Abstract
Background:
Developing effective strategies for reducing dementia risk requires a detailed understanding of the risk and protective factors associated with the progression of mild cognitive impairment (MCI) to dementia.
Objective:
We aimed to systematically review the evidence for sex differences in these factors.
Methods:
Five online databases (PubMed/CINAHL/EMBASE/PsycINFO/Cochrane) were searched from inception until 17 October 2022 for cohort studies that focused on sex differences in risk and protective factors in the progression of MCI to dementia.
Results:
A total of 2,972 studies were identified, of which 12 studies from five countries were included in the systematic review. There was substantial variability in study designs, study populations and outcome measures. Sex differences were present in the associations of sociodemographic, health, psychological factors, genetic and other biomarkers with the progression of MCI to dementia. APOE ɛ4 status and depression appeared to increase the risk of progression for females, whereas history of stroke, MRI markers and cerebrospinal fluid biomarkers appeared to increase the risk of progression for males. APOE ɛ2 status and marital status (unmarried) were observed to reduce risk of progression in males and females, respectively.
Conclusions:
The ability of studies to accurately detail risk factors for dementia are likely limited when solely controlling for the effects of sex. Although the heterogeneity and underpowered nature of the studies made it difficult to synthesize the findings for each risk factor, this study highlights the apparent need for further research examining risk factors for dementia in males and females with MCI separately.
Keywords
INTRODUCTION
Dementia is a clinical syndrome affecting cognition and functional capacity and is caused by neurodegeneration and cerebrovascular dysfunction [1, 2]. It has a substantial impact on the health and quality of life of people living with the condition [3]. Vascular disorders, neuropsychiatric symptoms, frailty, lower levels of education, and a sedentary lifestyle have been associated with a higher risk of dementia [4–7]. Conversely, a healthier lifestyle with increased physical activity and dietary antioxidants have been postulated to have a protective effect on dementia risk [8–10]. Dementia is a multiply determined, complex condition and its cure remains elusive. Therefore, it is imperative to develop effective preventive strategies that reduce risk in those at highest risk for dementia. One target population is people with mild cognitive impairment (MCI), who are at three to five fold higher risk compared to those with intact cognition [11].
MCI can be considered an intermediate cognitive state between the expected decline of normal aging and dementia and can be characterized further as amnestic (aMCI) and non-amnestic MCI (naMCI) [12, 13]. aMCI primarily affects memory while naMCI affects thinking skills other than memory [14]. Although it is often a prodromal stage of dementia, MCI can also remain stable or even revert to normal cognition in some people [15, 16]. The heterogeneity of the prognosis of MCI makes it an ideal intervention point for research on dementia prevention and risk reduction. Studies and meta-analyses have indicated that apolipoprotein E ɛ4 (APOE ɛ4), depression, diabetes, and female gender are risk factors that increase the likelihood of declining to dementia from MCI [17, 18].
Previous research suggests that sex differences are important in many aspects of dementia prevention and management [19]. However, our understanding of these differences is limited due to many studies adjusting for sex as a confounder instead of stratifying results by sex. Sex differences have been noted in cardiovascular risk factors and APOE ɛ4, which are both important risk factors of dementia [20]. Identifying sex-specific risk and protective factors for the progression of MCI to dementia would allow better identification of males and females who are at highest risk. It could also aid in the development of effective evidence-based prevention and management strategies to be applied at the individual and population levels. Here we aimed to systematically review the evidence for sex differences in risk and protective factors in the progression of MCI todementia.
METHODS
This systematic review was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. The study protocol was registered on PROSPERO database (registration numberCRD42021279701).
For the purposes of this review, a risk factor is any characteristic associated with an increased risk of progression to dementia while a protective factor is any characteristic associated with a reduced risk of progression to dementia. Sex and gender are often used interchangeably in the existing literature. For clarity, here the terms ‘male’ and ‘female’ will be used predominantly when discussing sex differences.
Search strategy
An electronic database search was conducted on PubMed, EMBASE, PsycINFO, CINAHL, and Cochrane, from inception until 17 November 2021. The search was updated on 17 October 2022. The MeSH headings included ‘cognitive dysfunction’, ‘dementia’, and ‘disease progression.’ ‘Sex’ was included a MeSH heading in the search terms as it was hypothesised that studies reporting sex differences in risk or protective factors would refer to sex/female/male in the title or abstract. The terms sex and gender are often used interchangeably in the literature; therefore, gender was used as a search term to ensure no studies were missed. Key words included ‘mild cognitive impairment’, ‘dementia progression’, and ‘sex differences’ combined with Boolean operators as appropriate (see Supplementary Table 1 for complete search strategy). The keyword ‘transition’ was added as an additional search term when rerunning the search as the initial search was exclusively based on ‘progression’ as the key word to describe the transition between diagnostic groups. A manual search was also carried out by perusing the reference lists of included articles to ensure that key papers were not missed.
Study selection
Published articles were included if they met the following criteria: 1) conducted in adults aged 50 years and older at baseline, 2) contained observational data (cohort and longitudinal studies), 3) included participants with a diagnosis of MCI (any subtypes), some of whom progressed to dementia (any subtypes), 4) assessed at least one sex-stratified risk factor or protective factor. Reviews, book chapters, editorials, dissertations, theses, poster presentations, and conference abstracts were excluded. Imaging studies solely comparing MCI and dementia cross-sectionally as well as animal studies were excluded (see Supplementary Table 2 for study selection criteria).
Screening process
Two reviewers independently assessed potentially relevant articles for eligibility on Covidence (JM and SK). The study title and abstract screening was used to eliminate obviously irrelevant articles. A full text review of the remaining articles was conducted, and studies were included for extraction based on inclusion and exclusion criteria. Conflicts were resolved by discussion between JM and SK and any discrepancies between the two reviewers was resolved by a third reviewer (NR).
Quality assessment
Two independent reviewers (JM and SK) assessed the methodological quality of all the included studies. The Newcastle Ottawa Scale (NOS) for cohort studies was used to assess the quality and risk of bias of the included studies [21]. The risk of bias was assessed in three main categories: selection, comparability, and outcome. The maximum possible score was 9; four stars for selection, two stars for comparability, and three stars for outcome. NOS criteria are outlined in Supplementary Table 3.
Data extraction and analysis
Data extractions were done by two independent reviewers (JM and SK). The data extraction template in Covidence was modified for extraction of data for the included studies. The data included information on: 1) author and year of publication, 2) study design, 3) follow-up duration, 4) population description, 5) sample size, 6) mean age, 7) MCI type and diagnostic criteria, 8) dementia type and diagnostic criteria, 9) progression/transition of MCI, 10) risk factors, 11) protective factors, 12) sex differences in risk or protective factors. The full list of data extraction items is available in Supplementary Table 4. Meta-analyses were not undertaken due to the heterogeneity of settings, study designs and outcome measures.
RESULTS
2,972 articles were identified through database searching (965 from PubMed, 995 from EMBASE, 200 from PsycINFO, 730 from CINAHL, and 82 from Cochrane). 810 duplicate records were removed before screening. 2,162 title and abstracts of articles were screened. 1,843 records were excluded at the title and abstract screening stage as they either addressed topics overtly irrelevant to dementia and MCI, used empirical approaches inappropriate for inclusion in this review (e.g., animal models of disease), were conference proceedings or other excluded article types (e.g., reviews), or were published in languages other than English. Information on the number of articles and the specific reasons they were excluded for is not available. 319 articles were then assessed for eligibility. 311 records were excluded for not meeting inclusion criteria (103 articles had no MCI diagnosis which remains stable, reverts to normal or progresses to dementia, 52 articles did not assess at least one risk or one protective factors, 46 articles did not have sex-stratified risk/protective factor results, 43 articles were reviews, book chapters, editorials, dissertation, thesis or conference abstracts, 31 articles were imaging studies, 21 articles did not have observational data, 14 articles did not have full text available, and one article was on adults aged under 50 years old. Four additional articles were identified through hand searching. 12 articles were included in the final review (Fig. 1).
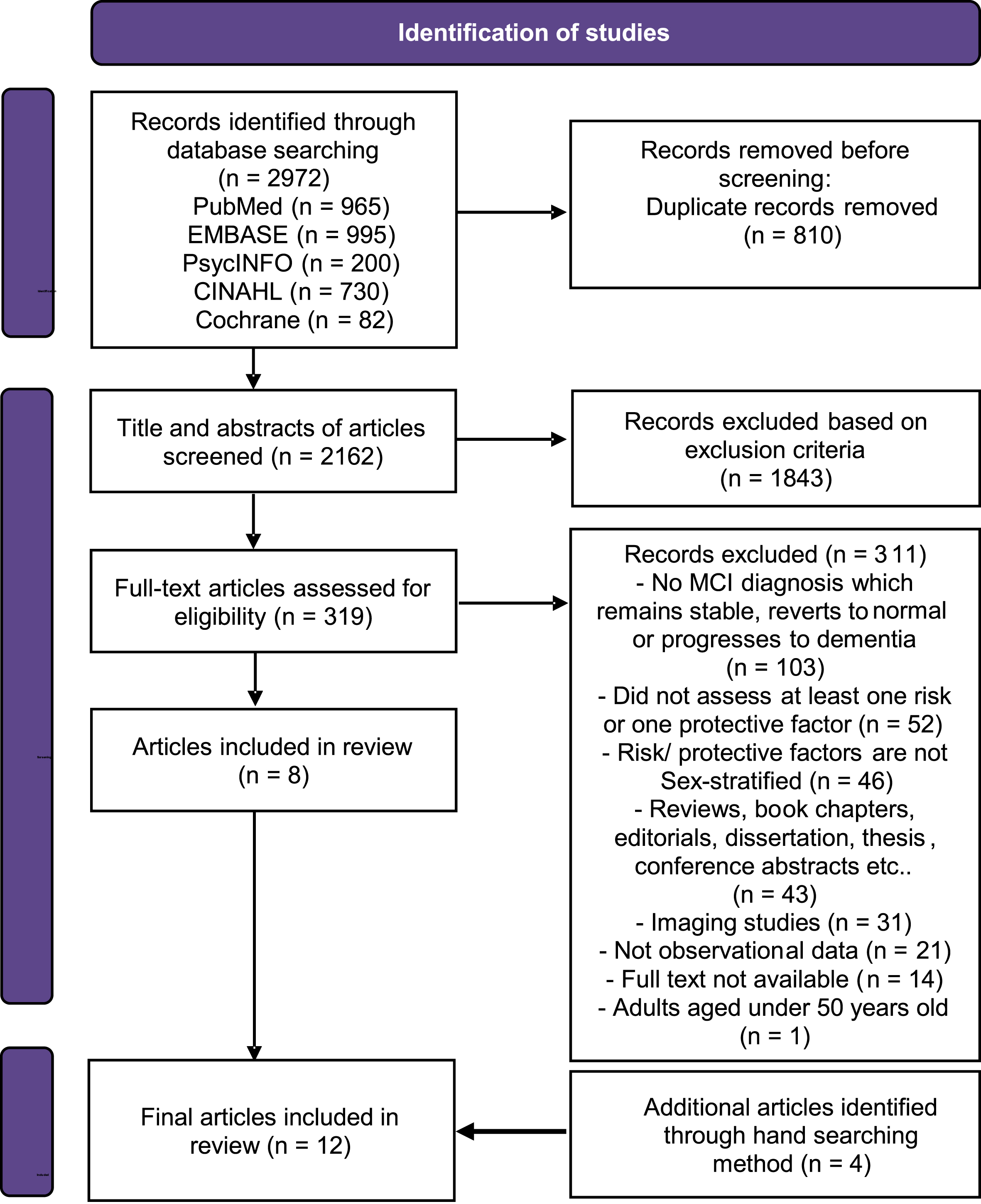
Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flow diagram for study selection.
Results are grouped under traditional risk and protective factors and diagnostic measures and progression from MCI to dementia.
Study and participant characteristics
Twelve studies published between 2008 and 2021 were included in the review with sample sizes ranging from 74 [22] to 8,084 [23]. This included four from the USA [23–26], three from South Korea [27–29], three from Canada [30–32], and one each from Italy [22] and France [19]. They were all published in English.
Six of the studies used Alzheimer’s Disease Neuroimaging Initiative (ADNI) dataset [24, 32]. The remaining studies used the National Alzheimer’s Coordinating Centre (NACC) Uniform Dataset (UDS) [23], Clinical Research Center for Dementia of South Korea (CREDOS) dataset [29], Alzheimer’s Disease Cooperative Study (ADCS) [32], Mayo Clinic Study of Aging dataset [26], and Three-City (3C Study) dataset [19]. One study looked at both the ADNI and ADCS datasets [32]. Two studies used data from hospital memory clinics[22, 31].
Overview of included studies
NACC, National Alzheimer’s Coordinating Centre; UDS, Uniform Dataset; ADNI, Alzheimer’s Disease Neuroimaging Initiative; HC, healthy control; MCI, mild cognitive impairment; AD, Alzheimer’s Disease; VaD, vascular dementia; LBD, Lewy body dementia; ICV, intracranial volume; MCI-nc, mild cognitive impairment non-converters; MCI-c, mild cognitive impairment converters; 3C Study, Three City Study; IADL, Instrumental Activities of Daily Living; RAVLT, Rey Auditory Verbal Learning Test; FAQ, Functional Activities Questionnaire; MMSE, Mini-Mental State Examination; aMCI, amnestic mild cognitive impairment; SCD, subjective cognitive decline; BDNF, brain-derived neurotrophic factors; K-MMSE, Korean Mini Mental State Examination; ADAS-cog 13, Alzheimer’s Disease Assessment Scale- Cognitive Subscale 13; CDR-SB, Clinical Dementia Rating Scale- Sum of Boxes; HIS score, Hachinski ischemic Score; WMH, white matter hyperintensity; CREDOS, Clinical Research Center for Dementia of South Korea; ADCS, Alzheimer’s Disease Cooperative Study; HMGCR G, 3-Hydroxy-3-Methylglutaryl-CoA Reductase gene; PRS, polygenic risk score; BMI, body mass index.
The mean or median age of cohorts within the included studies ranged from 65.6 to 77.8 years. Full details of the studies included are provided in Table 1. The terms ‘men’ and ‘women’ and ‘female’ and ‘male,’ as used in the studies, were included in Table 1.
Assessment of risk and protective factors in the progression of MCI to dementia
An overview of the risk and protective factors identified in the studies is detailed in Supplementary Table 5.
Genetic factors
Five out of 12 studies focused on the association between APOE ɛ4 gene and risk of dementia in individuals with MCI (Fig. 2). The studies assessed APOE ɛ4 status as the presence of one or two ɛ4 alleles or APOE ɛ4 carrier. One of the studies conducted a sub-group analysis on the heterozygote APOE ɛ3/ɛ4 genotype [23]. One study [32] which did not provide the confidence interval is not included inFig. 2.
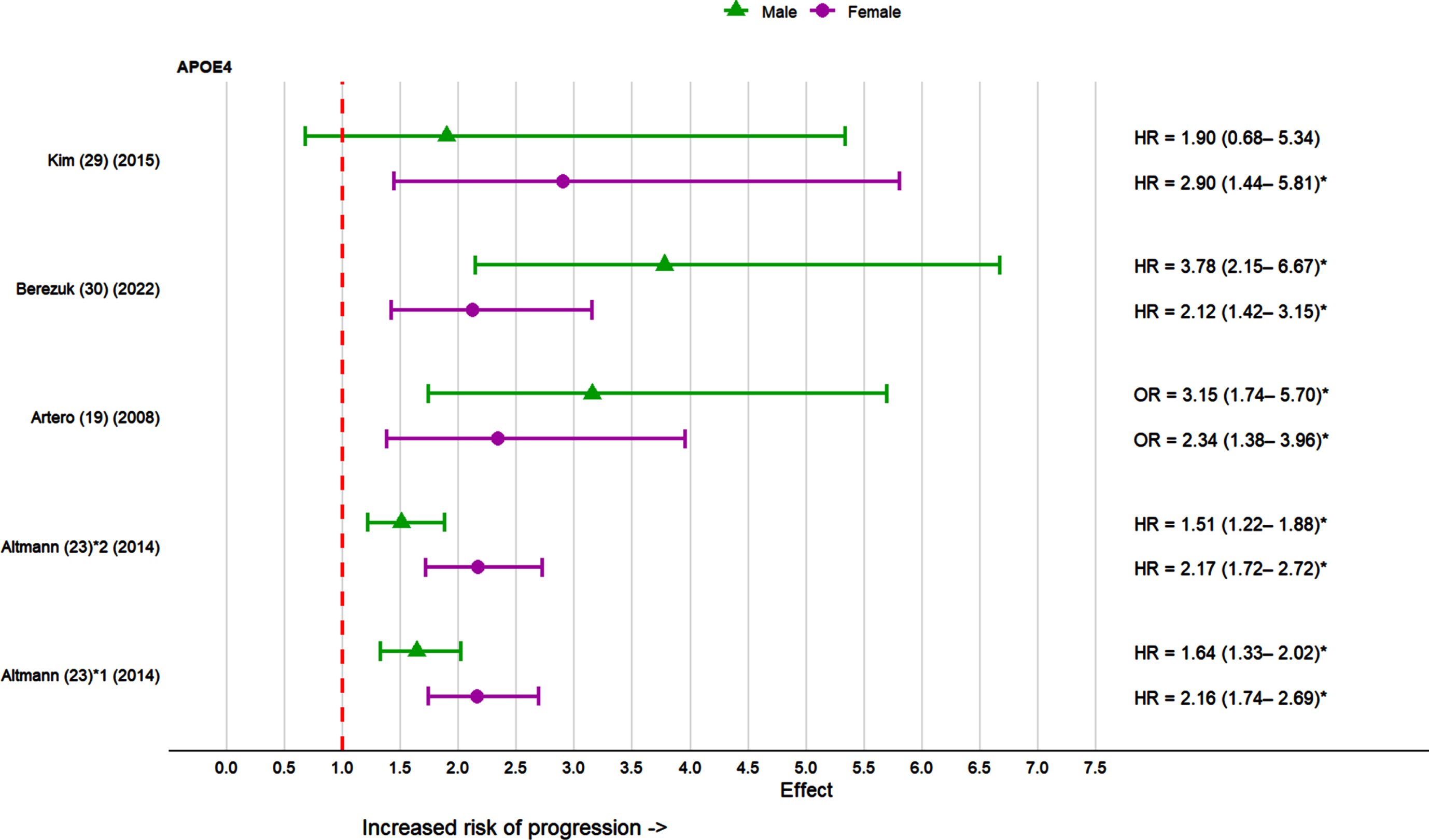
Effect sizes for APOE ɛ4 in the progression of MCI to dementia in females and males in individual studies. 1APOE ɛ4 allele carrier; 2APOE ɛ3/ ɛ4 heterozygote genotype. *p < 0.05.
In 3 out of 5 studies [19, 30], APOE ɛ4 was observed to be a significant risk factor in the progression of MCI to dementia for both sexes. One study found the effect was nominally stronger in women, but the interaction was not statistically significant (p = 0.136) [23]. However, in the sub-analysis restricted to APOE ɛ3/ɛ3 and APOE ɛ3/ɛ4 genotypes, the association of APOE ɛ4 with dementia risk was stronger among females than males (APOE ɛ4 by-sex interaction p = 0.022) [23]. One study [30] found that while APOE ɛ4 was significant in the sex-stratified analyses adjusting only for age and education, it was no longer an independent predictor of dementia progression in the fully adjusted sex-stratified models (p = 0.43 for females and p = 0.11 for males). As APOE ɛ4 modulates the risk of dementia through numerous mediating pathways, some of which were included as covariates in the authors’ fully adjusted model, only the initial results adjusting for age and education were included in Fig. 2.
One study [32] assessed the protective effect of APOE ɛ2 and found a significant reduction in risk of progression to dementia in females but not in males.
One study [22] which investigated the BDNF Val66Met polymorphism found an increased risk of Alzheimer’s disease (AD) only in females (effect size not available for males). Another study [24] on the genetic risk of AD assessed as a polygenic risk score (PRS) reported the PRS to be a significant predictor of AD for females but not males. However, sex-stratified analyses on the interaction between PRS and body mass index (BMI) revealed that a higher PRS and lower BMI predicted conversion to AD only in males [24].
Sociodemographic factors
Sociodemographic factors assessed in studies included education, age and marital status. Education was examined in three studies [19, 29] (Fig. 3). Education was assessed as low education level, higher education level, and years of education in the three studies. All three studies found education was significantly associated with dementia risk in females but only one study [19] found significant results for males. Two studies [19, 29] found a lower education level was associated with significantly increased dementia risk among females. In contrast, a different study found higher education level in females associated with higher rate of progression from MCI to AD [22].
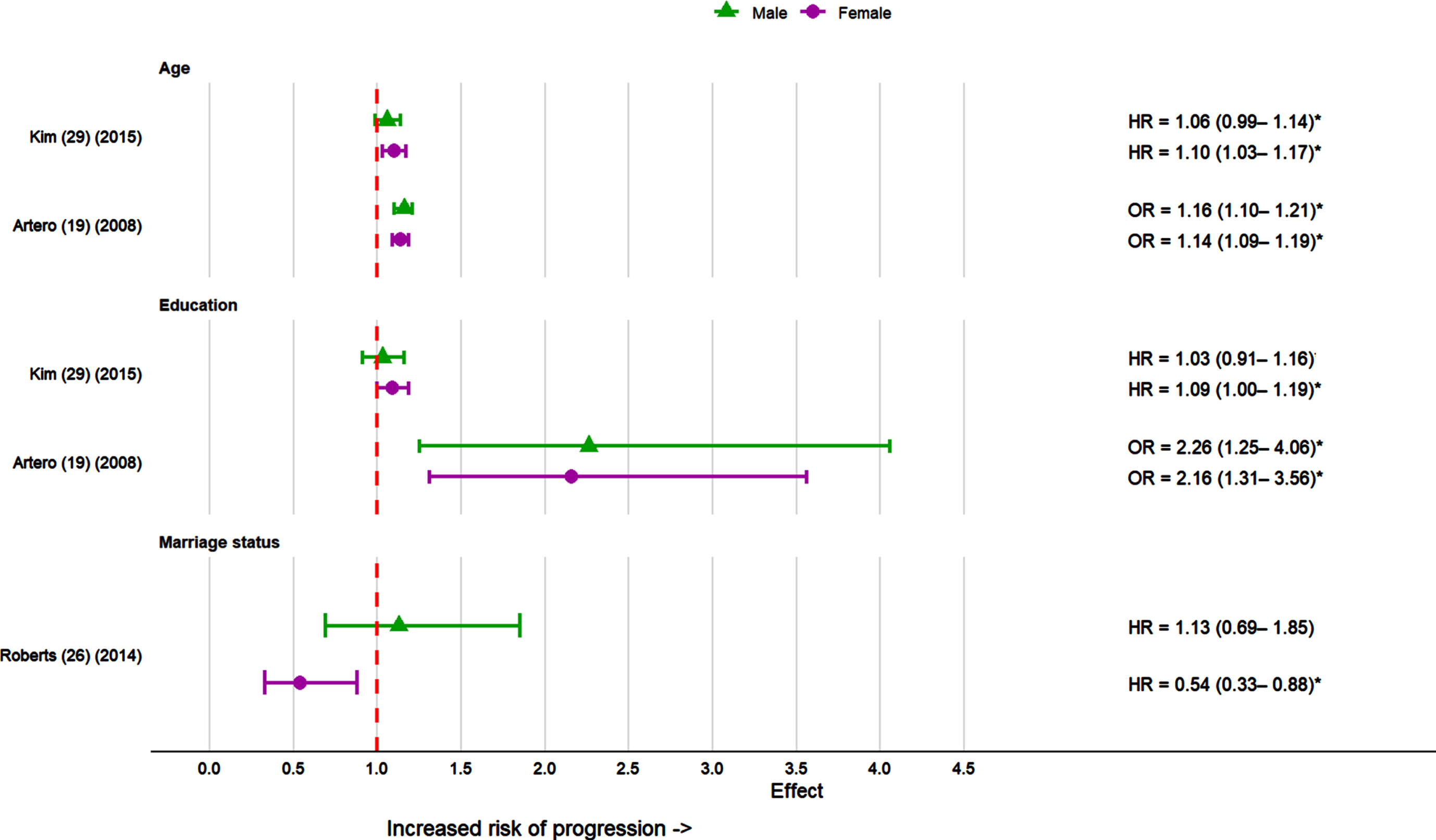
Effect sizes for sociodemographic factors in the progression of MCI to dementia in females and males in individual studies. Reference group for marriage status was married males and females. *p < 0.05.
Advanced age was significantly associated with greater risk of progression to AD in females in two studies [19, 29].Only one study [19], out of two [19, 29] found advanced age to be a significant risk factor of dementia for males.
Marriage status was assessed as a risk factor in one study [26]. Here, sex-stratified analyses found that females who were not married had a lower risk of progressing to dementia compared to females who were married. No difference in risk was identified between married and unmarried males.
Health factors
Four studies assessed health risk factors in the progression of MCI to dementia [19, 29] (Fig. 4). Vascular risk factors assessed included stroke, hypertension, diabetes mellitus, and hyperlipidemia. Three studies [19, 29] which looked at the history of stroke revealed varying results. One study [19] identified stroke as a significant risk factor only for males, another study [26] found stroke was a risk factor only for females, while a third study [29] found no significant association for both sexes.
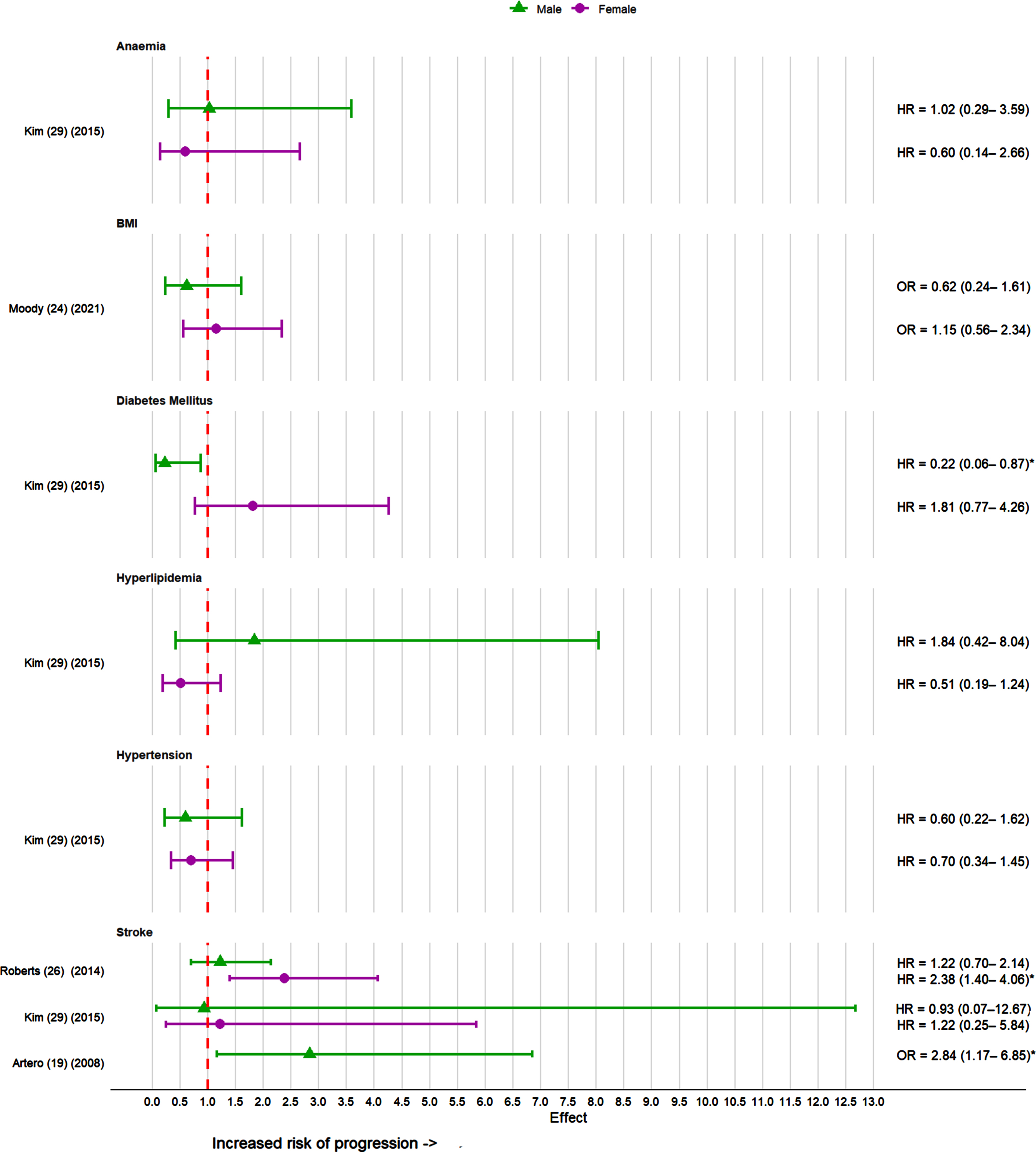
Effect sizes for health factors in the progression of MCI to dementia in females and males. *p < 0.05; BMI, body mass index. Effect sizes not available for stroke in females as it was not statistically significant in the study [19].
One study [29] assessed the risk of hypertension, diabetes, and hyperlipidemia in the progression of MCI to dementia with significant reduced risk noted for males with diabetes. The study [29] which also looked at anaemia and risk of progression to dementia found no significant results for both sexes.
A study [24] assessing BMI as a predictor for dementia identified no significant interaction between BMI and sex for dementia risk; however, subgroup analysis revealed that in males with lower BMI, PRS significantly predicated conversion to AD. Another study [25] examining the neuroprotective role of leptin in reducing risk of dementia found no association with reduced risk of dementia and weak association between leptin levels and cognitive ability in men.
Psychological factors and medications
Depression was the main psychological factor assessed in the progression of MCI to dementia (Fig. 5). Two studies [19, 29] examined depression as a predictor of dementia. Both studies [19, 29] found depression as a significant predictor of dementia only in females with MCI.
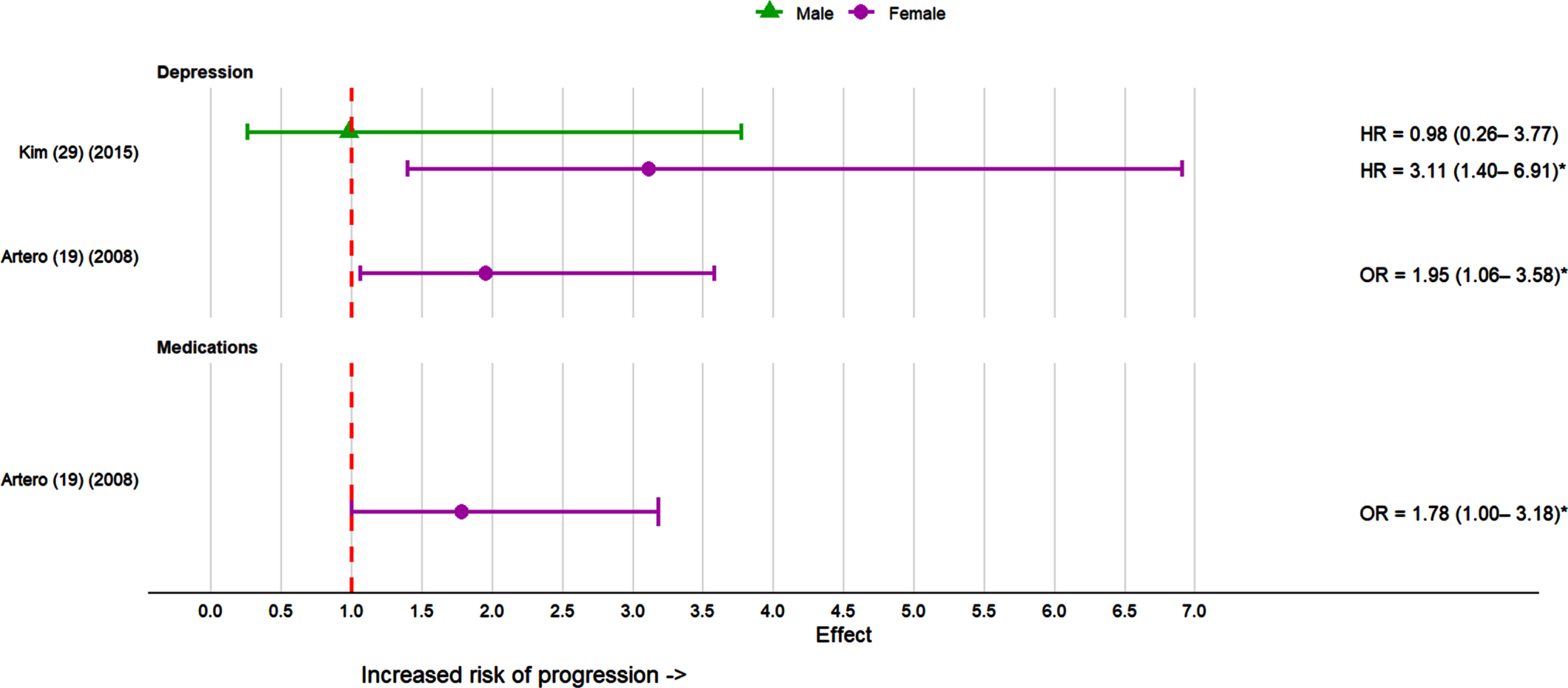
Effect sizes for psychological factors and medications in the progression of MCI to dementia in females and males. *p < 0.05. Effect sizes not available for depression and medications in males as it was not significant in the study [19].
One study examined whether the use of medications with anticholinergic effects was associated with dementia development and found significant positive associations with the odds of dementia among females but not males [19].
Assessment of diagnostic measures and risk of progression from MCI to dementia
An overview of the diagnostic measures and risk of progression from MCI to dementia is detailed in the Supplementary Table 6.
Imaging and biomarker measurements
Three studies [27, 30] analyzed MRI markers in predicting progression to dementia (Fig. 6). ICV was used as a proxy for premorbid brain volume and a measure of brain reserve in one of the studies [27]. Larger ICV was associated with higher risk of conversion to AD in males and in APOE ɛ4 non carriers but not in females or APOE ɛ4 carriers [27]. Hippocampal volume was assessed in two studies [27, 30]. While one study [30] found low baseline hippocampal volume to be a significant predictor of progression to AD in both sexes, another study [27] found it to be a significant predictor only in males. In that latter study, low medial temporal lobe volume was also associated with a higher risk of progression to AD only in males [27]. In a different study, low ventricular volume was a significant predictor for AD in both sexes [30]. Regarding white matter hyperintensities, one study [29] investigated their severity in periventricular (PWMH) and deep subcortical (DWMH) spaces and found higher severity in PWMH and DWMH to be associated with significantly higher and lower dementia risk, respectively, in males but not in females.
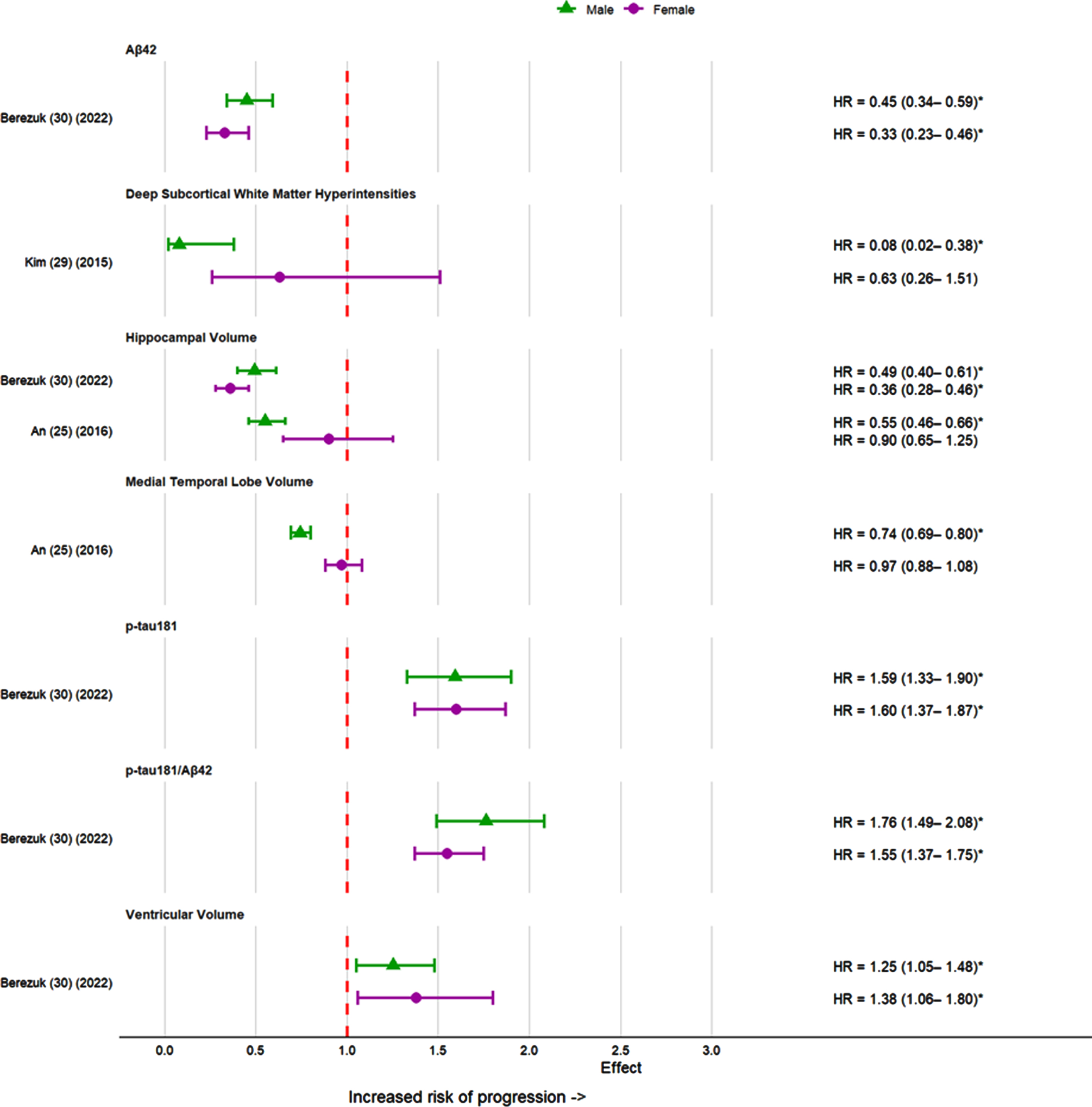
Effect sizes for imaging and biomarker measurements in the progression of MCI to dementia in females and males. *p < 0.05; HV, hippocampal volume; MTP, medial temporal lobe volume; VV, ventricular volume; DWMH, deep subcortical white matter hyperintensities.
One study [30] assessed cerebrospinal fluid markers and found Aβ42, p-tau181 and p-tau181/Aβ42 as significant predictors for progression to AD in both sexes (Fig. 6). Sex interactions were only significant for p-tau181/Aβ42 with stronger effects for men.
Functional and neuropsychological measures
Instrumental activities of daily living (IADL) and Functional Activities Questionnaire (FAQ) were two functional measure tools assessed in the risk of progression from MCI to dementia in two studies [19, 30] (Fig. 7). IADL deficit was a significant predictor of dementia in both sexes after adjusting for confounders [19]. A score of 6 or greater on the FAQ was associated greater risk of AD in both sexes after adjusting for age and education [30]. In this study, although associations appeared stronger among males than females, the sex interaction effect did not reach statistical significance.
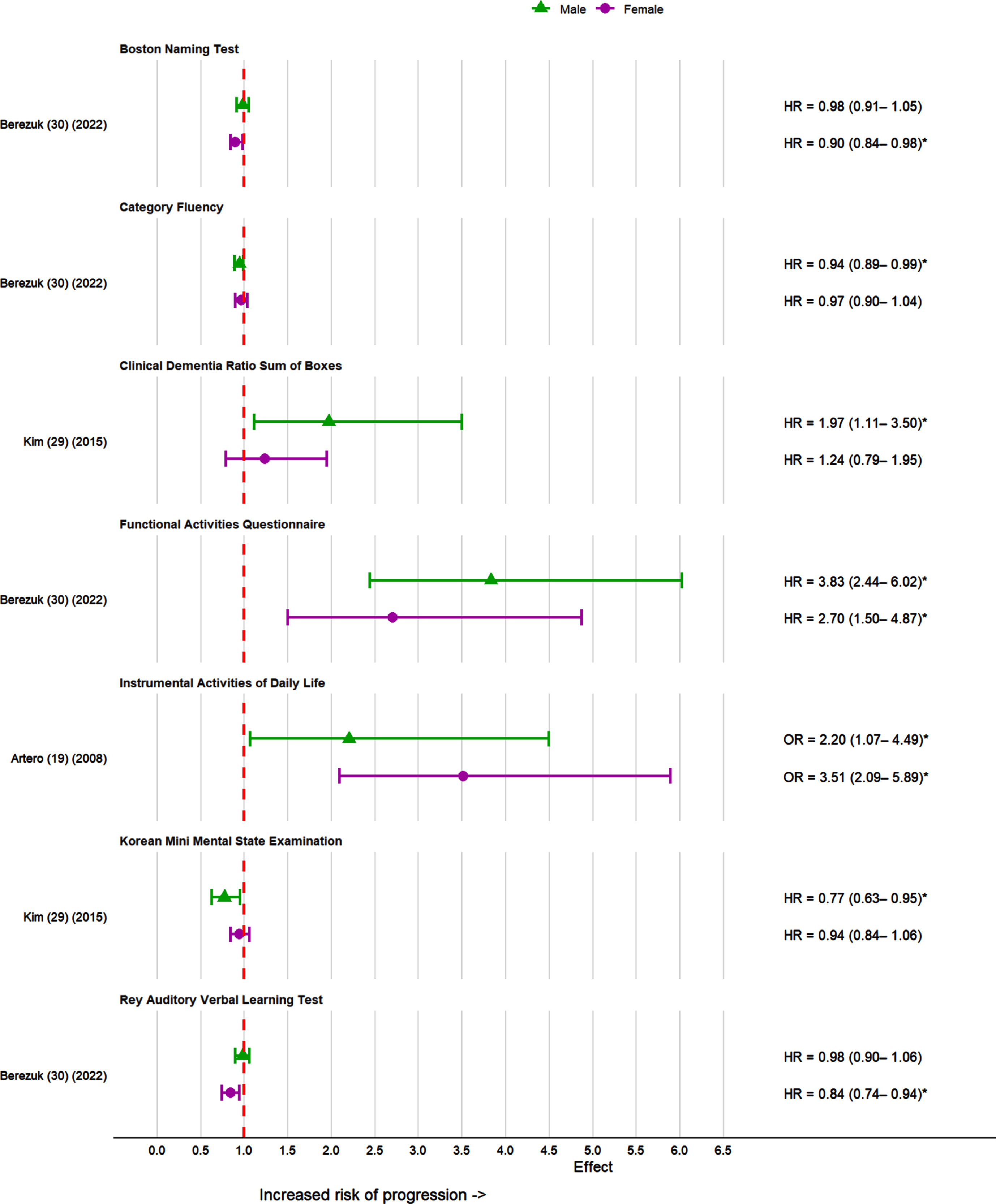
Effect sizes for functional and neuropsychological measures in the progression of MCI to dementia in females and males. *p < 0.05.
Two studies [29, 30] assessed the risk of neuropsychological measures in the progression of MCI to dementia (Fig. 7). In one study [29], baseline Korean Mini-Mental State Examination (KMMSE) and Clinical Dementia Rating scale sum of boxes (CDR-SB) scores were significantly associated with the risk of AD in males and not in females. In another study [30], poor Rey Auditory Verbal Learning Test (RAVLT) delayed recall, fewer words on category fluency and lower Boston Naming Test (BNT) scores were significant risk factors for dementia in both males and females. In the fully adjusted model, poor RAVLT delayed recall and lower BNT scores were significant for females while only category fluency was significant in males [30].
Risk of bias
The risk of bias was heterogenous in all the included studies ranging from 5 to 9 (the maximum possible score of 9 indicates a lower risk of bias). The risk of bias for all included studies is presented in Table 2.
DISCUSSION
This review aimed to systematically review the evidence for sex differences in risk and protective factors in the progression of MCI to dementia. Traditional risk factors and imaging and biomarker measures in the risk of progression to dementia were identified in the included studies. APOE ɛ4 status and depression were stronger and more consistently observed among females, whereas MRI markers and CSF biomarkers had significant associations in females but were more consistently observed among males. While sex differences were evident in males and females with history of stroke in the risk of progression to dementia, the direction was inconsistent and requires more research in the area. APOE ɛ2 status and unmarried marital status were noted to reduce risk of progression in males and females, respectively. While sex differences were evident in genetic risk factors, sociodemographic and psychological factors, as well as biomarkers, it is difficult to draw firm conclusions from the available literature due to the low number of suitable studies, some of which featured small sample sizes.
Genetic factors, especially APOE ɛ4 was assessed in most studies and showed increased progression from MCI to dementia for both sexes but more so in females [17, 28]. The results are consistent with previous studies that reported the same sex difference in APOE ɛ4 allele as a risk factor for AD [33–35]. The pathological hallmarks of AD are extracellular amyloid deposits (neuritic plaques) and intracellular neurofibrillary tangles (tau aggregates). An accumulation beyond normative levels of amyloid-β (Aβ) deposits and tau proteins, in addition to inflammation and excitotoxicity, is associated with progressive cognitive decline in AD [36]. The ɛ4 allele of the APOE gene has been consistently associated with the abnormal accumulation of Aβ plaques with the APOE ɛ4 protein that has been shown to be less effective at Aβ clearance [37, 38]. The ɛ4 allele of the APOE is the strongest genetic risk factor for AD, however, the effect varies by sex [39]. The vulnerability of the female brain to APOE ɛ4 allele has been substantially investigated and is postulated to be a mediator of the higher incidence of AD in females [40]. It also predicts a poorer prognosis for females with MCI as they tend to be more vulnerable to the effects of APOE ɛ4 with lower hippocampal volume and prominent cognitive decline than their male counterparts [34]. APOE ɛ4 is known to significantly increase brain atrophy and decrease brain connectivity much more strongly in females over males [41].
Risk of bias of included studies
★ indicates a score of 1 in that category.
APOE ɛ2 is a genetic modifier that has been documented to reduce risk of dementia. Only one of the included studies assessed APOE ɛ2 and it confirmed the protective effect of APOE ɛ2 specifically in the progression of MCI to dementia with a significant association noted for females [32]. Previous studies which examined APOE ɛ2 have consistently acknowledged the protective effect it has on dementia risk and the prominent effect in females [42–44]. APOE ɛ2 reduces AD risk through amyloid dependent and independent pathways. APOE ɛ2 carriers also have better baseline cognition which sets a higher threshold for cognitive impairment [45, 46].
Sociodemographic factors such as age and education are known risk factors of dementia and increased the risk of progression to dementia from MCI; however, the results were only significant for females in the studies [19, 29]. Age is one of the strongest risk factors for AD with females having a greater lifetime risk of developing AD which is attributed to longevity [47]. Nonetheless, the extent to which sex differences are primarily a matter of the increasing number of females relative to males at older ages or of greater risk in females than males at the same age remains to be resolved [47]. One study reported that unmarried women with MCI had a significantly reduced risk of progression to dementia when compared to married women [26]. The results were not significant for married versus unmarried men. An explanation for this protective effect could be attributed to the traditional gender roles assigned to women as caregivers in a relationship with reduced associated role strain for unmarried women [48]. This is not consistent with previous studies as majority of studies have found marriage and cohabitation to have a beneficial effect on dementia risk [49, 50].
Studies have suggested a sex-specific role of cardiovascular risk factors in the pathogenesis of dementia [51]. History of stroke has been noted as a potential risk factor in the progression of MCI to dementia; however, results were mixed in the studies which assessed stroke as a risk factor [19, 29]. Previous studies [52] have found stroke to be more prevalent in males and increase the risk of all cause dementia in males compared to females; however, the studies focused on participants with normal cognition or no MCI at baseline.
Depression and clinically significant depressive symptoms were significantly associated with greater risk of progression to probable AD in females but not males [19, 29]. This is consistent with previous studies that have found subclinical depression to be a significant risk factor for incident dementia in females [53]. Women are at a greater risk of developing depressive disorders compared to men from mid-puberty to adulthood and at times of dramatic sex hormone fluctuations such as a pregnancy and menopause [54]. Deleterious effects of depression on the brain have long been studied in terms of hippocampal atrophy that may be explained by glucocorticoid neurotoxicity and impaired neuroplasticity resulting from stress-induced reduction in neurotrophic factors [55, 56].
Volumetric measures derived via MRI can be used to support a diagnosis of dementia. In patients with AD, hippocampal volume is smaller than in people with normal cognition [57]. Larger hippocampal volume at baseline reduced risk of progression to dementia for both genders in one study and only in males in another study. This could be attributed to the sexually dimorphic nature of the hippocampus with its function in learning and reactivity to stress grossly favoring males [58]. Sex differences were also present in intracranial volume which is often used as a proxy of premorbid brain volume and brain reserve [59]. Gender differences in brain and cognitive reserve have been studied extensively with women being disadvantaged due to traditional gender and social roles limiting their access to education. Older women are usually more likely to have had lower education and subsequently simpler jobs than older men leading to lower levels of cognitive reserve [60]. Higher levels of cognitive reserve may delay the onset of cognitive impairment in subjects with AD pathology but once symptoms manifest, they more progress rapidly [61]. Large ICV increased the risk of conversion to AD in males and APOE ɛ4 non-carriers with MCI and not in females in the included study [27]. This contrasts with earlier findings which notes reduced risk for individuals with larger intracranial volume [62, 63]. This may be due to the influence of intracranial volume on disease trajectory, which shortens the duration of the MCI stage therefore having a protective effect in the earlier stages but act as a risk factor at later stages [27, 64].
The traditional risk and protective factors as well as the biomarker results apply to the high-risk population already demonstrating MCI and could be thought to serve different purposes. Sex differences identified in risk and protective factors may have implications at population, research, and clinical practice levels. It needs to be accounted for or addressed when designing studies and educating the general population on MCI and risk of dementia. Sex differences identified in imaging and biomarker measurements and risk of progression from MCI to dementia may be more applicable for informing clinicians and patients with MCI regarding their individual risk of progression to dementia.
Limitations of this systematic review include the small number of studies included with only one or two studies for some risk factors. The search was restricted to published articles written in English and therefore some studies may have been missed. A meta-analysis was deemed inappropriate so that each risk domain was handled consistently, regardless of the number of studies included, and due to the high risk that pooled estimates would be misleading. For these reasons, heterogeneity statistics were not computed. Although two risk factors did have three or more included studies, we deemed the clinical heterogeneity to be too high (setting, study design, outcome measure). Although the substantial heterogeneity in addition to the small number of included studies precluded any definitive conclusions being drawn about sex differences, it also highlights the need for further research in the area utilizing high powered studies with strong clinical diagnostic criteria of MCI and dementia.
Thorough investigation of sex differences in the context of risk factors and protective factors for the progression from MCI to dementia requires a nuanced approach that goes beyond simply adjusting for sex as a covariate [65]. Statistical adjustment for sex fails to acknowledge the heterogeneity between health factors, exposures, and outcomes between the sexes [66, 67] and assumes that the relationship between risk factors and outcomes is equal for both. Some resources already exist that provide guidance for researchers. For example, the Sex and Gender Equity in Research (SAGER) guidelines describe standardized approaches to reporting sex and gender information in study design, data analyses, results, and interpretation of findings [68]. General principles include the accurate use of the terms sex and gender and designing and conducting research in a way that can reveal sex-related differences even if these were not initially expected [68]. While SAGER provides broad guidelines, the Women’s Brain Project (WBP) provides more specific recommendations to accurately capture the impact of sex and gender in AD research. Recommendations are provided for different areas of AD research such as in vivo and in vitro research, sex-based disease modelling, clinical studies, sex-based patient journey and policy framework [69]. This includes providing sex related effects and stratification and inclusion of demographic, imaging, clinical, genetic and biomarker data for clinical studies [69]. Future research needs to consider both SAGER guidelines and WBP recommendations when planning, designing, and conducting studies on sex differences in MCI and dementia. This can contribute to current knowledge on the influence of sex on prevalence, risk factors and outcomes, as well as creating effective sex specific preventative and intervention strategies for MCI and dementia.
Conclusion
In conclusion, this is a comprehensive review addressing sex differences in the progression of MCI to dementia. Although the heterogeneity and underpowered nature of the studies made it difficult to synthesize the findings for each risk factor, this study highlights the importance of addressing sex differences in MCI and dementia in future research.
Footnotes
ACKNOWLEDGMENTS
The authors would like to acknowledge the assistance received from Marcos Riba (UQ librarian) in the development of the search strategy. JM is supported through the Australian Government Research Training Program (RTP) Scholarship.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
The authors have no conflicts of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article and/or its supplementary material.
