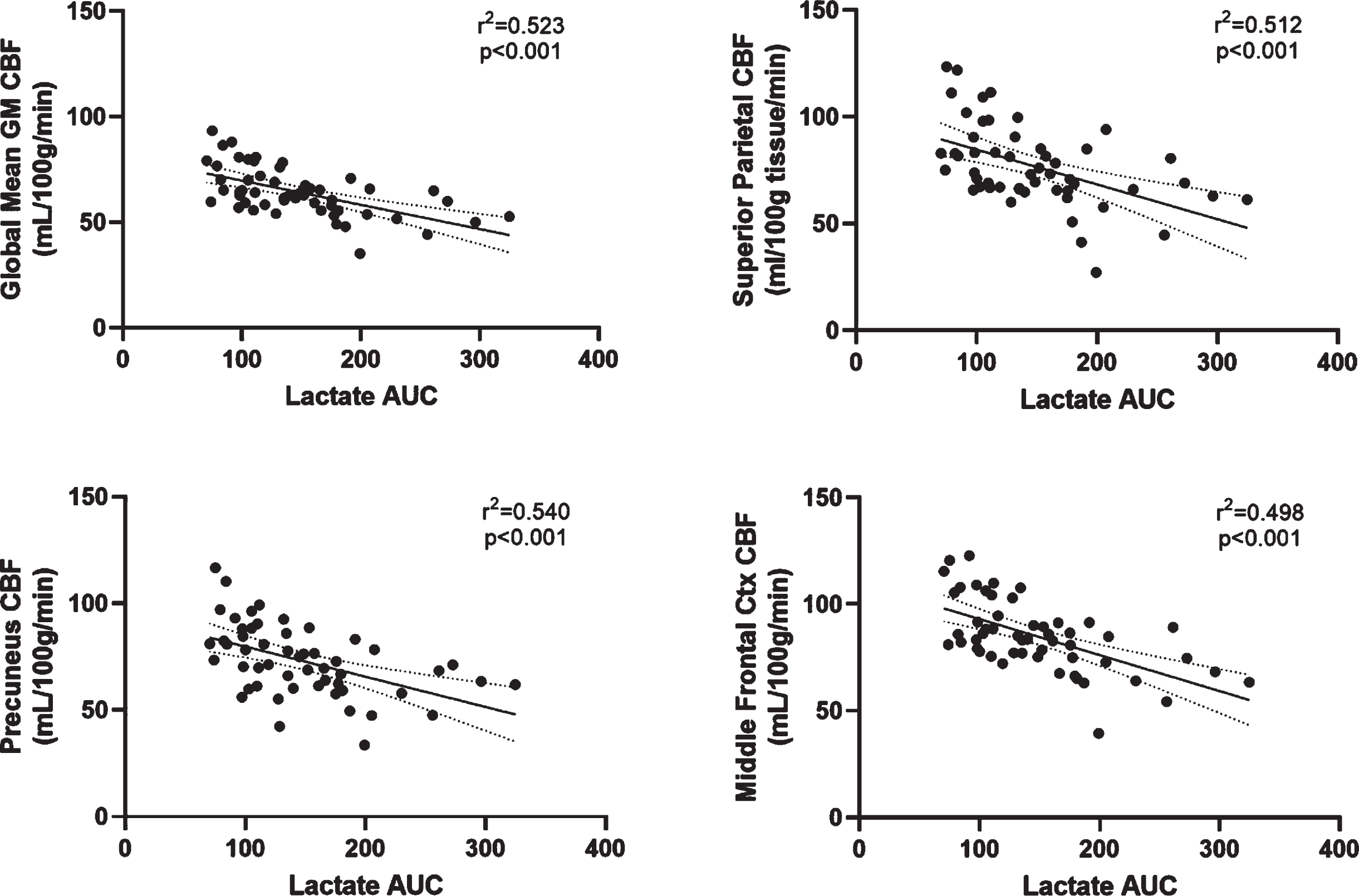
Post-exercise Cerebral Blood Flow tracks with lactate response. Striking relationships were observed between in global brain blood flow post exercise and lactate AUC (A) that were consistent within regions (B–D). CBF, cerebral blood flow; AUC, area under the curve. Scatterplots depict raw data values.
Because increased BDNF following acute exercise may be due to release from activated platelets, we also assessed the relationship between the BDNF response (BDNF AUC) and the response of platelet factor 4, a known marker of platelet activation (platelet factor 4 AUC). The relationship between change in BDNF and platelet factor 4 over the course of the experiment was positive (r2 = 0.530, p < 0.001; Fig. 3), suggesting that platelet activation is an important consideration in the BDNF response during acute exercise.
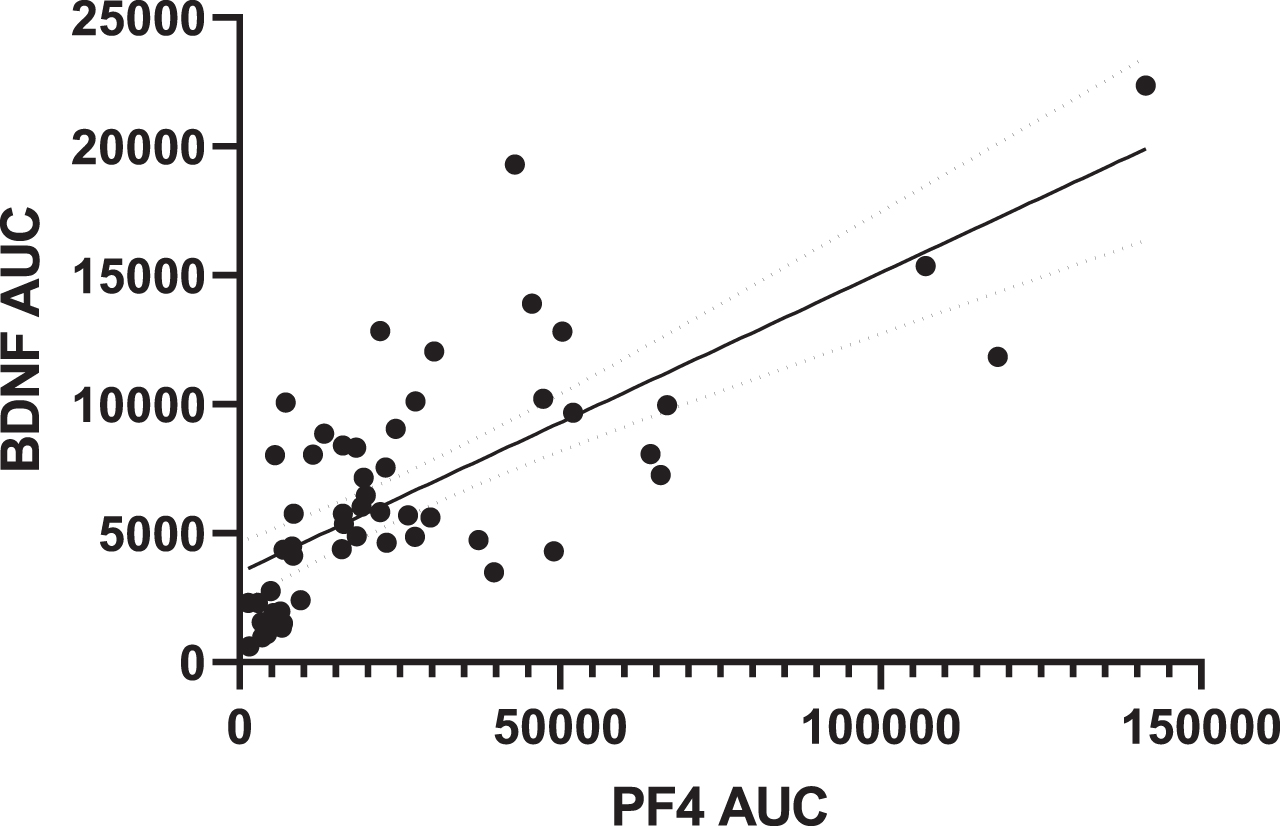
BDNF response tracks positively with response of platelet factor 4. The BDNF response tracks positively with changes in a marker of platelet activation, PF4. BDNF, brain-derived neurotrophic factor; PF4, platelet factor 4; AUC, area under thecurve.
There were no relationships between the acute lactate, BDNF, or glucose responses and change in cognitive performance measures in any domain (attention, memory, or processing speed) pre-post exercise (data not shown). The flow of the experiment is shown in Fig. 4.
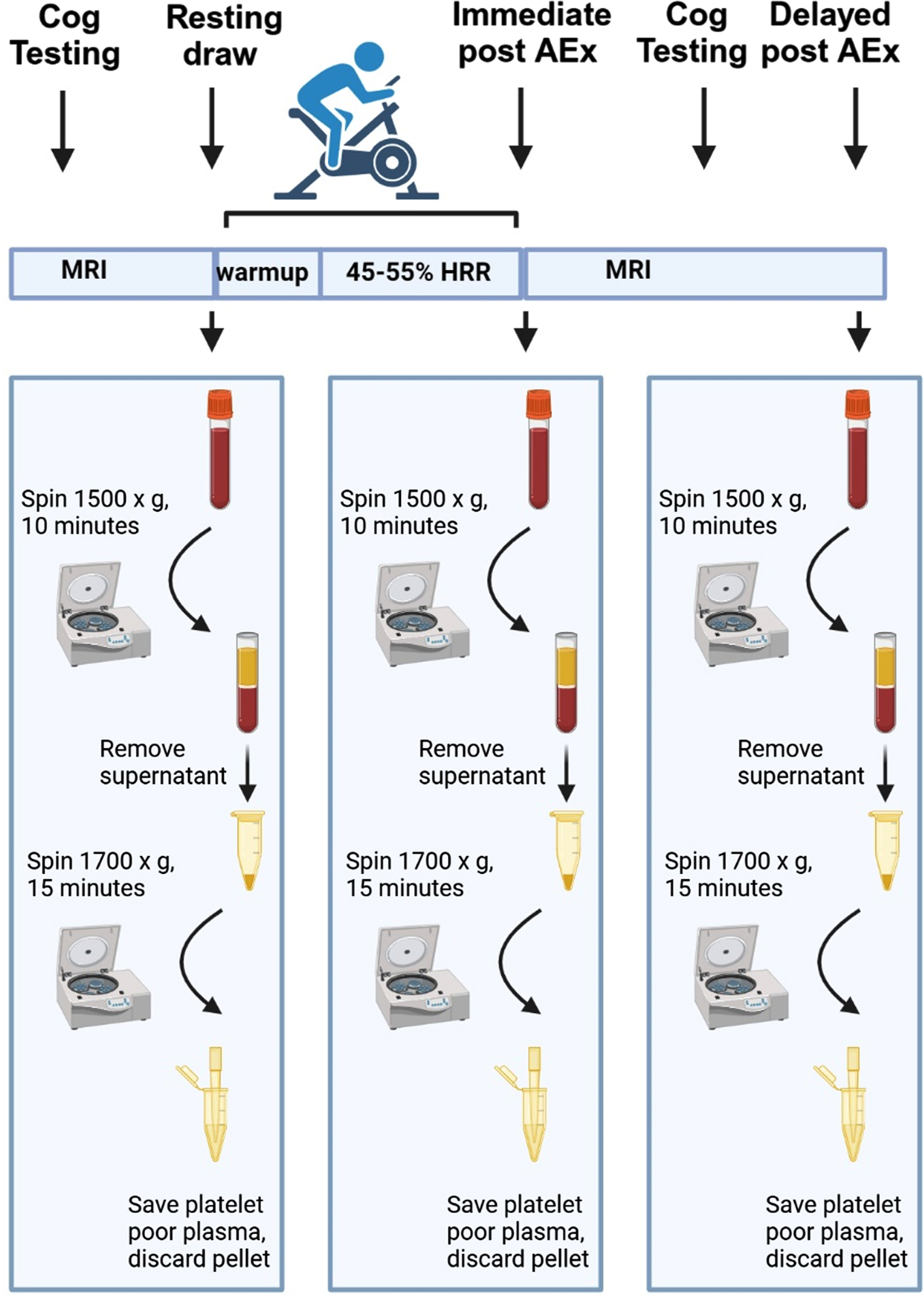
Schematic of exercise timings, blood draws and processing. Study flow in response to acute exercise and blood draws. Blood was immediately processed upon draw for both plasma and platelet poor plasma, then frozen and stored at –80°C prior to analysis. All processing steps occurred at 4°C. Immediate post draw occurred on average approximately 32 min post-exercise start, while the delayed post draw occurred on average approximately 69 min post-exercise start. Figure created with Biorender.com.
DISCUSSION
Given the variability in the literature regarding the brain benefits of exercise, it is imperative to understand the mechanisms that may dictate benefits, to ensure that future trials maximally engage these responses. Here, we characterize the acute responses of bioenergetic and neurotropic markers to acute exercise in cognitively healthy older adults. Our main finding of interest is that exercise significantly affected these biomarkers, but individual responses were variable and there were differential relationships of these biomarkers with brain blood flow.
Lactate and glucose are the most abundant carbon carriers in mammals. Lactate concentrations are known to increase acutely after exercise, although these increases are different between individuals due to factors such as lactate production and turnover. For decades, lactate was considered to be primarily a waste product, but it is now recognized that lactate serves as both a major energy source and a signaling molecule [18]. As expected, we observed a significant increase in lactate post exercise, with levels falling closer to baseline by the post-exercise timepoint. The calculation of AUC provides a metric that provides insight into the systemic exposure to our biomarkers of interest, including both production and clearance. Using this metric, we observed a striking relationship between the physiological lactate response and brain blood flow post exercise.
We previously showed that in this group of individuals, brain blood flow increased overall post-exercise [14]. However, large differences were observed between individuals, so we examined additional mechanisms that may explain this variability. Our most striking and somewhat surprising observation was a strong relationship between lactate response and brain blood flow. There are several potential mechanisms that may explain this link. For instance, nitric oxide synthase is expressed in muscle [19] and increases following exercise. In addition, lactate readily enters the brain, where it has been shown to locally reduce the clearance of prostaglandins released from astrocytes [11]. Finally, pH decreases due to ATP hydrolysis and other factors also have been shown to affect vascular tone [20]. However, those processes should promote vasodilation, and the directionality of the relationship we observed was opposite of what would be expected from these mechanisms. It is thus important to consider that most prior studies were performed in cellular and animal models, while whole-body lactate dynamics are more complex. Lactate uptake and clearance are important cellular functions which improve with training and would be reflected in a decreased AUC value [21]. This holds for individuals with disease; in subjects with chronic heart failure, for instance, lactate AUC significantly decreased with a 3-month exercise intervention, and was a sensitive marker for functional work capacity [22]. It is possible that higher AUC levels in individuals with lower post-exercise blood flow actually reflect poorer lactate systemic lactate metabolism (i.e., poorer clearance, turnover), potentially as a function of muscle mass, fitness level, etc. These potential mechanistic relationships warrant further investigation.
We also observed changes in glucose levels with time post exercise. This is not surprising, given that it has been known for many years that exercise is an effective way to facilitate glucose uptake [23–25]. Glucose dynamics during acute exercise are complex, and factors such as hepatic glucose production, glucose delivery to muscle, and glucose uptake can all affect concentrations [26]. Unlike lactate, glucose did not track with changes in brain blood flow, suggesting an effect unique to lactate.
Increased BDNF levels are thought to be beneficial to the brain, and BDNF increases following exercise have been observed in young individuals [27]. Given BDNF’s neuroprotective qualities, it is imperative to understand the factors that may affect its production and release. Importantly, serum BDNF has been linked to brain volume in cognitively healthy older adults [28] and brain connectivity in individuals who underwent an exercise intervention [29]. However, not all studies are consistent, with other showing that BDNF does not show a relationship with brain volume [30]. One potential contributor to inconsistent findings is the measurement of BDNF in serum, which contains BDNF levels that are up to 100 times higher than that of plasma due to release of BDNF from activated platelets during the clotting process [31]. Individual differences in the acute responses of physiological mediators such as BDNF to health interventions are an important but poorly understand factor to mediating long term benefits of such interventions, and careful measurement of these outcome measures is also an important consideration during analysis.
Here, we show that increases in BDNF levels are detectable following exercise in platelet poor plasma. Blood platelets are an important source of circulating BDNF [32]—about half of platelet BDNF is released upon platelet stimulation [33]. Platelets have multiple BDNF pools, with BDNF stored in alpha granules responsive to activation [34, 35]. It has been shown that mature BDNF is found in these alpha granules, which can account for as much as 70% of circulating BDNF [36]. Exercise related factors, such as increased blood flow, can increase shear stress and platelet activation during exercise. Because we show that BDNF response tracks with platelet activation, this could be an important mechanism of exercise related benefits. However, because post processing factors could potentially mask subtle changes in BDNF from being detected in intervention studies, we also suggest that future clinical trials should give thought to blood processing considerations and consider what type of fluid is being measured (i.e., plasma, platelet poor plasma, or serum) when making conclusions.
Interestingly, BDNF itself can further increase platelet activation, and in vitro studies suggest that exposure to BDNF can increase the release of angiogenic factors such as vascular endothelial growth factor (VEGF) [37]. It is also important to consider that chronically, other circulating factors may also play a role in potentiating the acute BDNF response, such as lactate [38]. It has been shown that, compared to saline, lactate injection in mice increased hippocampal gene expression of bdnf, as well as hippocampal levels of SIRT1, PGC1 alpha, and FNDC5, suggesting a potential pathway to mediate these effects [39]. Moreover, studies in human cell lines suggest that short-term lactate exposure increases BDNF expression in both cortical astrocytes and SY5Y cells [40]. Over the long term, exercise-stimulated increases in lactate may affect BDNF production [38]. Lactate has been linked to both BDNF and VEGF levels following chronic exercise [41, 42], although it is unclear whether lactate drives these responses, and we did not find a relationship between circulating lactate and BDNF. It is possible that acutely, platelet-mediated BDNF release is the primary driver of circulating concentrations, while chronically, lactate-stimulated increases in BDNF transcription will increase the pool of BDNF available for release, measurable only over an extended training period of weeks or months. The increase in lactate concentration immediately post exercise was highly variable between individuals (Supplementary Figure 3) and may be an important factor when considering the long-term effects of physiological responses to exercise.
Our study has strengths and weaknesses. We performed protocol optimization when designing this study [15] and found that BDNF was both higher in plasma collected without platelet removal and affected substantially by sitting time following centrifugation. Thus, we performed all steps as quickly and consistently as possible. A major strength of our study was the generation of platelet poor plasma, which has not been extensively studied in acute exercise. However, there is no easy way to tell the difference between platelet activation due to post processing procedures and activation due to exercise in the body. We also had limited vascular risk factor information and analysis of factors that may underly differences in shear stress or brain blood flow (such as nitric oxide). An additional limitation is that we did not collect baseline measures of physical activity or cardiorespiratory fitness measures, so we are unable to evaluate the effects of these factors on biomarker responses. We also were not powered to detect sex differences. The relationship between those outcomes and acute biomarker responses during exercise warrants further investigation.
Conclusion
We report a significant increase in circulating lactate and BDNF, as well as decreased blood glucose, following a relatively brief, discrete bout of aerobic exercise. There was a significant relationship between individual lactate responses and brain blood flow in the both the whole brain and within discrete regions of interest. Finally, the BDNF response tracked with platelet activation, providing important information regarding potential mechanisms of the acute BDNF response to exercise. This suggests that lactate response may be an important factor to consider when targeting brain neuroimaging outcomes such as cerebral blood flow. Additional work is needed to determine whether lactate response could be a surrogate biomarker that should be measured at multiple timepoints during an exercise intervention and used to adjust intensity or duration of exercise and potentially modulate benefit. Further investigation into the mechanisms that contribute to these changes and the relevance to additional fluid and imaging brain health biomarkers in this cohort iswarranted.
Footnotes
ACKNOWLEDGMENTS
We would like to gratefully acknowledge the research volunteers for their time and participation in our study.
FUNDING
This study was supported by NIH R21 AG061548 and R01 AG070036 (EDV), R01 AG062548 and R01 AG081304 (JKM), and P30 AG072973. Support was also provided by the Leo and Anne Albert Charitable Trust and the Margaret “Peg” McLaughlin and Lydia A. Walker Opportunity Fund.
CONFLICT OF INTEREST
Dr. Sandy Billinger is an Editorial Board Member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer-review.
DATA AVAILABILITY
All data will be made available in the Harvard Dataverse website upon publication.
