Abstract
Background:
In Alzheimer’s disease (AD), the gradual accumulation of amyloid-β (Aβ) and tau proteins may underlie alterations in empathy.
Objective:
To assess whether tau aggregation in the medial temporal lobes related to differences in cognitive empathy (the ability to take others’ perspectives) and emotional empathy (the ability to experience others’ feelings) in AD.
Methods:
Older adults (n = 105) completed molecular Aβ positron emission tomography (PET) scans. Sixty-eight of the participants (35 women) were Aβ positive and symptomatic with diagnoses of mild cognitive impairment, dementia of the Alzheimer’s type, logopenic variant primary progressive aphasia, or posterior cortical atrophy. The remaining 37 (22 women) were asymptomatic Aβ negative healthy older controls. Using the Interpersonal Reactivity Index, we compared current levels of informant-rated cognitive empathy (Perspective-Taking subscale) and emotional empathy (Empathic Concern subscale) in the Aβ positive and negative participants. The Aβ positive participants also underwent molecular tau-PET scans, which were used to investigate whether regional tau burden in the bilateral medial temporal lobes related to empathy.
Results:
Aβ positive participants had lower perspective-taking and higher empathic concern than Aβ negative healthy controls. Medial temporal tau aggregation in the Aβ positive participants had divergent associations with cognitive and emotional empathy. Whereas greater tau burden in the amygdala predicted lower perspective-taking, greater tau burden in the entorhinal cortex predicted greater empathic concern. Tau burden in the parahippocampal cortex did not predict either form of empathy.
Conclusions:
Across AD clinical syndromes, medial temporal lobe tau aggregation is associated with lower perspective-taking yet higher empathic concern.
INTRODUCTION
In Alzheimer’s disease (AD), the progressive accumulation of amyloid-β (Aβ) plaques and tau neurofibrillary tangles [1–3] can manifest as several distinct clinical syndromes [4]. Although the topography of Aβ aggregation in the neocortex is diffuse and not consistently related to symptoms or disease progression [5–7], regional tau deposition closely aligns with cognitive deficits [8–13] and atrophy (e.g., [14–16]) and predicts functional decline [17] and clinical severity [18, 19] better than Aβ. In AD, tau first aggregates in the brainstem (e.g., locus coeruleus) [20, 21] and medial temporal lobes (MTL) [3, 23]. In people with mild cognitive impairment (MCI)—the clinical phase that precedes functional impairment—and “typical” dementia of the Alzheimer’s type (DAT), MTL pathology often gives rise to episodic memory decline [24]. “Atypical” AD clinical syndromes emerge when pathological changes target other brain regions and cause deficits in language, visuospatial processing, or executive functioning [25, 26]. Whereas individuals with prominent language and phonological processing deficits may meet criteria for logopenic variant primary progressive aphasia (lvPPA) [27], those with predominant visual and spatial deficits may receive a diagnosis of posterior cortical atrophy (PCA) [28].
Cognitive symptoms in AD have been well characterized [24], but the changes in behavior and emotion that also arise remain poorly understood (e.g., [29–31]). Heightened social and emotional sensitivity may contribute to the neuropsychiatric symptoms that emerge in the symptomatic phase of AD [32–38] and to the gains in negative emotional reactivity, stress, and loneliness that characterize the preclinical period [39–42]. Neuropathological changes in AD may also affect how people understand or respond to others and thereby alter cognitive empathy (the ability to adopt the viewpoint of others) as well as emotional empathy (the ability to experience what others are feeling via physiological and motor mirroring systems) [43]. Tests of cognitive empathy often entail recognizing emotions in others [44], but cognitive empathy also includes perspective-taking abilities [45]. Although there is some evidence that people with AD have lower informant-reported perspective-taking than healthy older adults [29], it is unclear whether this impairment reflects more widespread cognitive deficits or problems with mentalizing per se [46]. There is accumulating evidence, in contrast, that emotional empathy climbs in the early stages of AD, even when people are cognitively asymptomatic. In our prior research, we have found elevations in two forms of emotional empathy—emotional contagion (a self-oriented form of emotional empathy that can be accompanied by feelings of distress) and empathic concern (an other-oriented form of emotional empathy that promotes prosocial actions) [47, 48]—in people on an AD trajectory. While emotional contagion is higher in MCI and AD than in healthy older adults [49], empathic concern increases more over time in cognitively healthy older adults with higher cortical Aβ than in those with lower levels [50].
In people with AD, tau deposition in the MTL may contribute to changes in empathy. Cognitive and emotional empathy have largely distinct neuroanatomical underpinnings [43, 51], but both rely on the MTL [51, 52]. Cognitive empathy, which allows individuals to step outside of their own minds and to take the perspective of another, often engages MTL structures (primarily the entorhinal cortex and hippocampus) as well as other regions in the default mode network [43, 51–56]. Tau pathology in the MTL, therefore, may disrupt perspective-taking as the default mode network declines in AD [53–55, 57–59] but have the opposite effect on emotional empathy. Unlike cognitive empathy, which does not necessarily elicit changes in subjective experience [51], emotional empathy evokes shared feeling states between people by activating the MTL (primarily the amygdala) and other structures in the salience network [51, 60], a system that supports emotion generation and interoception [61–64]. In early AD, default mode network dysfunction is accompanied by heightened salience network connectivity, a functional gain that relates to neuropsychiatric symptoms in the clinical phase [65, 66] and increasing empathic concern in the preclinical period [50]. While there is some evidence that early tau deposition in the MTL relates to heightened stress, anxiety, and depression [41, 42], whether MTL tau aggregation also relates to higher emotional empathy in AD is unknown.
Here, we investigated cognitive and emotional empathy in AD clinical syndromes and their associations with MTL tau pathology. As neuropathological changes in AD can give rise to a diverse set of syndromes that vary in the extent to which they affect the MTL [67, 68], we included participants with a wide range of symptoms who met criteria for MCI, DAT, lvPPA, or PCA. All participants with AD were in the very mild to mild clinical stages of disease and were Aβ positive (Aβ+) on molecular positron emission tomography (PET) imaging. Given that previous studies have found diminished cognitive empathy and enhanced emotional empathy in people along the AD continuum [29, 50], we expected that symptomatic Aβ+ participants would have lower perspective-taking (a measure of cognitive empathy) yet higher empathic concern (a measure of emotional empathy) than Aβ negative (Aβ-) healthy older controls. While elevated Aβ accumulation may set the stage for empathy alterations in AD, regional tau aggregation may play a critical role in shaping empathy over the course of the disease. We hypothesized that, just as the default mode and salience networks show divergent functional changes in the setting of AD pathophysiology [69–71], tau aggregation in the MTL would also have opposing effects on empathy. We expected that greater tau burden in the entorhinal cortex and amygdala would relate to lower perspective-taking but higher empathic concern in Aβ+ participants. In contrast, we anticipated that tau burden in the parahippocampal cortex, an MTL region that is less involved in empathy but more involved with cognition [72], would not relate to cognitive or emotional empathy.
MATERIALS AND METHODS
Participants
A total of 105 older adults (48.0–85.5 years old) recruited from the University of California, San Francisco (UCSF) Memory and Aging Center participated in the present study. The sample included 68 symptomatic participants who met criteria for MCI (n = 14) [73], DAT (n = 33) [74], lvPPA (n = 10) [27], or PCA (n = 11) [28] and 37 healthy older controls. In the MCI group, 12 participants had an amnestic-predominant presentation, and two had a non-amnestic, multi-domain syndrome [73, 75]. The study was approved by the UCSF Human Research Protection Program, and informed consent was given by all participants, or their surrogates, before participating.
Participants underwent multidisciplinary diagnostic evaluations that included a neurological examination, neuropsychological testing, neuroimaging, and an informant-based assessment of daily functioning using the Clinical Dementia Rating Scale (CDR) [76]. The neuropsychological assessment included tests of episodic memory, executive functioning, visuospatial processing, language, and mood [77]. All symptomatic participants had CDR total scores of 1 or less, which suggests they were in the very mild to mild stages of impairment. The healthy controls underwent the same diagnostic evaluation as those in the symptomatic group. They were free of current psychiatric or neurological disorders, had a score of 0 on the CDR (indicating no functional impairment), and a score of 27 or greater on the Mini-Mental State Examination (MMSE), an assessment of overall mental status [78].
Cognitive and emotional empathy
Informants rated participants’ current levels of empathy using the Interpersonal Reactivity Index (IRI), a multidimensional empathy measure [79]. Informants assessed participants’ behavior using a scale of 1 (does not describe well) to 5 (describes well). Each IRI subscale contains scores ranging from 7 to 35 where higher scores indicate greater levels of empathy. Informant reports are a reliable technique to evaluate empathy and personality in individuals who are healthy [80, 81] and in those with dementia [29, 82].
In line with our prior work on AD [29, 50], we used the Perspective-Taking IRI subscale as our measure of cognitive empathy and the Empathic Concern IRI subscale as our measure of emotional empathy. While the Perspective-Taking subscale measures the tendency to adopt others’ points of view (e.g., “Likely to try to understand others better by imagining how things look from their perspective”), the Empathic Concern subscale evaluates other-oriented feelings of compassion and concern (e.g., “Would show tender, concerned feelings for people less fortunate than them”).
Molecular PET scan acquisition and processing
Amyloid-PET scans
Participants underwent Aβ-PET imaging with either 11C-Pittsburgh compound B (PIB) or 18F-AV-45 (Florbetapir) PET ligands. The Aβ-PET scans were used to dichotomize Aβ levels into Aβ+ and Aβ-, which indicated the presence or absence of AD pathology following previous methods (e.g., [83, 84]). All participants in the symptomatic group were Aβ+ as determined by a visual read [83, 85] of their PIB (n = 67) or Florbetapir (n = 1) Aβ-PET scans at the time of their empathy assessment (mean [M] = 8.4 days after IRI, standard deviation [SD] = 239.3 days). All healthy controls were Aβ- on visual reads of their Florbetapir (n = 36) or PIB (n = 1) Aβ-PET scans. An additional 23 healthy controls had available empathy data but were Aβ+, and thus were excluded. For the Aβ- healthy controls, the Aβ-PET imaging occurred after the empathy assessment (M = 6.5 years after IRI; SD = 2.5 years), at which time they were still cognitively normal and functionally intact.
Tau-PET scans
Symptomatic participants also underwent 18F-AV-1451 (Flortaucipir) tau-PET imaging using established protocols [10, 87], and these scans were used to correlate regional MTL tau aggregation with empathy. Standardized uptake value ratio (SUVR) values were calculated according to previous methods [10, 87], using PET data acquired between 80- and 100-minutes post tracer injection, and using the inferior cerebellum grey matter as a reference region. Freesurfer segmentation [http://surfer.nmr.mgh.harvard.edu; 88] was performed to acquire regional values in right and left MTL regions of interest (i.e., entorhinal cortex, amygdala, and parahippocampal cortex) using the Desikan-Killiany atlas. All the included imaging data passed our visual quality control checks that assessed the magnetic resonance imaging (MRI) to PET scan registration and the Freesurfer segmentation. The data were not partial volume corrected because this entails making multiple assumptions, and our previous work has shown that SUVR values computed with and without partial volume correction are highly correlated (r = 0.98) [89].
We hypothesized that the entorhinal cortex and amygdala would relate to empathy in AD because these structures are hubs in networks that support empathy and typically have more tau signal than the parahippocampal cortex [43, 60]. The parahippocampal cortex plays a central role in contextual associations [72] and, while it can be related to socioemotional behaviors (e.g., [90]), is less critical for empathy. Thus, we included the parahippocampal cortex as a control region within the MTL as we did not expect tau burden in this area would relate to empathy in AD. The tau-PET scans were obtained in close proximity to the Aβ-PET scans (M = 26.7 days after Aβ-PET scans, SD = 225.2 days) and empathy assessments (M = 35.1 days after IRI; SD = 106.0 days).
Analyses
All analyses were conducted in R v.4.0.3 [91].
Empathy analyses
We used multivariate linear regression analyses to compare the Aβ+ symptomatic and Aβ- healthy control groups on perspective-taking and empathic concern. Group (Aβ+ = 1, Aβ- = 0) was the independent variable, and the perspective-taking or empathic concern subscale score was the dependent variable. Covariates of non-interest included gender, age at IRI, and the contrasting IRI subscale score (i.e., the empathic concern score in the perspective-taking analysis and the perspective-taking score in the empathic concern analysis) to isolate the effects of cognitive or emotional empathy in each model.
We next conducted exploratory multivariate linear regression analyses to compare each clinical group (MCI, DAT, lvPPA, and PCA) to the healthy controls on perspective-taking and empathic concern (using the same covariates as above). Follow-up Type-II analyses of variance were conducted to determine the significance of the fixed effects. Results were considered significant if the p-values of the post hoc pairwise comparisons survived Bonferroni correction for four analyses (to account for pairwise comparisons between each clinical group and the healthy controls).
Tau and empathy analyses
We conducted forward-selection hierarchical regression models [92] using the MASS package in R [93] to examine associations between regional tau burden and empathy in the Aβ+ group. This approach avoids problems with multiple comparisons as it allowed the statistical program to determine the regions in which tau SUVR accounted for a significant amount of variance in empathy above and beyond the covariates of non-interest that were forced into the model in step one [94]. This is a conservative test of our hypothesis that the entorhinal cortex and amygdala are important for empathy because these regions must emerge as significant predictors from a group of candidate predictor regions (i.e., tau SUVR in the entorhinal cortex, amygdala, and parahippocampal cortex). To minimize collinearity between homologous regions and to assess potential effects of lateralization on emotion-related processes [95], we ran separate hierarchical regression models in each hemisphere. Covariates of non-interest included gender, age at IRI, CDR total score (to account for disease severity), and the contrasting IRI subscale (as described above). To account for the time interval between the tau-PET scan and IRI assessment, we computed a “time interval” variable (IRI date –tau-PET scan date) for each participant and included this variable as an additional covariate of non-interest in these analyses. Only variables that were significant predictors of empathy at p < 0.05 were eligible to enter the final regression model whereas variables that did not account for significant variance were excluded. All histograms and P–P plots of standardized residuals for the final regression models demonstrated normally distributed error terms, indicating that the assumptions of regression models were met. All variables in the final regression models demonstrated very weak collinearity with one another such that variance inflation factors < 2.0.
RESULTS
Aβ+ and Aβ- groups had similar demographics but different cognitive profiles
The Aβ+ symptomatic and Aβ- healthy control groups were similar in age, gender, and education (all p > 0.05). On average, the two groups were in their mid-sixties, highly educated, and predominantly White/European American. The Aβ+ participants were in the very mild to mild range of functional impairment, as measured by the CDR, and had lower scores on neuropsychological tests of episodic memory, language, visuospatial processing, and executive functioning than the Aβ- healthy controls. Depressive symptoms were minimal across the sample but somewhat higher in the Aβ+ than in the Aβ- group. Follow-up analyses that compared the MCI, DAT, lvPPA, and PCA groups to the Aβ- healthy controls showed expected cognitive profiles with milder deficits in MCI, episodic memory impairment in DAT, language difficulties in lvPPA, and visuospatial dysfunction in PCA. See Table 1.
Group demographic and clinical data
Analyses included Pearson’s Chi-squared tests and Welch’s t-tests. aIndicates a significant pairwise difference in comparison with Aβ- healthy controls (p < 0.05). bIndicates a significant pairwise difference in comparison with MCI (p < 0.05). cIndicates a significant pairwise difference in comparison with DAT (p < 0.05). dIndicates a significant pairwise difference in comparison with lvPPA (p < 0.05). eIndicates a significant pairwise difference in comparison with PCA (p < 0.05). MCI, mild cognitive impairment; DAT, dementia of the Alzheimer’s type; lvPPA, logopenic variant primary progressive aphasia; PCA, posterior cortical atrophy.
Lower perspective-taking and higher empathic concern in the Aβ+ group
There was a main effect of group on both perspective-taking, b = –5.339, t(100) = –4.824, p = 5.039 × 10–6, model adjusted R2 = 0.431, n = 105 (Fig. 1A), and empathic concern, b = 3.550, t(100) = 4.925, p = 3.344 × 10–6, model adjusted R2 = 0.513, n = 105 (Fig. 1B), such that the Aβ+ participants had lower perspective-taking but higher empathic concern than the Aβ- healthy controls. There was also a main effect of gender in the empathic concern regression model, b = 3.202, t(100) = 4.609, p = 1.197 × 10–5, model adjusted R2 = 0.513, n = 105, where women had higher empathic concern than men, which is consistent with prior studies (e.g., [45, 79]). In addition, there was a main effect of perspective-taking in the empathic concern model, b = 0.397, t(100) = 7.628, p = 1.419 × 10–11, model adjusted R2 = 0.513, n = 105, and of empathic concern in the perspective-taking model, b = 0.927, t(100) = 7.628, p = 1.419 × 10–11, model adjusted R2 = 0.431, n = 105, which suggested a positive association between these two empathy subscales.
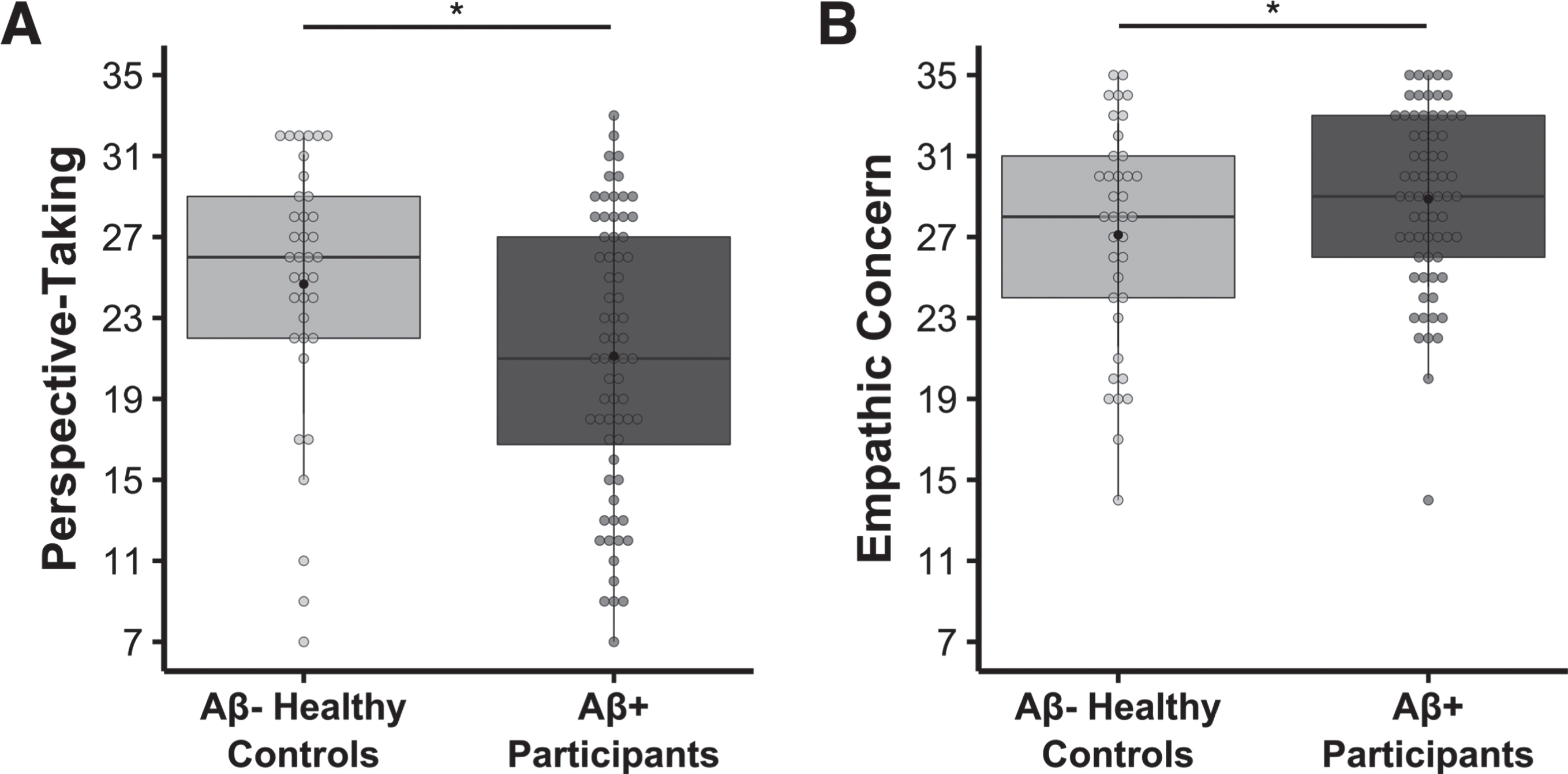
Lower perspective-taking, but higher empathic concern, in the Aβ+ symptomatic participants relative to the Aβ- healthy controls. Multivariate linear regression analyses found that, compared to Aβ- healthy controls, the Aβ+ symptomatic group had A) lower perspective-taking (b = –5.339, t(100) = –4.824, p = 5.039 × 10–6, model adjusted R2 = 0.431, n = 105) and B) higher empathic concern (b = 3.550, t(100) = 4.925, p = 3.344 × 10–6, model adjusted R2 = 0.513, n = 105). Covariates of non-interest in these models included age at IRI, gender, and the contrasting IRI subscale (i.e., empathic concern or perspective-taking). Raw perspective-taking scores and empathic concern scores are shown in the figure.
To investigate the role of gender in more detail, we conducted additional analyses to examine whether there was an interaction between group and gender. These analyses (which included all the same covariates as the previous models in addition to the interaction term between group and gender) revealed no interactions for either the perspective-taking, interaction b = 0.612, t(99) = 0.284, p = 0.777, model adjusted R2 = 0.426, n = 105, or empathic concern, interaction b = –1.636, t(99) = –1.173, p = 0.243, model adjusted R2 = 0.515, n = 105, models. All findings in the original analyses remained significant, however, including the main effect of group on perspective-taking, b = –5.696, t(99) = –3.391, p = 0.001, model adjusted R2 = 0.426, n = 105, and empathic concern, b = 4.467, t(99) = 4.206, p = 5.715 × 10–5, model adjusted R2 = 0.515, n = 105, such that Aβ+ participants had diminished perspective-taking but elevated empathic concern relative to the Aβ- healthy controls.
We next conducted a follow-up exploratory analysis of perspective-taking that compared each of the Aβ+ clinical syndromes to the Aβ- healthy controls. In this model (which included the same covariates as our original analyses), there was a main effect of group, F(4, 97) = 7.571, p = 2.374 × 10–5, model adjusted R2 = 0.449, n = 105. Bonferroni-corrected post hoc analyses revealed lower perspective-taking in the DAT, t(97) = –4.651, p BONFERRONI = 4.183 × 10–5, Cohen’s d = 1.157; lvPPA, t(97) = –4.176, p BONFERRONI = 2.590 × 10–4, Cohen’s d = 1.579; and PCA, t(97) = –2.658, p BONFERRONI = 0.037, Cohen’s d = 0.974, groups than in Aβ- healthy controls (Supplementary Figure 1A). Perspective-taking in the MCI and Aβ- healthy control groups did not differ, t(97) = –1.726, p BONFERRONI = 0.350, Cohen’s d = 0.556, however. There was also a main effect of empathic concern, F(1, 97) = 56.497, p = 2.817 × 10–11, model adjusted R2 = 0.449, n = 105, in line with the previous analysis.
In a similar follow-up exploratory analysis of empathic concern, there was also a main effect of group, F(4, 97) = 6.437, p = 1.228 × 10–4, model adjusted R2 = 0.507, n = 105. Bonferroni-corrected post hoc analyses revealed higher empathic concern in the MCI, t(97) = 2.825, p BONFERRONI = 0.023, Cohen’s d = 0.889; DAT, t(97) = 4.763, p BONFERRONI = 2.673 × 10–5, Cohen’s d = 1.180; and lvPPA, t(97) = 2.996, p BONFERRONI = 0.014, Cohen’s d = 1.177, groups than in the Aβ- healthy controls (Supplementary Figure 1B). Empathic concern in the PCA and Aβ- healthy control groups did not differ, however, t(97) = 2.095, p BONFERRONI = 0.155, Cohen’s d = 0.778. Additional main effects included gender, F(1, 97) = 21.850, p = 9.518 × 10–6, model adjusted R2 = 0.507, n = 105, and perspective-taking, F(1, 97) = 56.497, p = 2.817 × 10–11, model adjusted R2 = 0.507, n = 105, consistent with the previous analysis.
MTL tau burden had divergent associations with perspective-taking and empathic concern in Aβ+ participants
Forward-selection hierarchical regression models in the Aβ+ group revealed that greater tau burden in the left amygdala (final model b = –6.349, final model coefficient p = 0.003, final model R2 = 0.514, R2 change from preliminary model = 0.074, final model adjusted R2 = 0.466, n = 68; Fig. 2A) and right amygdala (final model b = –6.305, final model coefficient p = 0.009, final model R2 = 0.501, R2 change from preliminary model = 0.060, final model adjusted R2 = 0.451, n = 68; Fig. 2B) was associated with lower perspective-taking. Other main effects included empathic concern and the time interval between participants’ tau-PET scans and IRI (significant in both models), which suggested that a greater time interval between the IRI assessments and the tau-PET scans related to lower perspective-taking. See Table 2. Tau burden in neither the left nor the right parahippocampal cortex predicted perspective-taking.
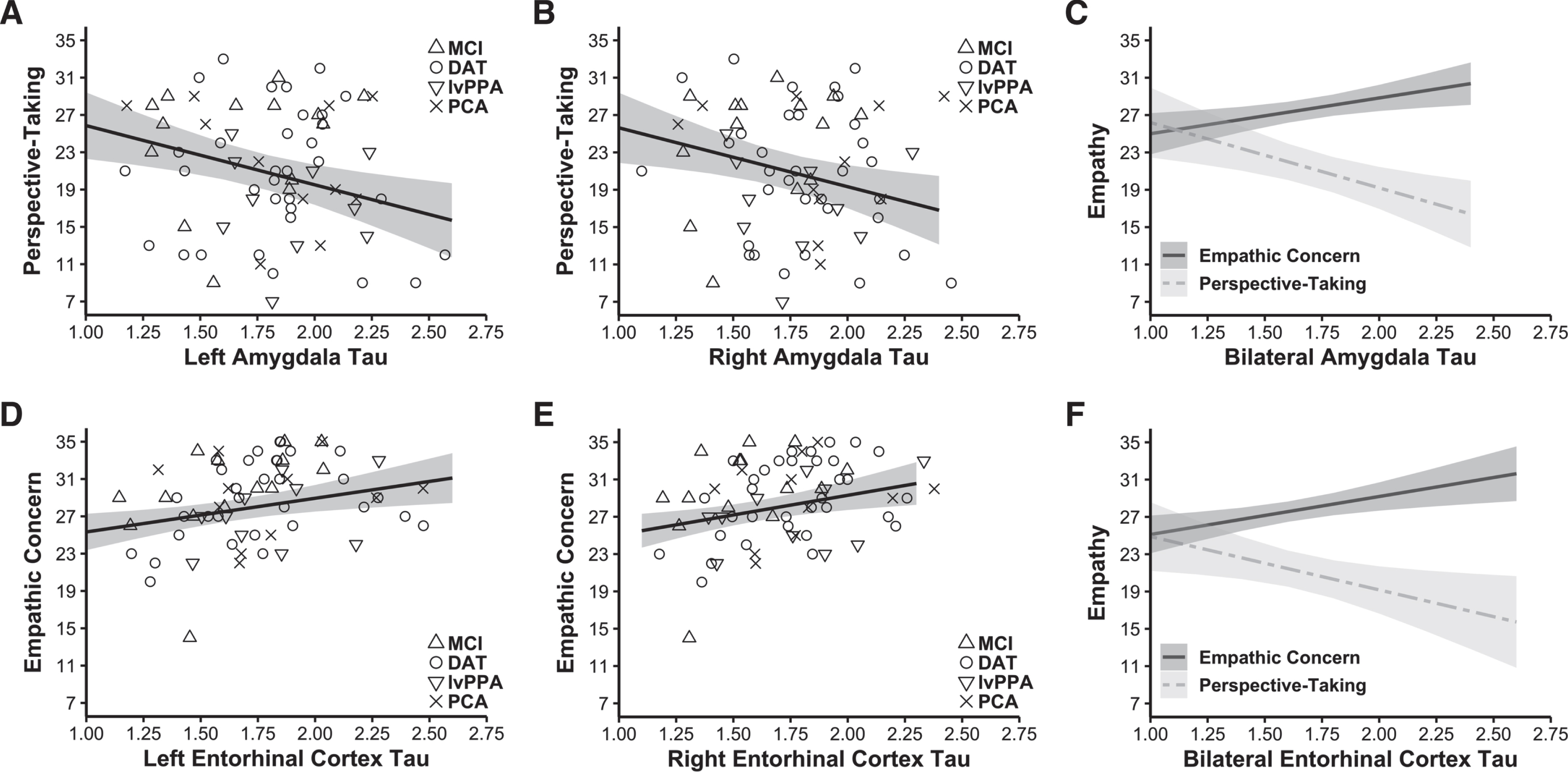
Tau burden in the amygdala had a negative association with perspective-taking, but tau burden in the entorhinal cortex had a positive association with empathic concern. Forward-selection hierarchical regression models conducted in the Aβ+ group found tau burden in the MTL was differentially related to empathy. Greater tau burden in the A) left (final model b = –6.349, final model coefficient p = 0.003, final model adjusted R2 = 0.466, n = 68) and B) right amygdala (final model b = –6.305, final model coefficient p = 0.009, final model adjusted R2 = 0.451, n = 68) related to lower perspective-taking. In contrast, greater tau burden in the D) left (final model b = 3.610, final model coefficient p = 0.007, final model adjusted R2 = 0.518, n = 68) and E) right entorhinal cortex (final model b = 4.217, final model coefficient p = 0.006, final model adjusted R2 = 0.520, n = 68) was associated with greater empathic concern. Panels C and F indicate perspective-taking and empathic concern as they relate to tau burden in the bilateral amygdala and entorhinal cortex (tau burden in these bilateral regions were calculated as weighted averages using the number of voxels present in each MTL region). Covariates of non-interest in these analyses included gender, age at IRI, time interval in days between the tau-PET scan and IRI, CDR total score, and the contrasting IRI subscale (i.e., empathic concern or perspective-taking). Plotted regressions reflect the predicted fits from the analysis models, while the scatterplots indicate raw data grouped by diagnosis. MCI, mild cognitive impairment; DAT, dementia of the Alzheimer’s type; lvPPA, logopenic variant primary progressive aphasia; PCA, posterior cortical atrophy.
Forward-selection hierarchical regression analyses assessing the impact of tau burden in the left and right amygdala on perspective-taking. Gender is coded such that men = 0 and women = 1
Forward-selection hierarchical regression models in the Aβ+ group found that greater tau burden in the left entorhinal cortex (final model b = 3.610, final model coefficient p = 0.007, final model R2 = 0.561, R2 change from preliminary model = 0.056, final model adjusted R2 = 0.518, n = 68; Fig. 2D) and right entorhinal cortex (final model b = 4.217, final model coefficient p = 0.006, final model R2 = 0.563, R2 change from preliminary model = 0.059, final model adjusted R2 = 0.520, n = 68; Fig. 2E) was associated with greater empathic concern. Additional main effects included perspective-taking (significant in both models) and gender (significant in the left hemisphere model), which suggested that higher perspective-taking was associated with higher empathic concern. See Table 3. Tau burden in neither the left nor the right parahippocampal cortex predicted empathic concern.
Forward-selection hierarchical regression analyses assessing the impact of tau burden in the left and right entorhinal cortex on empathic concern. Gender is coded such that men = 0 and women = 1
DISCUSSION
The present study uncovered novel associations between MTL tau burden and empathy in symptomatic Aβ+ participants. Compared to their cognitively healthy Aβ- counterparts, Aβ+ participants with a broad range of cognitive symptoms had diminished perspective-taking but greater empathic concern. In exploratory analyses of the different clinical syndromes, perspective-taking was lower in DAT, lvPPA, and PCA (but not MCI) than in Aβ- healthy controls while empathic concern in MCI, DAT, and lvPPA (but not PCA) was higher. In the Aβ+ participants, regional tau burden had opposing associations with empathy such that greater MTL tau pathology was associated with lower cognitive empathy yet higher emotional empathy. While greater MTL tau aggregation in the amygdala related to lower perspective-taking, greater MTL tau aggregation in the entorhinal cortex related to higher empathic concern. Tau aggregation in the parahippocampal cortex, however, was not associated with either form of empathy.
These findings build on our previous studies that revealed enhanced emotional empathy in AD [49, 50]. Although perspective-taking may decline in people with AD [29] as they lose their ability to take another’s point of view [96, 97], emotional empathy—a more automatic form of empathy that does not require higher-order cognition—climbs. Unlike emotional contagion, which can elicit self-oriented feelings of distress or being overwhelmed during negative emotional situations [48], empathic concern focuses attention outward onto the needs of others and promotes prosocial actions such as helping and consolation [43, 47]. In a prior study, we found greater gains in empathic concern over time in Aβ+ than Aβ- cognitively healthy older adults [50], which suggested there are enhancements in this form of emotional empathy in the preclinical stages of AD. In the present study, we expand on this work by showing that empathic concern is also higher in the clinical phase of AD. In general, symptomatic Aβ+ participants had higher empathic concern than Aβ- healthy controls, and follow-up pairwise comparisons revealed this difference was driven primarily by elevations in the MCI, DAT, and lvPPA groups. Although the emotional empathy enhancement in PCA failed to reach significance, our results suggest heightened emotional empathy is a common feature of AD pathophysiology.
Our results suggest that, during the early symptomatic phase of AD, tau deposition in the MTL contributes to alterations in empathy. The MTL plays important roles in empathy and other socioemotional processes including perception of emotional and social cues [98–101], emotion generation [63, 102], interpersonal sensitivity [103], and affiliative behavior [104]. Often a prominent site of early tau pathology in DAT and MCI [3, 23], the MTL is also affected in lvPPA and PCA, albeit to a lesser extent [10, 106]. Taken together with our prior study that found empathic concern gains in cognitively healthy Aβ+ older adults [50], our research suggests that Aβ and tau may both contribute to emotional empathy increases in people on an AD trajectory. Previous research has suggested that Aβ and tau interact through both local and remote connections and that Aβ deposition facilitates the spread of tau beyond the MTL [107]. Just as the synergistic effects of tau and Aβ lead to cognitive decline [108], they may also drive empathy alterations in AD. We speculate that tau accumulation in the MTL may impede perspective-taking as the default mode network declines in AD [109–113] but accentuate empathic concern, a form of emotional empathy that depends on the salience network [51, 60]. Our findings are consistent with a picture of early AD in which neuropathological changes in the MTL and its connected networks [65, 114] lead to heightened social sensitivity and emotional empathy.
The present study has several important limitations to consider. First, our data were cross-sectional, and we were unable to determine how longitudinal tau aggregation relates to changes in empathy over time. Neurofibrillary tau tangles in the MTL arise early in the AD pathophysiological cascade [115–119] and then spread to other vulnerable regions [107, 120–122]. Within the MTL, tau first aggregates in the entorhinal cortex before spreading into the hippocampus, amygdala, and parahippocampal cortex and then affecting cerebral neocortical regions [3, 23]. As tau in the entorhinal cortex was associated with elevated empathic concern, and tau in the amygdala was associated with lower perspective-taking, our results suggest the possibility that emotional empathy begins to increase before cognitive empathy declines in those on an AD course. Future longitudinal studies will be needed to investigate this question in detail, however. Second, we quantified tau deposition in MTL regions of interest, but these structures are comprised of smaller subregions that differ in their functions and connections [116, 123]. Whereas the basolateral amygdala plays an important role in evaluating affective and social information [124], the central nucleus generates affective responses via its connections to subcortical pattern generators [125–129]. Human and non-human animal studies of the entorhinal cortex have found anatomical subdivisions [130–132] such that the medial and lateral portions have different functions [133–136] and connectivity patterns [135, 137–139]. As the limited spatial resolution of PET did not allow us to differentiate between amygdala nuclei or entorhinal subregions, we were unable to determine whether tau burden in these areas had unique associations with perspective-taking or empathic concern. More fine-grained functional or structural MRI analyses may help to resolve the anatomical correlates of empathy change in AD. Third, as we could not include task-free neuroimaging in our study, we could not test whether expected changes in default mode network or salience network functional connectivity related to MTL tau burden and empathy in this sample. Additional research is warranted to address this question and to elucidate the neural mechanisms underlying cognitive and empathy alterations in AD.
With recent advances in AD biomarkers, studies can now link cognitive and behavioral measures with in vivo neuropathological changes in the brain and body. Relatively little is known about emotions and social behavior in typical and atypical AD syndromes [140], but our findings contribute to an emerging picture of the empathy changes that characterize early AD. The results of the present study suggest that lower perspective-taking and higher empathic concern may be common features of AD pathophysiology. As disease-modifying therapies become increasingly available for people with AD, new clinical outcomes will be needed to assess improvement, stability, or decline. Empathy may be an overlooked area of change in AD that, if evaluated with rigor, could help to expedite detection and to improve monitoring by offering an additional window into disease progression.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
This study was supported by the Larry L. Hillblom Foundation, the UCSF Alzheimer’s Disease Research Center (P30AG062422, P50AG023501), the John Douglas French Alzheimer’s Foundation, and grants from the National Institute on Aging (K23AG040127, K99AG065501, R01AG057204, and R01AG073244).
CONFLICT OF INTEREST
Dr. Rabinovici receives research support from Avid Radiopharmaceuticals, GE Healthcare, and Life Molecular Imaging; has received consulting fees or speaking honoraria from Axon Neurosciences, Avid Radiopharmaceuticals, GE Healthcare, Johnson & Johnson, Roche, Eisai, Genentech, and Merck; and is an associate editor of JAMA Neurology. Dr. Seeley received consulting fees or speaking honoraria from BridgeBio, Corcept Therapeutics, Biogen Idec, Bristol Myers-Squibb, Guidepoint Global, and GLG Council.
All other authors have no conflict of interest to report.
