Abstract
Background:
Increasing evidence is demonstrating that degeneration of specific thalamic nuclei, in addition to the hippocampus, may occur in Alzheimer’s disease (AD) from the prodromal stage (mild cognitive impairment – MCI) and contribute to memory impairment.
Objective:
Here, we evaluated the presence of macro and micro structural alterations at the level of the anterior thalamic nuclei (ATN) and medio-dorsal thalamic nuclei (MDTN) in AD and amnestic MCI (aMCI) and the possible relationship between such changes and the severity of memory impairment.
Methods:
For this purpose, a sample of 50 patients with aMCI, 50 with AD, and 50 age- and education-matched healthy controls (HC) were submitted to a 3-T MRI protocol with whole-brain T1-weighted and diffusion tensor imaging and a comprehensive neuropsychological assessment.
Results:
At macro-structural level, both the ATN and MDTN were found significantly smaller in patients with aMCI and AD when compared to HC subjects. At micro-structural level, instead, diffusion alterations that significantly differentiated aMCI and AD patients from HC subjects were found only in the ATN, but not in the MDTN. Moreover, diffusion values of the ATN were significantly associated with poor episodic memory in the overall patients’ group.
Conclusions:
These findings represent the first in vivo evidence of a relevant involvement of ATN in the AD-related neurodegeneration and memory profile and strengthen the importance to look beyond the hippocampus when considering neurological conditions characterized by memory decline.
INTRODUCTION
The progressive deterioration of episodic memory has been proven to be the earliest and most reliable cognitive marker of Alzheimer’s disease (AD) [1]. In fact, poor learning ability and rapid memory decay over a relatively short period of time have been consistently documented in patients with AD and in patients with amnestic mild cognitive impairment (aMCI) in the prodromal stage of AD dementia [2–5]. The medial temporal lobe (MTL), including the hippocampal and parahippocampal cortices, is critical for episodic memory functioning and is particularly vulnerable to AD from the prodromal stage of the disease [6–8]. Consequently, it is not surprising that most previous neuroimaging studies in aMCI and AD have mainly focused on such cortical areas.
In the last few years, however, greater evidence demonstrates that degeneration of other brain structures inside the Papez circuit and in addition to the MTL can also occur early in the process of AD and can contribute to the episodic memory impairment [9–11]. Inside the Papez circuit, the anterior thalamic nuclei (ATN) are prominent due to their central contribution to episodic memory and their vulnerability to AD starting from the prodromal stage of the disease. Specifically, through connectivity with the MTL and cingulate cortices, the ATN are assumed to be involved in learning and memory. In fact, vascular damage to the ATN and/or the mammillo-thalamic tract (connecting the mammillary bodies to the ATN) is a core feature of diencephalic amnesia [1, 13]. Regarding the vulnerability of these thalamic nuclei to AD, pathological studies have shown that the earliest accumulation of neurofibrillary tangles starts in the ATN [7, 11]; these nuclei also show the clearest structural changes with normal aging [14]. Recently, Perry and colleagues [15] quantified neurons and glial cells in the ATN of four older patients affected by Down syndrome (a population with a significantly higher prevalence of AD than the general population). The authors found a striking volume reduction of ATN in these patients, which reflected the loss of about 70% of neurons. They concluded that the pathology in this region likely contributes to the memory impairments observed in these patients.
Another thalamic structure which has been implicated in episodic memory functioning is the medio-dorsal thalamic nucleus (MDTN), owing to its dense connections with parahippocampal regions, especially the perirhinal cortex [9, 17]. Aggleton and Brown [9] proposed that the rostral parahippocampal-medial dorsal thalamic axis is critical for familiarity-based recognition. This nucleus, together with various midline and intralaminar thalamic nuclei, is also assumed to have broader, indirect effects on recollective-based recognition [16]. However, whether damage restricted to the MDTN is sufficient to impair declarative memory is still unclear. Two systematic reviews of reported cases with diencephalic amnesia [13, 18] failed to find a direct link between MDTN damage and declarative memory impairment. In both cases, the authors concluded that although memory deficits are quite consistently observed following mammillo-thalamic tract/ATN damage, executive deficits are more frequently associated with damage to the medio-dorsal thalamic region. In fact, the MDTN is linked to the prefrontal cortex via a thalamo-frontal pathway [17].
Despite the importance of the thalamus for normal memory functioning and the evidence of pathological changes occurring early in the thalamus of aMCI and AD patients, thalamic nuclei have received little attention in imaging studies of AD. Recently, Bernstein and colleagues [19] investigated the presence of significant differences in the volume of thalamic nuclei across the continuum of disease progression from prodromal to clinical AD. In fact, in a large cohort of biomarker confirmed patients with early MCI, late MCI, AD, and healthy elderly controls, the volumes of 11 specific thalamic nuclei were obtained for each subject using a novel MRI-based thalamic segmentation technique [19]. The authors found significant between-group differences in several thalamic nuclei. In particular, the anteroventral nucleus was significantly smaller in late MCI and AD patients when compared to healthy control (HC) subjects, and the mediodorsal nucleus was also significantly smaller in subjects with early MCI, late MCI, and AD when compared to HC subjects. Interestingly, the anteroventral nucleus and the mediodorsal nucleus, together with the pulvinar, were the only nuclei whose volumes significantly correlated with both biomarker levels (i.e., tau, p-tau, or Aα) and cognitive measures (i.e., Montreal Cognitive Assessment, word-list recall, Clinical Dementia Rating, and Alzheimer’s Disease Assessment Scale–Cognitive Subscale-13) in the whole group of participants.
In light of the foregoing, here we aimed to assess whether there are differences in the structural integrity of specific thalamic nuclei in patients with aMCI or AD compared to elderly subjects without cognitive impairment and whether such changes are related to the severity of episodic memory impairment. For this purpose, we analyzed grey matter volumes at the level of thalamic nuclei, in order to determine whether significant atrophic changes in the ATN and MDTN are present in aMCI and AD patients compared to elderly controls. In addition to volumes, we also assessed microstructural alterations using diffusion tensor imaging (DTI). In particular, the presence of pathological processes at the level of the ATN and MDTN was explored by investigating whether there are associated changes in the overall magnitude of diffusion (mean diffusivity, MD) or in the degree of its directionality (fractional anisotropy, FA). Increased mean diffusivity and decreased fractional anisotropy are thought to reflect loss of integrity of cellular structures and are commonly observed in aging and neurodegenerative diseases [20, 21]. Finally, we assessed the potential correlation of these structural brain alterations with the severity of the episodic memory impairment in both groups of patients.
MATERIALS AND METHODS
Participants
A cohort of 50 patients with aMCI, 50 with probable AD and 50 HC were enrolled in this cross-sectional study. The appropriateness of our sample size was established using G*Power software. Considering the effect size obtained in previous studies [10], a power of 0.8 and an alpha of 0.05, we estimated that about 50 subjects per group would be needed. Sample recruitment and data collection were performed from January 2018 to March 2020. AD and aMCI patients were consecutively recruited from the specialist dementia clinics of Santa Lucia Foundation (Rome, Italy). They were submitted to a formal clinical, neuropsychological, behavioral, and functional evaluation and a computed tomography (CT) or a magnetic resonance (MR) scan as part of the diagnostic process. Diagnosis of aMCI was made according to established criteria [22], which includes: (a) a complaint of memory decline (described by the patient and confirmed by an informant); (b) objective memory impairment (revealed by scores below age/education-adjusted norms on at least one of the standard episodic memory tests administered); (c) normal general cognition, as indicated by a Mini-Mental State Examination (MMSE) score above the normality cut-off (>23.8); (d) normal activities of daily living (as confirmed by a total Clinical Dementia Rating scale score less than or equal to 0.5); (e) CT or MR brain imaging negative for focal lesions (minimal diffuse changes or minimal lacunar lesions were allowed). Furthermore, patients did not fulfill the clinical criteria for the diagnosis of dementia and had no history of drug/alcohol abuse or any psychiatric or neurological disease. Patients with AD met the clinical criteria for Alzheimer’s dementia established by the National Institute on Aging and the Alzheimer’s Association [23]. HC were recruited through local advertisements. They were submitted to a formal clinical and neuropsychological evaluation to exclude cognitive decline (for details, see “General Neuropsychological Examination” section). Inclusion criteria were (a) absence of neurological or psychiatric disorders; (b) no history of alcohol or drug abuse; (c) age-appropriate cognitive functioning as confirmed by performance above the normality cut-off scores on the MMSE (>24) and each neuropsychological test administered (Table 2).
All participants gave their written informed consent in accordance with the Declaration of Helsinki in a protocol approved by the Joint Ethics Committee of the Santa Lucia Foundation.
Neuropsychological assessment
The tests comprising the neuropsychological battery are listed below according to the cognitive domains they examine: (a) Verbal episodic long-term memory: 15-Word List recall test [24]; (b) Visuo-spatial episodic long-term memory: the Rey-Osterrieth complex figure recall test [25]; (c) Executive functions: Phonological and Categorical Word Fluency test [24], Stroop Test [26] and Modified Card Sorting Test [27]; Reasoning: Raven’s Coloured Progressive Matrices [24]; Constructional praxis: Copy of simple drawing and Copy of drawing with landmarks [24], Copy of the Rey-Osterrieth complex figure [25]. For all tests we used Italian normative data for score adjustment (sex, age, and education) and to define normality cut-off scores, which were established as the lower limit of the 95% tolerance interval for a confidence level of 95% (which corresponds to approximately – 1.3 SD). For each test, normative data are reported in the corresponding references.
In the neuropsychological battery, the tests of interest for the correlation analyses with brain measures were the 15-word-list recall test [24] and the Rey-Osterrieth complex figure recall test [25].
The 15-word-list recall test [24] consists of a list of 15 unrelated names of concrete objects. The examiner reads the wordlist aloud five times. Immediately following each presentation and 15 min after the last one, the subject is required to recall as many words as possible without a time limit and in any order. The immediate recall score consists of the total number of words recalled in the five immediate trials (range 0–75) and the delayed score consists of the number of words recalled after the 15-min delay (range 0–15).
The Rey-Osterrieth complex figure recall test [25] consists of three trials. First, a copy task, in which the subject is required to copy a complex figure placed in front of him/her as accurately as possible. Immediately after the copy, an immediate recall trial is administered; here, the model is removed from sight and the subject is required to retrieve the figure from memory. After 20-min delay, the subject is asked again to recall the figure from memory (i.e., delayed recall trial). The figure consists of 18 units and the maximum score for each of the three trials (copy, immediate, and delayed recall) is 36 (max 2 points for each unit).
For both memory tests, we used delayed recall scores as variables of interest in the correlation analyses, because of the widely-demonstrated higher sensitivity of these scores in identifying AD patients from the prodromal stages compared to immediate recall scores [1–5].
MRI protocol and image processing
All participants underwent the same imaging protocol, which included standard clinical sequences (FLAIR, DP-T2-weighted), whole-brain T1-weighted and diffusion-weighted scanning using a 3T Allegra MR imager (Siemens, Erlangen, Germany). Diffusion-weighted volumes were acquired using echo-planar imaging (TE/TR = 89/8500 ms, bandwidth = 2126 Hz/vx; matrix size 128×128; 80 axial slices, voxel size 1.8×1.8×1.8 mm3) with 30 isotopically distributed orientations for the diffusion-sensitizing gradients at a b-value of 1000 s/mm2 and 2 b = 0 images. Scanning was repeated three times to increase the signal-to-noise ratio. Whole-brain T1-weighted images were obtained in the sagittal plane using a modified driven equilibrium Fourier transform (MDEFT) [28] sequence (TE/TR = 2.4/7.92 ms, flip angle 15°, voxel-size 1×1×1 mm3).
Thalamic nuclei volumetry was obtained using the FreeSurfer (version 7.1.1) pipeline of Iglesias and colleagues [29], which is a Bayesian segmentation algorithm based on a histologically derived probabilistic thalamic atlas, leading to volume quantification of ATN and MDTN volume. Raw volumes were normalized upon the total intracranial volume estimated by FreeSurfer segmentation pipeline, in order to correct measures for individual total head-size.
DTI processing was performed using FSL 5 (http://www.fmrib.ox.ac.uk/fsl/). Image distortions induced by eddy currents and head motion were corrected by applying a 3D full affine alignment of each image to the mean b0 image. Then, DTI data were averaged and concatenated into 31 (1 b0 + 30 b1000) volumes. A diffusion tensor model was fit at each voxel, generating FA and MD maps. The FA maps were co-registered to T1-weighted images using a full affine (correlation ratio cost function) alignment with nearest-neighbor resampling, in order to rescale microstructural information into the smaller spatial resolution of T1-weighted images, and thus making DTI maps more compatible with the limited sizes of thalamic nuclei. The calculated transformation matrix was applied to the MD maps with identical resampling options. FA and MD values within ATN and MDTN were obtained as follows: first, anatomical T1-weighted images were processed with the segmentation tool FIRST [30] integrated in the FSL software [31] in order to segment participants’ thalami (as a whole), which were saved as regions of interest (ROI) (Fig. 1). Then, for each subject, the segmented ROIs and the coregistered FA map were superimposed on the original T1-weighted volume and the resulting images were visually assessed by two experienced researchers (FP and GS) to exclude misregistration or erroneous ROIs identification. These segmented ROIs defined the binary masks where mean values of FA and MD were calculated for each individual. Finally, in order to extract microstructural measures of thalamic ATN and MDTN, we used a published atlas (“The Thalamus Atlas” [32, 33]), which previous research found consistent with histologic sections [34], focal stroke localization [35] and with functional cluster localization [36]. The Atlas provides individual NIfTI files in stereotactic space (MNI) for ATN and MDTN, which were used as ROIs to extract mean FA and MD for each subject. Increased MD and decreased FA are thought to reflect loss of integrity of cellular structures [20, 21].
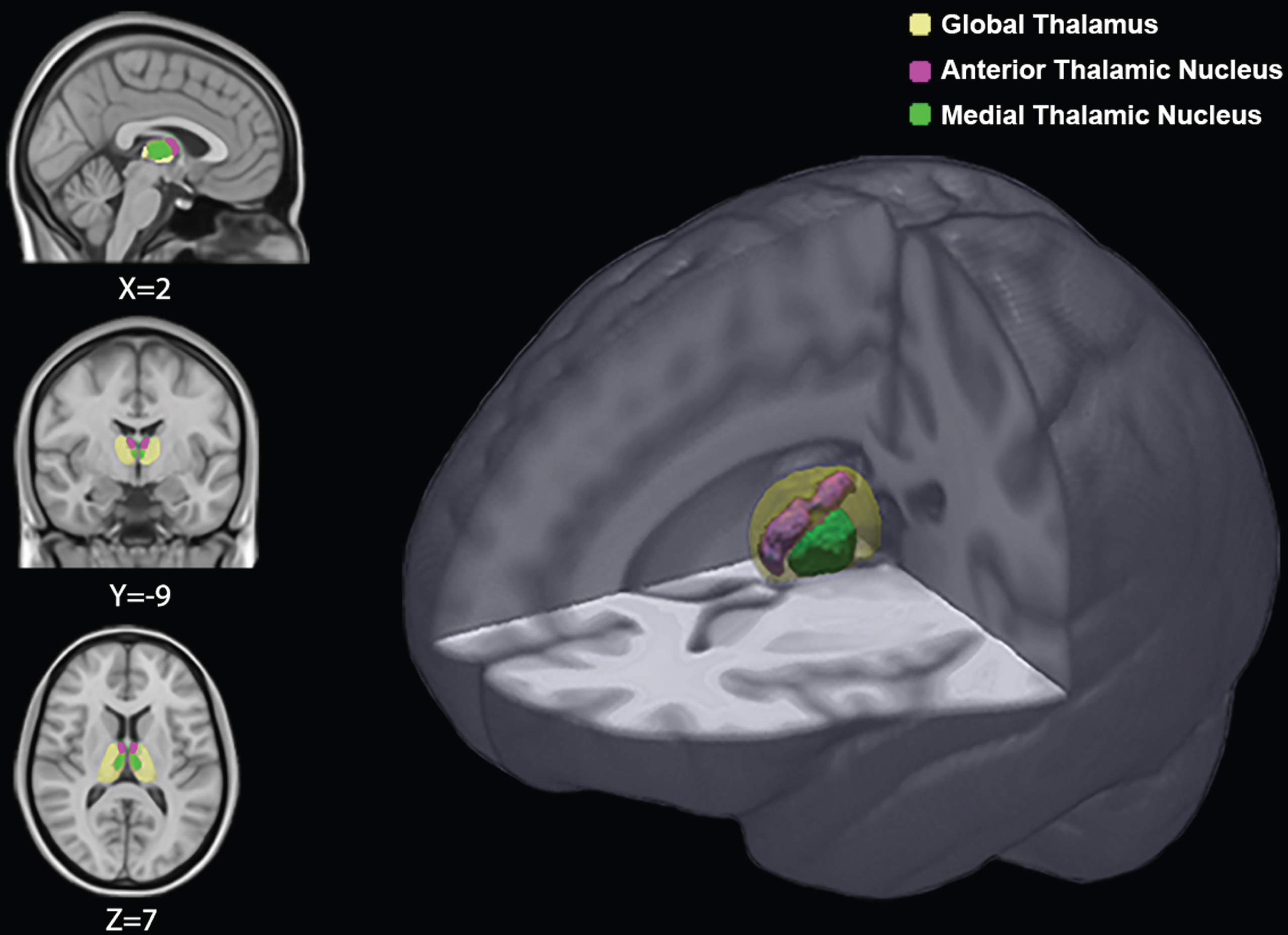
Global thalamus and thalamic nuclei superimposed on a reference MRI scan. Coordinates are in Montreal Neurological Institute space.
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences (IBM SPSS, Version 22.0, Inc., Chicago, IL).
Considering that most of the variables showed non-normal distribution, the non-parametric Kruskal-Wallis H test was used to detect between-group differences in demographic (age and education) and neuropsychological test performance. For comparing volumes and diffusivity data (FA and MD) of each individual thalamic nucleus (left ATN, right ATN, left MDTN, right MDTN) among the aMCI, AD, and HC groups, Quade’s rank analysis of covariance (non-parametric ANCOVA) was used in order to control for age. When significant effects were obtained, post-hoc comparisons were conducted using the Least Significant Difference (LSD) test. To account for multiple comparisons, the Benjamini–Hochberg procedure was used to control the false discovery rate (FDR). Finally, the non-parametric Kendall’s tau correlation coefficients (τ) with FDR correction for multiple comparisons was applied to determine whether thalamic volumes or diffusion FA and MD values of ATN and MDTN were significantly associated with neuropsychological memory scores (15-word list delayed recall scores and Rey-Osterrieth complex figure delayed recall scores) in aMCI and AD participants as a whole group. In addition, partial non-parametric Kendall’s correlation was performed in order to evaluate whether significant associations that emerge from previous analysis also stand after controlling for the potentially confounding effect of age and education.
RESULTS
Demographic and neuropsychological data
As shown in Table 1, the three groups were comparable for mean age, education and gender distribution. As expected, a significant difference for the MMSE emerged, with HC performing better than aMCI and AD and aMCI obtaining higher scores than AD. Moreover, HC performed better than both aMCI and AD on all tests included in the neuropsychological battery with the exception of the copy of simple drawing and the copy of drawing with landmarks, in which scores of HC and aMCI were comparable. In turns, aMCI patients obtained higher scores than AD patients on all tests included in our neuropsychological battery.
Demographic and neuropsychological data of HC, aMCI, and AD patients
Mean scores with standard deviations in parentheses. Range in square brackets. HC, healthy controls; aMCI, amnestic mild cognitive impairment; AD, Alzheimer’s disease; MMSE, Mini-Mental State Examination; WL, word list test; ROCF, Rey-Osterrieth complex figure; Copy drawing I, Copy of simple drawing; Copy drawing II, Copy of Drawing with landmarks; MCST, modified card sorting test; Stroop-I, Stroop test interference scores. aSignificantly different from HC subjects (p < 0.05). bSignificantly different from aMCI patients (p < 0.05).
Correlations between volumetric and diffusivity data of ATN and MDTN and memory scores in aMCI and AD patients
Kendall tau correlation coefficient with associated FDR-corrected p value in parenthesis. ATN, anterior thalamic nuclei; MDTN, medio-dorsal thalamic nuclei; L, left; R, right. ROCF, Rey-Osterrieth complex figure. *Statistically significant (FDR-corrected p < 0.05).
Thalamic nuclei volumes: Between group differences and correlations with memory scores
As reported in Fig. 2, Quade’s rank ANCOVA showed significant between group differences in grey matter volumes in the left ATN (F[2,147] = 6.85, FDR p = 0.002), right ATN (F[2,147] = 5.09, FDR p = 0.012), left MDTN (F[2,147] = 7.29, FDR p = 0.002), and right MDTN (F[2,147] = 7.79, FDR p = 0.002). In all analyses, post-hoc comparisons evidenced significantly smaller thalamic nuclei in AD and aMCI patients when compared to HC subjects, whereas patients’ groups did not differ each other. However, as reported in Table 2, no significant association between thalamic nuclei volumes and memory performance scores was found in the aMCI and AD patients as a whole group.
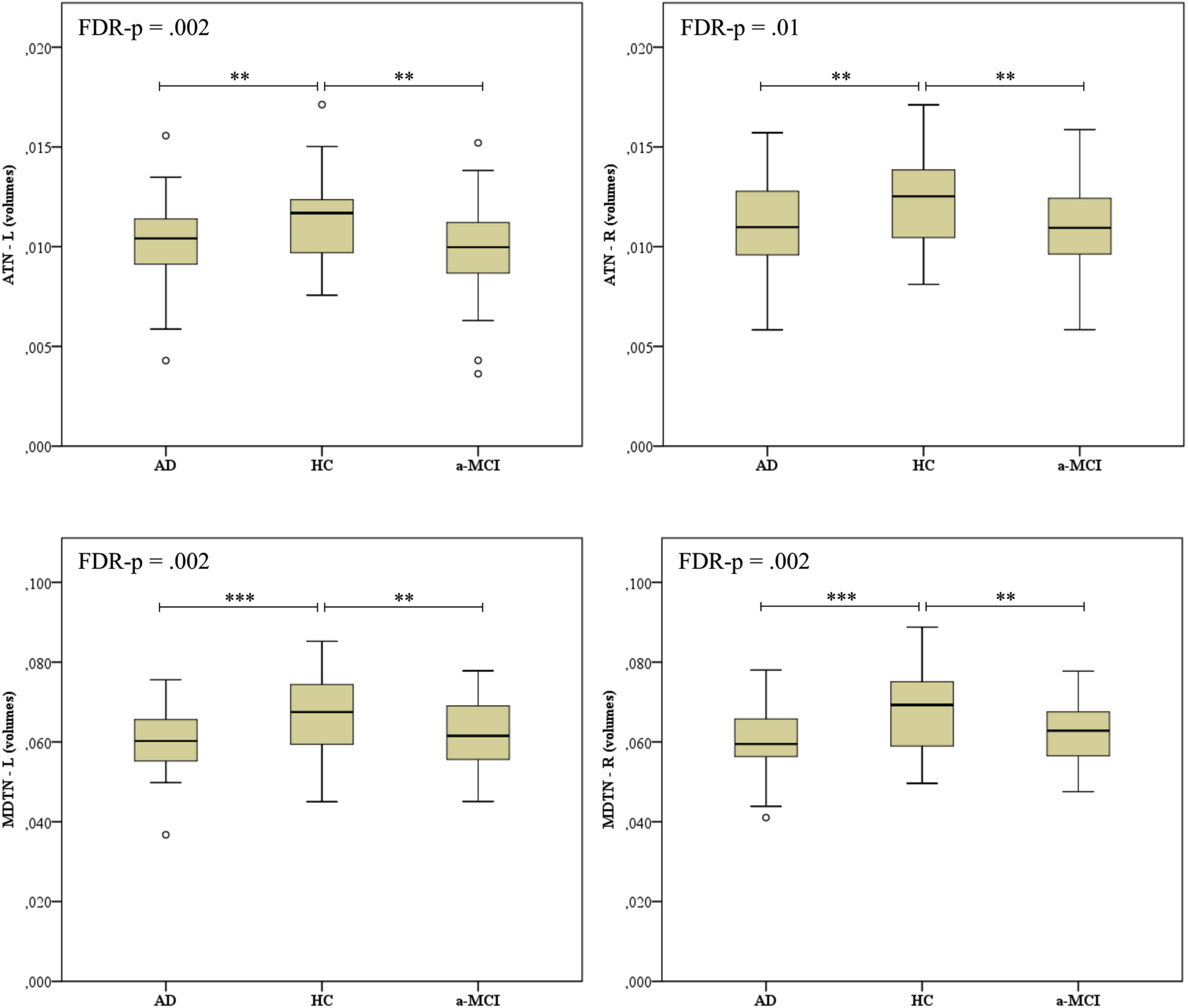
Box plots of ATN and MDTN volumes in the HC, aMCI, and AD groups. Volumes were corrected for individual total intracranial volume (TIV). The bottom, the middle and the top of the box represent the 25th, 50th, and 75th percentile, respectively; the dots outside the upper and lower whiskers represent out of 10th and 90th percentile. ATN, anterior thalamic nuclei; MDTN, medio-dorsal thalamic nuclei; L, left; R, right; AD, Alzheimer’s disease; aMCI, amnestic mild cognitive impairment; HC, healthy controls. The p-value shown is based on the three-sample Kruskal-Wallis test and corrected for FDR. Post-hoc comparisons are based on the Mann-Whitney test: ***p < 0.001, **p < 0.01, *p < 0.05.
Thalamic nuclei diffusivity: Between group differences and correlations with memory scores
Fractional anisotropy (FA)
Quade’s rank ANCOVA applied to FA data showed significant differences among groups in the left ATN (F[2,147] = 11.4, FDR p < 0.001) and right ATN (F[2,147] = 4.51, FDR p = 0.017) (Fig. 3). In both thalamic nuclei, post-hoc analyses indicated that patients with aMCI and AD had lower FA values than HCs, whereas the patient groups did not differ each other. No significant difference emerged in the left MDTN (F[2,147] = 0.722, FDR p = 0.487) or right MDTN (F[2,147] = 0.764, FDR p = 0.487). Regarding correlation analysis, no significant association was found between FA values and memory scores in the aMCI and AD groups as a whole (Table 2).
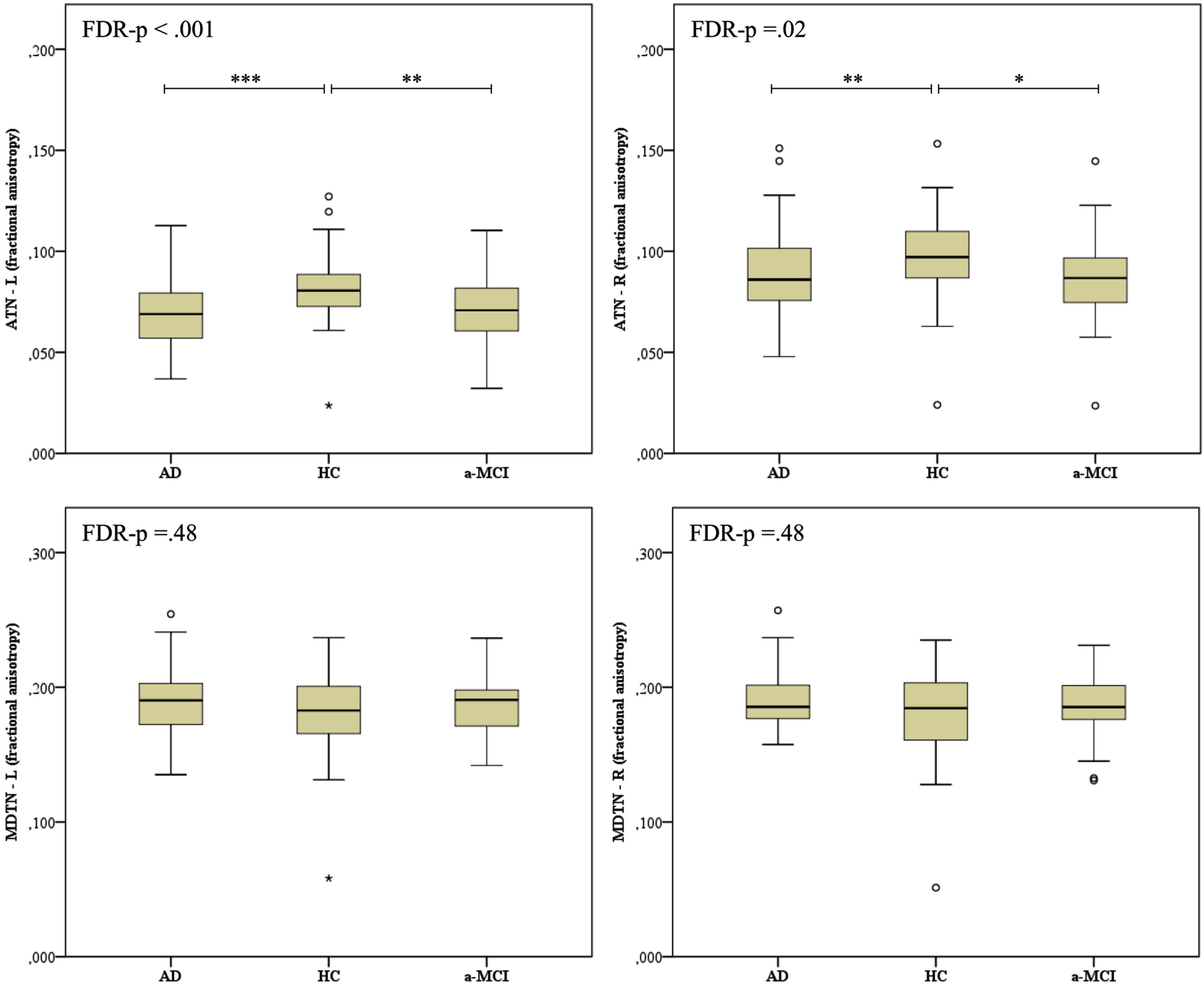
Box plots of FA values of the ATN and MDTN in HC, aMCI, and AD groups. The bottom, the middle and the top of the box represent the 25th, 50th, and 75th percentile, respectively; the dots outside the upper and lower whiskers represent out of 10th and 90th percentile. FA, fractional anisotropy; ATN, anterior thalamic nuclei; MDTN, medio-dorsal thalamic nuclei; L, left; R, right; AD, Alzheimer’s disease; aMCI, amnestic mild cognitive impairment; HC, healthy controls. The p-value shown is based on the three-sample Kruskal-Wallis test and corrected for FDR. Post-hoc comparisons are based on the Mann-Whitney test: ***p < 0.001, **p < 0.01, *p < 0.05.
Mean diffusivity (MD)
Quade’s rank ANCOVA applied to MD data has showed significant differences among groups in the left ATN (F[2,147] = 14.29, FDR p < 0.001) and right ATN (F[2,147] = 16.42, FDR p < 0.001), which were due to higher MD values observed in the aMCI and AD groups when compared to HCs, whereas the patient groups did not differ each other (Fig. 4). A significant difference in MD values was found also in the left MDTN (F[2,147] = 4.78, FDR p = 0.015), but not in the right MDTN (F[2,147] = 2.645, FDR p = 0.089). Regarding correlation analysis, we found that lower scores on the delayed recall of the word list were significantly associated with higher MD values in the left ATN (Table 2 and Fig. 5). This result remained significant after Kendall’s partial correlation controlling for age and education (r = –0.175, p = 0.042). No other significant association between MD values and memory scores was found.
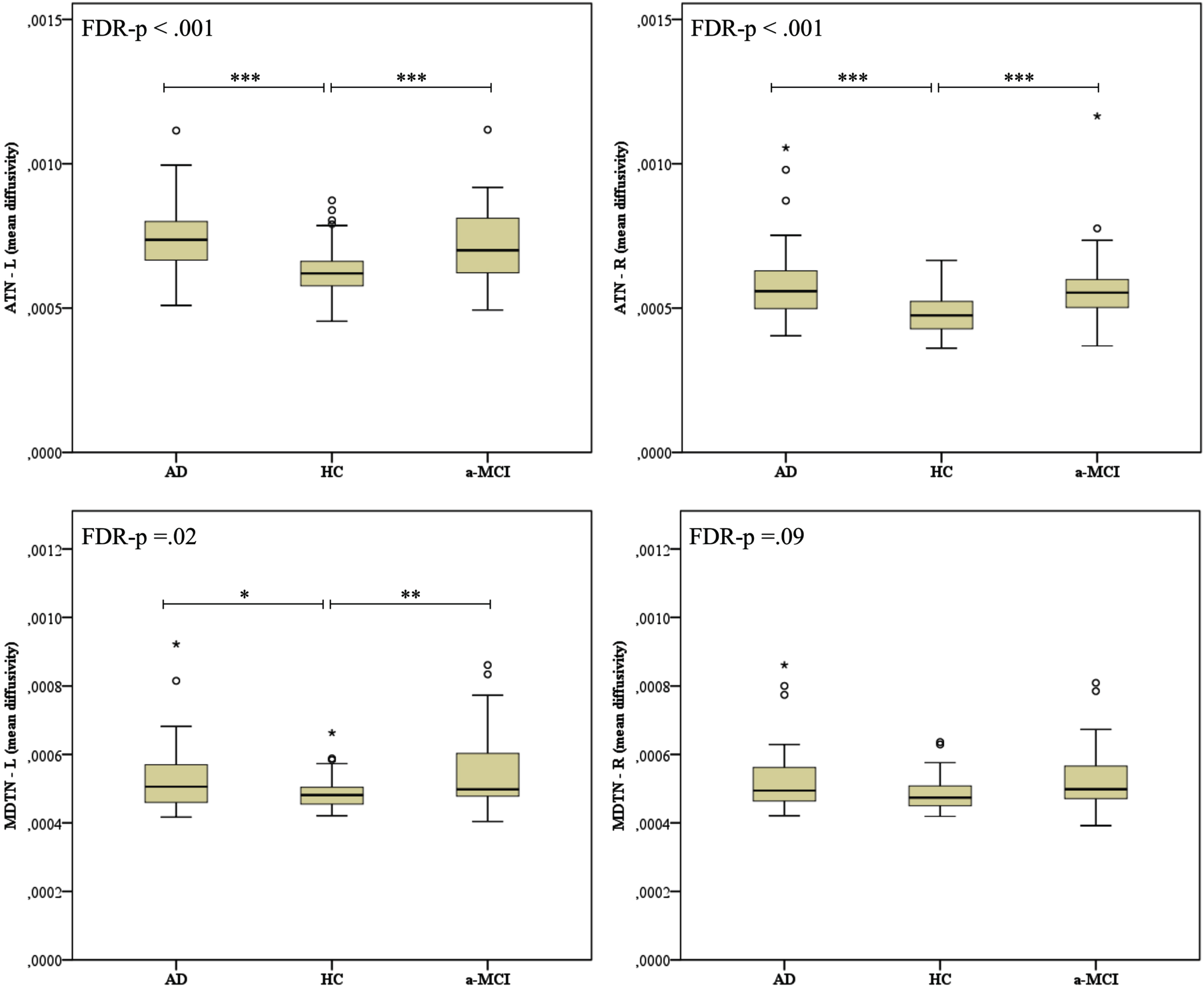
Box plot of MD values of the ATN and MDTN in HC, aMCI, and AD groups. The bottom, the middle and the top of the box represent the 25th, 50th, and 75th percentile, respectively; the dots outside the upper and lower whiskers represent out of 10th and 90th percentile. MD, mean diffusivity; ATN, anterior thalamic nuclei; MDTN, medio-dorsal thalamic nuclei; L, left; R, right; AD, Alzheimer’s disease; aMCI, amnestic mild cognitive impairment; HC, healthy controls. The p-value shown is based on the three-sample Kruskal-Wallis test and corrected for FDR. Post-hoc comparisons are based on the Mann-Whitney test: ***p < 0.001, **p < 0.01, *p < 0.05.
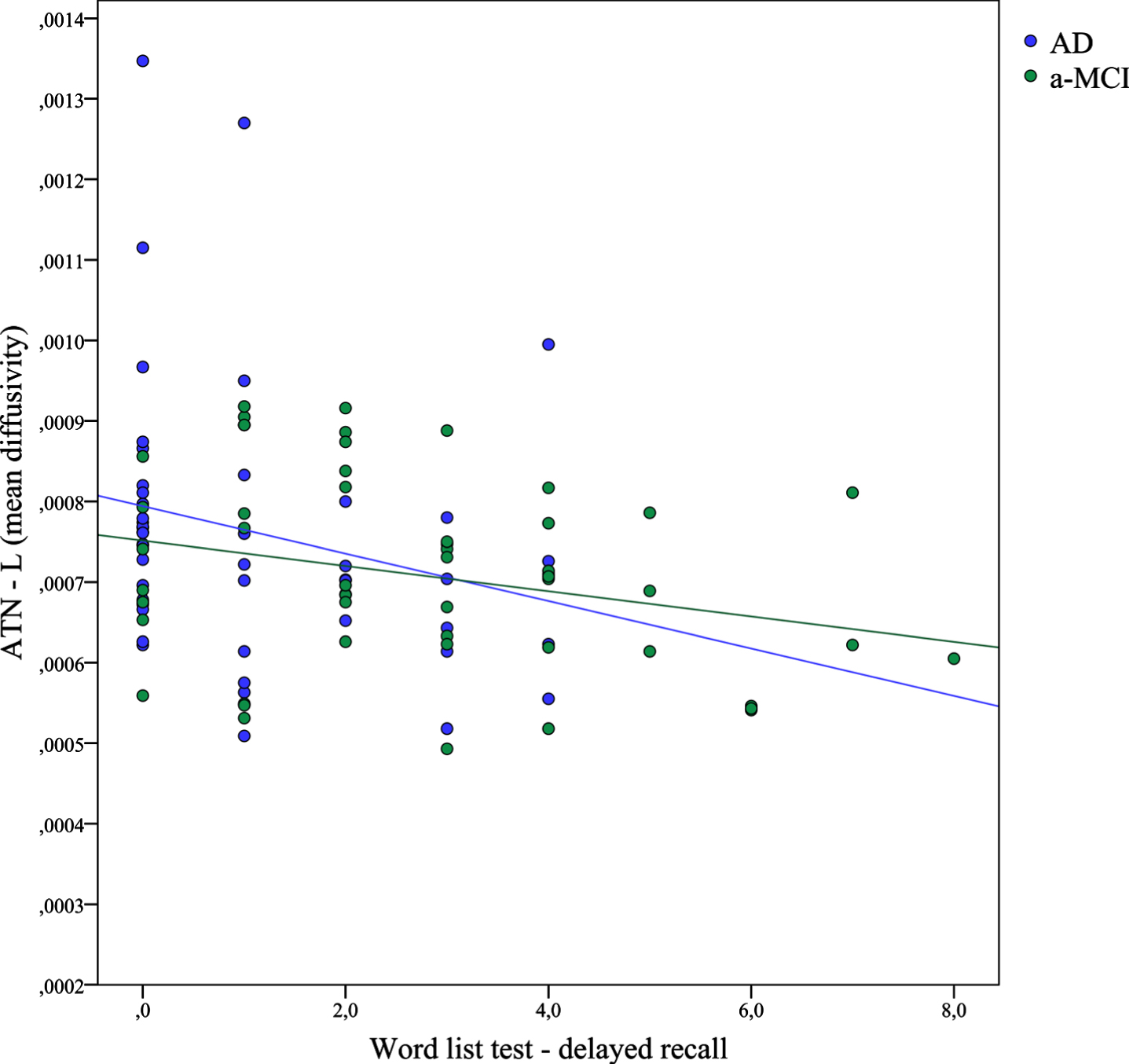
Scatterplot showing the significant linear association between memory scores on the word list delayed recall and MD values of the left ATN in the aMCI and AD group.
DISCUSSION
In this study, we investigated whether grey matter volumetric and white matter diffusivity alterations could be found in specific thalamic nuclei of patients with aMCI and AD in comparison to HC subjects and whether these alterations were associated with episodic memory impairments.
The novel finding of this study is that, in addition to the medial temporal lobe areas, other cerebral structures relevant for episodic memory, such as specific thalamic nuclei, are compromised in AD even in the early phases of the disease and contribute to memory dysfunctions.
In particular, at the macrostructural level our analyses revealed that volumes of the ATN and MDTN were significantly smaller in the AD and aMCI groups with respect to the HC group. This is consistent with the results reported by Bernstein et al. [19], who found smaller volumes in both anteroventral nucleus and mediodorsal nucleus in patients with early and late MCI and AD when compared to HC subjects. However, in contrast with Bernstein et al., we found no significant association between the volumes of ATN and MDTN and memory scores in our patients’ sample. This failure in replicating previous results [19] was probably due to methodological differences between the two studies; in fact, Bernstein assessed possible associations between cognitive scores and thalamic volumes in the participants’ sample as a whole group (early and late-MCI, AD, HC) and in bilateral thalamic nuclei volumes; conversely, we assessed correlations between volumes and memory scores specifically in the patients’ group (AD and aMCI) and in lateralized thalamic nuclei volumes (right and left nuclei, separately).
Interestingly, at the microstructural level, consistent diffusion alterations, in terms of both lower FA and higher MD that significantly differentiated aMCI and AD patients from the group of HCs, were found only in the ATN, but not in the MDTN. Similarly, significant associations between diffusion metrics and memory scores were found only in the ATN, in that increased MD values in left ATN were inversely correlated with delayed recall performance on the word list delayed recall test.
Overall, the results of our study corroborate previous findings reported in the neuroimaging literature concerned with early thalamic changes in AD, which demonstrated thalamic volume loss and shape changes at both a presymptomatic stage in familial AD and in the symptomatic stage of sporadic AD [37, 38], as well as in MCI patients prior to the onset of AD [10]. This is suggested to be the consequence of axonal degeneration and amyloid/tau accumulation and that was prominent in the thalamus due to the large number of white matter tracts the thalamus is connected with [10]. However, the fact that the ATN, but not the MDTN, was the site of both macro- and microstructural alterations, in that the difference between controls and the two groups of patients was observed for both volumes, FA and MD metrics only in the ATN, is in line with neuropathological studies which report that the earliest accumulation of neurofibrillary tangles starts in the ATN [6, 11], thus supporting the precocious vulnerability of these specific thalamic nuclei to AD.
Finally, consistently with the demonstrated central contribution of the ATN to episodic memory functioning [9, 12], we found that microstructural changes at the level of ATN, but not at the MDTN, were significantly associated with poor episodic memory in the overall aMCI and AD groups. This failure to find a direct link between MDTN damage and declarative memory impairment is consistent with previous literature [12, 18], which overall demonstrated that episodic memory deficits are quite consistently observed following mammillo-thalamic tract/ATN damage, whereas it is not still clear if damage restricted to the MDTN is sufficient to impair declarative memory [11]. The finding that only the left ATN correlated with verbal episodic memory impairments in the patients’ group supports the widely-demonstrated left hemispheric lateralization of verbal memory abilities [39] and is consistent with previous studies [40]. Instead, the lack of any significant association between visuo-spatial memory scores and volumetric or diffusion metrics is quite unexpected; it probably reflects a reduced specificity of the Rey-Osterrieth figure test at detecting pure memory deficits, in view of the executive and constructive abilities implicated in reproducing the complex figure [41].
Taken together, the results of our study provide a clear in vivo demonstration of the relevant involvement of thalamic nuclei, and particularly the ATN, in the AD-related neurodegeneration and of the role of these changes in the genesis of the episodic memory impairment. Although the hippocampus has traditionally been regarded as the key structure contributing to episodic memory impairment related to early AD neuropathology, our findings support recent literature suggesting that the Papez circuit could be considered as an extensive cortico-subcortical network, in which critical limbic regions in addition to the medial temporal lobe could be crucial for memory functioning and involved in the pathophysiology of AD and symptoms progression [42, 43]. In this framework, Papez’ original conception of a serially processing circuit has been recently revised in light of recent evidence supporting the idea of a more complex network of parallel and reciprocal connections between critical limbic structures, with the hippocampus, ATN and retrosplenial cortex serving as key hubs [42]. According with this new conceptualization of an integrated mnemonic circuit, recently Forno and colleagues found significant correlation between ATN volumes and both hippocampal atrophy and episodic memory performance in late-onset AD patients [44]. Despite the emerging recognition of the importance of the ATN for memory processing and its vulnerability to AD pathology, it is still unclear whether thalamic abnormalities in AD are a secondary process due to medial temporal lobe dysfunction or occur in the initial stages of the disease [43]. In fact, some authors claim that thalamic and extra-hippocampal alterations could result from a “disconnection process”, according to which hippocampal grey matter atrophy progressively leads to white matter changes that spread to targeting cerebral regions of a same brain network [45, 46]; this view is supported by studies demonstrating that the hippocampal formation is the first region affected in aMCI, followed by late changes in the limbic thalamus in AD [47]. Conversely, other authors claim that brain alterations in related areas may occur in parallel as distinct effects of a common pathological process; this view is supported by studies demonstrating that thalamic abnormalities occur in the early stages of AD [11].
One limitation of this study is the lack of prospective data or biomarkers about our aMCI. It has been widely documented that aMCI is a heterogeneous etiological entity that includes both patients at the prodromal stage of AD as well as patients in the preclinical phase of AD or non-converter subjects [1, 5]. Given the explorative nature of the present study, additional research including aMCI patients in the prodromal stage of AD is definitely needed in order to better explore the contribution of the thalamus, and specifically of ATN, to the genesis of memory disorders. A second possible limitation is related to what discussed above and regard the cross-sectional nature of the study that does not allow to answer the question whether alterations of the thalamus are a primary or secondary phenomenon to hippocampal tissue loss and memory impairment. Further studies are needed to shed light on this issue.
In conclusion, our data show striking volumetric and diffusion alterations affecting specific thalamic nuclei, in particular the ATN, which can be identified early in the course of AD neuropathology. The pathology in this region likely contributes to memory impairments given its importance for mnemonic processes. These findings represent the first in vivo evidence of a relevant involvement of ATN in the AD neurodegeneration and strengthen the importance to look beyond the medial temporal lobe when considering neurological conditions characterized by memory decline.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The study was partially supported by the National Research Council (CNR) “A multifactorial intervention for successful aging” grant, CUP J84I20000250005 and by the Italian Ministry of Health, grant RC 19, 20 and 23.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
