Abstract
Background:
Constipation is a common symptom in dementia, and the cause is controversial. Rare clinical studies focused on plasma orexin-A levels and constipation in dementia.
Objective:
To evaluate the associations between orexin-A and constipation in patients with cognitive impairment.
Methods:
A total of 21 patients with mild cognitive impairment (MCI), 142 with Alzheimer’s disease (AD), and 57 with Lewy body dementia (LBD) were conducted. Besides informant-based history, neurological examinations or neuropsychological assessments, plasma levels of orexin-A, and constipation were assessed. The associations between orexin-A and constipation were evaluated by logistic regression models.
Results:
There were 47/220 (21.36%) cognitive impairment patients having constipation, and the proportion of constipation in LBD (61.40%) was significantly higher than AD (5.63%) and MCI (19.05%). No significant age or sex differences in the prevalence of constipation were found in the MCI, AD, and LBD groups. We found the cognitive impairment patients with constipation had lower levels of plasma orexin-A [1.00 (0.86, 1.28) versus 1.29 (1.01, 1.50) ng/ml, p < 0.001] than those without. And the plasma levels of orexin-A were significantly associated with the occurrence of constipation after adjusting for all variables in all patients with cognitive impairment (OR = 0.151, 95% CI: 0.042–0.537, p = 0.003). And the same finding was more prominent in the LBD group (p = 0.048).
Conclusions:
The decrease of plasma level of orexin-A is closely associated with the occurrence of constipation. Orexin-A has an intestinal protective effect and is involved in the gastrointestinal symptoms of patients with cognitive impairment.
Keywords
INTRODUCTION
The general prevalence of constipation worldwide ranges from 0.7 to 79% [1–4], with a latest prevalence rate of 10.1% (8.7–11.6) when Rome IV criteria were used [2]. Gender, age, social psychology, diet structure, drugs, alteration of intestinal flora, mucosal immune function changes (low-grade intestinal inflammation, increased intestinal permeability, immune activation), visceral hypersensitivity, and abnormal gastrointestinal motility have an impact on constipation [2, 3]. The alternation of intestinal flora interferes with the metabolism of toxic substances, resulting in impaired intestinal motility and chronic inflammation, which can cause or aggravate constipation [5, 6].
Constipation has been observed in variety of neurodegenerative diseases, such as Lewy body dementia (LBD), Parkinson’s disease (PD), and Alzheimer’s disease (AD) [2, 4]. The pathogeneses of constipation and dementia are unclear, while the brain-gut axis dysfunction and changes in intestinal flora both influenced the two diseases. The gut and CNS play key role in regulating the CNS function via gut microbiome, given that its relationships with regulating neuroendocrine, autonomic nervous system, host immune response, neurobehavioral effects, and detoxification mechanism [7, 8]. In addition to amyloid-β (Aβ) and tau, the postmortem of AD brain also showed bacteria, viruses, fungi, and protist parasites [9]. The mouse models of AD revealed that amyloid produced by the bacterial strain contributes to AD pathology by promoting misfolding of Aβ oligomers and fibrils [9–11]. Alteration in gut microbial composition enhances the release of amyloids, which may contribute to the pathogenesis of AD [12], especially during aging when both the intestinal epithelial barrier (IEB) and blood-brain barrier (BBB) become more permeable [13]. There were many questions remained regarding the mechanisms by which gut dysfunction leads to the development of α-synucleinopathies. Pathological α-synuclein can be found in CNS and enteric nervous system (ENS), which supports the Braak’s initial theory [14] that “α-synuclein aggregation occurs in the gut and spreads in a prion-like way to the brain”. The increased intestinal permeability and translocation of bacteria and their products are also associated with α-synuclein pathology in LBD and PD [15, 16]. Sampson et al. [17] found short-chain fatty acids and extracellular fibers produced from microbes in the gastrointestinal tract could affect α-synuclein aggregation, indicating the systemic inflammatory processes driven by gastrointestinal dysfunction contribute to α-synuclein.
The neuropeptides orexins A and B, also called hypocretins, derive from prepro-orexin and selectively bind to two types of receptors, OX1 R and OX2 R. Orexin has been observed in neurons and enteroendocrine cells of the gut [18]; thus, it is one of the peptides localized in both the CNS and the ENS [19]. It has been proposed that orexin-A is an intrinsic factor influencing the composition of gut microflora, as well as a chemical mediator in the bidirectional gut-brain axis. The neuroprotective role of orexin-A has been proved in several disease [20–23]. Tunisi et al. [18] showed the “peripheral” orexin-A released by myenteric neurons acting on OX1 R in enterocytes, could inhibit lipopolysaccharide (LPS)-induced IEB leakage as well as microglia activation when the composition of the microbiome is altered. And the “central” orexin-A also improves LPS-induced intestinal hyperpermeability via the vagal cholinergic pathway, again demonstrating that orexin mediates in bidirectional gut-brain communication [24]. Previous studies have only explored the relationship between intestinal flora and orexin-A or constipation, but there are few studies on orexin-A and constipation.
Due to the overlap in neuropathology and clinical symptoms between LBD and AD [25, 26], as well as the commonality of constipation in AD, LBD, and mild cognitive impairment (MCI) [4], we aimed to explore the association between orexin-A and constipation in patients with AD, LBD, and MCI in current study. These findings may 1) provide new biomarkers for the differential diagnosis of AD, LBD, and MCI to some extent, 2) provide new research targets and clinical evidence for the mechanism of brain-gut axis in dementia, and 3) be beneficial for constipation management in dementia.
MATERIALS AND METHODS
Study participants
The present study included 220 patients with cognitive impairment (21 patients with MCI, 142 patients with AD, and 57 patients with LBD) at the cognitive impairment clinics, Beijing Tiantan Hospital, from January to October 2022.
Diagnoses were confirmed by two experienced doctors and if there was disagreement, the subject was excluded. MCI was diagnosed if the participants had 1) subjective cognitive impairment; 2) objective impairment in one or more cognitive domains; 3) slightly impaired complex instrumental daily abilities, but independent daily living abilities; and 4) no dementia according to the clinical criteria recommended by the Winblad & Peterson [27, 28]. Probable AD was diagnosed based on the criteria of the National Institute on Aging and the Alzheimer Association workgroup, and 11C-PIB PET scans (only for assessing Aβ deposition, n = 112) or cerebrospinal fluid tests (n = 52) for AD neuropathological biomarkers were performed [29].
LBD comprised with dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD), probable DLB was diagnosed according to the criteria of McKeith in 2017 [30], and probable PDD was diagnosed according to the clinical criteria of the Movement Disorder Society in 2007 [31]. A probable DLB diagnosis can be made with two or more core symptoms with or without indicative biomarkers, or only one core symptom with one or more indicative biomarker. International consensus suggests that DLB should be diagnosed when cognitive impairment precedes parkinsonism or begins within a year of parkinsonism and PDD should be diagnosed when parkinsonism precedes cognitive impairment by more than one year.
Exclusion criteria were as follows: diagnosis of any other neurological disease except MCI, AD, or LBD; currently using orexin receptor antagonists; patients with hearing loss, aphasia, or an inability to complete a clinical examination or scale assessment; history of mental disorders and illicit drug abuse; patients with acute or chronic liver and kidney dysfunction, malignant tumors, or other serious underlyingdiseases.
The study was designed and conducted in accordance with the Declaration of Helsinki, received ethical approval from the Beijing Tiantan Research Ethics Committee (KYSQ 2021-068-01), and all participants had completed written informed consent.
Assessments of constipation
Constipation was diagnosed according to the revised Rome IV criteria in 2016 [32], and the standard references were all followed constipation assessment indicators. The typical clinical symptoms including straining, having lumpy or hard stools, having sensation of incomplete evacuation, having sensation of anorectal obstruction/blockage, needing manual maneuvers to facilitate defecation more than one quarter of times or having fewer than three spontaneous bowel movements per week. The criteria were fulfilled for the last 3 months, with two or more defecation difficulties onset at least 6 months prior to diagnosis, as our previous report[3].
Clinical assessments
All participants underwent a standardized diagnostic workup that included a semi-structured medical history interview, collection of an informant-based history [including demographic characteristics, lifestyles, comorbidities, family history of cognitive impairment, course of disease, and body mass index (BMI, kg/m2)], clinical and neurological examinations, neuropsychological assessments, as previously described [33]. Neuropsychological assessments included the Mini-Mental State Examination [34], the Montreal Cognitive Assessment [35], and Clinical Dementia Rating [36] used to evaluate global cognitive function and severity of cognitive impairment. The Activities of Daily Living scale [37] was used to measure the functional status.
Sample collection and measurement
After a 12–14 h overnight fast and between 07:30 and 08:30 am before breakfast, peripheral blood from each participant was collected by venipuncture into 6-mL EDTA-containing test tubes. Immediately (within 2 h) after collection, blood was centrifuged (2,200 rpm, 10 min) and stored at –80°C until measurement. Smoking, alcohol consumption, and vigorous activity were prohibited 24 h before the study.
Plasma orexin-A levels were measured using an enzyme-linked immunosorbent assay kit (EK-003-30; Phoenix Pharmaceuticals, Burlingame, CA, USA). The detection range of this kit is 0–100 ng/mL, with a sensitivity of 0.2 ng/mL, and the intra- and inter-assay variability were <10% and <15%, respectively. Samples were analyzed twice on the same plate and the concentration values obtained from all samples were within the detection linearity range (0.12–2.0 ng/mL) of the kit.
Genomic DNA was extracted from peripheral blood stored at –80°C and the Apolipoprotein E (APOE) gene was amplified using polymerase chain reaction [38].
All measurements were determined without knowledge of the patient status.
Statistical analyses
Descriptive analyses were conducted using frequency for qualitative variables and mean [±standard deviation (SD)] or median [interquartile range (IQR)] for quantitative variables. We classified all participants into two groups according to the presence or absence of constipation (“with constipation” or “without constipation”) or three groups according to their diagnosis (“MCI”, “AD” or “LBD”), and then subgrouped them based on sex (“male” and “female”) or age (“≤70 years old” or “>70 years old”). To assess differences in different groups, Student’s t-test was performed to compare two independent groups for normally distributed data and Mann-Whitney U test was used for nonparametric data; the qualitative variables were assessed using a chi-squared test. Binary logistic regression models [odds ratio (OR) with 95% confidence intervals (95% CIs)] were used to analyze the associations between plasma orexin-A levels and constipation.
For statistical analyses, the IBM Statistical Package for the Social Sciences (SPSS) for Windows (version 25.0; IBM Corporation, Armonk, NY, USA) was used. A p-value < 0.05 was considered statistically significant.
RESULTS
Demographic and clinical characteristics of all participants
We conducted 220 patients with cognitive impairment (21 patients with MCI, 142 patients with AD, and 57 patients with LBD) in this study, and the clinical characteristics in the different diagnostic groups are shown in Supplementary Table 1.
In total, there were 47 (21.36%) patients with constipation. We found that patients with constipation had more hypertension (59.57% versus 33.53%, p = 0.001) than those without, while there was no significant difference in other demographic information or scores of neuropsychological assessments (Table 1).
Sample characteristics
We also showed that the proportion of constipation in LBD (61.40%) was significantly higher than AD (5.63%, p < 0.001) and MCI (19.05%, p = 0.001). Males had slightly higher proportions of constipation than females in all patients, while we did not find significant differences between female and male subgroups in Fig. 1. In MCI and AD groups, patients≤70 years old had slightly higher proportions of constipation than those > 70 years old; while in LBD group, patients≤70 years old had lower proportion of constipation than those > 70 years old; however, there were no significant differences between age subgroups in each diagnosticgroup.
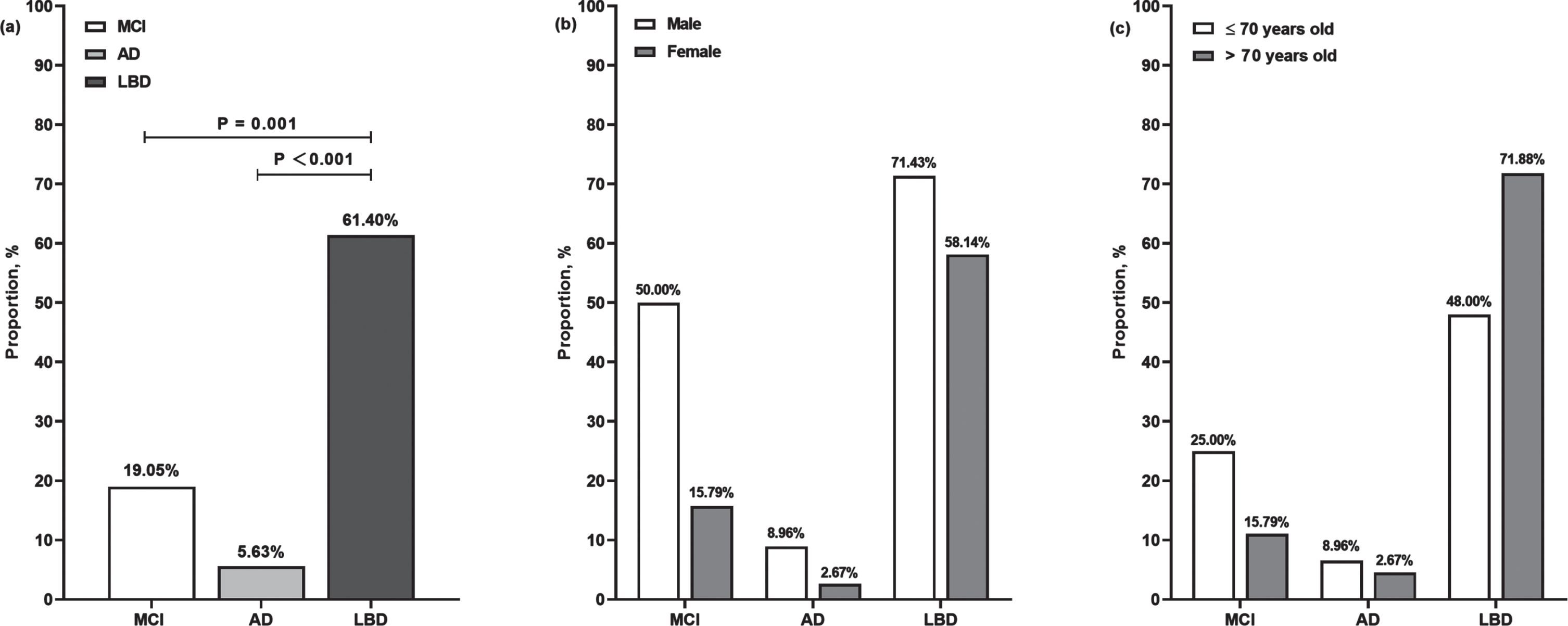
The proportions of constipation in three diagnostic groups. Panel a showed the proportions of constipation in MCI (4/21), AD (8/142), and LBD (35/57) group, the proportion in LBD was significantly higher than MCI and AD. Panel b showed the proportions of constipation according to diagnosis-gender and there was no significant difference between female and male subgroups in each diagnostic group. Similarly, there was no significant difference between age subgroups in each diagnostic group according to diagnosis-age in c. MCI, mid cognitive impairment; AD, Alzheimer’s disease; LBD, Lewy body dementia.
As shown in Table 1, we found that patients with constipation had lower levels of plasma orexin-A [1.00 (0.86, 1.28) versus 1.29 (1.01, 1.50) ng/ml, p < 0.001] than those without. When we did a subgroup analysis of the three diagnostic groups (Table 2), we found that plasma orexin-A was highest in patients with AD than those with MCI and LBD (p < 0.001), with a median level of 1.31 (IQR: 1.01, 1.52) ng/ml. We found that the patients with higher plasma levels of orexin-A were more likely to be diagnosed with AD, but not LBD, after adjusting all confounders in regression models (Supplementary Table 2, LBD versus AD: OR = 0.213, 95% CI: 0.074–0.613, p = 0.004). Additionally, there were no significant differences of plasma orexin-A levels between gender and age in each diagnostic group. When subgrouping according to diagnostic-constipation, plasma levels of orexin-A in MCI, AD and LBD patients with constipation were found to be lower than those in the non-constipation group, but there was no significant difference.
The distributions of plasma levels of orexin-A (ng/ml) in three diagnostic groups
IQR, interquartile range; SD, standard deviation; MCI, mid cognitive impairment; AD, Alzheimer’s disease; LBD, Lewy body dementia; y, years.
Unless otherwise indicated, the continuous variables conforming to normal distribution were described by mean (±SD), and the non-normal distribution continuous variables were expressed by median (IQR). Comparison between the “without constipation” and “with constipation” groups was performed by t-test or Mann–Whitney U test, respectively. The Chi-square test was used to compare the categorical variables between groups.
SD, standard deviation; IQR, interquartile range; T2DM, type 2 diabetes mellitus; APOE, Apolipoprotein E; MMSE, the Mini-Mental State Examination; MoCA, the Montreal Cognitive Assessment; ADL, the Activity of Daily Living Scale; CDR, the clinical dementia rating; y, years; mo, months
Associations between plasma levels of orexin-A and constipation
In order to explore the associations between plasma levels of orexin-A and constipation, we established different binary regression analysis models in Table 3. We found that plasma levels of orexin-A were significantly associated with the occurrence of constipation in all patients with cognitive impairment (both in Model 1 and Model 2, p < 0.001), even after adjusting for gender, age, hypertension, and diagnostic variables in Model 3 (OR = 0.151, 95% CI: 0.042–0.537, p = 0.003). We also found that the plasma levels of orexin A were significantly associated with constipation in the LBD group, with the OR = 0.147 (95% CI: 0.025–0.867, p = 0.034) in Model 1 and OR = 0.162 (95% CI: 0.026–0.987, p = 0.048) in Model 2. Unfortunately, we did not find a significant association between plasma levels of orexin-A and constipation in the MCI and AD groups.
Associations between plasma levels of orexin-A and constipation in variable models
MCI, mild cognitive impairment; AD, Alzheimer’s disease; LBD, Lewy body dementia; ORs, odd ratios; CI, Confidence Interval. *p < 0.05; **p < 0.01; ***p < 0.001. Model 1: Crude model; Model 2: Adjusted gender, age, history of hypertension; Model 3: Adjusted gender, age, history of hypertension and diagnosis.
DISCUSSION
With regard to the role of central orexin in digestive functions, it had been proved to act centrally to stimulate gastrointestinal secretion or motility [39, 40]. To our knowledge, we firstly explored the association between orexin-A and constipation based on clinical level in the present study, and pointed out that in patients with cognitive impairment, lower plasma levels of orexin-A was associated with the occurrence of constipation. This finding was particularly strong among people with LBD, and it provides a new biomarker for the differential diagnosis of AD, LBD and MCI to some extent.
In this study, we found 21.36% patients with cognitive impairment had constipation, the proportion was higher than the prevalence rate of general population [10.1% (8.7–11.6)] [2], as well as our previous report (17.6%) in old population≥65 years old based on community study [3]. A recent view had emerged that degenerative component increases the occurrence of constipation, and constipation had been observed in a variety of neurodegenerative diseases, especially in DLB and PD [41, 42]. We found that the proportion of constipation in patients with LBD were 61.4%, and significantly higher than AD and MCI groups (both p = 0.001). LBD is a major neurocognitive dementia with α-synuclein deposition/Lewy bodies, constipation is the most frequently dysautonomia symptom and the earlier manifestation of LBD. We previously reported that 71/185 patients with LBD had constipation in their mild-moderate stage, with PDD (50/93) being more pronounced than DLB (21/92) [43]. In the cohort of Honolulu Heart Program, the prevalence of PD increased significantly with decreasing bowel movement frequency, indicating that a history of constipation was associated with PD [44]. Additionally, our multicenter cross-sectional survey revealed [4] that the prevalence rates of constipation were 19.2% (95% CI, 17.3–21.0), 19.1% (95% CI, 16.8–21.5), 14.4% (95% CI, 12.8–15.9), and 13.8% (95% CI, 13.0–14.6) in the all-cause dementia, non-amnestic-MCI, amnestic-MCI and normal cognition populations, all the prevalence rates were significantly lower than LBD. Traditionally, loss of dietary fiber consumption and reduction of physical exercise can lead to constipation, and the prevalence of constipation increases significantly with age [45]. After subgrouping by age, we found the patients≤70 years old had slightly higher proportions of constipation than those > 70 years old in MCI and AD groups; while in LBD group, patients≤70 years old had lower proportion of constipation than those > 70 years old. However, there were no significant differences between age or gender subgroups in each diagnostic group. In addition to LBD, we found similar trends but without significant difference in AD and MCI as in previous studies; In addition, the results in the LBD group in our study were inconsistent with previous studies, possibly because of the small sample size.
The potential mechanism of high proportion of constipation in LBD is “LBD pathology might originate in the bowel” [46], the brain-gut axis dysfunction can affect the occurrence of constipation. A series of pathological reports by Braak [46, 47] showed that PD pathology in the brain starts in the dorsal vagal nucleus (that might regulate bowel function) earlier than in the substantia nigra (that regulates motor function). Gelpi et al. [48] also demonstrated that α-synuclein aggregated in the peripheral nerves, like vagus nerve (86.7%), myenteric plexus (86.7%), and cardiac sympathetic nerve (100%), earlier than in the brain. The same findings were proved by Beach et al. [49] by immunohistochemically staining, they verified that 12/26 LBD (46%) and 32/36 PD (89%) patients performed pSyn-positive in the vagus, 5/30 (17%) LBD and 42/52 (81%) of PD subjects were pSyn-positive in the stomach. And many clinical studies have proven that constipation appears about 10 to 20 years prior to motor symptoms [50], and 3.7 (±9.2) years preceding the onset of memory loss [51]. Meanwhile, recent studies have pointed out that changes in intestinal flora are associated with the occurrence of constipation [7, 52], but the “causal relationship” between them is still worth discussing. Constipation can cause intestinal flora disorder, then disturb the structure and function of IEB, leading to the abnormal aggregation and metabolism of Lewy bodies, as well as aggravating constipation symptom. Moreover, the alteration in gut microbial composition enhances the release of amyloids, which may contribute to the pathogenesis of AD, especially during aging when both the IEB and BBB become more permeable [9–11].
Orexin-A was initially generated by hypothalamic neurons and participated the neuroprotection mostly through the effect on microglia activation [53]. Many studies have found orexin-A and OX-1 R also presented in peripheral tissues including the gut [54]. The orexin-A-producing neurons in the myenteric plexus can project to OX-1R-expressing enterocytes of the intestinal villi in the IEB [55], possibly indicating their involvement in gut motility, intestinal absorption, and/or secretion of nutrients [56]. Thus, we analyzed plasma levels of orexin-A in different MCI, AD, and LBD groups and found that the plasma levels of orexin-A in patients with AD were significantly higher than those in LBD and MCI. We have previously reported that the plasma levels of orexin-A in MCI-LB and DLB was higher than that in the normal elderly, but there was no statistical difference between the MCI-LB and LBD groups [38]. Our findings indicated that plasma levels of orexin-A have no significant relationship with sex and age.
However, the pathogeneses of constipation and cognitive impairment are unclear, but they both share similar risk factors, such as age, sex, economic status, dietary structure, etc., and several studies have pointed that the neuroendocrine factors, including orexin-A, can affect the occurrence of the two diseases [18, 57–59]. The present results strongly suggested the decreased plasma levels of orexin-A significantly increased the risk of constipation. Previous literature pointed that orexin-A facilitates distal stomach motility and relaxation of the proximal stomach via the vagus nerve [18, 57]. Kobashi et al. [60] found orexin-A can induce relaxation of the proximal stomach and phasic contractions in the distal stomach, but the distal gastric contractions fail in animal models with vagal resection. Mouse studies using both mechanical and electrophysiological techniques also confirmed a direct effect of orexin-A on the smooth muscle of the duodenum that may reinforce or compensate for orexinergic signals from neurons of the dorsal motor nucleus of the vagus (reviewed in [24]). Alterations of gut microbiota is also involved in the link between orexin and constipation. The alterations of gut microbiota causing by constipation can increase IEB permeability, then aggravate the leakage of LPS into the systemic circulation. Orexin-A can block LPS translocation to the blood, weaken metabolic endotoxemia and activation of immune cells, as well as the subsequent systemic and central inflammation, which are responsible of BBB dysfunction and microglia activation. Also, orexin-A released by Myenteric neurons can prevent occludin decreasing through interacting with OX-1 R expressed on the basolateral side of enterocytes, thereby preventing intestinal barrier impairment induced by LPS. It follows then that peripheral orexin-A may act as a protective factor of the epithelial barrier, preventing LPS transfer from the gut to the CNS and reducing the occurrence of neuroinflammation. We speculate that the increased permeability of the IEB leads to α-synuclein aggregation within the ENS [61], which further affects the gastric motility, which may be the reason for the high proportion of constipation in LBD.
Although we have verified the effect of orexin-A on constipation in the clinical population for the first time, the universality of the results is limited due to the small sample size of the single-center study and its concentration in the cognitive impairment population. In addition, due to the lack of control group and the lack of records of lifestyle and other information, we cannot determine other causes affecting constipation. We need to further expand the sample size for more detailed and in-depth comparable studies.
The decrease of plasma level of orexin-A is closely related to the occurrence of constipation, and the orexinergic nervous system is involved in the gastrointestinal symptoms of patients with cognitive impairment. These findings suggest that orexin-A acts as a neuroendocrine medium through which the brain-gut axis co-regulates symptoms of cognitive impairment, confirming the role of the brain-gut axis in neurodegeneration. In addition, we also suggest that orexin-A has an intestinal protective effect, focusing on plasma levels of orexin-A can effectively determine the constipation complications. Clinicians should pay attention to the orexinergic nervous system in the treatment and management of cognitive impairment, which will help to improve the quality of life of patients with cognitive impairment.
Footnotes
ACKNOWLEDGMENTS
The authors are grateful to all those who participated in this study and wish to acknowledge the valuable assistance obtained from all specialized physicians, as well as the Tianjin Key Medical Discipline (Specialty) Construction Project for their help. We sincerely gratitude Jing Li (Tianjin Huanhu Hospital, Tianjin, China), Xia Yang (Beijing Tiantan Hospital, Capital Medical University), Zhichao Chen (Beijing Friendship Hospital, Capital Medical University), and Moyu Li (Beijing Tiantan Hospital, Capital Medical University) for the data collection and input. The ELISA tests were sponsored by Dr. Sen Liu and his research team at Beijing Pason Pharmaceuticals Inc., including the experimental methods, purchase for diagnostic reagents, and technical support. We would like to express our sincere gratitude to Dr. Sen Liu and his team. All these authors accept responsibility for all aspects of the manuscript and approved the final version of the manuscript.
FUNDING
This work was supported by the National Natural Science Foundation of China [grant number 82171182], Tianjin Science and Technology Plan Project [grant number 22ZYCGSY00840], Tianjin Health Research Project [grant number ZC20121 and TJWJ2023QN060], and Tianjin Key Medical Discipline (Specialty) Construction Project [grant number TJYXZDXK-052B].
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Dr. Ji is an Editorial Board Member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
