Abstract
Background:
The mediating roles of neuropathologies and neurovascular damage in the relationship between early-life education and later-life cognitive function are unknown.
Objective:
To examine whether Alzheimer’s and neurovascular biomarkers mediate the relationships between education and cognitive functions.
Methods:
Data were from 537 adults aged 55–94 in the Alzheimer’s Disease Neuroimaging Initiative 3. We tested whether the relationships between education (continuous, years) and cognitive function (memory, executive functioning, and language composites) were mediated by neuroimaging biomarkers (hippocampal volumes, cortical gray matter volumes, meta-temporal tau PET standard uptake value ratio, and white matter hyperintensity volumes). Models were adjusted for
Results:
Hippocampal volumes and white matter hyperintensities partially mediated the relationships between education and cognitive function across all domains (6.43% to 15.72% mediated). The direct effects of education on each cognitive domain were strong and statistically significant.
Conclusions:
Commonly measured neurobiomarkers only partially mediate the relationships between education and multi-domain cognitive function.
Keywords
INTRODUCTION
In their Strategic Directions for Research, 2020–2025, the National Institute on Aging (NIA) identified that understanding “how social and economic factors throughout the lifespan affect health and well-being at older ages” is critical to informing interventions and policy decisions [1]. Education has long been identified as a protective factor against cognitive aging and Alzheimer’s disease and related dementias (ADRD) risk [2, 3]. Greater educational attainment is linked to higher levels of performance across cognitive domains, including semantic memory, executive functioning, and episodic memory, and findings are consistent across cross-sectional studies [4–6].
However, mechanisms accounting for the protective effect of education on cognitive performance are not fully understood, and longitudinal findings are mixed with some observing a continued protective effect of education against cognitive declines [7], others reporting null findings [8–10], and still others identifying a sharper decline in cognition among those with higher education as individuals progress to ADRD [11]. Education as it relates to late-life cognition is often viewed through a cognitive reserve lens [12, 13]. That is, higher experiential resources, such as education preserves cognitive functioning in the presence of brain atrophy and/or ADRD pathology potentially through differences in neural activation patterns and recruitment of brain networks [12–17]. However, in studies that incorporate biomarkers of brain atrophy or pathology, the relationships between education and cognition vary and may contradict the cognitive reserve hypothesis [11, 19]. For example, Bauer and colleagues detected direct relationships between education and cognition, but higher education did not mitigate the relationship between ADRD biomarkers and cognition in a sample of individuals with varying levels of cognitive impairment [18]. Although education may not modify the relationships between ADRD biomarkers with cognition, it may preserve cognition by increasing neurogenesis early in life which could then increase brain reserve [12, 21]. In the presence of greater brain reserve (e.g., larger brain volumes), one may sustain more structural change before reaching a level of brain atrophy/pathology that correlates with notable cognitive declines or clinically significant symptoms of ADRD [13].
The pathway by which established ADRD biomarkers link education to cognitive functioning is somewhat unclear. Albeit mixed, the existing literature suggests that higher education is associated with larger global and regional (e.g., hippocampal) brain volumes and fewer neurofibrillary tangles (NFTs), particularly in cognitively healthy or preclinical ADRD stages, but education is less likely to be associated with white matter hyperintensity (WMH) volumes [22–26]. Several of these neuroimaging measures, including cerebral atrophy, presence of WMHs, and tau NFTs (measured as tau deposition from positron emission tomography (PET)) are associated with increased risk for cognitive impairment and ADRD [27–32]. NFTs found in Alzheimer’s disease (AD) are known to result from hyperphosphorylation of tau, with the abnormal accumulation of tau resulting in NFTs [33]. NFTs and hyperphosphorylated tau are typically first detected in the transentorhinal region of the mesial temporal lobe (Braak Stage I) [34] and correlate with cognitive symptoms later in the progression of AD [31, 35]. Prior studies related to this topic have been small but suggest that the relationship between education and cognitive function was not mediated by total brain nor hippocampal volumes [6] but other markers of brain structure and function (e.g., white matter integrity) may play a role[36].
Conflicting evidence of an ADRD biomarker pathway may also partially reflect heterogeneity in the cognitive domains assessed across studies or chance due to small and selected samples [6, 37]. The relationships between education and cognitive function may differ by domain as a function of biomarker-specific mediating pathways. Ischemic damage to the cerebrovasculature in the form of WMHs is more strongly associated with speed-dependent measures of perceptual-motor skills and executive functioning than measures of memory [38–41]. In contrast, greater hippocampal atrophy is more strongly associated with declines in episodic memory than in executive functioning [42, 43]. Language abilities are strongly linked to education, and certain tasks (e.g., word reading) may function as a proxy for education [44]. Although some language abilities (e.g., confrontation naming, word reading) are relatively well preserved in healthy aging and typical AD, others (e.g., verbal fluency) are susceptible to declines with age and preclinical ADRD [45, 46]. Therefore, we aimed to investigate whether ADRD biomarkers mediate the relationships between education and three cognitive domains (i.e., memory, executive functioning, and language).
MATERIALS AND METHODS
Study population
We used data from the third cohort of the Alzheimer’s Disease Neuroimaging Initiative (ADNI 3) database, collected from 2016 to 2021. Data were obtained from the ADNI database (http://adni.loni.usc.edu). The ADNI was launched in 2003 as a public-private partnership, led by Principal Investigator Michael W. Weiner, MD. The primary goal of ADNI has been to test whether serial magnetic resonance imaging (MRI), PET, other biological markers, and clinical and neuropsychological assessment can be combined to measure the progression of mild cognitive impairment (MCI) and early AD. For up-to-date information, see http://www.adni-info.org. ADNI 3 is a continuation of the initial ADNI multi-site study. ADNI 3 retained some participants from ADNI 2 and enrolled new participants (ages 55–90 years) including individuals who were cognitively healthy or were living with amnestic MCI or dementia. A complete description of the ADNI 3 study protocol was reported by Weiner and colleagues [47]. For the current study, only baseline data from newly enrolled ADNI 3 participants are used. From a total of 819 newly enrolled ADNI 3 participants who participated in neuroimaging, participants were excluded if they were missing information on education (
Education (exposure)
Years of education was self-reported by the participant or study partner.
Neuroimaging measures (mediators)
The present study used processed neuroimaging values from the ADNI database. Detailed procedure on scan acquisition and optimization are provided elsewhere (www.adni.lon.i.usc.edu/pages/access/studyData.jsp). In brief, participants underwent whole-brain MRI scanning using 3.0- Tesla MRI. Scans of interest were the T1-weighted scan to measure whole and regional brain volumes and to provide spatial information for PET scans. ADNI MRI processing methods, including cortical reconstruction and volumetric segmentation, were based on previously published FreeSurfer techniques [48, 49] using FreeSurfer image analysis (version 5.1; http://surfer.nmr.mgh.harvard.edu/). Regional segmentations (e.g., the hippocampus) were identified according to an atlas defined by FreeSurfer. WMH volumes also required fluid-attenuation inversion recovery (FLAIR) sequences. The WMH measurement approach has been described in detail on the ADNI site (http://adni.loni.usc.edu/pages/access/studyData.jsp). In short, 1) non-brain structures were removed from the 3D T1 images prior to measurement using an automated atlas based method; 2) Preserved parallel relationships of the FLAIR image is then transformed to the 3DT1 image using the FLIRT method from the FSL tool box; 3) nonuniform correction of the 3D T1 is performed; 4) Next, the image is molded using a spatially smooth thin-plate spline interpolation; 5) Prior to WMH calculation, each 3D T1 image is non-linearly aligned to a common template atlas, and each of the accompanying images are transformed onto the same atlas; 6) estimation of WMH is then performed using a modified Bayesian probability structure based on a previously published method of histogram fitting [50]. Raw WMH volumes were log-transformed. Raw gray matter volumes (i.e., total cortical gray and bilateral hippocampal volumes) and log transformed WMH volumes were each adjusted for total brain volume by regressing each imaging measure on total brain volume. Each adjusted imaging measure was then z-standardized to a mean value of 0 and standard deviation of 1.
Participants underwent PET imaging at their initial ADNI 3 visit. Tau PET data was obtained using the ligand [18F]-T807 (AV-1451). ADNI investigators applied a region of interest based approach to measure standard uptake value ratio in areas that represented Braak stages [47]. For the purposes of this study, we examined tau within the meta-temporal region of interest [51].
Cognitive scores (outcomes)
Participants completed a battery of neuropsychological tests and standardized, composite scores for memory, executive functioning, and language were previously developed by investigators using item-response theory methods and confirmatory factor analysis [45, 53]. In brief, measures for the memory composite included the Rey Auditory Verbal Learning Test (RAVLT; including List B, immediate and delayed recalls, and recognition); Alzheimer’s Disease Assessment Scale-Cognitive subscale (ADAS-Cog) list learning, recall, and recognition; Logical Memory immediate and delayed recalls; and three word recall items from the Mini-Mental State Exam (MMSE) [52]. Measures for the executive functioning composite included Wechsler Adult Intelligence Scale (WAIS-R) Digit Symbol Substitution, Digit Span Backwards, Trails Making Test A & B, Category Fluency (Animals and Vegetables), and Clock Drawing [53]. Measures for the language composite included the Boston Naming Test, Animal Fluency, language items from the MMSE (i.e., object naming, sentence repetition, sentence reading, writing, and following a 3-step command), language items from the ADAS-Cog (i.e., commands, object naming, and ideational praxis), and language items from the Montreal Cognitive Assessment (i.e., phonemic fluency and sentence repetition) [45].
Covariates
Covariates considered to be possible confounders of the relationship between education and cognitive function were: age at the ADNI 3 baseline interview (continuous, years) sex/gender (male; female), self-reported race (considered as White or non-White due to insufficient sample sizes of individuals from distinct non-White backgrounds). We conceptualize racial identity as a social construct, developed and maintained to sustain a social hierarchy. It is relevant in this analysis because the historical and ongoing processes of structural racism create substantial race-based inequality in educational access and numerous other exposures that may influence brain aging. We also considered additional possible confounders of the mediator-outcome relationships: depressive symptoms within the past week (continuous score on the 15-item Geriatric Depression Scale), history of cardiovascular disease (a binary indicator of any self-reported cardiovascular disease, hypertension, stroke, or diabetes), body mass index (kg/m2, continuous), and
Statistical analysis
We fit linear regression models to assess the total effect (TE) of education on each cognitive outcome with each domain evaluated in separate models. We then used causal mediation analyses to estimate the neuroimaging measures as mediators between education and each cognitive domain [54, 55]. Each neuroimaging measure was included in separate models to estimate the natural direct effect (NDE) of education not mediated by that neuroimaging measure. The natural indirect effect (NIE) of education on cognitive functioning mediated by each neuroimaging measures was estimated by calculating the TE minus the NDE. We sequentially added covariates to assess each mediator. Model 1 included age, race,
RESULTS
Descriptives
Participants were 73 years of age on average, 53% were women, the majority were White (91%), and highly educated, with 73% of participants completing at least a 4-year college degree (Table 1).
ADNI 3 Descriptive Characteristics
*Non-White participants consisted of 1 American Indian or Alaskan Native (0.19%) participant, 9 Asian (1.68%) participants, 30 Black or African American (5.59%) participants, and 9 multiracial (1.68%) participants. ADNI, Alzheimer’s Disease Neuroimaging Initiative;
Primary analysis
Education was associated with better performance on memory (est = 0.08, 95% CI: 0.06, 0.11), executive functioning (est = 0.10, 95% CI: 0.07, 0.14), and language composites (est = 0.11, 95% CI: 0.08, 0.14) in models adjusting for age, sex/gender, race, and
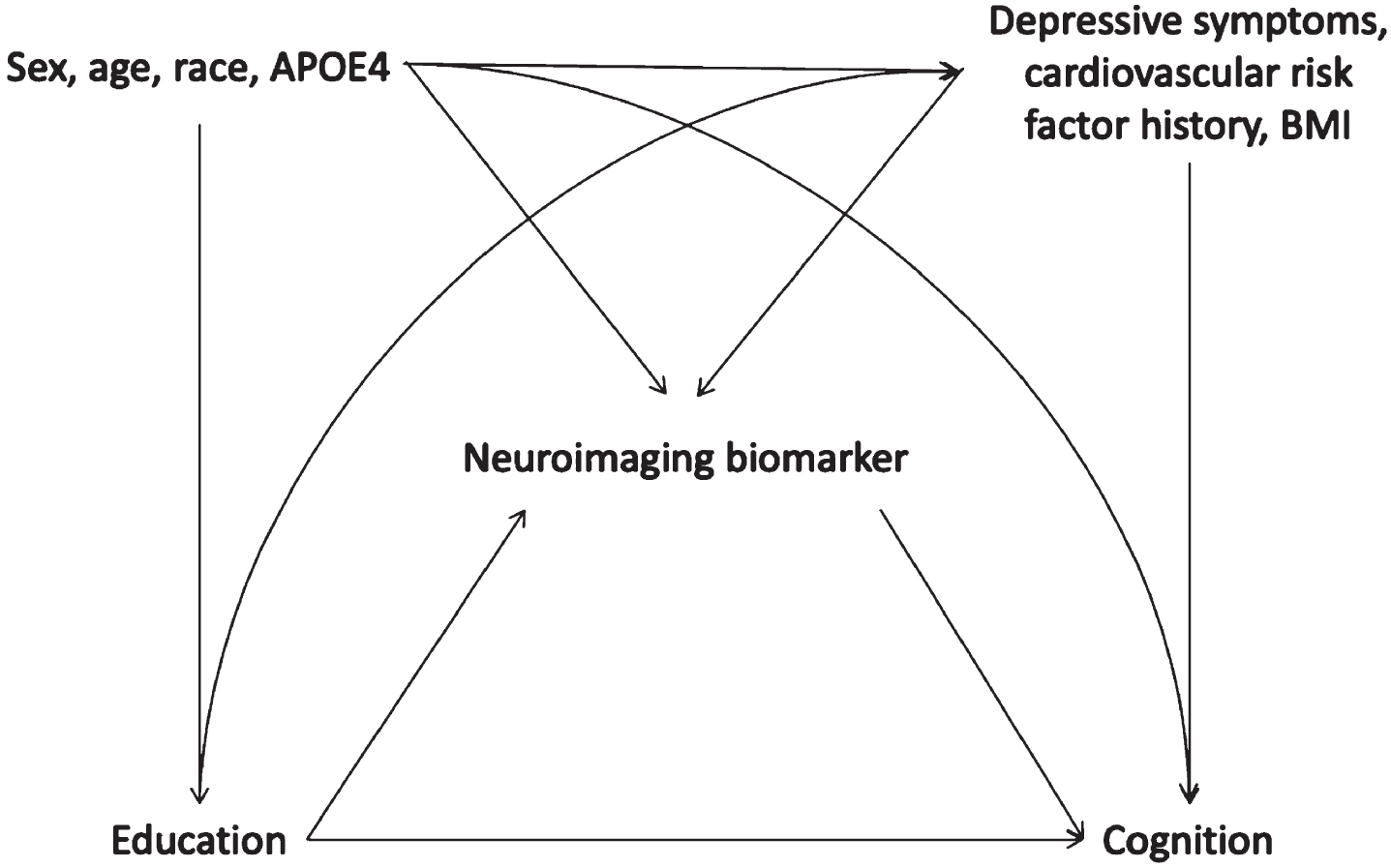
Hypothesized causal structure guiding choice of covariates in models to test mediation of the relationship between education and cognition by neuroimaging biomarkers. APOE4, Apolipoprotein E
In fully adjusted models, hippocampal volume was estimated to mediate 15.7% (95% CI: 3.23, 28.22) of the relationship between education and memory and WMH volume to mediate 9.2% (95% CI: 1.73, 16.72) (Fig. 2 and Supplementary Table 1). Cortical gray matter volume was estimated to mediate 6.3% of the relationship between education and memory but the fully adjusted confidence interval (95% CI: –0.70, 13.27) included zero, consistent with no mediating role for gray matter volume. Likewise, presence of tau was estimated to mediate 14.3% of the relationship between education and memory but the confidence interval (95% CI: –0.30, 28.80) also included zero.
Hippocampal volume was estimated to mediate 14.5% (95% CI: 2.34, 26.64) of the effect of education on executive function and WMH to mediate 9.1% (95% CI: 1.18, 16.95) (Fig. 3 and Supplementary Table 2). Estimated mediation fractions were similar for cortical gray matter (11.9%, 95% CI: –0.24, 24.00) and tau (15.4%, 95% CI: –0.36, 31.21), although both CIs were wide and includedzero.

Direct and indirect effects between education, ADRD biomarkers, and memory in fully adjusted models. Fully adjusted models included the following covariates: age, sex/gender, race, apolipoprotein

Direct and indirect effects between education, ADRD biomarkers, and executive functioning in fully adjusted models. Fully adjusted models included the following covariates: age, sex/gender, race, apolipoprotein

Direct and indirect effects between education, ADRD biomarkers, and language in fully adjusted models. Fully adjusted models included the following covariates: age, sex/gender, race, apolipoprotein
Hippocampal and WMH volumes were estimated to partially mediate the relationship of education with language in fully adjusted models (hippocampus: 11.4%, 95% CI: 1.91, 20.97; WMH: 6.4%, 95% CI: 0.75, 12.10) (Fig. 4 and Supplementary Table 3). Neither cortical gray matter nor tau significantly mediated the relationship between education with executive functioning after accounting for health risk factors in fully adjusted models (cortical gray matter: 8.0%, 95% CI: –0.38, 16.38; tau: 9.8%, 95% CI: –0.74,20.39).
Secondary analyses
DISCUSSION
In the ADNI 3 cohort, higher education was associated with better cognitive functioning, an association partly mediated via larger hippocampal and smaller WMH volumes. Patterns were similar across cognitive domains of memory, executive functioning, and language. Our findings suggest major mechanisms via which education influences later life cognition are not captured in typical neuroimaging measures of brain health. Further, these results underscore the importance of education for later-life brain and cognitive health, suggesting that education continues to play a significant role in preserving brain integrity as one ages. Our findings were robust to control for multiple factors associated with poor brain health, including
Our data support the notion that education may confer brain reserve as evidenced by lower ADRD biomarkers which were associated with better cognition. Given that ADRD biomarkers only partially mediated the relationship between education with cognitive functioning, education may largely operate through additional mechanisms, such as cognitive reserve which should be tested using longitudinal data [12, 13]. Existing literature corroborates a protective effect of education on cognitive function even in the presence of brain atrophy/pathology [16, 58], although findings are inconsistent [18].
Individual ADRD biomarkers were relatively similar in the extent to which they connected education to cognitive functioning with two out of four biomarkers (hippocampal and WMH volumes) reaching significance. Further, accounting for multiple ADRD biomarker pathways resulted in only slight increases in the percent of the relationships between education and each cognitive domain mediated, suggesting more overlap than independence across ADRD biomarkers. The hippocampus has a critical role in memory functioning and, to a lesser extent, has been associated with executive functioning and language [43]. Individuals with more years of education also had larger hippocampal volumes in a population of cognitively normal older adults [59] and across the lifespan [25]. Likewise, extensive training in spatial navigation was associated with larger posterior hippocampus volumes among London Taxi drivers, underscoring the connection between cognitive engagement and temporal lobe structure [60]. Education may trigger a feedback process, in which higher education preserves hippocampal volume and preserved hippocampal integrity promotes continued cognitive engagement. This in turn preserves hippocampal integrity and promotes plasticity.
Small vessel cerebrovascular damage (i.e., WMH volumes) also linked education to cognitive functioning with a nominally greater impact on memory, followed by executive functioning, and then language. Existing literature on WMHs indicates a stronger, negative relationship between white matter hyperintensities and executive functioning relative to memory [38–40]. Parks et al. [61] examined a mediational model of the relationships among WMHs and cognition in older adults and proposed an indirect effect of WMHs on episodic memory, one mediated by executive function and hippocampal volumes. With regards to education, there appears to be a negative effect of WMH on cognition particularity among those with lower education and not those with higher education with the same severity of WMH [62, 63].
Although point estimates suggest that NFTs in the temporal lobe and whole brain cortical gray matter volumes mediated a similar percentage of the association between education with cognition as hippocampal and WMH volumes, these relationships were not statistically significant. Larger samples are needed to precisely estimate the role of NFT and tau in mediating education effects on cognition, although our results rule out that these variables account for a large fraction of the effect.
Notably, when we excluded individuals with dementia, WMH volumes, but not hippocampal volumes, remained a robust mediator of the relationship between education and cognition. It is possible that the individuals with dementia were more likely to have an AD etiology which, when excluded, lowered our ability to detect mediation by hippocampal volumes to a greater extent than WMH volumes. Alternatively, by excluding a cognitive status group (i.e., individuals with dementia), other statistical challenges (e.g., a collider variable) may have been introduced [64]. This is possible given that, 1) higher education is linked to higher likelihood of dementia diagnosis due to greater access to healthcare and research studies, 2) neurobiomarkers are considered in dementia status/diagnosis, and similarly 3) dementia status/diagnosis is defined by cognitive status. Future studies with larger sample sizes are needed to test potential modification by cognitive status and the role of varying etiology (e.g., AD, vascular, mixed) among individuals with dementia.
In the present study, education was directly related with performance in each cognitive domain (memory, executive functioning, and language). Though not formally tested, the direct association of education with cognition was nominally largest for language, followed by executive functioning, then memory. Education may confer the most protection on the language domain which is consistent with other findings in the literature concerning the protective effects of education on cognition via preservation of white matter integrity [65]. However, it is more likely that the brain regions involved in language processing are less susceptible to changes from aging and ADRD pathology [34]. Additionally, education and specific language tests which measure crystallized abilities (e.g., vocabulary/semantic knowledge, a component of the ADNI language composite score) have a strong premorbid relationship. In fact, such crystallized language measures are often used to operationalize education and language [66]. Therefore, longitudinal data, separate analyses by cognitive status, and/or data breaking down the relationship between education with each language test (i.e., separating crystalized from fluency measures) would be necessary to clarify whether language abilities are uniquely protected by education in the face of ADRD pathology.
Limitations
ADNI 3 is a non-representative, highly educated sample of individuals who nearly all identify as White race/ethnicity. Generalizability to more diverse samples is unclear [67–69]. Future studies should incorporate more diverse samples. Educational attainment may be confounded by other social and economic factors that were not accounted for in this study. Multiple social factors such as low economic stability, lack of health care access, and increased stress are proposed to interact with one another in producing limited psychosocial resources or allostatic load [70], ultimately affecting later-life cognition and brain integrity [71, 72]. The ADRD biomarker mediation estimates in our study were relatively small. Given this limitation, we were also unable to conduct sensitivity analysis by other relevant demographic factors, such as sex/gender which may modify the associations between education, ADRD biomarkers, and cognition [73]. Finally, we examined cognition at one timepoint, so we are unable to determine whether ADRD biomarkers mediate the associations between education with rates of cognitive decline or conversion to dementia. Despite these limitations, our study has a number of strengths, including examination of a relatively large sample with data on multiple ADRD biomarkers derived from multimodal neuroimaging data. Additionally, cognitive outcomes were well-characterized with each cognitive domain reflecting a composite score; thus, improving stability and reducing the likelihood of a type I error [52]. Finally, our use of mediation analytic methods goes beyond simple regression to explain potential pathways by which education impacts late life cognition.
Conclusion
Among a predominantly White sample of middle-aged and older adults in the US, higher education was associated with better memory, executive functioning, and language domains. These relationships were partially attributable to larger hippocampal and smaller WMH volumes among those with higher education. Hippocampal volumes were a consistent mediator of the relationships between education with cognitive function across multiple domains. Our findings warrant further investigation into the relationships between education, ADRD biomarkers, and later-life cognitive functioning in more diverse and population-based cohorts.
Footnotes
ACKNOWLEDGMENTS
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the NIA, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.;Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
FUNDING
This work is supported by Advanced Psychometrics conference (NIA grant R13 AG030995). PAA is supported by funds from the Alzheimer’s Diseases and Related Dementias T32 training fellowships (NIH/NIA grant 5T32AG061892-04, T32AG020499-18). AMS is supported by funds from the NIH (K08AG075351, L30AG074401, U54CA267789). RC is supported in part by the NIA (4K00AG068431-02). CWE is supported by the NIA (F31AG062114).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
