Abstract
Background:
Subjective cognitive decline (SCD) refers to the self-reported persistent cognitive decline despite normal objective testing, increasing the risk of dementia compared to cognitively normal individuals.
Objective:
This study aims to investigate the attributes of SCD patients who demonstrated memory function improvement.
Methods:
In this prospective study of SCD, a total of 120 subjects were enrolled as part of a multicenter cohort study aimed at identifying predictors for the clinical progression to mild cognitive impairment or dementia (CoSCo study). All subjects underwent 18F-florbetaben PET and brain MRI scans at baseline and annual neuropsychological tests. At the 24-month follow-up, we classified SCD patients based on changes in memory function, the z-score of the Seoul verbal learning test delayed recall.
Results:
Of the 120 enrolled patients, 107 successfully completed the 24-month follow-up assessment. Among these, 80 patients (74.8%) with SCD exhibited memory function improvements. SCD patients with improved memory function had a lower prevalence of coronary artery disease at baseline and performed better in the trail-making test part B compared to those without improvement. Anatomical and biomarker analysis showed a lower frequency of amyloid PET positivity and larger volumes in the left and right superior parietal lobes in subjects with improved memory function.
Conclusions:
Our prospective study indicates that SCD patients experiencing memory improvement over a 24-month period had a lower amyloid burden, fewer cardiovascular risk factors, and superior executive cognitive function. Identifying these key factors associated with cognitive improvement may assist clinicians in predicting future memory function improvements in SCD patients.
INTRODUCTION
Subjective cognitive decline (SCD) refers to the condition concerning the self-experienced persistently reduced cognitive function but remaining in the cognitively unimpaired status on the standardized neuropsychological tests [1, 2]. According to the development of tests for the biomarkers of Alzheimer’s disease (AD), it was reported that the presence of amyloid pathology was associated with subjective cognitive concerns and SCD might be regarded as the very early stages of AD [3–5]. Also, patients with SCD have a higher risk to develop incident mild cognitive impairment (MCI) or dementia than controls [6–8]. Various factors contribute to the risk of developing dementia in SCD; these include specific characteristics of SCD including worry about cognitive decline, the informant-reported SCD, biomarkers indicative of AD and neurodegeneration, psychiatric factors like depression and anxiety, the presence of apolipoprotein E (APOE) ɛ4 allele, and findings from neuropsychological test, including poorer performance on the Trail Making test part B and lower scores on the Mini-Mental State Examination (MMSE) score [7–9]. Thus, recently physicians and researchers had paid attention to SCD and identify those prognostic factors among them. Moreover, multidisciplinary interventions including aerobic exercise and cognitive training are adopted for patients with SCD to prevent progression to the next stage [10–12] and those SCD patients, especially those with higher amyloid burden, are considered good candidates for amyloid-modifying therapy [13].
Generally, the rate of reversion to normal status in MCI is reported to be between 24% to 31% [14–16]. The main factors affecting reversion include younger age, absence of the APOE ɛ4 allele, fewer comorbidities, higher MMSE score, and greater cognitive reserve [16, 17]. However, defining recovery from SCD is difficult due to its characteristic absence of objective memory decline. Nevertheless, some SCD patients report improvement in subjective cognition during follow-up, and in fact, some patients demonstrate improved scores on the subsequent neuropsychological assessments. Understanding the factors that contribute to improved cognitive function is important for guiding patients with SCD in clinical settings. Therefore, we investigated the clinical features, neuroanatomical status, and AD biomarkers of SCD patients who demonstrated improved memory function.
METHODS
Subjects
A total of 120 subjects were included in this prospective study of SCD. All participants were recruited as part of the multicenter cohort study to identify predictors for the clinical progression to MCI or dementia from SCD (CoSCo study) [18]. Patients were enrolled from memory clinics in six institutions across South Korea between November 2018 and November 2019.
SCD was diagnosed according to the research criteria of SCD [19]. Inclusion criteria were as follows: 1) self-reported persistent cognitive decline compared to a previously normal status, 2) normal performance relative to the individual’s age and education on the standardized neuropsychological tests, and 3) scores between the 7th and 50th percentiles on delayed recall of the verbal memory task. We defined normal neuropsychological performance as a score higher than the 7th percentile in the delayed recall subtest of the Seoul Verbal Learning Test (SVLT) and higher than the 16th percentile in all other subtests within the Seoul Neuropsychological Screening Battery (SNSB) [20]. The last criterion was determined to increase the likelihood of capturing progression to MCI or dementia within a short observation period. Exclusion criteria included 1) space-occupying lesions, previous stroke, or major psychiatric disorders, 2) use of drugs affecting cognitive function, and 3) fulfillment of the criteria for MCI defined by Petersen [21] or dementia according to the Diagnostic and Statistical Manual of Mental Disorders V criteria [22]. We obtained informed consent from all participants, and the study was approved by the local Research Ethics Committee of each institution.
All subjects underwent annual neuropsychological assessments. We compared the results of the 2-year follow-up test to the baseline, and defined participants who scored higher on the delayed recall of the Seoul Verbal Learning Test (SVLT), the task of memory function, at the 2-year follow-up than at baseline as the “improvement group”, while those who scored the same or worse in the SVLT test at the 2-year follow-up were classified as the “no improvement” group.
Clinical assessment
Clinical assessments were conducted to collect demographic and clinical information at baseline. Height, weight, body mass index (BMI), and waist circumference were measured. Patients underwent the InBody test (InBody Co., Ltd, Seoul, Korea) to obtain body fat percentage, skeletal muscle mass, and the visceral fat area. To assess physical function, all participants underwent a hand grip strength test of each hand (kg), five successive chair rise tests (total time, sec), the 6-meter walking test (averaged from two sessions, sec), and the dual-task walking test with phonemic word fluency task (averaged from two sessions, sec). Two patients who complained of difficulty due to sustained fatigue and personal reasons in the walking test were excluded from the analysis related to the item. Patients also underwent pure tone audiometry (PTA). It was calculated using weighted four-frequency average (W4FA = (0.5 kHz+(1 kHz)x2+(2 kHz)x2+4 kHz)/6). The ‘hearing impairment’ of each ear was defined when the PTA average was more than 25 dB. In addition, patients underwent routine laboratory tests and apolipoprotein E (APOE) genotyping.
Neuropsychological tests and self-report questionnaires
Neuropsychological tests and self-report questionnaires were administered annually to follow up with patients. All patients received standardized neuropsychological batteries, Seoul Neuropsychological Screening Battery (SNSB) [20], which included the digit span test forward for attention, the Korean version of the Boston Naming test for language function, the Rey-complex figure test (RCFT) copy for visuospatial function, delayed recall of the Seoul verbal learning test (SVLT) and RCFT for memory function, and the phonemic word fluency test, digit symbol coding, trail-making test part B, and Stroop test color naming for executive function.
Patients completed the SCD questionnaire [23] and the Korean-Everyday Cognition (K-ECog) scale [24]. The 10-item SCD questionnaire was designed based on core features of SCD-plus [19] and preclinical AD pathology, resulting in a score ranging from 0 to 29; a higher score indicates a more severe perceived cognitive decline [23]. The K-ECog consisted of 39 questions, scored on a scale of 1 (better or no change) to 4 (consistently much worse). It provided a global score and six sub-scores of cognitive domains; memory, language, visuospatial function, and executive function (planning, organization, divided attention) [24].
Depressive symptoms were assessed using the Patient Health Questionnaire-9 (PHQ-9), which ranges from 0 to 27 (a higher score indicating a greater degree of depressive mood). Perceived stress was measured using the Korean version of the Brief Encounter Psychosocial Instrument (BEPSI-K) with 5 items [25]. The total score of BEPSI-K ranges from 5 (the lowest level of stress) to 25 (the highest level of stress). The Pittsburgh Sleep Quality Index (PSQI) was used to evaluate the overall sleep quality [26]. The total score of the PSQI ranges from 0 to 21, with a higher score indicating poorer sleep quality. Subjective hearing difficulty was measured using the Korean version of the Hearing Handicap Inventory for the Elderly (K-HHIE) [27]. The K-HHIE comprises 25 items, and the total score ranges from 0 to 100, with a lower score indicating improved hearing ability over unaided status.
Brain MRI
At baseline, all participants underwent brain MRI scans using a 3.0 T magnetic resonance scanner. T1- and T2-weighted images, fluid-attenuated inversion recovery imaging (FLAIR), susceptibility-weighted imaging (SWI), and three-dimensional T1-weighted images were obtained. Experienced neurologists conducted the assessment of brain MRI. Lacunes were counted when the lesion was small (≤15 mm in diameter) with a high signal on T2-weighted images, low signal on T1-weighted images, and a perilesional halo on FLAIR in the basal ganglia, thalamus, and white matter. Cerebral microbleeds (CMB) were defined as homogenous round lesions (≤10 mm in diameter) with low signals on SWI. The severity of white matter hyperintensities (WMH) was assessed using a visual rating scale of axial FLAIR images based on the previously described method [28]. Deep and Periventricular WMHs were rated as minimal (grade 1), moderate (grade 2), or severe (grade 3), respectively. Medial temporal lobe atrophy (MTA) was evaluated according to Scheltens’ visual rating scale using T1-weighted coronal images.
Image processing for volumetric measurement of 3D T1-weighted images was performed using AQUA 2.0 software (Neurophet Inc., Seoul, Korea), a deep learning-based whole brain segmentation tool [29, 30]. The extracted regional gray matter volumes were adjusted for intracranial volume. The corresponding percentiles of each area was calculated by adjusting for the individual’s age and sex using East Asian normative data [30].
Amyloid positron emission tomography scans
All participants underwent 18F florbetaben amyloid positron emission tomography (PET) and computed tomography (CT) scans at each institution, following a standardized protocol [31]. Scanned images were interpreted by a trained nuclear radiologist who was blinded to the patients’ clinical information. The results were classified as either positive or negative based on previous studies [32]. The quantitative regional amyloid burden was calculated using SPM8 (http://www.fil.ion.ucl.ac.uk/spm/software/spm8) and MATLAB 2014b (The MathWorks Inc., Natick, MA, USA) as described in earlier methods [23]. The standardized uptake value ratio (SUVR) was measured by calculating the median uptake across voxels in the regions of interest on PET images, with reference to the cerebellar cortex [33]. The average SUVR of the 90 regional uptake values was computed to obtain a global SUVR.
Statistical analysis
The demographic, clinical, neuropsychological, and biomarker results were presented as means with standard deviations (SD) for continuous variables and frequencies with percentages for categorical variables. The chi-square test or Fisher’s exact test was employed for categorical variables, while the Mann-Whitney U test was employed for continuous variables. Statistical analysis was performed using SPSS 20.0 for windows (SPSS Inc., Chicago, IL, USA). Statistical significance was considered p < 0.05.
RESULTS
Demographics and clinical characteristics
We included 120 patients in this CoSCo study, with 107 participants followed-up and analyzed at a 24-month follow-up. After 24 months, 80 (74.8%) patients with SCD demonstrated an improvement in their memory function as assessed by the SVLT, while 27 (25.2%) SCD patients did not. (Fig. 1) Nine patients (8.4%) progressed to MCI or dementia at the 24-month follow-up.
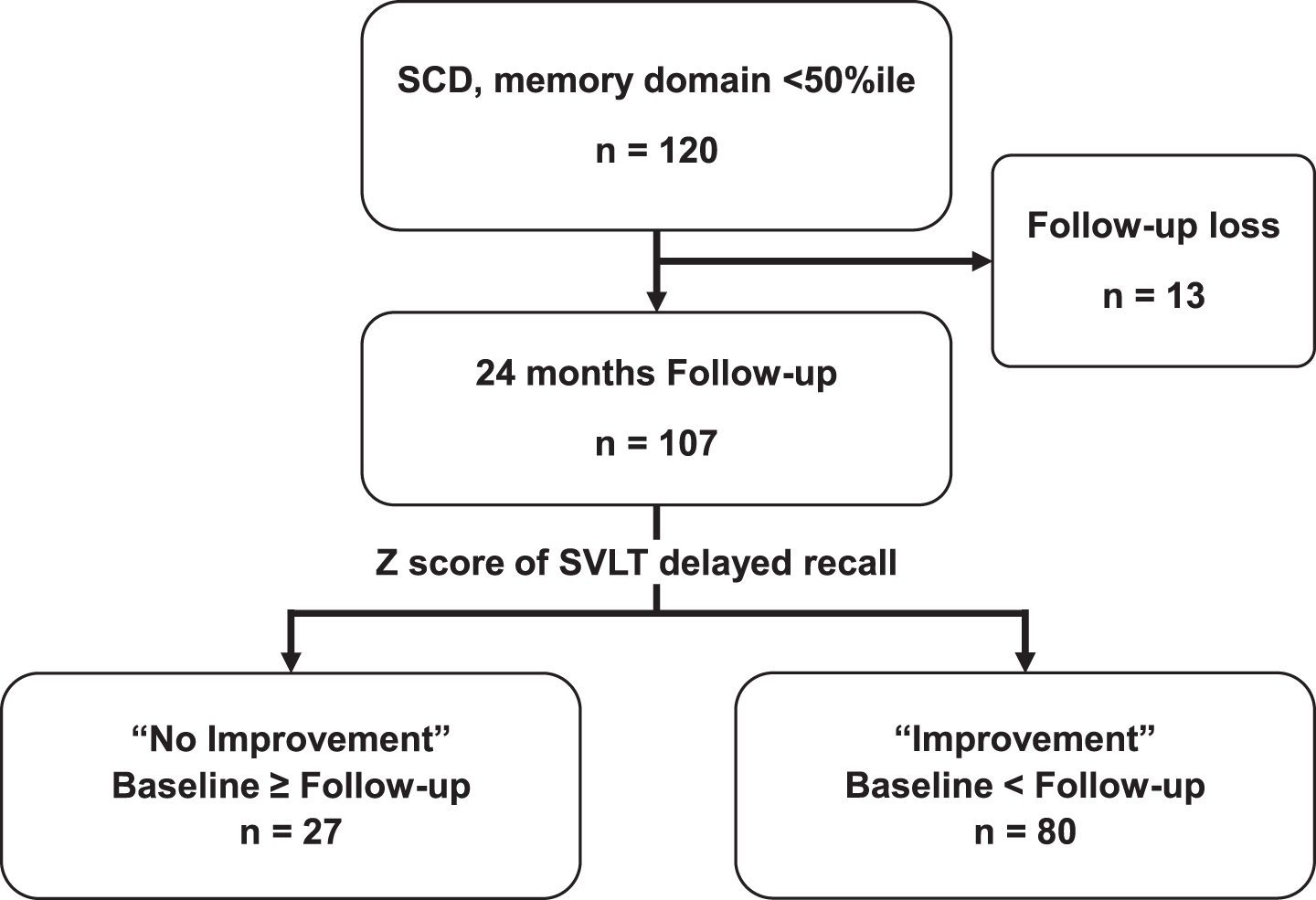
Flow diagram of study participants.
Regarding baseline clinical and demographic characteristics, there were no significant differences in age, sex, and education level between SCD patients with and without memory improvement. SCD patients with memory function improvement exhibited a significantly lower frequency of coronary artery disease (1/80 (1.2%) versus 3/27 (11.1%), p = 0.049) compared to those without improvement (Table 1). The frequency of bilateral hearing impairments was higher in the improvement group, though not significant (36/80 (45.5%) versus 7/27 (25.9%), p = 0.062). Physical status indicators, including BMI, waist circumference, skeletal muscle mass, five successive chair rise test, the 6-meter walking test, and dual-task walking test did not significantly differ between SCD patients with memory improvement and those without improvement (see Supplementary Table 1). Various questionnaires assessing cognitive concerns and psychiatric symptoms, including K-ECog total scores, PHQ-9 total scores, K-HHIE, BEPSI-K, and PSQI scores, showed no significant difference between the two groups (see Supplementary Table 2).
Baseline clinical and demographic characteristics of SCD patients with improved memory function at 24-month follow-up
Data are expressed as the mean with standard deviation or number with percentages in parenthesis. *p < 0.05.
Baseline neuropsychological tests revealed significantly better performance in the trail-making test part B among SCD patients with memory improvement (0.445±0.566 versus 0.099±0.748, p = 0.035). There was no significant difference in memory, visuospatial, attention, or global cognition (MMSE) between the two groups (Table 2).
The neuropsychological test outcomes for SCD patients demonstrated improvements in memory function at the 24-month follow-up
Data are expressed as the mean with standard deviation. All subtests of neuropsychological test were expressed as z scores. The scores of MMSE were described as the raw score. COWAT, Controlled oral word association test; MMSE, Mini-Mental Status Examination; RCFT, Rey complex figure test; SVLT, Seoul verbal learning test. *p < 0.05.
The anatomical and biomarker results
The severity of WMHs, as well as the number of lacunes and microbleeds, showed no significant difference between the two SCD patient groups (Table 3). The MTA grade for both right and left sides in SCD patients with improved memory function were not significantly different from those in SCD patients without memory improvement. The frequency of positive amyloid PET scans was significantly lower in SCD subjects with memory improvement (12/80 (15.0%) versus 13/27 (48.1%), p = <0.001). Although the average SUVR of amyloid PET scans in SCD patients with memory improvement was lower, it was not significantly different compared to those without improvement (1.261±0.207 versus 1.358±0.325, p = 0.074). The analysis of regional SUVR of amyloid burden on PET demonstrated that patients with improved memory function exhibited significantly lower SUVR in the left dorsolateral superior frontal gyrus, as well as in certain regions of the bilateral temporal cortex, including Heschl’s gyri, and in the superior and a portion of inferior parietal gyri (see Supplementary Table 3). Upon analyzing age, sex, and intracranial volume-adjusted regional brain volume using AI-based volumetric analysis SegPlus and the AQUA program, only the volumes of the right superior parietal lobe (13.79±1.73 versus 13.21±1.86, p = 0.029) and left superior parietal lobe (14.22±1.83 versus 13.47±1.88, p = 0.022) were significantly higher in the SCD patients with memory improvement than those without improvement (see Supplementary Table 4).
Baseline anatomical and biomarker results SCD patients with improved memory function at the 24-month follow-up
Data are expressed as the mean with standard deviation or number with percentages in parenthesis. MTA, the medial temporal lobe atrophy; SUVR, standardized uptake value ratio. ¶White matter hyperintensities were shown by a grade which was as previously reported in a nationwide Clinical Research Center for Dementia of South Korea (CREDOS) study. *p < 0.05.
Changes in various neuropsychological assessment outcomes at 24-month follow-up
We examined the alterations in multiple neuropsychological assessments among SCD patients with or without memory enhancement (Table 4). SCD subjects exhibiting improvement in verbal memory function also demonstrated significant enhancements at the 24-month follow-up in Stroop test – color reading (6.35±22.79 versus –5.65±20.34, p = 0.003), attention and calculation subtest within MMSE (0.26±1.09 versus –0.3±0.78, p = 0.008), and MMSE total score (0.9±2.08 versus –0.56±2.01, p = 0.003) (Fig. 2). The frequencies of improvement in each task were significantly different in the trail-making test part B (46 (57.5%) versus 9 (33.3%), p = 0.044), Stroop test – color reading (54 (67.5%) versus 10 (37.0%), p = 0.007), attention and calculation subtest within MMSE (25 (31.3%) versus 3 (11.1%), p = 0.045), and MMSE total score (48 (60.0%) versus 9 (33.3%), p = 0.025) between the SCD patients with and without memory improvement.
The changes of various neuropsychological tests between the baseline and 24-month follow-up
Data are expressed as the mean with standard deviation or number with percentages in parenthesis. COWAT, controlled oral word association test; MMSE, Mini-mental status examination; n.a., not applicable; RCFT, Rey complex figure test; SVLT, Seoul verbal learning test. *p < 0.05.
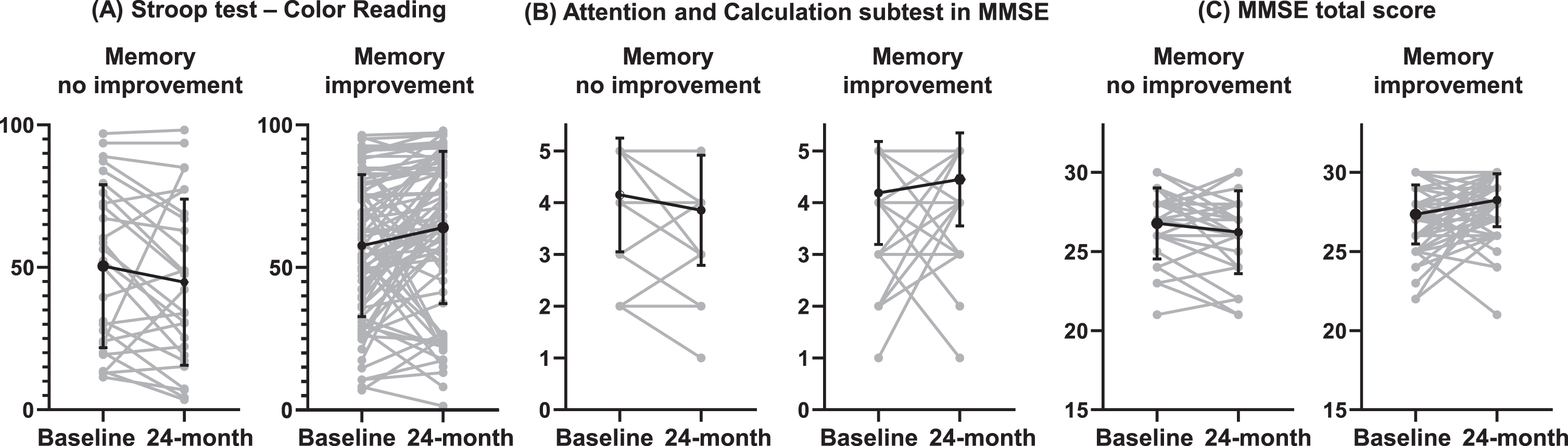
Changes in various neuropsychological evaluation test results at 24-month follow-up. A) Stroop test – color reading. B) Attention and calculation subtest in MMSE. C) MMSE total score.
DISCUSSION
In this prospective study, we identified the characteristics of SCD patients who demonstrated memory function improvement over a 24-month period. Our study revealed that 80 out of 107 (74.8%) SCD patients exhibited enhanced performance in verbal memory tasks during the 24-month follow-up. At baseline, SCD patients with memory improvement displayed a decreased prevalence of coronary artery disease, a reduced likelihood of a positive amyloid PET scan, and superior performance in executive function, as assessed by the trail-making test part B. Furthermore, these patients exhibited larger volumes in both the left and right superior parietal lobes comparison to SCD patients who did not experience memory function improvement.
Many previous studies have revealed that the practice effect can be seen in cognitively intact individuals and even patients with cognitive impairment [34–40]. It was estimated that a performance gain of approximately 0.25 effect size (Cohen’s d) would be observed in healthy individuals aged 70–75 years who underwent three assessments within a 6- to 12-month period [37]. A meta-analysis of the practice effect found that it was sustained over test-retest intervals of up to 5 years [35]. A longitudinal study of the practice effect and cognitive changes in normal individuals and incident MCI or dementia patients, with an average interval of 15 months between assessments, revealed that the stable normal group showed significant improvement in memory up to the third assessment, whereas the incident MCI/dementia group showed significant memory improvement only from baseline to the second test, followed by subsequent decline [38]. Similarly, when neuropsychological tests were administered repeatedly with a short interval, the practice effect was particularly pronounced in the early phase (3 months) of the tests and reached a plateau during the later phase (12 months) [34]. Declining performance on repeated neuropsychological tests can be observed in patients with AD and MCI. Among the MCI group, the lack of a practice effect at the very early phase (about 1 week) could predict later deterioration on the neuropsychological profiles [36].
Given these findings, we hypothesized that the memory improvement of the patients with SCD in our study could be partly attributed to the practice effect. However, the reason why the cognitive change in SCD patients was not solely due to the practice effect is that neuropsychological tests were conducted annually in our study, and the memory improvement group was classified based on the third assessment, 24 months after baseline. Notably, the practice effect was not observed in all patients with SCD, as 74.8% of SCD patients showed improvement in memory function but the others did not. Considering that the lack of a practice effect among MCI patients has been suggested to be a risk factor for clinical deterioration, SCD patients without memory improvement, or the practice effect, may be at risk of progression.
The presence of cardiovascular risk factors, including hypertension [41], diabetes [42, 43], dyslipidemia [44], and coronary heart disease [45], has been associated with cognitive decline and dementia. These comorbidities are well-established risk factors for dementia [46], mediated by neuropathological damage and vascular insufficiency, which result in cerebral hypoperfusion [44, 47]. Our study demonstrated that patients exhibiting improved memory function at the 24-month follow-up had a significantly lower prevalence of coronary artery disease at baseline. Although it remains unclear whether SCD patients with cardiovascular disorders exhibit a higher progression rate to MCI or dementia compared to those without such disorders, it was found that the burden of cerebrovascular damage such as white matter hyperintensities was higher in SCD than in controls [48]. Our study did not provide neuroanatomical evidence corroborating the relationship between coronary artery disease (CAD) and alterations in the memory function of SCD patients—specifically in the context of small vessel disease or microbleeds severity. Furthermore, the underlying cardiovascular risk factors have been shown to gradually and persistently impact brain structure through neuroinflammation, microvascular damage, and even psychological factors [42, 47]. It was observed that CAD patients with cognitive impairment showed elevated platelet activity relative to their cognitively intact counterparts CAD patients, and the heightened platelet activity could induce perivascular inflammatory responses and compromise the integrity of the blood-brain barrier [49, 50]. The damaged blood-brain barrier might hinder amyloid-β clearance via endocytosis and cerebral hypoperfusion could precipitate tau hyperphosphorylation, engendering neurofibrillary tangles [50]. Moreover, in this study, the presence of amyloid-beta pathology was identified as another significant factor related to changes in memory function among patientswith SCD.
In our study, we found that SCD patients exhibiting memory improvement were associated with negative amyloid PET scan results. The prevalence of amyloid pathology in SCD patients has been reported to span a wide range, from 10% to 76%, and a 27% amyloid positivity rate in SCD patients at the age of 70 [51]. Our findings indicated that SCD patients who experienced memory improvement demonstrated a 15.0% positive scan rate (70.5 years old on average), while those without memory improvement exhibited a 48.1% positive scan rate (71.0 years old on average). Individuals with preclinical AD, who are cognitively normal yet present evidence of amyloid pathology in the brain, are at an increased risk of developing dementia due to AD [52]. The presence of positive amyloid biomarkers in SCD patients serves a robust prognostic factor for cognitive decline and heightened dementia risk compared to those without an amyloid profile [53, 54]. Our findings are consistent with and support the previous research in this area [9]. Although the global SUVR of amyloid PET was not significantly different between the SCD patients with and without memory improvement, the analysis of regional SUVR showed that patients with improved memory function exhibited significantly lower amyloid burden, focused on the temporal and parietal areas, especially in the left hemisphere. The spatial pattern of amyloid burden may imply neurodegeneration and further cognitive impairment related to amyloid deposition. The brain amyloid deposition in the left precuneus and medial-temporal regions and bilateral parietal regions were related to the brain atrophy in the temporoparietal regions, which are vulnerable to AD pathology in MCI population [55]. In our study, the volumetric analysis showed that the left and right superior parietal volumes were significantly larger in SCD patients with memory improvement, similar to the result of regional brain amyloid SUVR analysis. These findings might clarify that the presence of AD signature imaging patterns would suggest how the prognosis in the aspect of memory function of SCD patients will be.
Regarding cognitive assessment, SCD patients exhibiting an improvement in memory function at baseline demonstrated superior performance on Trail-Making Test B, pertaining to executive function; however, no significant differences were observed in the results of delayed recall of verbal and visual memory tasks. Furthermore, at 24-month follow-up, SCD subjects with enhanced memory also displayed improved performance in executive function tasks and higher MMSE scores in comparison to baseline measurements, whereas those without memory amelioration exhibited worsening trends of statistical significance (Table 4, Fig. 2). Minor cognitive deficits, encompassing memory, executive, and language domains are discernible in patients with SCD [56]. Additionally, poorer performance on Trail-Making Test B was identified as a contributing factor for SCD patients who progressed to MCI or dementia during follow-up evaluations [57]. SCD patients exhibiting evidence of AD pathology (preclinical AD), who are considered at an elevated risk for future dementia, also demonstrated a decline in executive function over time [58]. Furthermore, executive function was found to be associated with amyloid deposition among SCD patients with or without objective subtle cognitive difficulties [59]. Moreover, executive function has been identified as intricately connected to cognitive reserve [60, 61]. A heightened cognitive reserve served as a protective factor against the progression to MCI in SCD patients and was found to influence daily activity performance through its association with executive function [60, 62]. Collectively, these findings suggest that memory improvement in SCD patients may transpire in conjunction with alterations in executive function.
At baseline, the severity of medial temporal atrophy or WMHs did not exhibit any significant difference between SCD with or without memory improvement. Nevertheless, the volumetric brain analysis revealed that SCD patients who demonstrated enhancement in memory function possessed larger volumes in the right and left superior parietal lobes compared to those without improvement. Moreover, the regional SUVR analysis of amyloid PET revealed that the regional amyloid burden in multifocal temporo-parietal cortices, including superior parietal gyrus, is significantly lower in SCD patients with memory improvement compared to those without. The superior parietal area, a constituent of the AD-signature regions [63], was associated with episodic memory retrieval [64] and a limited subsequent memory effect in SCD patients when contrasted with cognitively healthy controls [65]. It is well-established that reduced brain volume, a biomarker of neurodegeneration, is linked to a steeper decline of cognitive function among individuals with SCD [53, 67].
Our study has several limitations. Firstly, we introduced a novel definition for improvement in SCD, characterized by enhanced scores in the verbal memory delayed recall task. Nevertheless, in comparison to the definition of reversion from MCI to normal, establishing a concrete concept for improvement in SCD status remains challenging due to the fundamental features of SCD itself. Secondly, the small sample size of this study (total 107 patients, 80 from the improvement group, and 27 from the no improvement group) is a limitation that may reduce its statistical power. However, considering that this study investigated the novel concept of memory improvement in SCD, this result could serve as the foundation for a larger and more extensive study on SCD. Finally, our research did not account for all specific behaviors that patients may engage in to bolster their cognitive function, such as nutritional prevention of dementia, physical activity, or adopting hearing aids for hearing impairment. This study was designed to observe the natural progression of SCD and identify the characteristics of patients with SCD who either advance to MCI or dementia status or exhibit changes over time. Despite these limitations, our investigation boasts several strengths. We conducted a prospective two-years follow-up study, encompassing neuropsychological, neuroanatomical, AD biomarkers with amyloid PET, and lifestyle characteristics of SCD patients.
In conclusion, our findings suggested that the memory function enhancement in SCD may be attributed to the absence of significantly increased amyloid burden at baseline, decreased cardiovascular risk factors, and abundant executive function. Understanding the pivotal characteristics associated with cognitive improvement can aid clinicians in predicting which SCD patients will demonstrate improvement in memory function in the future. A comprehensive examination of biomarker profiles, underlying risk factors, and cognitive status may provide valuable insights into the prognosis of the cognitive function in SCD.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
This study was supported by the Ministry of Health and Welfare, HI18C0530.
CONFLICT OF INTEREST
All authors declared no conflict of interest.
DATA AVAILABILITY
Raw data will be made available upon request.
