Abstract
Background:
Gut microbiota could affect the onset and development of vascular cognitive impairment (VCI) through modulating metabolic and immune pathways. However, the vascular mechanisms involved remain unclear.
Objective:
To investigate the gut microbiota associated with VCI and examine the mediating effects of regional cerebral blood flow (CBF) to explore potential therapeutic targets for VCI.
Methods:
This prospective study enrolled patients with VCI (n = 16) and healthy controls (n = 18) from the Chinese Imaging, Biomarkers, and Lifestyle study between January 1 and June 30, 2022. The gut microbiota composition and diversity were determined by 16 S ribosomal RNA gene sequencing. The association between gut microbiota and Montreal Cognitive Assessment (MoCA) scores was determined using Spearman’s correlation analysis. Regional CBF was calculated using pseudo-continuous arterial spin labeling. The mediating effects of regional CBF on the relationship between specific gut microbiota and cognition in VCI were investigated using mediation analysis.
Results:
Compared to healthy controls, patients with VCI had significantly greater abundance of Bifidobacterium, Veillonella, R uminococcus gnavus , Fusobacterium, and Erysipelatoclostridium and smaller abundance of Collinsella. The abundance of Ruminococcus gnavus was negatively associated with MoCA scores in patients with VCI, with the CBF in the left hypothalamus, right hypothalamus, and left amygdala accounting for 63.96%, 48.22%, and 36.51%, respectively, of this association after adjusting for confounders.
Conclusions:
Ruminococcus gnavus is associated with cognition in VCI, which is strongly mediated by CBF in the bilateral hypothalamus and left amygdala. These findings highlight the potential regulatory roles of nutrition and metabolism-related areas of the brain in VCI.
Keywords
INTRODUCTION
Vascular cognitive impairment (VCI) refers to a group of clinical syndromes in which at least one cognitive domain is damaged by clinical stroke or subclinical vascular injury [1]. With the changing lifestyles of the global population, vascular risk factors are becoming more prevalent in the rapidly growing aging population, and the incidence and disability rates of VCI are increasing [2]. VCI is the second most common type of dementia, accounting for approximately 15% –30% of cases [3]. It negatively affects quality of life and increases the burden on society and families [4, 5]. Despite its increasing prevalence worldwide, the diagnosis and management of VCI remain inconclusive [6]. Moreover, there is limited understanding of the underlying pathophysiology of VCI, although factors such as inflammation and endothelial dysfunction have been found to play a role [7]. Therefore, it is crucial to identify early diagnostic biomarkers and explore the potential mechanisms involved in the development of VCI to slow its progression.
The microbiota–gut–brain axis has recently received attention as an essential pathway in the development of VCI, suggesting that gut microbiota (GM) can affect brain cognition through neural, immune, endocrine, and metabolic pathways [8, 9]. Recently, a study reported that dysbiosis of several GM, including Enterobacteriaceae and Proteobacteria and their metabolites, could be linked to VCI and useful for distinguishing patients with VCI from normal controls [10]. However, the potential mechanisms behind the effects of GM on cognition, neuropsychiatric symptoms, and the development of VCI remain to be elucidated.
A recent review suggested that VCI was characterized by cerebral hemodynamic alterations ranging from decreased cerebral blood flow (CBF) to small vessel disease and involved a multifactorial process that led to demyelination and gliosis, including blood-brain barrier disruptions, hypoxia, hypoperfusion, oxidative stress, neuroinflammation, alterations in the neurovascular unit coupling, cerebral microbleeds, or superficial siderosis [11]. A retrospective study of 108 patients with ischemic cerebrovascular disease demonstrated that non-invasive 3D arterial spin labeling (ASL) imaging could be used to detect cerebral hemodynamics and predict prognosis in patients with VCI [12]. Another observational study using 3D pseudo-continuous ASL (pCASL) found that deficits in brain perfusion in the hippocampus, thalamus, and insula were related to the degree of cognitive impairment in patients with VCI [13]. However, evidence from recent research is limited, and findings have been inconsistent. Moreover, it remains unclear whether changes in GM affect cognition in VCI through the regulation of specific brain areas. Thus, this study aimed to investigate the relationship between specific GM and cognition in patients with VCI and to examine whether this association is mediated by regional CBF to explore the potential mechanisms of and intervention targets for VCI.
METHODS
Dataset and study design
This study enrolled consecutive patients with VCI and healthy controls (HCs) from the Chinese Imaging, Biomarkers, and Lifestyle (CIBL) Study between January 1 and June 30, 2022. The CIBL study has been described previously [14], which was approved by the Institutional Review Board of the Beijing Tiantan Hospital of Capital Medical University (KY-2021-028-01) and registered at Chictr.org.cn (ChiCTR2100049131). All participants or their relatives provided informed consent before enrollment.
The inclusion criteria for patients with VCI were as follows: 1) patients were 60–85 years old; 2) patients met the 2018 standardized diagnosis of VCI guidelines [15]; 3) objective cognitive impairment was identified by Montreal Cognitive Assessment (MoCA) scores: illiteracy≤13 points; 1–6 years of education≤19 points; at least 7 years of education≤24 points; 4) patients had evidence of vascular brain injury (moderate to severe vascular brain injury on MRI defined as WMH Fazekas score > 1 and/or lacunar infarcts and/or intracerebral microhemorrhages, or mild vascular brain injury defined as WMH Fazekas score 1); 5) vascular injury was clearly a dominant factor affecting patients’ cognitive impairment. Excluded patients 1) had immediate and/or delayed cognitive decline that began within 6 months after a stroke and that does not reverse; 2) stroke within the previous 6 months and displayed moderate or severe stenosis in large vessels by MR angiography; 3) cognitive impairment caused by other neurodegenerative diseases including Alzheimer’s disease (AD), frontotemporal dementia, dementia with Lewy bodies, etc., intracranial tumors, or traumatic brain injury; 4) had history of a mental disorder in accordance with the Diagnostic and Statistical Manual of Mental Disorders-5 [16]; 5) taken antibiotics or probiotics, or other drugs that may affect gut microbiota, within one month before examination; and 6) an inability to cooperate to complete the data collection.
Healthy older participants from the CIBL cohort were recruited from the community and served as the HC group. These healthy individuals had no history of major medical, neurological, or psychiatric illnesses, and no apparent abnormal findings or cognitive impairments were identified by MRI or neuropsychological testing, and did not take antibiotics or probiotics, or other drugs that may affect gut microbiota, within one month before examination. A flowchart of the participant selection process is shown in Fig. 1.
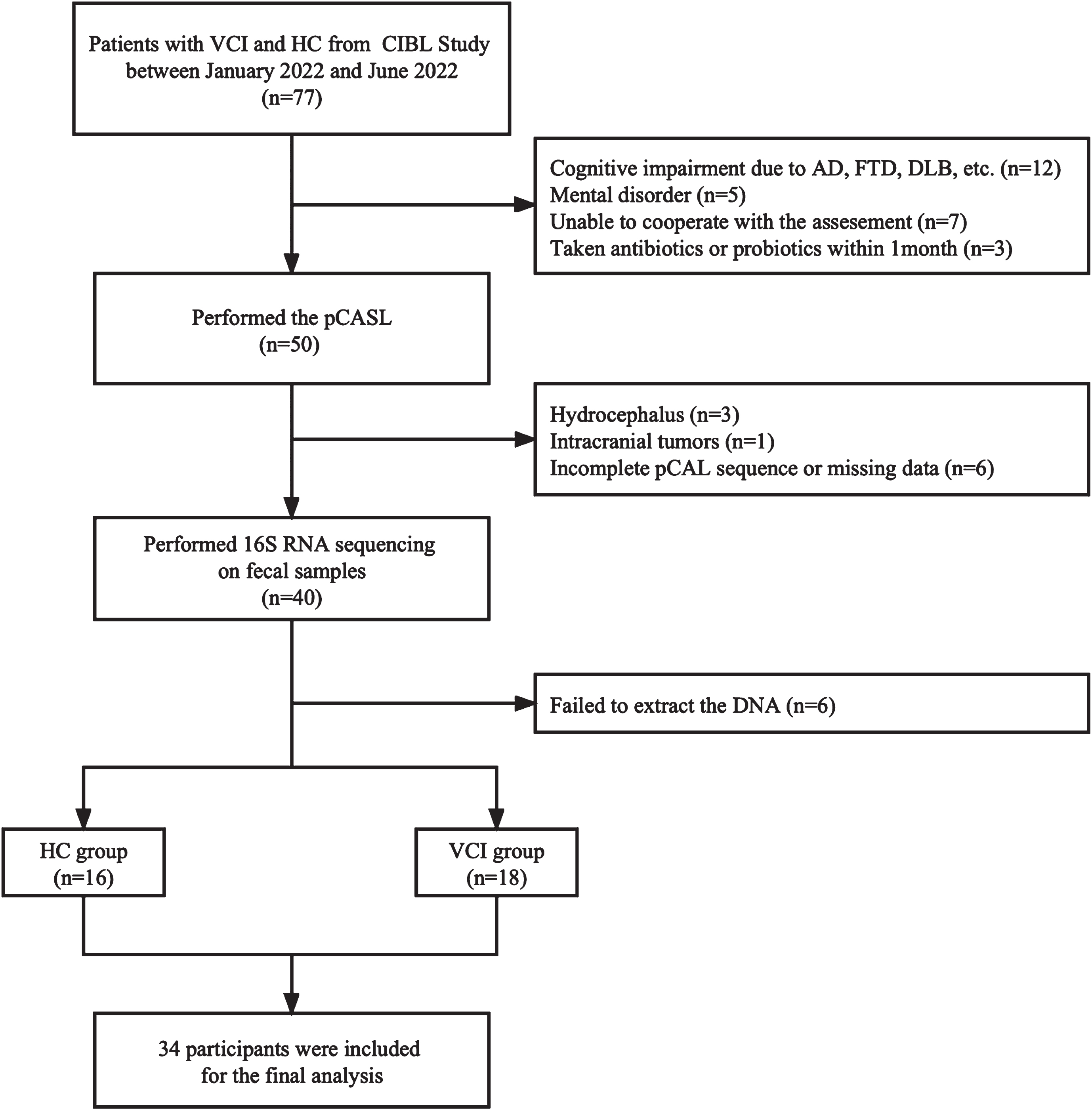
A flowchart of the inclusion and exclusion processes. VCI, vascular cognitive impairment; HC, healthy control; AD, Alzheimer’s dementia; FTD, frontotemporal dementia; DLB, dementia with Lewy bodies; pCASL, pseudo-continuous arterial spin labeling; RNA, ribose nucleic acid; DNA, deoxyribonucleic acid.
Data collection and neuropsychological assessments
Information on the patient’s age, sex, years of education, body mass index (BMI), diagnoses, and medical history, including hypertension, diabetes, coronary heart disease, smoking, drinking, and family history of dementia, were collected. Global cognitive function was evaluated using the MoCA, the severity of depression and anxiety were assessed using the Hamilton Depression Rating Scale (HAMD) and Hamilton Anxiety Rating Scale (HAMA), and the nutritional status and dietary habits were assessed using Mini Nutritional Assessment (MNA) and Dietary Diversity Score (DDS), respectively.
Fecal sample collection and 16 S ribosomal RNA (rRNA) sequencing
Fecal samples were collected on the morning of admission. Fresh fecal samples were immediately provided to doctors or nurses and collected in sterile tubes. These samples were stored at –80°C prior to microbial analysis.
16S rRNA gene sequencing was performed using an Illumina MiSeq PE300 platform (Shanghai Majorbio Bio-Pharm Technology Co., Ltd., Shanghai, China). Bacterial genomic DNA was extracted using an E.Z.N.A. Soil DNA Kit (Omega BioTek, Norcross, GA, USA). The V3–V4 hypervariable regions of the bacterial 16S rRNA genes were amplified using the forward primer 338F (5′-ACTCCTACGGGAGGCAGCAG-3′) and the reverse primer 806R (5′-GGACTACVVGGGTATCTAATC-3′) in a thermocycler polymerase chain reaction (PCR) system. All PCR amplicons were mixed and sequenced using an ABI GeneAmp 9700 PCR thermocycler (Applied Biosystems, Waltham, MA, USA). To study the constituent diversity of the species in the samples, the effective sequences of all samples were clustered into operational taxonomic units with 97% similarity. Shannon’s diversity index, the Chao1 richness estimate, the abundance-based coverage estimator index, and Simpson’s Diversity Index were used to assess diversity. Partial least squares discriminant analysis and principal coordinate analysis based on the weighted UniFrac algorithm were used for data analysis.
MRI acquisition and processing
Subjects were instructed not to use alcohol, caffeine, and nicotine for at least six hours prior to MRI measurement. Image acquisition was performed on a 3-T magnetic resonance imaging scanner (SIGNA Premier; GE Healthcare, Chicago, IL, USA) with a 48-channel head coil. High-resolution 3D T1-weighted scans were performed using the magnetization-prepared rapid gradient echo sequence: repetition time = 7.3 ms, echo time = 3.0 ms; flip angle = 12°, field of view = 256 × 256 mm2, acquisition matrix = 256 × 256, slice thickness = 1.0 mm, slice number = 176, and scan time = 4 min 56 s. ASL was performed using a 3D pCASL sequence: axial acquisition, repetition time = 4849 ms, echo time = 10.6 ms, field of view = 220×220 mm, acquisition matrix = 512×512, slice thickness = 4 mm, slice number = 36, and post-labeling delay = 2025 ms. Brain MRI examination, including T2-weighted images, fluid-attenuated inversion recovery, pCASL, diffusion weighted imaging, and susceptibility-weighted imaging were performed for all the participants in this study. The imaging results were independently interpreted by a neurologist and a radiologist to achieve consistency, if their interpretations were inconsistent, they will subsequently be determined by a higher-level expert.
PCASL Data processing was performed using CereFlow software (Anying Technology [Beijing] Co., Ltd., China), with the following steps: 1) CBF was calculated from the ASL proton density images and perfusion-weighted images using the standard simple compartment model with the assumption that the arterial transit time was equivalent to the post-labeling delay; 2) the M0 image (obtained with pCASL acquisition within the same image space as the CBF) was matched with the anatomical T1-weighted image; 3) the T1-weighted images were normalized to the Montreal Neurological Institute template; 4) the forward transformation matrix derived from T1-weighted images was used to warp the CBF image into the Montreal Neurological Institute space; 5) the regional CBF was extracted using the automated anatomical labeling atlas. Six brain areas involved with eating, metabolism, and inflammation were analyzed, including the bilateral hypothalamus, amygdala, and insula [17–19].
Statistical analysis
All statistical analyses were performed using SPSS statistical software (v26.0; IBM Corp., Armonk, NY, USA). Categorical variables are presented as the total number (n) and percentage (%) per group, and the chi-squared or Fisher’s exact test was used to assess statistical differences. The mean and standard deviation were calculated for continuous variables with normal distributions, whereas the median and interquartile range were used for continuous variables with non-normal distributions. Similarly, the independent t-tests were used for normally distributed data, whereas the Mann–Whitney U test was used for non-normally distributed data. The Wilcoxon Rank Sum test was used at the genus level to determine changes in the abundance of GM in both groups. The false discovery rate approach was used to adjust for multiple tests. Spearman’s correlation analysis was performed to examine the association between GM abundance and MoCA scores in patients with VCI.
Mediation models were developed using Model 4 in PROCESS macro (v3.5) with a 95% confidence interval and 5,000 bootstrap samples to analyze the effects of regional CBF on the association between the abundance of Ruminococcus gnavus and MoCA scores [20]. The total effect (TE) was decomposed into two components: the natural direct effect (NDE) and the natural indirect effect (NIE). The NDE represented the effect of Ruminococcus gnavus on MoCA scores that were independent of CBF. The NIE represented the effects of Ruminococcus gnavus on MoCA scores that could be explained by changes in regional CBF. The mediating effect was considered significant if the 95% confidence interval (CI) was not zero. The percentage of the mediated effect was calculated using the following formula: (NIE/TE)×100. Under this framework, three multivariate linear regression models were fitted: one model for MoCA scores (outcome) on Ruminococcus gnavus (exposure) and all study confounders, one model for regional CBF (mediator) conditional on the abundance of Ruminococcus gnavus (exposure) and all study confounders, and one model for MoCA scores (outcome) conditional on the abundance of Ruminococcus gnavus, regional CBF, and all study confounders. Confounders included age and the CBF of the whole brain, which was found in the univariate analysis. Iterative reweighted least squares algorithm was used to evaluate sensitivity and robustness of the model confounders. The threshold for statistical significance was set at a two-tailed p < 0.05.
RESULTS
Baseline characteristics of clinical and regional CBF in both groups
The demographic and neuropsychological characteristics of the 16 patients with VCI and the 18 HCs are shown in Table 1. Patients with VCI were older (p = 0.026) and had lower MoCA scores (p < 0.001) than HCs. There were no significant differences between the two groups in terms of sex, years of education, BMI, hypertension, diabetes, coronary heart disease, family history of dementia, HAMD scores, HAMA scores, MNA scores, or DDS scores. (all p > 0.05).
The regional CBF values of the six brain areas, namely the bilateral hypothalamus, amygdala, and insula, and the whole brain were significantly lower in the VCI group than in the HC group (all p < 0.001; Table 1).
Baseline characteristics of clinical and regional CBF in both groups
Data were evaluated using the χ2 or Mann–Whitney U test. ap values were evaluated using Fisher exact tests. HC, healthy control; VCI, vascular cognitive impairment; BMI, body mass index; MoCA, Montreal cognitive assessment; HAMD, Hamilton depression rating scale; HAMA, Hamilton anxiety rating scale; CBF, cerebral blood flow; MNA, Mini Nutritional Assessment; DDS, dietary diversity scores.
GM diversity and composition
The β-diversity of GM was higher in patients with VCI than in HCs in the partial least squares discriminant analysis model (Fig. 2B), whereas there was no significant difference between the two groups in the principal coordinate analysis model (Fig. 2A). There was no significant difference in α-diversity between the two groups (all p > 0.05, Fig. 2C–F). At the genus level, the abundance of Bifidobacterium (p = 0.023), Veillonella (p = 0.022), Ruminococcus gnavus (p = 0.002), Fusobacterium (p = 0.026), and Erysipelatoclostridium (p = 0.021) was greater in patients with VCI than in HCs, whereas the abundance of Collinsella (p = 0.002) was smaller in patients with VCI than in HCs (Fig. 2G, H). There was no significant difference in the abundance of GM at the phylum level between the two groups (all p > 0.05).
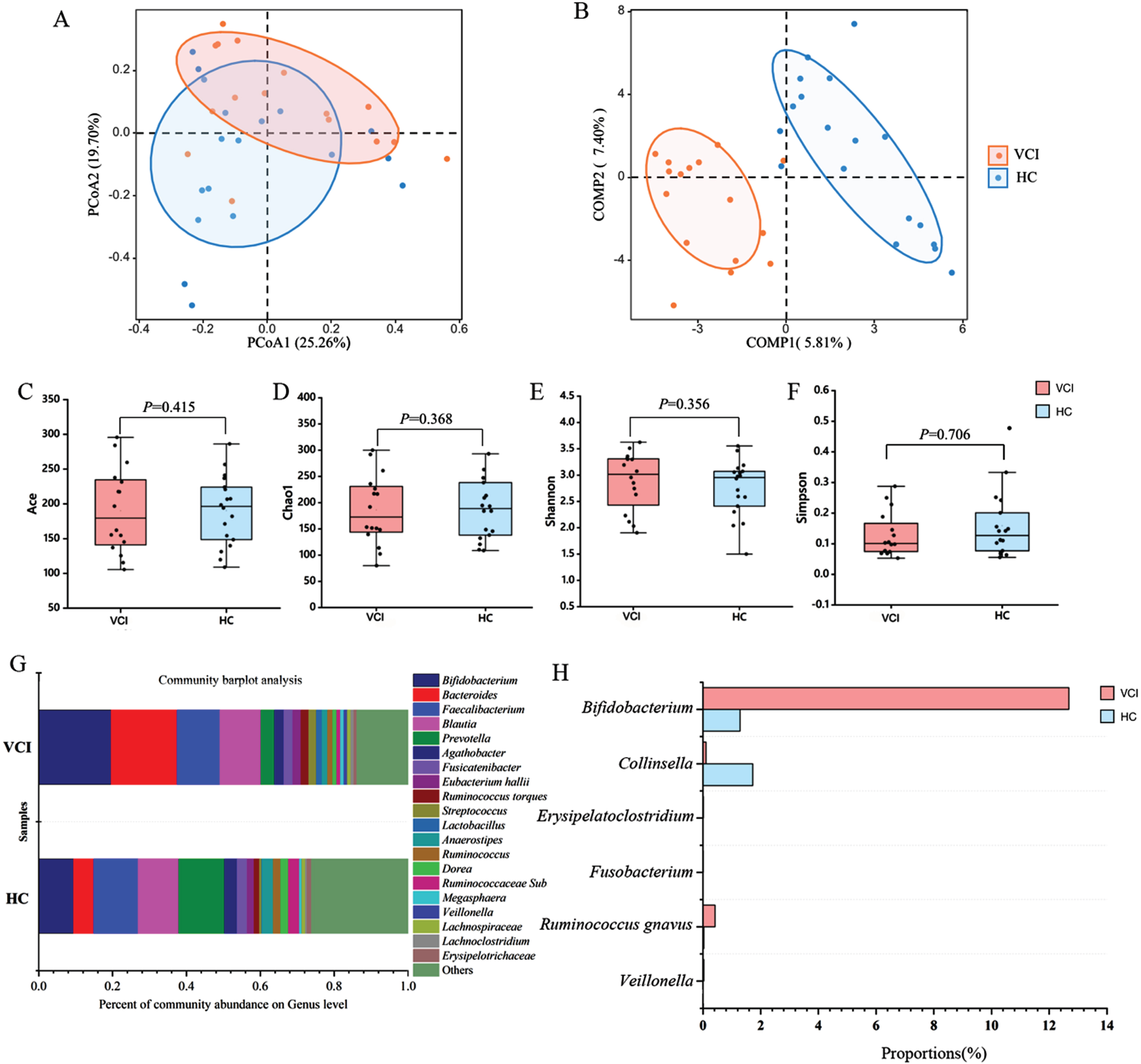
The diversity and composition of GM in both groups. The β-diversity of GM on the PCoA model (A) and the PLS-DA model (B). No difference in α-diversity between two groups, identified by the Ace index (C), Chao1 index (D), Shannon index (E), and Simpson index (F). The composition of GM in two groups (G). Genus-level gut microbial taxa comparisons showed there were significant difference in the relative abundance of Bifidobacterium, Veillonella, Ruminococcus gnavus, Fusobacterium, Erysipelatoclostridium, and Collinsella between two groups by using Wilcoxon rank sum test (H). VCI, vascular cognitive impairment; HC, healthy control; GM, gut microbiota; PCoA, principal coordinate analysis; PLS-DA, Partial least squares discriminant analysis; Ace, Abundance-based coverage estimator.
Spearman’s correlation analysis between the above-mentioned six genera of GM and MoCA scores demonstrated that the abundance of Ruminococcus gnavus was significantly negatively associated with MoCA scores in patients with VCI (r = –0.793, p = 0.009) and was not related to MoCA scores in HCs (p > 0.05; Fig. 3A). There was no significant association between the abundance of the other five GM and MoCA scores (all p > 0.05, Fig. 3B–F).
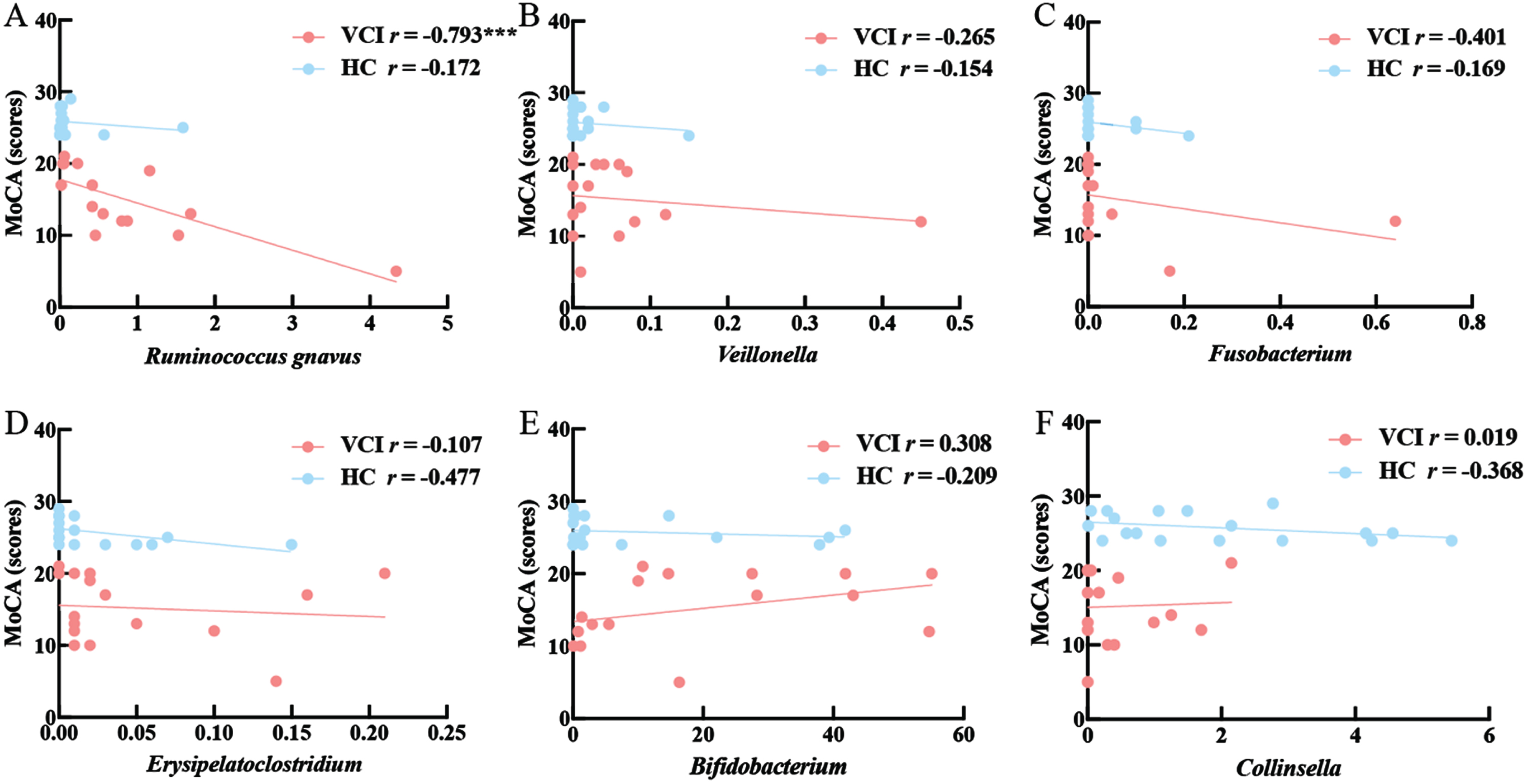
Spearman correlation analysis between the abundance of six genera in the GM and MoCA scores in VCI and HC. Six genera include Ruminococcus gnavus (A), Veillonella (B), Fusobacterium (C), Erysipelatoclostridium (D), Bifidobacterium (E), Collinsella (F).VCI, vascular cognitive impairment; HC, healthy control; MoCA, Montreal Cognitive Assessment; r, Correlation Coefficient; ***p < 0.001.
Mediating effects of regional CBF on the association between GM and cognition
Table 2 shows the mediation effect of the regional CBF on the association of the abundance of Ruminococcus gnavus with MoCA scores. The abundance of Ruminococcus gnavus was negatively associated with MoCA scores (β= –3.405, p = 0.002). The regional CBF of the left hypothalamus (β= 380, p = 0.002), right hypothalamus (β= 0.418, p = 0.004), and left amygdala (β= 0.309, p = 0.006) were significantly positively associated with MoCA scores in VCI, whereas there was no significant association between the regional CBF of the right amygdala or bilateral insula and MoCA scores after adjusting for age and the CBF of the whole brain (all p > 0.05). The proportion of the association between the abundance of Ruminococcus gnavus and MoCA scores mediated by the CBF of the left hypothalamus was 63.96%, the right hypothalamus was 48.22%, and the left amygdala was 36.51% in patients with VCI (Fig. 4).
Mediation effect of the regional CBF on the association of the abundance of Ruminococcus gnavus with MoCA scores
MoCA, Montreal cognitive assessment; SE, standard error; CI, confidence interval.
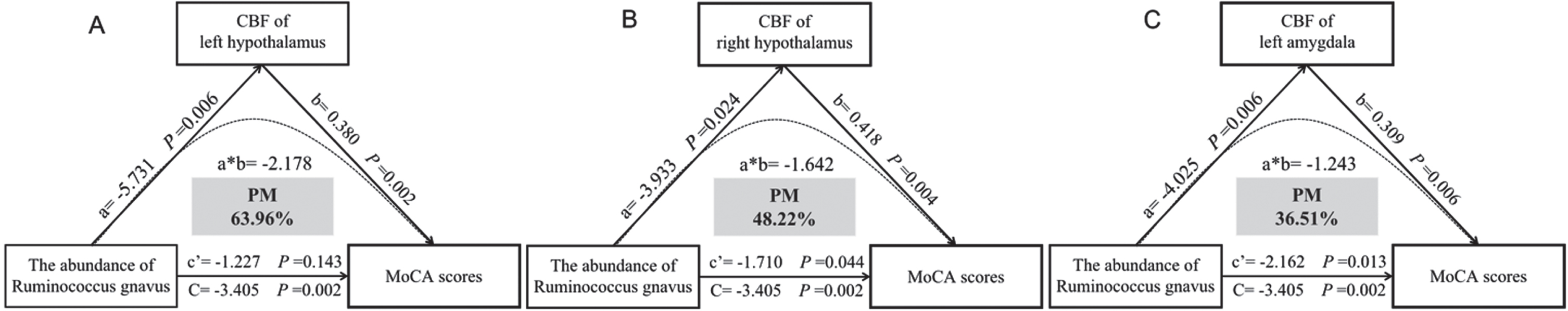
Mediation analyses of regional CBF on the association between the abundance of Ruminococcus gnavus with MoCA scores in patients with VCI after adjusting confounders. Confounders include age, CBF of whole brain. CBF, cerebral blood flow; MoCA, Montreal Cognitive Assessment; PM, percentage mediated.
In the sensitive analysis, the correlation between the CBF of the left hypothalamus and MoCA scores (coefficient = –0.380) and between the abundance of Ruminococcus gnavus and CBF of the left hypothalamus (coefficient = –5.731) remain unchanged with different parameter values. The correlation between the CBF of the right hypothalamus and MoCA scores (coefficient = –0.426 to –0.411) and between the abundance of Ruminococcus gnavus and CBF of the left hypothalamus (coefficient = –5.731) were not significantly different within the full sample analysis results. The correlation between the CBF of the left amygdala and MoCA scores (coefficient = –0.399 to –0.410) and between the abundance of Ruminococcus gnavus and CBF of the left hypothalamus (coefficient = –4.025) also were stable. These findings suggested that the model was robust and stable.
DISCUSSION
This study found that the CBF of the bilateral hypothalamus and the left amygdala were significant mediators in the relationship between the abundance of Ruminococcus gnavus and MoCA scores in patients with VCI, which revealed potential metabolic mechanisms and therapeutic targets for this disease. First, we found that the abundance of Ruminococcus gnavus in the GM was negatively correlated with cognitive function in patients with VCI. Previous evidence demonstrated that Ruminococcus gnavus, a prevalent member of the human GM, was related to autism, depression, and other neurological disorders [21, 22]. Despite the lack of direct evidence connecting Ruminococcus gnavus and VCI, some studies have described changes in this bacterium in AD-related dementia. For instance, an observational study showed that a higher abundance of Ruminococcus gnavus was related to higher plasma levels of neurofilament light chain, regarded as a biomarker for cognitive impairment in patients with AD-related dementia [23]. Park et al. found that the abundance of Ruminococcus gnavus was higher in transgenic APP/PS1 model mice than in wild-type mice [24]. An animal study showed that metabolites of Ruminococcus gnavus, such as isoamylamine, accelerated the expression of genes related to aging and promoted the apoptosis of microglial cells, resulting in a decline in cognitive function [25]. Additionally, it has been established that Ruminococcus gnavus influences metabolic and neuroinflammatory processes [26, 27]. Thus, we speculated that the increased abundance of Ruminococcus gnavus could lower MoCA scores in patients with VCI via the regulation of metabolic and neuroinflammatory pathways.
We also found that the association between the increased abundance of Ruminococcus gnavus and decreased MoCA scores were strongly mediated by the decreased CBF in the bilateral hypothalamus and left amygdala in patients with VCI. As part of the hypothalamic–pituitary–adrenal axis (HPA), the hypothalamus plays important roles in neuroendocrine, cognitive, and metabolic functions [28]. HPA axis dysfunction can elevate proinflammatory cytokines and chemokines, reduce synaptic plasticity, and persistently activate microglia, which can further cause amyloid beta plaque accumulation and Tau hyperphosphorylation, typical pathological markers of AD [29]. A previous study showed that the increased abundance of R uminococcus gnavus is related to intestinal and central nervous system inflammatory response activation and oxidative stress, which could decrease CBF in regions regulating eating, such as the hypothalamus [30]. Moreover, and importantly, dysfunction of the HPA axis also leads to GM dysbiosis, forming a vicious cycle of GM dysbiosis, disrupted HPA axis regulation, and cognitive impairment [31]. In addition, reduced amygdala CBF was a mediator of the association between Ruminococcus gnavus dysbiosis and cognitive impairment in VCI. Similar to the present findings, previous studies showed that the basolateral amygdala is directly related to short and long-term memory associated with significant experiences [32, 33], and learning and episodic memory are regulated by the central amygdala and its output to the hypothalamus [34]. Previous evidence demonstrated that reduced amygdala activity could lead to memory deficits via metabolic dysregulation and, particularly, insulin resistance [35]. Insulin resistance can cause deficits in neuroplasticity, pathological amyloid-beta deposition, increased tau protein phosphorylation, oxidative stress and neuroinflammation, leading to cognitive decline [36]. In an observational study, a greater abundance of Ruminococcus gnavus was found in people with insulin resistance, which may be associated with chronic inflammation and damage to the intestinal and blood–brain barriers [37]. Therefore, we concluded that the cognitive decline associated with a higher abundance of Ruminococcus gnavus and amygdala hypoperfusion could be related to both insulin resistance and other metabolic syndromes.
Considering the role of GM and metabolism-related brain regions on cognition, dietary nutrition could provide a novel therapeutic route for the improvement of cognition in patients with VCI. A randomized controlled trial identified that the Mediterranean diet could generate consistent metabolome changes, including lower plasma and urinary carnitine levels and decreased levels of potentially proinflammatory Ruminococcus gnavus. In addition, previous studies showed that the Mediterranean diet could significantly increase CBF in the left inferior frontal cortex, parahippocampal gyri, posterior cingulate, precuneus, and hippocampus regions, thereby improving cognitive function [39, 40].
The present study had some limitations. First, this was a single-center study with a relatively small number of patients; therefore, our findings may not be generalizable to other settings due to inherent selection bias. Second, we did not perform further metabolism-related laboratory indicators and other multiomics research to validate the relationship among regional CBF, cognitive function, and GM.
Conclusions
This study demonstrated that the abundance of Ruminococcus gnavus was significantly negatively associated with cognitive function in patients with VCI, and this association was strongly mediated by CBF in the bilateral hypothalamus and left amygdala. These findings suggested that Ruminococcus gnavus could be considered an important biomarker for early screening and intervention in VCI. Moreover, increased CBF of the three metabolism-related brain regions more directly reflected the activation pathway of the microbiota–gut–brain axis and highlighted that dietary nutrition interventions targeting Ruminococcus gnavus and the bilateral hypothalamus and left amygdala could provide an opportunity for improving cognition in patients with VCI. Prospective longitudinal studies are needed to evaluate the diagnostic value of Ruminococcus gnavus in the early detection of VCI in addition to translational studies to elucidate the potential metabolic mechanisms of the association among Ruminococcus gnavus, regional CBF, and cognition in patients with VCI.
Footnotes
ACKNOWLEDGMENTS
We would like to thank AnImage (Beijing) Technology Co., Ltd for pCASL consultation.
FUNDING
This work was supported by the National Key Research and Development Program of China (2021YFC2500100 and 2021YFC2500103) and the National Natural Science Foundation of China (82071187 and 81870821).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author.
