Abstract
Alzheimer’s disease (AD) affects more women than men, with women throughout the menopausal transition potentially being the most under researched and at-risk group. Sleep disruptions, which are an established risk factor for AD, increase in prevalence with normal aging and are exacerbated in women during menopause. Sex differences showing more disrupted sleep patterns and increased AD pathology in women and female animal models have been established in literature, with much emphasis placed on loss of circulating gonadal hormones with age. Interestingly, increases in gonadotropins such as follicle stimulating hormone are emerging to be a major contributor to AD pathogenesis and may also play a role in sleep disruption, perhaps in combination with other lesser studied hormones. Several sleep influencing regions of the brain appear to be affected early in AD progression and some may exhibit sexual dimorphisms that may contribute to increased sleep disruptions in women with age. Additionally, some of the most common sleep disorders, as well as multiple health conditions that impair sleep quality, are more prevalent and more severe in women. These conditions are often comorbid with AD and have bi-directional relationships that contribute synergistically to cognitive decline and neuropathology. The association during aging of increased sleep disruption and sleep disorders, dramatic hormonal changes during and after menopause, and increased AD pathology may be interacting and contributing factors that lead to the increased number of women living with AD.
INTRODUCTION
Alzheimer’s disease (AD) and related dementias are neurodegenerative conditions common to the aging population, affecting an estimated 55 million people worldwide [1]. In the United States (US) alone, AD affects 6.5 million individuals [2], of which 2.5 million are men and 4 million are women. This means that women account for two out of every three cases of AD in the US [3]. When broken down by age, almost every group exhibits more women than men with AD [4]. Compared to men, women are on average: diagnosed later in the disease stage [5]; have increased rates of brain atrophy [6, 7]; experience faster rates of cognitive decline [7, 8]; and have a 70% higher lifetime cost of living with the disease [9]. Moreover, women account for more than 60% of caregivers for those living with AD or related dementias [10]. In recent years, growing evidence has suggested the most at-risk group for developing AD may be the roughly 1.5 million women, aged 45 to 55, undergoing the transition through menopause each year in the US [11]. Universal symptoms experienced during menopause are repeatedly and consistently being linked to poor outcomes in women’s brain health [6, 12–18], and unsurprisingly, women have a one in five lifetime risk of developing AD by age 45 as compared to a one in ten risk for age-matched men [19].
While age is the largest risk factor for developing AD, there are countless others ranging from genetic and unmodifiable (APOE4 genotype, Down syndrome, genetic mutations) to “modifiable” behavioral and environmental (smoking, physical/social/mental activity, diet, education, pollution, sleep, etc.) that have been linked with disease development and progression [2]. While most risk factors are prevalent in both men and women, decades of multidisciplinary research have shown that there are sex and gender differences within each risk category. According to Rocca et al. (2014), there are three categories that should be considered with regards to risk factors and disease: 1) factors that are present at equal frequencies in both sexes but have a greater effect in one sex over the other (e.g., APOE4 genotype); 2) factors that occur more often culturally/socially in one sex, thus having a greater effect size on one sex (e.g., low education); 3) factors that are exclusive to one sex (e.g., menopause) [20]. In addition to risk contribution, differences in presentation and response to treatments and medications in men and women are well known and documented across many diseases, and as such, it is critically important to account for these differences in AD research.
SEX AND GENDER BIAS IN MEDICAL RESEARCH
To date, the majority of medical research in animals and humans has been conducted on males, owing to the alleged difficulty in using female subjects with fluctuating hormones [21]. Indeed, changing hormone patterns across the female lifespan, menstrual cycles, pregnancy, and menopause can, and do, affect clinical outcomes and responses to therapies in regards to safety and efficacy [22]. The exclusion of female subjects in early to recent medical history has left an enormous gap in the healthcare system between men and women and has led to women being underdiagnosed and inappropriately treated in medicine [21]. Although almost 80% of drugs in the US are consumed by women, women are still being prescribed drug dosages derived from the average weight and metabolism of men, leading to increased risk of complications for women [23]. It was not until 1993 that Congress passed into law the National Institute of Health (NIH) Revitalization Act requiring women and minorities be included in clinical research [24], and not until 2014 that the NIH started requiring sex to be considered as a biological variable in preclinical research in humans and vertebrate animals [25].
While slow progress has been made (as of 2019, women account for about half of participants in clinical research funded by the NIH [26]), many areas, including brain research, are still behind. A 2020 study showed that in the three years prior to its publication date, only 52% of articles in leading neuroscience journals included female as well as male test subjects, and only 5% of studies used only females as compared to 26% that used only males [27]. When looking at AD and dementia research specifically, only one-third of participants in clinical trials are women; only 12% of the allocated NIH budget for AD and dementia is allotted to women-centric research; and as of 2019 the NIH Alzheimer’s budget equates to $24 per man and only $3 per woman [28].
Although both sex and gender can, and do, influence health and disease status in humans, the two are not interchangeable. In very simplistic terms, “sex” by definition is a biological trait encoded in DNA that characterizes differences in reproductive organs and hormones, while “gender” accounts for cultural, social, and psychological traits that differ between sexes through social context [29, 30]. Although multiple sex-and gender-related factors could conceivably make a compounding contribution to a person’s risk for developing AD, most research to date has focused on sex-related mechanisms at the hormonal and molecular level, and less on gender-related factors. Preclinical research in non-human animal models is also limited to sex specific exploration as non-human animals do not have gender. Owing to these limitations, this narrative review will focus primarily on sex differences in AD and the numerous impacts that biological sex has on the relationship between sleep disruptions and AD risk. We employed the use of PubMed to search more broadly for the three main topics “Alzheimer’s disease + sleep + sex”. This search was further refined by cross referencing each main topic with other subtopics including: hormones (estrogen, progesterone, follicle stimulating hormone, luteinizing hormone), age, menopause, amyloid, tau, and individual sleep disorders/health conditions (insomnia, sleep apnea, depression, chronic pain, etc.). Citations from manuscripts and other published works were also examined.
INTERRELATIONSHIPS BETWEEN SLEEP, SEX, AND AD
Decades of research have elucidated the complex and bidirectional relationship between sleep disruptions and AD, but how biological sex influences the genetic and molecular processes underlying the sleep-AD connection is largely unknown. There is considerable evidence that sleep disruptions may be one of many AD risk factors that occur at a higher frequency and have an overall greater effect in women as compared to men. The basis for this argument, as with most sex related discussions of health and disease, hinges on hormonal changes throughout the menopausal transition. Most hormone-based research regarding AD risk in women has been gonad-centric, focusing on the loss of circulating estradiol-17-β from the ovaries following natural or surgical (hysterectomy) menopause. Falling estrogen levels from perimenopause, menopause, and hysterectomy have been associated with cognitive decline [12], elevated AD pathology [13], and a rise in sleep disruptions [14] (Fig. 1). While unsurprising, due to the broad expression of estrogen receptors throughout memory and sleep-regulating centers of the brain [31, 32], alterations in circulating levels of gonadal steroids are not the only, or even the most, dramatic hormonal change that accompanies menopause.
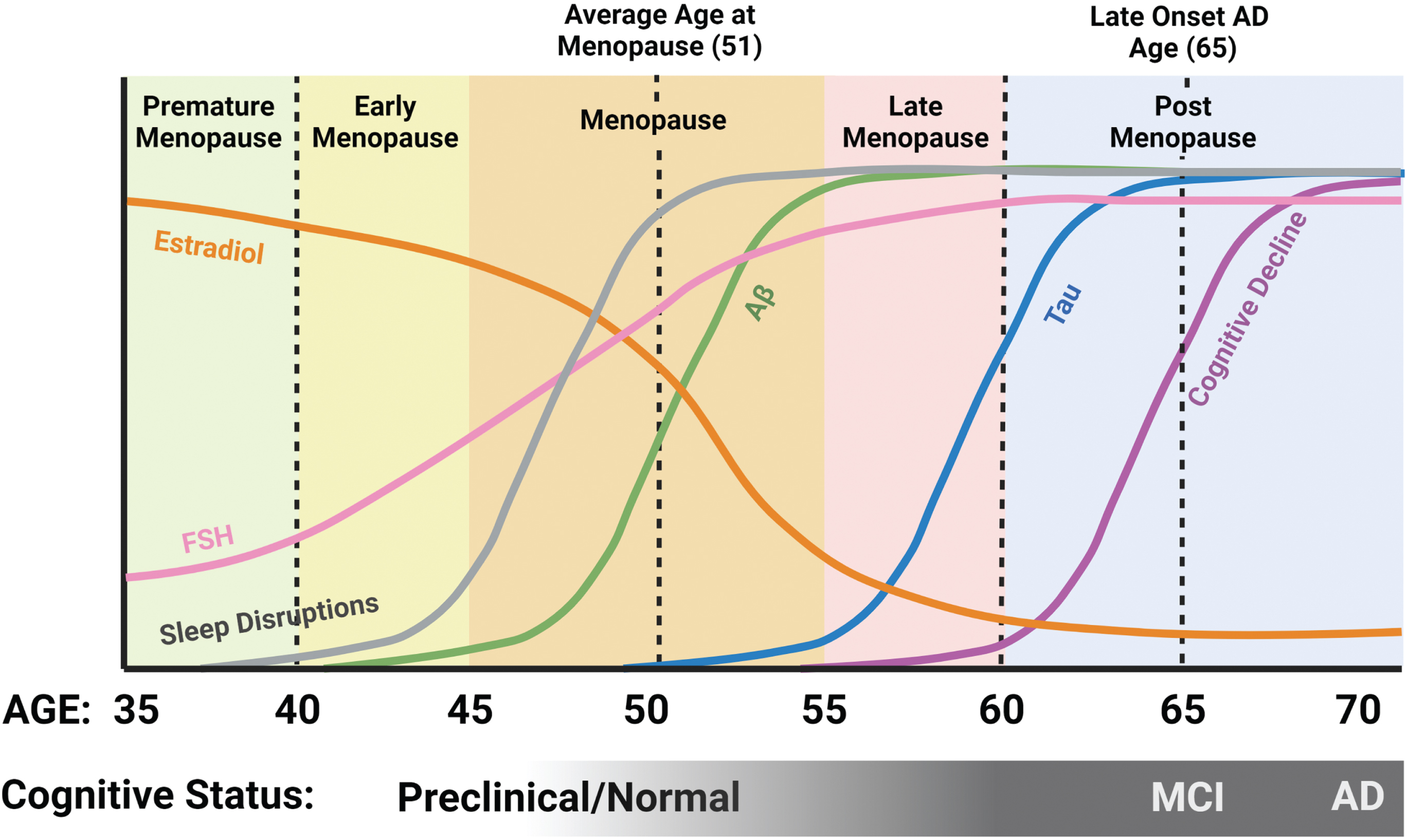
Relationship between sleep, hormones, and AD pathology in women. Estradiol levels begin to decline pre-menopause, while FSH levels begin to rise. Sleep disruptions become prominent during the menopausal transition, about the time when Aβ levels in the brain begin to increase, followed later by an increase in tau. Although some cognitive issues may start in late menopause, the majority of AD-related cognitive decline occurs post-menopausal. Created with BioRender.com.
Profound increases in circulating levels of pituitary gonadotropins, especially follicle stimulating hormone (FSH), are a hallmark of the menopausal transition. An increase in circulating levels of FSH is the earliest detectable hormonal alteration in the transitional period of perimenopause [33]. Throughout the transition from peri- to post-menopause, a sharp and continuous increase in FSH is inevitable, and levels of FSH remain high in the years following. Compelling studies have implicated elevated FSH levels as well as estrogen loss in menopause-related neuropathological changes including cognitive decline and AD pathology as well as sleep disruptions [34]. These neuropathological changes appear to coincide with and be exacerbated by the substantial hormonal fluctuations throughout the menopausal transition (Fig. 1).
Much like hormonal fluctuations, sleep disruptions have been identified in the preclinical stages of AD and precede cognitive impairment and AD pathology by a decade or more [35]. For those who will develop late onset AD, clinical symptoms typically appear around age 65, with pathology developing up to 20 years before clinical symptoms are present [36]. When considering both this preclinical AD window and the average age of menopausal transition between 45 and 55, it becomes evident that the two overlap substantially in middle age (Fig. 1). Accordingly, a drastic increase in sleep disruptions is seen in women as compared to age matched men during this same timeframe [37, 38]. Sleep disruption commonly exemplified in AD patients includes loss of total sleep, loss of rapid eye movement (REM) sleep, loss of non-REM (NREM) sleep, loss of deep (slow wave) sleep, as well as changes in the daily sleep-wake rhythm. Increased fragmentation of the daily sleep-wake rhythm and loss of a consolidated circadian rhythm are strong AD risk factors [39–41] that may worsen as AD progresses and may contribute to AD pathology [35]. Consequently, due to its increasingly disruptive nature for both caretakers and patients, dysregulated sleep is the primary contributing factor in placing older adults (persons aged 65+ [42, 43]) into full-time care facilities [44]. Given that both AD and sleep disturbances are more prevalent in women, it is critical to unravel the complicated and bi-directional interactions between the two. Further efforts to investigate underlying biological mechanisms driving sex differences in sleep and disease will not only serve to identify potential therapeutic targets and strategies, but also to improve the quality of women’s health throughout the lifespan.
SLEEP AND ITS ROLES IN HEALTH AND AGING
To date, every studied animal species partakes in some form of sleep, a behavior which has persisted throughout millions of years of evolution. Despite its hazards and evasive critical function, humans spend about one-third of their lives asleep [45]. Research has demonstrated that sleep is necessary for survival [46] and plays widespread regulatory roles in almost every system in the body. Obtaining an appropriate amount of sleep is a proven necessity for maintaining overall health, with brain health being no exception to this rule. Sleep enhances learning and memory [47, 48]; removes waste buildup from the brain [49–51]; improves mental health and stabilizes emotions [52]; boosts the immune system and prevents infection [53]; balances insulin and glucose levels [54] and regulates appetite [55]; maintains a healthy gut microbiome [56]; facilitates healthy growth and development [57]; aids in cellular repair [58]; lowers blood pressure [59]; and more.
Failing to acquire the necessary amount of sleep can have detrimental effects on overall health and well-being. Alarmingly, lack of sleep can not only shorten life span [60], but can also lead to: disruptions in general cognitive processes [61]; increases in psychiatric conditions including depression, anxiety and suicide [62]; weakened immune function [63, 64]; and increased risk for diseases including certain cancers [65], diabetes [66], obesity [67], hypertension [68], cardiovascular disease [69], stroke [65], congestive heart failure [70], systemic inflammation and pain [53], and cognitive diseases including AD [39, 72]. Currently, the CDC recommends a minimum of 7 hours of sleep per day for the average adult, however one-third of adults report getting less than 7 hours each night [73]. While a multitude of factors contributing to a chronic lack of sleep are modifiable, the general attributes of modern lifestyles (child-care, shift work, sleep disorders, noisy environments, artificial light exposure, stress, etc.) are not conducive to consistently obtaining adequate amounts of sleep. Compounding this issue, both sleep quantity and quality naturally change and decline with advancing age.
More than half of adults by the age of 65 will experience some form of sleep problem [74] in addition to having increased sensitivity to outside factors that can alter the normal sleep-wake cycle such as jet lag, shift work, stress hormones, noise, caffeine, medications, health problems, and psychiatric disorders [75–80]. Even without health issues, sleep quality in healthy individuals naturally declines with age starting as early as age 20 and is typically significant by age 60 [81, 82]. Throughout adulthood, reduced sleep duration and increased awakenings for longer periods of time are common, and often result in increased daytime drowsiness and napping [83].
With advancing age, it is common for changes in sleep patterns to occur [78]. While sleeping each night, humans cycle through two phases of sleep known as NREM and REM which can be distinguished based on differences in recordings of electrical activity of brain and muscle activity. NREM sleep is further subdivided into three stages (N1-N3), with N3 sleep often referred to as deep or slow wave sleep (SWS) characterized by very slow, synchronized brain waves. In REM sleep, the electrical activity recordings are indistiguishable from those of wake, but muscle tone is absent, i.e., the brain appears awake but the body is paralyzed. During a night of sleep, an individual cycles through these stages approximately 4–6 times with occassional brief awakenings between bouts (for more detailed information on sleep stages, see [84]). This pattern of progression of sleep stages throughout a sleep period is called sleep architecture. The most prominent age-related change in sleep architecture typically happens within NREM. Most older adults will experience a considerable decline in deep SWS sleep, along with a rise in the lighter sleep stages—N1 and N2 [82, 85]. Ultimately, time spent in REM decreases as well [86, 87]. All of these age related sleep changes occur in both sexes, with studies showing mixed outcomes on which sex experiences stronger age related decreases in sleep quality when measuring NREM and REM sleep [88–90].
While not a perfect model for sleep or aging, animal models have provided much insight into the basic physiological processes surrounding sleep and age (for an in depth review, see [91]). Briefly, both rats and mice experience age-related changes with sleep similar to humans including an increase in fragmented sleep patterns [92–98] and changes in distribution of sleep throughout the 24-hour day [93, 95–100]. Changes in NREM and REM sleep varied by rodent strain with some showing no change with age and some showing increases or decreases with age [93–97, 100–102]. Unlike humans, most mouse strains have been shown to have increased total sleep with age [98, 103].
SEX DIFFERENCES IN SLEEP
Many sex differences exist in both subjective and objective measurements of sleep. Subjectively, women report having poorer sleep quality compared to men [38, 104–108] and two out of every three adult women do not attain the CDC recommended 7 hours of sleep per night [73]. Chief complaints include increased instances of insomnia, more difficulty falling and staying asleep, increased awakenings at night, increased sleep disturbances due to noise, not feeling rested the following morning, and increased daytime sleepiness [109–116]. Women also may experience a greater sleep debt, or a greater discrepancy in the amount of sleep needed versus the amount of sleep acquired, as compared to men [117]. Overall, women have been shown to seek treatment for sleep problems and to use sleep medications more often than men [118, 119].
Contrary to subjective measures of sleep, objective sleep measures obtained with polysomnography—a multiparameter diagnostic laboratory sleep test that combines recording of sleep stages, eye movements, and muscle activity [120]—have typically shown that women obtain “better” sleep than men in young, middle, and old age [90, 121–123]. Young adult women have been shown to fall asleep faster and stay asleep longer than men of similar ages [38, 124]. Middle-aged women spend more time asleep and exhibit higher levels of REM sleep than middle aged men [122, 124–126]. Women across all ages have be shown to have more SWS sleep [90, 127], and more restful and stable sleep than age-matched men [128, 129]. While objective measures point to women experiencing better sleep overall, the opposing outcomes of subjective sleep studies are indicative of underlying sex-related differences in how sleep is utilized in the body across the lifespan. These discrepancies could also be attributable to experimental bias and design or other sociological factors. For example: sex differences in brain wave measurements may be due to sex differences in skull thickness [123, 131]; men and women may respond differently to study environment (i.e., hospital, laboratory, home) when it comes to polysomnography; and men and women report medical issues and seek medical intervention for sleep issues at different rates [118, 119]. Any additional or combination of these factors have the potential to heavily influence sleep study outcomes and warrant further investigation.
SLEEP AND THE INFLUENCE OF FLUCTUATING HORMONES ACROSS THE FEMALE LIFESPAN
From birth until old age, women experience more hormonal fluctuations and transitional periods than men, and it is throughout these regular hormonal cycles and transitory periods that sex differences in sleep are the most pronounced. There is a substantial amount of research demonstrating the role of gonadal hormones such as estrogen and progesterone on sleep, and, to a lesser extent, pituitary hormones such as FSH and luteinizing hormone (LH) on sleep. During periods of extreme hormonal fluctuations such as puberty, pregnancy, and the menopausal transition, sleep problems are reported at an increased frequency [112, 132–139]. The similar trajectories of changes in circulating hormone levels and changes in sleep architecture and sleep quality throughout the female lifespan suggest that sleep is influenced by reproductive hormones and that this phenomenon may contribute to the vast discrepancies that have been observed between sleep in both sexes. Unsurprisingly these fluctuations in ovarian hormones have yet again pushed women and non-human animals to the margins of sleep and circadian rhythm research. A review by Kuljis et al. (2013) found that less than 20% of rhythm studies included females [140], and a review by Gervais et al. (2017) found that only about 25% of animal studies published in the previous five years included females [91]. As sleep loss over time is a compounding issue and AD develops over a decades long timespan, it is important to understand the extent of sleep disruption throughout the lifespan. It is possible that the increased rates of sleep disruption from adolescence to old age in women contributes to their increased susceptibility to AD.
Reproductive years
During puberty, when gonadal and pituitary hormones rise to high levels inducing sexual maturity, young women are more likely to develop insomnia as compared to age-matched men [137]. After menses begins and throughout reproductive years (ages 15–44 [141]), it is common for women to experience changes in sleep architecture throughout the menstrual cycle (for details on menstrual cycle and hormones, see [142]). Studies have shown that most sleep problems occur during the luteal phase (characterized by decreasing estrogen, progesterone peaks, decreasing FSH and LH (Fig. 2)) and that these sleep problems include lower subjective sleep quality [143–146], longer times to fall asleep (i.e., increased sleep latency), less time asleep compared to the total amount of time spent in bed (i.e., lower sleep efficiency) [143, 147], less REM sleep [134, 139], less SWS [139, 148], and more daytime sleepiness [148, 149]. These sleep problems are accompanied by, and potentially worsened by elevation of core body temperature [150, 151]. Women who experience pain from premenstrual syndrome (PMS) are more likely to experience sleep disruptions including insomnia and daytime sleepiness during the luteal phase [144, 152]. It is also of note that sleep appears to be more affected in women of late reproductive age with more reports of sleep disruption in both the early follicular and luteal phase [153], and more difficulty staying asleep plus less total time asleep during the luteal phase [154].
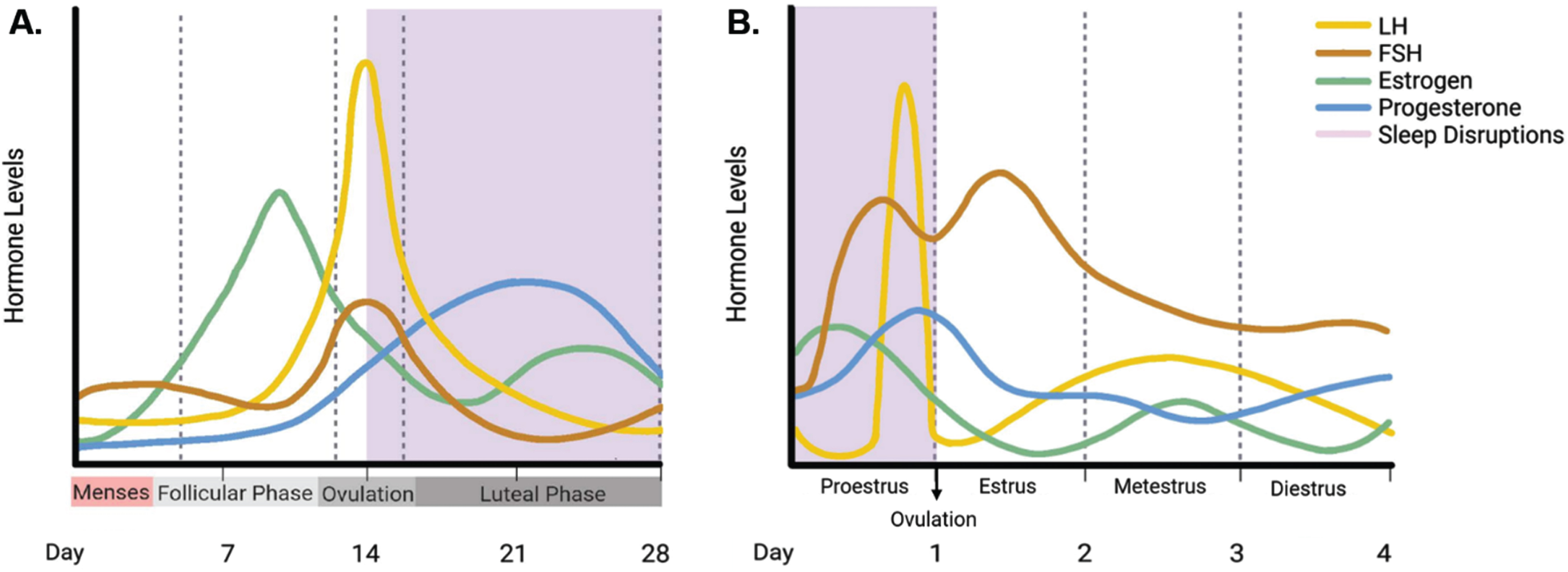
Relationship between circulating hormone levels and sleep disruptions during reproductive years in women and female rodents. In reproductive years of women and female rodents, circulating levels of gonadal hormones (estrogen and progesterone) and gonadotropins (LH and FSH) rise and fall in accordance with the menstrual (A) and estrous (B) cycle. The majority of sleep disruptions (shown in purple shading) occur in the luteal phase for women in the menstrual cycle, and in the proestrus phase of the estrous cycle for rodents. Comparison of the two suggests that high circulating levels of LH and FSH, and rising levels of progesterone may be correlated with periods of increased sleep disruptions. Created with BioRender.com.
Although laboratory animal models such as rodents have different reproductive cycles than humans, they share many characteristics and have observable sex differences in sleep patterns. Like humans, female rodents experience changes in sleep patterns across their reproductive cycles. Unlike humans, rodents have a four to five day “estrous cycle” which consists of four phases—proestrus, estrus, metestrus, and diestrus [155] (for estrous cycle and hormones, see [156] and for a summary, see Fig. 2). During proestrus, when estrogen, LH, and progesterone peak and ovulation occurs, female rodents experience the least amount of sleep, and spend most of their time awake [157–160] (Fig. 2). Both REM and NREM are lower in this phase than in any other phase [159, 161–166]. During the estrus phase when estrogen, LH, and FSH are declining and progesterone is low, mice sleep the most in comparison to any other phase [159].
Pregnancy
Pregnancy is another period of substantial hormonal fluctuations that can cause sleep disruption and negatively impact sleep quality in women. Throughout pregnancy, the concentrations of estrogen and progesterone increase 50 to 60-fold [167], and this is accompanied by a large increase in self-reported sleep disturbances. A clear majority (greater than two-thirds) of pregnant women report some degree of disrupted sleep [167, 168], and causes for disruptions cover a wide range of physiological symptoms including increased urge to urinate, physical discomfort, restless sleep, difficulty falling asleep, difficulty staying asleep, increased daytime sleepiness, sleep apnea, and restless legs syndrome (RLS) [168–173]. Additionally, both REM and NREM [136, 174] sleep decrease throughout pregnancy. In the time immediately following childbirth, hormones typically return to normal levels within 2–5 months [175] and sleep typically returns to normal within 3–5 months [155, 176]. Sleep studies in pregnant rodents are all but non-existent, with one study showing increased total sleep and increased NREM sleep during late pregnancy [177].
Menopause
Menopause is a universal physiological event experienced by aging women who live to midlife and beyond. On average, menopause occurs at age 51 meaning that women will spend about one-third of their lives in a post-menopausal state [178]. Throughout the transition from peri- to post-menopause, menstruation ceases permanently as the ovaries stop producing estrogen and progesterone. Without inhibition from estrogen and progesterone, the anterior pituitary produces higher circulating levels of FSH and LH, which remain high post-menopause [178] (Fig. 1). Sleep disruptions and changes in sleep architecture accompany these dramatic hormonal shifts, such that the percentage of women reporting sleep problems nearly doubles in the post-menopausal state to 61% [15, 179]. In fact, post-menopausal women report the highest rate of insomnia in the US [15]. During menopause the most common sleep complaints include trouble falling and staying asleep, frequent nighttime awakenings, daytime sleepiness, and less satisfaction with sleep quality [133, 180–182].
One of the main drivers of sleep loss throughout menopause is thought to be the loss of estrogen. Decreasing levels of estrogen are associated with increased nighttime awakenings and difficulty falling asleep [181]. Higher FSH levels in association with decreasing estrogen levels have been associated with increased nighttime awakenings in peri- and post-menopausal women [183], and poorer sleep quality and trouble sleeping in peri-menopausal women [181, 184–186] (Fig. 1). Interestingly, women who undergo surgical menopause by full or partial hysterectomy have more subjective sleep complaints than those who experience natural menopause [187]. Menopausal symptoms can last for ten or more years [188], and in view of these findings, it is not surprising that women have a higher risk than men for experiencing age-related changes in sleep [37, 38]. These findings point to an intriguing but understudied topic—the potential role of endocrine changes during menopause in driving sex differences in age-related sleep disruption and AD risk.
Sex differences in sleep and their dependence on gonadal steroids has been demonstrated in laboratory rodents. In general, female rodents sleep less and have reduced NREM and REM sleep during the dark phase, as compared to males [91, 189–194]. Ovariectomy of mice and rats eliminates sex differences in their sleep patterns, making females virtually indistinguishable from males in total and REM sleep amounts. Treatment of ovariectomized females with estrogen to restore their circulating levels to physiological range recreates the expected sex differences in total sleep and REM sleep [162, 195–197]. Furthermore, various approaches to restoring physiologically appropriate hormone levels after ovariectomy, including treatment with estrogen, or a combination of estrogen and progesterone, induced wakefulness and reduced dark phase sleep in females [159, 195], and decreased both REM and NREM sleep in females [161, 199]. Concerning fragmentation versus consolidation of sleep, studies in mice reveal that males generally have more fragmented sleep than females [189, 201], and that gonadectomy removes this sex difference [189]. In rats, the results are more variable and largely dependent on estrous cycle phase with males having more fragmented sleep patterns unless compared to female rats in proestrus [194, 202].
SLEEP-RELATED CHANGES IN AD
It is widely accepted that sleep impacts memory and cognition, and as such it has been postulated that disturbances in sleep are a risk factor for cognitive decline and neurodegenerative diseases such as AD [203–207]. Individuals with AD frequently have disrupted sleep-wake patterns compared to cognitively normal older adults [208–210]. It is estimated that up to half of those individuals with mild cognitive impairment (MCI)—a state between normal aging and early dementia [211]—and two-thirds of those with AD suffer from dysregulated sleep [203–205, 212–214]. Common sleep problems in AD patients range from increased napping, excessive daytime sleepiness, sundowning, insomnia, sleep disordered breathing, less time spent asleep while in bed (decreased sleep efficiency), decreased total sleep, decreased SWS during NREM, and increased latency to REM sleep [215, 216]. Previously, it was thought that sleep disruptions were side effects caused by the AD process; however, mounting evidence in recent years has shown that sleep disruptions occur early in the course of the disease and probably contribute to the progression of pathological changes (Fig. 1). It is likely that a bi-directional relationship exists between the two phenomena [217].
Long before cognitive symptoms are readily noticeable, in a preclinical stage of AD that can last as long as 15–20 years, amyloid-beta (Aβ) begins to build up in the brain [36, 218]. The accumulation of Aβ is believed to be a key driver of the disease (Fig. 3). It is during this time frame that sleep problems begin to manifest and to progress alongside AD pathology [208] (Fig. 1). This hypothesis is supported by several studies. In cognitively normal individuals, a decrease in time spent asleep and an increase in the number of daytime naps has been correlated with amyloid status [219]; shorter fragments of sleep duration and overall poor sleep quality have been associated with increased amyloid pathology in specific brain regions [219–222] (Fig. 3); and individuals with increased sleep fragmentation have been shown to have a 1.5-fold increased risk of developing AD [39, 223]. Both amyloid and tau must be accounted for when considering the relationship between AD pathological changes and sleep disturbances. Tau is a well-known correlate of cognitive decline in AD [224, 225], and has also been noted as the earliest observable AD-related pathology in sleep-wake regulating brain regions [226] (Fig. 3). Changes to sleep architecture have been observed both in cognitively normal older adults and those with MCI. For example, both amyloid and tau deposition are associated with decreased SWS [227–230] and increased CSF levels of Aβ42 are seen in patients with more frequent arousals during SWS [231]. It has been proposed that poor sleep quality in middle-age may be predictive of amyloid and tau burden in the brain, and thus directly tied to AD risk [232].
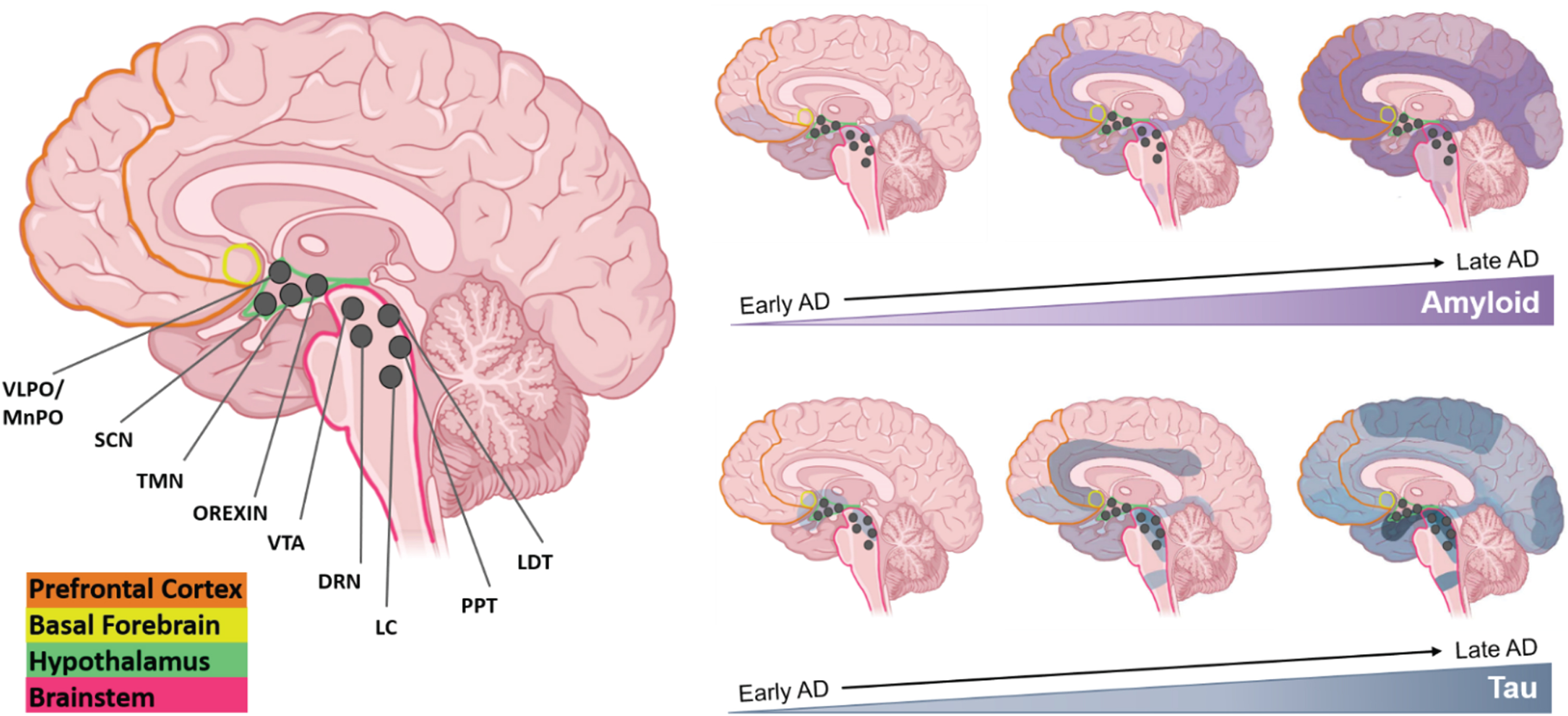
Sleep-wake influencing brain regions and AD pathology progression. Several major brain regions are involved in maintaining states of sleep and wake—PFC (orange), BF (yellow), hypothalamus (green), and the BS (pink). Within the hypothalamus and BS, many sleep-wake influencing nuclei have been identified and are represented as dark grey circles—VLPO/MnPO, SCN, TMN, Orexin neurons, VTA, DRN, LC, PPT, and LDT. Visualizing the progression of both Aβ and tau pathology may be useful in understanding how sleep is affected throughout the course of AD. Many of the areas/nuclei involved in maintaining states of sleep and wake are affected by both Aβ and tau pathology early on in the disease, and worsening pathology with time is often correlated with more dysregulated sleep patterns. Created with Biorender.com.
The specific biological mechanisms that cause changes in sleep architecture with AD are unknown, and much research has explored the relationship between amyloid and tau burden and sleep. Sleep regulates diurnal fluctuations in Aβ levels in the hippocampal interstitial fluid (ISF) of mice and the cerebrospinal fluid (CSF) of humans—soluble amyloid is high during wakefulness, and much lower during sleep [233, 234]. When healthy young adults are deprived of sleep, even for just one night, levels of soluble Aβ38, Aβ40, and Aβ42 are significantly increased by 25–30% in the CSF, potentially due to increased overnight amyloid production when compared to control subjects [235, 236]. In contrast to the body’s lymphatic system which aids in removal of metabolic waste products, the brain has its own unique system dubbed the glymphatic system that is especially active during sleep [49–51]. During wakefulness, waste products like the AD-related proteins, amyloid and tau, accumulate in the interstitial space around brain cells [237, 238]. During sleep, pulses of CSF continuously enter the interstitial space and interchange with ISF [239]. This constant flux of CSF drives waste build-up out of the interstitial space and towards the lymphatic system to be removed from the brain and body [240, 241]. Rodent studies have shown that the interstitial space increases by more than 60% during sleep, and during this time both amyloid and tau are cleared more efficiently than during wake [49, 242].
Sleep studies in AD mouse models, despite their limitations, have proven informative in elucidating how sleep dysregulation relates to AD. For example, many mouse lines have shown that increasing AD pathology is associated with increased sleep disruption. Mouse lines with increased amyloid deposition (APP/PS1, 5xFAD, PDAPP TgCRND8, and Tg2576) have more fragmented sleep wake cycles, increased wake time, and decreased levels of both NREM sleep and REM sleep [243–248]. Conversely, APP/PS1 mice that were actively immunized against amyloid regained a normal sleep-wake cycle [243]. Mouse lines with increased tau levels have also shown disrupted sleep patterns including increased wake bout duration and decreases in both NREM and REM sleep [96, 250]. Alternatively, many studies have attempted to demonstrate the opposite by measuring the effect of sleep disruption on AD pathology. Common models of sleep disruption or deprivation in mice include: 1) the “flowerpot technique” (a method in which mice are placed on a small platform over water such that loss in muscle tone during REM sleep causes the mouse to fall into the water and wake up) [234, 251], 2) intermittent tactile stimulation [234], or 3) light-dark cycle changes that disrupt circadian rhythm [252]. These techniques are not reflective of the sleep fragmentation into shorter bouts that are seen in AD patients, and only a few studies to date have attempted to model more intermittent sleep fragmentation with either presentation of wheel running or toys and gentle tactile stimulation [253, 254]. In AD mouse models, acute sleep deprivation increases Aβ levels in the hippocampal ISF, while chronic sleep restriction accelerates amyloid plaque deposition in many brain regions [234, 255]. Studies comparing sleep fragmentation versus sleep deprivation have found enhanced amyloid deposition with the sleep fragmentation approach [253, 254]. Sleep deprivation in mice has also been found to increase tau in ISF and to accelerate tau spreading in specific brain regions [256].
AGE-RELATED SEX DIFFERENCES IN AMYLOID AND TAU
Many studies have investigated sex and gender differences in amyloid and tau pathology in individuals who are cognitively normal, and individuals with MCI or AD. Overall, women have been shown to have more global AD pathology than men [257, 258]. When looking at cognitively normal individuals ranging from middle-age to advanced age, outcomes have been mixed with some showing no difference between men and women [259–263], and others showing a greater amyloid burden in women [257, 264]. Tau pathology, including neurofibrillary tangles, as well as total and phosphorylated tau in CSF, has consistently been shown to be higher in women than in men [257, 264–269], especially in the rostral middle frontal, inferior parietal, temporal, and lateral occipital lobes [270–272]. Additionally, among cognitively normal women, tau pathology has been shown to be higher in those with greater amyloid pathology [259, 274]. In those with MCI and AD, several studies have shown higher tau in women as compared to men [258, 276]. Sex also influences the effect of APOE4 on AD pathology. Among cognitively normal older adults as well as older adults with MCI, women who carry the APOE4 allele have higher levels of phosphorylated and total tau in CSF than men, and one study found increased phosphorylated tau in women who carry the APOE4 allele in addition to higher levels of amyloid [268, 277].
Despite the mixed outcomes of measuring amyloid pathology over a wide age range or in very old adults postmortem, earlier and more dramatic changes in amyloid have been exemplified in multiple studies involving women undergoing the menopausal transition [6, 16–18]. The same trends are seen with tau. Menopause has long been hypothesized to increase the risk of AD in women [34, 279]. Symptoms during menopause are largely neurological and include disruptions in many processes regulated by estrogen, including thermoregulation, sleep and circadian rhythms, mood disorders, and cognitive decline [278]. Throughout menopause, women have increased levels of amyloid and tau, show a loss of white matter integrity and reduced grey matter volume, and exhibit decreased glucose metabolism [6, 16–18]. In all of these studies, comparisons of pre-, peri-, and post-menopausal women to age-matched men found that these pathological changes were significantly higher in post-menopausal women than in men or pre-menopausal women, while the latter two groups were not significantly different [6, 16–18]. Women who experienced menopause before age 50 had increased levels of tau [280] compared to those who underwent menopause later. Additionally, women who experienced hysterectomy before 50 years of age experienced an increased rate of cognitive decline with advancing age and exhibited greater amyloid pathology postmortem [281, 282]. Overall, the changes in these neuropathological features in menopausal women precede any clinical AD symptoms and are independent of other risk factors such as age, APOE4 status, and other comorbidities [283].
In animal models of AD, loss of circulating estradiol after ovariectomy is associated with increased levels of amyloid and decreased glucose metabolism in the brain [284, 285]. Furthermore, the concomitant increase of FSH accelerates amyloid and tau pathology and impairs cognition, while blocking FSH receptors ameliorates these disease phenotypes [34]. These findings suggest that the AD process begins earlier in women than in men, and that the menopausal transition is a critical time window when sex differences in AD may emerge, potentially putting women at an elevated risk for progression to AD [261, 283].
AD NEUROPATHOLOGY IN SLEEP/WAKE REGULATING AREAS OF THE BRAIN
Many studies have looked to pathological damage by Aβ or tau in centers of the brain that regulate the sleep-wake cycle to explain the connection between AD pathology and sleep disruption and most, if not all, of these regions show some sort of AD pathology or neuronal loss [226, 286]. Multiple regions and nuclei in the brain are responsible for maintaining states of sleep or wake including: the hypothalamus containing the suprachiasmatic nucleus (SCN), tuberomammillary nucleus (TMN), ventrolateral preoptic area (VLPO)/median preoptic area (MNPO), and orexin/hypocretin neurons; the brain stem containing the dorsal raphe nuclei (DRN), locus coeruleus (LC), pedunculopontine nucleus (PPT), and laterodorsal tegmental nucleus (LDT); the midbrain which contains the ventral tegmental area (VTA); and the basal forebrain (BF) [287] (Fig. 3). The interrelationship between these areas is complex and while each region may be involved in more than one process, the LDT, PPT, TMN, DRN, and LC are generally classified as the major “wake promoting” regions [288, 289]; and the VLPO is classified as the major “sleep promoting” region. While many other regions are involved in both sleep and wake promotion, the following section will focus on sexually dimorphic areas involved in sleep promotion (for a review on all sleep/wake promoting areas, see [287]).
The VLPO in rodents and its human homolog, the intermediate nucleus, is a major sleep promoting neural substrate that stimulates sleep and inhibits wake by releasing neurotransmitters γ-aminobutyric acid (GABA) and galanin [290]. GABA and galanin bind to receptors in the hypothalamus and brainstem and serve to dampen or inhibit the wake promoting neural circuits of the brain while promoting sleep [291]. Amyloid pathology has been found in this region of AD patients, while tau pathology has been found infrequently [292]. In older adult humans, AD was associated with decreased expression of galanin-immunoreactive neurons in the intermediate nucleus [293]. Furthermore, sleep fragmentation in subjects with or without AD was inversely associated with the number of galanin-immunoreactive neurons in the area [293]. It is interesting to note that the VLPO has been proposed to be sexually dimorphic [294]. In addition to being influenced by estrogen [158, 295] the VLPO in females also has a smaller neuronal volume, and more dramatic cell loss throughout the lifespan as compared to males [294, 296].
The hypothalamic SCN, dubbed the “master clock” of the body, synchronizes circadian rhythms with the external light-dark cycle and influences the propensity to fall asleep [297]. With normal aging, circadian rhythms are altered, often showing a reduction in amplitude, meaning the difference between the peak and trough of the 24-hour rhythm is reduced. In AD, these changes are exacerbated and sleep may become fragmented; with some patients suffering a complete loss of sleep-wake rhythms [298–300]. The SCN undergoes functional and morphological changes with age, and these changes are exacerbated in AD, where degeneration of SCN neurons may also occur [301–305]. Additionally, the SCN has been shown to be sexually dimorphic in shape (longer and thinner) in women as compared to men [304, 307]; and women with AD have been shown to have a greater loss of vasoactive intestinal peptide expressing neurons which make up the core of the SCN [308].
In addition to circadian regulation, propensity to sleep is strongly regulated by the amount of time spent awake, often referred to as “sleep pressure” or the homeostatic sleep drive [309]. Sleep pressure is mediated by the neuromodulator adenosine, a product of adenosine triphosphate degradation, which accumulates throughout the day. The more adenosine is accumulated, the greater sleep pressure will be. Throughout wake, adenosine is hypothesized to be produced and accumulate at higher levels in specific brain regions, one of which is the BF [310, 311]. In AD, the BF exhibits degeneration of cholinergic neurons, which appears to be one of the earliest pathological events of the disease [312, 313] (Fig. 3). In studies of cognitively normal, MCI and early AD patients, increases in tau pathology in the BF correlate with degree of dementia [314–316]. Additionally, loss of cholinergic neurons in this region has been associated with loss of sleep-wake regulation in AD, specifically loss of REM sleep [231, 317]. The BF is also sexually dimorphic, at least in rats, as a larger neuronal volume is found in this area in male rats compared to females, and it is influenced by estradiol [318–320].
The prefrontal cortex (PFC) plays an important role in sleep as it is one of the most common sites of origin of slow-wave activity that predominates in the deepest stage of NREM sleep [321]. With normal aging, the density of slow waves declines predominantly in frontal brain regions [322–324] and, interestingly, loss of slow wave sleep occurs early in the course of AD and contributes to its neuropathological progression [325, 326]. Amyloid aggregates in cortical regions in both cognitively normal adults and those with AD [322, 327] and its increase correlates not only with memory impairment, but with decreased slow wave activity in cognitively normal older adults [227] (Fig. 3). As the glymphatic system functions predominately during sleep, and Aβ42 levels have been shown to increase with decreasing SWS, it is probable that increasing Aβ and decreasing SWS exacerbate one another in a cyclic fashion, increasing neuronal degradation in AD [328]. It is of interest to note that the PFC is sexually dimorphic during early development with larger volumes present in females than in males [329, 330].
While sexual dimorphism exists in many sleep-wake regulating areas of the brain, it is possible that sex differences in the major sleep regulating region (VLPO) in combination with sex differences in multiple sleep influencing areas (SCN, BF, and PFC) have an additive effect on sleep changes in women with age. Smaller neuronal volume, more dramatic cell loss with age and AD, influence of circulating estrogen, and variance with development all have the potential to alter biological processes including sleep as well as AD progression in women.
SEX DIFFERENCES IN APOE4 AND SLEEP
One area that is still under investigation is the relationship of apolipoprotein E (APOE) and sleep. Sex differences have been reported regarding APOE4, demonstrating that females carrying this allele are at greater risk of developing AD earlier in life, have more rapid disease progression, and have more severe cognitive decline compared to males [261, 331]. Sleep may also be influenced by APOE4 genotype, with some studies reporting a relationship between APOE4 and sleep apnea [332]. For example, studies have shown that individuals with APOE4 alleles can be at increased risk for sleep apnea [333]; individuals with both APOE4 alleles and sleep apnea may have higher rates of impaired spatial working memory [334]; and individuals with APOE4 alleles may have significantly worse sleep quality than those without [335]. These studies suggest that women may be uniquely vulnerable to the combination of sleep disorders and APOE4 genotype as a risk factor for AD.
HORMONES AND THEIR EFFECTS ON SLEEP AND AD IN WOMEN
Gonadal steroids
The scientific literature regarding sex differences in disease and aging commonly discuss sex steroid hormones—estrogen and progesterone—with most research focusing on 17β-estradiol, the predominant form of estrogen. Most studies demonstrate an increased risk of AD and related dementia with loss of circulating 17β-estradiol, citing menopause or the surgical removal of ovaries prior to natural menopause as the main driver [281, 337]. Interventions and treatment strategies have logically followed a hormone replacement therapy (HRT) approach to mitigate symptoms from hormone loss as well as risk of certain diseases (it is important to note that while hormonal contraceptives have had both positive and negative associations with many diseases and health conditions throughout the female lifespan it is incredibly complex and beyond the scope of this review. See [338–340] for an overview of HRT and perimenopause). These therapies have employed either estrogen alone or a combinatorial approach using estrogen and progesterone. Results for these studies have been mixed depending on a multitude of factors such as age at administration, menopausal status, dosing strength, duration of administration, hormone formulation, and more [341]. Most studies have shown that AD risk is lower in those who used some form of HRT, with decreasing risk associated with increased dose and duration [13, 341–346]. Overall, these findings suggest that there may be a critical window, route of administration, and treatment plan in which HRT should be administered to be maximally effective in protecting the brain during aging from negative effects of reproductive hormone loss, including sleep disruption and neurodegeneration [347].
Estrogen
While estrogen is most commonly associated with female reproduction, it is also the most studied hormone in regards to AD. Women produce three different forms of estrogen: estradiol, estrone, and estriol [348]. Estradiol is the primary form of estrogen in circulation throughout the reproductive years; however, it has many functions extending beyond reproduction, including roles in sleep. There are two subtypes of estrogen receptors, ERα and ERβ, and they are distributed widely throughout the brain in areas associated with sleep and/or AD, including but not limited to, the hippocampus, cortex, amygdala, SCN, VTA, LC, VLPO, and pineal gland [349–351]. Estrogen replacement therapy is often utilized throughout menopause to combat unwanted symptoms.
The effects of estrogen on sleep are complex because it regulates many systems in the body. In a one-month study in women of reproductive age, those with low variation in sleep duration (i.e., routinely obtaining 7-8 hours of sleep per day) were shown to have 60% lower levels of estradiol as compared to those with higher variations in sleep durations [352]. Another study showed a correlation between amount of sleep and estrogen levels with estradiol increasing by almost 4% with every one additional hour of sleep obtained by participants [353]. Throughout the menopausal transition, women using HRT report fewer sleep complaints including: fewer hot flash and movement-related arousals [354]; reduced insomnia symptoms and improved sleep quality [355]; increased REM sleep [356], reduced time to fall asleep, reduced nighttime awakenings, increased total sleep time, and increased SWS [357].
In women with AD, plasma levels of estrogen are lower when compared to age-matched controls [358]. Animal models of AD have shown that loss of circulating 17β-estradiol after ovariectomy is associated with increased AD-like neuropathological changes including amyloid [359, 360] as well as decreased cognitive function [361, 362]. Treatment with 17β-estradiol after ovariectomy mitigated these effects to varying degrees [359–361]. Changes in the expression of estrogen receptors in the brain has also been implicated in AD. Declines in ERβ in female rats have been associated with a decline in cognitive function [363]. Cell and animal models have been beneficial in demonstrating the neuroprotective effects of estrogen as it has been shown in these models to: protect against neuronal cell loss from amyloid toxicity [364, 365]; prevent apoptosis [366]; reduce excitotoxic cell death [367, 368]; prevent oxidative damage [369]; suppress inflammation [370]; promote synaptogenesis [371]; reduce tau hyperphosphorylation [372, 373]; reduce amyloid production [362, 375]; induce AβPP processing via the non-amyloidogenic pathway [376]; inhibit AβPP processing via the amyloidogenic pathway [377, 378]; promote amyloid clearance [379]; increase neurogenesis [380]; and enhance learning and memory by increasing dendritic spine density [381]. While estrogen has been widely studied and shown to have protective effects on the brain, many gaps in research still remain, especially pertaining to estrogen in both men and women living with AD in general, and more specifically how it pertains to sleep and AD.
Progesterone
Progesterone, the ovarian steroid that often works in conjunction with estrogen and supports pregnancy, has been much less studied than its counterpart. Past studies have yielded mixed results, likely due to the complexities of several interacting factors. First, progesterone often works in combination with estrogen, and the combined effects are often difficult to tease apart. Second, there are multiple forms of synthetic progesterone, or progestin, rather than one standard progestin used across all scientific studies. Currently, there are three different groups of synthetic progestins divided by their structural properties, and within each of these groups there are 2–4 subtypes [382, 383]. Clinical availability of progestin as a therapeutic is dependent on country, thus making the form used or prescribed highly variable, which can affect preclinical and clinical outcomes. Progesterone and its metabolites are thought to regulate sleep and produce changes in sleep architecture through agonistic modulation of GABA receptors, much like benzodiazepines [384–386]. Additionally, progesterone is a primary regulator of LH pulse frequency in women. As LH has been shown to affect sleep patterns, changes in progesterone levels with age may have cascading effects [387].
The majority of studies involving progesterone and sleep have shown that progesterone has sedative and sleep promoting qualities similar to benzodiazepines [388]. Treatment with progesterone increases NREM and REM sleep, decreases the amount of time to fall asleep, decreases awakenings during sleep, and increases total sleep time [386, 388–391]. In one study of women, increasing levels of progesterone during the luteal phase were associated with increased levels of sleep, with each hour increase in sleep corresponding with increased levels of progesterone [353]. It has been hypothesized that increased levels of progesterone at the beginning of pregnancy, which are much higher than normal physiological range, may be the culprit behind the increased levels of tiredness, decreased concentration, poor coordination, and memory complaints that are often experienced [386, 392]. Alternatively, decreased levels of progesterone throughout menopause, which are much lower than normal physiological range, are associated with sleep disruption, while administration of intranasal progesterone during the same time period has been shown to induce sleep [393, 394].
Interestingly, there is little to no research on the sole effects or levels of progesterone in the brain of AD patients. Despite this, progesterone has been shown to have positive effects on cognition. In post-menopausal women, progesterone administration has been associated with better concentration, verbal memory, and executive function [395, 396]. All forms of progesterone are often prescribed for HRT during menopause in conjunction with estrogen. Combination HRT has shown some negative effects on short and long-term verbal memory [397], but have had positive effects on sleep [398]. Multiple studies show that progesterone is an important neuromodulator in the central nervous system and has a neuroprotective effect. In vitro and in vivo, progesterone has been shown to exert multiple neuroprotective effects in several animal models of brain injury [399, 400], improve learning and memory [401], increase neuronal survival rate [402, 403], decrease neuronal apoptosis [404, 405], improve glucose metabolism of neurons [406], decrease oxidative stress [407, 408], increase synaptogenesis [409], protect against amyloid-induced toxicity [364], and reduce tau hyperphosphorylation [361]. An extensive amount of research is still needed on progesterone in regards to men and women with AD, as well as progesterone and its involvement in sleep for both men and women throughout the lifespan and in those with AD.
Gonadotropins
Until recently, the impact of increasing levels of gonadotropins during reproductive aging on age-related changes in the brain was virtually ignored. However, mounting studies in cells, rodents, and humans demonstrate that elevated levels of circulating gonadotropins throughout normal aging may have important roles in cognition and neuronal disease. Gonadotropin levels are regulated through the hypothalamic-pituitary-gonadal (HPG) axis through a feedback loop. The hypothalamus releases gonadotropin-releasing hormone (GnRH) which stimulates secretion of the gonadotropins LH and FSH, which act sequentially to stimulate follicular growth and trigger ovulation, respectively, during the menstrual cycle. Release of 17β-estradiol from the growing ovarian follicles at first inhibits LH and FSH, but very high levels then trigger a surge release of LH, which induces ovulation and the formation of the corpus luteum that then secretes progesterone [410]. During menopause, ovarian follicles are depleted, leading to the cessation of menstrual cycles and the loss of circulating levels of estrogen and progesterone. This causes disinhibition of LH and FSH secretion such that circulating gonadotropin levels increase greatly [411, 412]. The menopausal loss of ovarian steroids causes a 3-4 fold increase in LH and a 4–18 fold increase in FSH levels [413] (Fig. 1). In contrast to women, age-related changes in reproductive hormones in men are more gradual and moderate, as decreases in androgens with age are associated with a 2-3 fold increase in LH and FSH [414]. Several studies have shown that AD patients have higher levels of FSH and LH compared to cognitively normal age matched controls [415–417]. Interestingly, as both LH and FSH receptors have been localized in the brain, age-related increases in circulating gonadotropin levels may have roles in neurological symptoms connecting menopause, sleep, and AD [34, 419].
Luteinizing hormone
The effects of sleep on LH have been somewhat characterized, but the converse, the effects of LH on sleep, have been understudied. Receptors for LH have been found in the brain in areas involved in memory and sleep such as the hippocampus, hypothalamus, cortex, and brainstem [420–425]. Throughout puberty, in both males and females, pulsatile LH secretion is increased dramatically only during sleep [426–428] with studies showing that the increase occurs during SWS and is accompanied by brief episodes of wakefulness [429]. During the early follicular phase of the menstrual cycle, LH pulses decrease in frequency but increase in amplitude which has been linked to nighttime awakenings [430]. Additionally, menopausal hot flashes which disrupt sleep are associated with increasing levels of LH [431, 432].
There is some evidence that LH may influence neurodegenerative disease processes. Several studies associate increasing levels of LH with a decline in cognition and poor memory recall in cognitively normal individuals [433–435]. A small study of aged men showed that increasing plasma LH levels were correlated with increasing amyloid levels [435, 436]. When looking at AD brains compared to control brains, LH has been found to accumulate intracellularly in pyramidal neurons in those with AD [437]. When looking at individuals with Down syndrome, who are already at a higher risk for developing AD, levels of LH were shown to be elevated [438]. More extensive research is needed to determine whether the correlations observed in these studies are indicative of causation.
In rats, studies have shown that LH can cross the blood brain barrier [439], and that LH receptors are found at high levels in the hippocampus [425]. Neuronal cells treated with LH were shown to increase secretion of Aβ; similarly, animal models have shown that LH increases levels of amyloid in the brain, increases AβPP expression, and impairs hippocampal associated behavior [440–443]. Additionally, when rats are chronically treated with leuprolide acetate, a gonadotropin-releasing hormone agonist that lowers LH levels, Aβ in the frontal cortex and hippocampus decreases, and cognitive performance increases [440, 444]. In view of these findings, gonadotropins have been proposed as a potential target for treating AD [445]. Overall, clinical research is extremely sparse in regards to LH in AD in both men and women, as well as in LH and its involvement in sleep throughout the lifespan of men and women.
Follicle stimulating hormone
Similarly to LH, studies on FSH are limited and inconsistent when it comes to the effects of FSH on sleep. Multiple studies show no influences of increased circulating FSH levels on sleep architecture in menopausal women [153, 447] or positive influences of FSH on sleep duration and SWS [186, 448]. One study has shown that women who sleep for shorter periods of time have 20% less urinary FSH than those who are long sleepers [449]. Alternatively, other studies have shown that rising circulating FSH levels during the menopausal transition are associated with increased wakefulness after initially falling asleep, increased numbers of awakenings, and decreased sleep quality [153, 450]. Women with a more rapid increase in FSH throughout menopause reported lower overall sleep quality [186]. Additionally, hot flashes which are a prevalent feature of the menopausal transition and a source of frequent awakenings [451] have been shown to increase in prevalence and severity with increasing FSH levels [183, 432].
Typically, circulating FSH levels increase substantially about 2 years before the final menstrual period, but can begin rising up to 6 years beforehand [452]. A few studies have shown that increasing FSH levels are associated with AD. In women, an increased FSH/estradiol ratio during menopause has been associated with cognitive impairment [453], may have a negative effect on verbal memory [454], and has been associated with a reduction in amygdala volume and working memory [455]. Interestingly, specific variants of the FSH receptor gene have a protective effect while other variants are associated with an earlier age at menopause [456].
In one study using several mouse models of AD, FSH was shown to cross the blood-brain barrier, increase amyloid and tau levels in the hippocampus and cortex, and to reduce cognitive function and spatial memory [34]. The same study showed that blocking FSH receptors ameliorates AD like pathology and restores cognition [34]. This study, as well as the studies blocking LH with leuprolide acetate (described above), support the concept that attenuating the dramatic post-menopausal increases in circulating gonadotropins may be a promising therapeutic strategy for AD. Future research should aim to confirm the role of FSH in AD in women and in men, as well as extend upon the knowledge of FSH and its role in sleep throughout the lifespan in both men and women.
SEX DIFFERENCES IN SLEEP DISORDERS AND THEIR ASSOCIATION WITH AD
When it comes to sleep disruption and disorders, studies have shown that there appears to be a sex bias, with women experiencing sleep disorders at a higher frequency. As sleep disorders negatively impact overall health over time, the increased prevalence of sleep disorders in women could lead to a substantial negative impact on women’s overall health, especially brain health, with age. While most sleep disorders as well as comorbidities that impair sleep occur in both sexes, women experience higher rates of insomnia [37, 457–460], depression and anxiety [461, 462], RLS [173, 464], chronic pain [465], and thyroid disorders [466], all of which have been shown to worsen with age. Obstructive sleep apnea has traditionally been regarded as a male-dominated disease; however, recent research has shown that this disease presents differently in women and as such may be underdiagnosed [467]. Additionally, sleep apnea rates rise dramatically in women after menopause, and both sleep apnea and menopause increase AD risk [393, 468]. Other than menopause, female-specific diseases, such as polycystic ovary syndrome (PCOS) that are highly prevalent yet often overlooked, have been heavily linked to sleep disorders due to their comorbidities that disrupt sleep [469]. As all these sleep disorders and associated comorbidities are indirect drivers of AD risk, their increased prevalence in women may be a contributing factor to the increased rate of AD in women (Fig. 4).
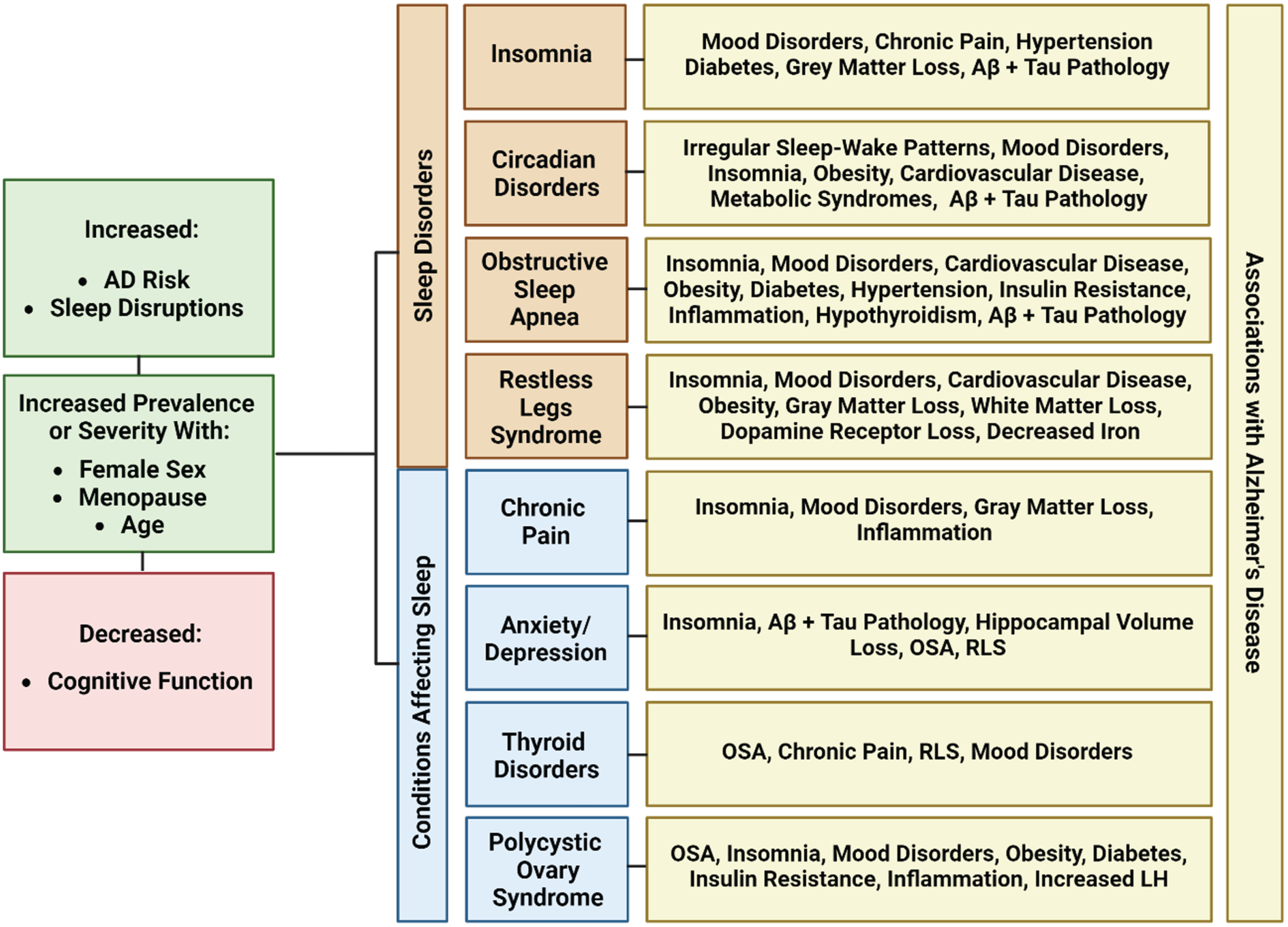
Shared characteristics between common sleep disorders and health conditions with increased prevalence in women. All conditions have been associated with an increased AD risk, increased sleep disruptions, and decreased cognitive functions. They are all increased in prevalence or severity with age, especially throughout the menopausal condition. Additionally, each condition shares many overlapping symptoms, comorbidities, and pathology with AD. Created with BioRender.com.
Insomnia
Insomnia is a common sleep disorder that has been linked to AD and has known sex differences. People with insomnia struggle with falling asleep, staying asleep, or achieving quality sleep. The buildup of these deficits night after night results in impaired daytime functioning [470]. It is the most commonly occurring sleep problem [471] in the US, and it typically occurs in conjunction with a variety of physical and mental health diseases [471, 472] such as depression and anxiety [473, 474], pain disorders [475], and other sleep disorders [476–479]. Additionally, it increases risk of hypertension, type 2 diabetes, neurocognitive disorders, and mortality [480, 481]. Insomnia prevalence increases with age with 30–40% of older adults reporting symptoms [482, 483]. It occurs more often in women than in men [37, 457–460], and as a result, women are more likely to report impaired daytime functioning [484, 485] and unsatisfactory sleep [486–488].
Like many other diseases, insomnia may be affected by fluctuating gonadal hormones. In girls who are transitioning from a pre-menses to post-menses state at age 10, insomnia risk was found to have a 2.75-fold increase as compared to girls who had not yet experienced menses. When compared to boys of the same age, the risk of insomnia for post-menses girls was found to be 2.5-fold [137], and this risk remains higher in women throughout middle age [482]. Continuing into old age and throughout menopause, insomnia rates only continue to increase [489, 490].
Insomnia increases the risk of developing dementia and occurs in up to one-third of dementia patients [491, 492]. In older adults, insomnia impairs cognitive function [493, 494] and has also been shown to reduce grey matter volume in areas related to AD including the hippocampus and cortex [495, 496]. Insomnia induced experimentally by sleep deprivation and/or chronic sleep restriction have been shown to promote AD neuropathology in both humans and rodents. In rodents, sleep deprivation has been shown to impair cognition, and increase levels of amyloid and phosphorylated tau in the brain [251, 498] (Fig. 4). Interestingly, in a study of cognitively normal middle-aged individuals, those with insomnia showed an increase in Aβ42 in CSF [499], possibly indicating that changes in Aβ metabolism related to sleep disruption might precede AD by several decades.
Restless legs syndrome
RLS is a common sensorimotor sleep disorder thought to be exacerbated by low iron, and characterized by prickling sensations, involuntary limb movements, and the urge to move the legs while at rest [500–502]. Oftentimes symptoms last for hours and can be partially or completely relieved by movement such as walking [503]. RLS symptoms occur predominantly at nighttime, making sleep disruption a serious consequence of this disease as it ultimately results in decreased daytime functioning and decreased overall health [173, 503–508].
RLS typically worsens with time and increases in prevalence with age [509, 510] and is twice as common in women as in men [173, 464]. During reproductive years, RLS increases in severity during menstruation, and may be linked to low iron levels [511–513]. RLS is also experienced at an increased rate by many women during pregnancy [172, 515], has a higher rate of prevalence in older adult women [510], and increases throughout menopause [508].
Studies have shown that risk of dementia may be 1.74 times higher in those with RLS [516], and that RLS is associated with several indirect drivers of AD including sleep disturbances, mood disorders, obesity, and cardiovascular disease [517–523]. Similarly to AD, RLS can cause patients to have trouble with concentrating on tasks, short term memory, and learning [524]. Several studies have shown that patients with AD and RLS share some common pathophysiology such as decreased white and gray matter volumes [525, 526], and decreased dopamine receptors [527, 528]. It is also thought that iron deficiency, which is a core contributing factor to RLS, could induce cognitive impairment or dementia [516, 529] (Fig. 4).
Chronic obstructive sleep apnea
Chronic obstructive sleep apnea (OSA) is the most frequently occurring form of sleep disordered breathing in the US [530], occurring in 26% of the population between ages 30 and 70 [531]. During sleep, individuals who experience OSA have repetitive collapses or near collapses of the upper airway which cause momentary cessation of breathing and episodes of hypoxia [532]. Eventually, these nighttime symptoms become so disruptive that neurological impairments arise during the daytime [532–534]. As with many other diseases, OSA presents differently in women versus men and can vary with age and hormonal status including menopause and pregnancy. These differences in clinical presentation contribute to underdiagnosis and undertreatment in women [467] in a “traditionally male” disease. Early studies suggested that OSA might be much more prevalent in men (i.e., 3–5 times more [535]); however, more recent studies suggest that women: 1) are nearly as likely as men to have OSA (current averages are 27% in men and 22% in women) [536, 537]; and 2) represent 40–50% of the current OSA patients in sleep clinics [538].
Women with OSA tend to be more symptomatic and [539] have increased rates of: insomnia, daytime fatigue, headaches in the morning, mood related symptoms and nightmares; and lower energy levels the following day [467, 540–543]. Women also have more comorbidities associated with OSA [544] including hypothyroidism, sleep disorders, and AD [542]. When looking at OSA and menopause, there is a lack of research as well as a lack of defined clinical definitions and presentations for the disease. OSA doubles in prevalence in women after menopause, and peaks at age 65, which is on average, 10 years later than the peak age of onset in males [545–547]. Additionally, women who undergo surgical menopause as compared to natural menopause, have an increased rate of OSA [548].
Not only is OSA more prevalent in older adults, it has also been associated with decreased cognitive function and AD [549–551]. OSA has been shown to increase risk of developing AD by three times and may lead to earlier onset of MCI [551–553]. Almost half of AD patients experience OSA [554] and the severity of OSA is often indicative of severity of AD symptoms [555]. Clinical studies have shown that cognitively normal individuals with OSA have increased levels of Aβ42 in CSF, and those with cognitive impairment with untreated OSA had higher total tau/Aβ42 ratio and lower levels of Aβ42 in CSF than those with treated OSA [556]. Similarly, in mouse models of OSA, chronic intermittent hypoxia increased brain amyloid and phosphorylated tau levels [557] (Fig. 4). When OSA is treated: executive function in cognitively normal individuals is improved; and in those with mild to moderate AD, cognitive decline is slowed and memory, executive function, sleep and mood are improved [558–561].
CONDITIONS WITH INCREASED PREVALENCE IN WOMEN AND THEIR RELATIONSHIP TO SLEEP AND AD
Chronic pain
It has been well documented that sleep disorders are prevalent in those suffering from chronic pain, which are defined as pain lasting more than 3 months [562]. Chronic pain is associated with negative effects on many aspects of cognitive and behavioral health [563]. Up to 88% of people living with chronic pain experience sleep disorders [564, 565] and the two conditions have been shown to have a bi-directional relationship [566, 567]. Furthermore, chronic pain, sleep disturbances, and AD share a wide array of comorbidities including obesity [568–570], type 2 diabetes [569–573], depression [574–580], and cardiovascular disease [581, 582]. In large population-based research it is consistently estimated that women have a higher prevalence of pain than men due to increased rates of chronic conditions (e.g., fibromyalgia, chronic tension type headache, irritable bowel syndrome, etc.) [465], self-report pain more often, and have a greater sensitivity to pain [583–585]. Additionally, women who are experiencing menopausal symptoms are more likely to experience chronic pain [586].
Insomnia is the most common sleep disorder associated with pain occurring at a rate of 50% [587]. Additionally, insomnia has been shown to increase risk of developing chronic pain [588], and increase pain in those already living with chronic pain [567, 588–591]. Alternatively, improvement in sleep quality is associated with a remission in chronic pain [565, 588–592].
In older adults, chronic pain is common and has been associated with cognitive deficits [593]. Estimates show that up to 50% of those living with chronic pain experience cognitive issues [594, 595] and with increasing pain comes increasing cognitive impairment [596–598]. Unfortunately, it is common for chronic pain to be underdiagnosed and undertreated in patients with AD due to difficulties in communication/expression of pain with decreased cognitive abilities, and difficulties in the perception of pain by providers [599, 600]. What is more disturbing is that those with both AD and chronic pain may actually be having worse chronic pain [601]. Those experiencing chronic pain have declines in grey matter volume [563, 602–606] in several brain areas, most commonly the PFC, an area also involved in sleep and affected in AD [607–609]. Furthermore, chronic pain has been found to be comorbid with AD in approximately half of patients [610], with those having more prevalent and severe pain having more severe dementia [611–614]. Recent research has shown that some of the underlying neurological changes associated with chronic pain or AD pathology may overlap. Neurological changes can include deficits in noradrenergic neurons in the LC [615–621], as well as increased inflammation [617, 623] and microglial activation [623–626] in brain areas affected by AD (Fig. 4).
Thyroid disorders
Thyroid disorders are common in the general population [627] and can have expansive effects in the body. The two most common forms of thyroid dysfunction, which are hypothyroidism (underactive thyroid) and hyperthyroidism (overactive thyroid) [628], are up to ten times more common in women than in men [466, 629–632].
Both too much and too little thyroid activity have been associated with sleep disruptions. Some studies have shown an association between hypothyroidism and poor sleep quality, longer times to fall asleep, and shorter duration of sleep [633]. Hypothyroidism may contribute to OSA [634–637] as it may cause an increase in thyroid size, which could cause or worsen upper airway obstruction and alter respiratory muscle function [638, 639]. Additionally, low thyroid hormone levels have been associated with musculoskeletal pain, cold intolerance, RLS, and increased anxiety, all of which have been linked to sleep disturbances and insomnia [640], and several of which show increased prevalence in women. In a study of patients with hyperthyroidism, two-thirds of patients had difficulties falling asleep [641] and additional studies have associated hyperthyroidism with prolonged times to fall asleep, difficulty staying asleep, increased daytime sleepiness, tremor which caused difficulty falling and staying asleep [642], RLS [643], and insomnia [644]. Hyperthyroidism can also cause or exacerbate other health conditions that have been known to cause sleep disruptions and insomnia, such as depression and anxiety [645, 646].
Thyroid diseases are not only a risk for sleep disorders but have also been linked to AD risk. During routine testing of older adults for dementia, screening for thyroid function is conducted since both short term hypo- and hyperthyroidism can cause reversible cognitive impairment [647–650], while long-term hypo- and hyperthyroidism can cause irreversible dementia [651–655]. One longitudinal study showed that either abnormally low or high circulating levels of thyroid stimulating hormone (TSH) were linked to increased AD risk, but only in women [656]. Studies have also shown an association between the APOE4 allele in older people with Down syndrome such that women with hypothyroidism and APOE4 had increased AD risk [657]. Another study found that higher levels of TSH were associated with increased atrophy of the hippocampus and amygdala [658]. Several studies have shown that low TSH levels may contribute to AD pathology such as increased amyloid levels and increases tau phosphorylation [659, 660] (Fig. 4).
Anxiety and depression
Sleep disturbances are the most commonly reported symptoms experienced by those with anxiety and depression [661–663] and there is much evidence for a bidirectional relationship between sleep disturbances and these disorders [664]. High prevalence of sleep disturbances is a strong predictor of future anxiety and depression, which in turn increase the risk for future insomnia [661, 665–668]. Additionally, when sleep disturbances are treated, it has been shown that individuals are more likely to experience a decrease in anxiety and depression [669, 670]. For both anxiety and depression, sleep disruption has been associated with more severe symptoms, higher rates of relapse, and lower rates of remission [666, 671–677]. The most commonly seen sleep disturbances in those with anxiety and depression, either independently or in combination, are insomnia, hypersomnia, OSA, RLS, and evening circadian disorders [678–685].
Women are twice as likely as men to suffer from depression and most anxiety disorders [461, 462]. Anxiety and depression occur at increased rates in women in every age group, with anxiety being twice as likely in girls as young as age 6 [686], depression being twice as likely in girls beginning at puberty [687], and both remain higher in women throughout menopause and beyond [462, 688]. Women appear to be more susceptible to anxiety and depression during periods of extreme hormonal fluctuation from monthly menstrual cycles, pregnancy, and menopause [689–691], and increased levels of FSH and LH throughout menopause have been linked with depression [692].
Although the rates of anxiety and depression do not increase substantially with age [693–697], almost all AD patients exhibit some sort of behavioral and psychological symptoms of dementia, with anxiety and depression being present in up to 75% of cases [698]. Oftentimes, they are early symptoms of AD and have been associated with more severe cognitive impairment and faster rates of cognitive decline [699–706]. In addition to behavioral aspects of AD, patients with anxiety and depression display increased amyloid and tau pathology, along with atrophy in certain brain regions [707–717] (Fig. 4). As depression, anxiety, and AD are all correlated with increased sleep disruptions, the three could have a synergistic effect in patients with more than one comorbidity [718] and could potentially have a more detrimental effect in women due to their increased prevalence. One potential contributing factor to this complex relationship could be orexins, which are neuropeptide integral in regulating wakefulness. Orexins have also been implicated in anxiety and depression [719, 720] as individuals with depression display a disrupted 24-hour orexin rhythm [721]. Sex-based orexin differences have been observed as orexin immunoreactivity in the PFC is higher in women with depression [722], and CSF levels of orexin are reported to be higher in women with AD [723].
Polycystic ovary syndrome
PCOS has recently had many documented interactions with sleep disruptions and other AD risk factors [469]. PCOS is a lifelong endocrine disorder with unknown causes that affects not only reproduction, but also metabolism and behavioral health. It affects 5–10% of women of reproductive age [724, 725] and is driven by increased male sex hormone production, or hyperandrogenism, in the ovaries [726]. Patient symptoms and comorbidities vary, ranging from hormonal imbalance, insulin resistance, obesity, inflammation, irregular menstruation, ovarian cysts, fertility problems, high blood pressure, cardiovascular diseases, worsened cognition, as well as sleep disorders [469, 726–729]. The varied presentation of the disease makes it difficult to treat and diagnose [730], and most physicians managing PCOS are trained very little in the management of the accompanying sleep disorders [731, 732].
Sleep problems occur up to twice as often in those with PCOS compared to age-matched controls and include poor sleep quality, trouble falling asleep, trouble staying asleep, excessive daytime sleepiness, insomnia, and sleep disordered breathing [733–742]. The most common sleep disorder in those with PCOS is OSA, which affects up to three-quarters of those with the condition [730, 744], followed by insomnia [742]. Behavioral disorders like anxiety and depression are also elevated in women with PCOS [745–748], and one study of women with PCOS and depression showed a three-fold increase in sleep disturbances [736]. Similar to other diseases, chronic poor sleep is thought to have a bidirectional relationship with PCOS and both may exacerbate many of the symptoms and comorbidities of the other.
Many of the comorbidities of PCOS that play a role in sleep disturbances are known risk factors for AD; furthermore, many of them interact with each other in a circular fashion. For example, insulin resistance, which is present in the majority of PCOS patients [749], is strongly associated with excessive daytime sleepiness [750], and is the strongest risk factor for sleep apnea [739]. Both insulin resistance and OSA increase the risk of AD [751–753], OSA is higher in women after menopause [468, 545–547], and menopause increases risk of insulin resistance [754]. Lastly, women with PCOS often have a high LH/FSH ratio with overproduction of LH [755–758] and as previously mentioned, a correlation between increased LH, a decline in memory, an increase in amyloid levels, and an increase in AD risk may exist [433–435, 759] (Fig. 4). Further confirmatory research is needed to determine whether there is a direct link between PCOS and AD risk in women.
CONCLUSION
Over the past several decades it has become clear that women face increased disease risk and burden owing to inadequate attention in healthcare. This imbalance stems not only from intrinsic biological factors related to female sex, but is also a result of exclusion from research, underfunding of woman-centric research, and underdiagnosis and less than optimal treatment in medicine. With the increasing aging population, gaps in knowledge of women’s brain health are a glaring example of this shortcoming as women with AD far outnumber the men. The same hormonal fluctuations that caused women and female animals to be excluded from clinical and scientific research are proving to be highly relevant and critical in the treatment and prevention of age-related diseases.
Throughout the lifespan, girls and women experience significantly more hormonal fluctuation compared to boys and men with the onset of puberty, pregnancy, and menopause. While it is possible that hormonal fluctuation from these life events has an additive effect on disease risk for women over time, it is certain that the menopausal transition is contributing to an overall decrease in brain health for most, if not all, middle-aged women. Numerous studies have linked menopause with increased AD risk, which may help to partially explain the increased number of women with AD.
Coinciding with menopause is a drastic increase in sleep disturbances which is a known risk factor for AD. Both menopause and sleep disturbances have been shown to increase AD pathology in the brain, and both pathology and sleep disturbances may be worsened by the loss of gonadal steroids and the increase of gonadotropins during menopause. Studies have shown that women have more global amyloid and tau pathology than men and increased brain atrophy in almost all age groups, and the interaction between sex, hormones, and sleep disruption may be a contributing factor.
What’s more, women are also prone to having more subjective sleep complaints throughout the lifespan and are either more likely to suffer from sleep disorders and related conditions throughout the lifespan, or to face a drastic increase in both throughout menopause. Women are at increased risk for insomnia, RLS, thyroid disorders, chronic pain, anxiety, and depression from adolescence to late adulthood, and the risk for all these plus OSA increases throughout menopause and persists into late life. All these health conditions have in turn been shown to have a negative impact on brain health and may influence AD pathology and neurodegeneration in a bidirectional manner. What is lacking is a better understanding of what the downstream mechanisms are for these interactions at the molecular level. Partially, this is due to the complex nature of the problem, although it is also at least somewhat reflective of the lack of attention to sex differences in the biomedical literature until relatively recently. A deeper examination of these understudied processes could lead to sex and gender-based therapeutics that might bridge the gap between men and women living with AD and related dementias.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
This work was supported in part by AG068215 (MJD and MPM); C.E. Johnson is supported by T32 AG078110.
CONFLICT OF INTEREST
M. Paul Murphy, MA, PhD, is an Editorial Board Member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer- review.
