Abstract
Background:
Trisomy 21 causes Down syndrome (DS) and is a recognized cause of early-onset Alzheimer’s disease (AD).
Objective:
The current study sought to determine if premorbid intellectual disability level (ID) was associated with variability in age-trajectories of AD biomarkers and cognitive impairments. General linear mixed models compared the age-trajectory of the AD biomarkers PET Aβ and tau and cognitive decline across premorbid ID levels (mild, moderate, and severe/profound), in models controlling trisomy type,
Methods:
Analyses involved adults with DS from the Alzheimer’s Biomarkers Consortium-Down Syndrome. Participants completed measures of memory, mental status, and visuospatial ability. Premorbid ID level was based on IQ or mental age scores prior to dementia concerns. PET was acquired using [11C] PiB for Aβ, and [18F] AV-1451 for tau.
Results:
Cognitive data was available for 361 participants with a mean age of 45.22 (SD = 9.92) and PET biomarker data was available for 154 participants. There was not a significant effect of premorbid ID level by age on cognitive outcomes. There was not a significant effect of premorbid ID by age on PET Aβ or on tau PET. There was not a significant difference in age at time of study visit of those with mild cognitive impairment-DS or dementia by premorbid ID level.
Conclusion:
Findings provide robust evidence of a similar time course in AD trajectory across premorbid ID levels, laying the groundwork for the inclusion of individuals with DS with a variety of IQ levels in clinical AD trials.
INTRODUCTION
Down syndrome (DS), which is caused by trisomy 21, is the most common known genetic cause of intellectual disability [1]. Trisomy 21 is also recognized as a genetic cause of Alzheimer’s disease (AD) [2, 3], with a 90% or higher lifetime incidence of AD dementia [4–6]. The triplication of the amyloid precursor protein, located on chromosome 21, leads to an overexpression of amyloid-beta (Aβ), which accumulates over time into extracellular Aβ plaques, an early pathologic feature of AD. These plaques are followed by the formation of intracellular neurofibrillary tangles of the protein tau [2, 7]. Natural history studies are documenting the timeline of AD biomarkers and clinical AD dementia in large DS cohorts [4, 8]. In positron emission tomography (PET) studies, 40–60% of individuals with DS have elevated Aβ across various cortical regions in their early- to mid-40 s [9] and the estimated age of onset of AD dementia is 53.8 years [10]. However, there is marked variability in these data: elevated PET cortical Aβ is present in many individuals with DS before the mid-30 s [9] and age of onset of AD dementia spans 32 years in DS [5, 10]. As clinical trials become available to people with DS field, it is critical to identify factors that influence this individual variability. Premorbid intellectual disability (ID) may be one of these factors.
People with DS vary in ID level prior to onset of AD (referred to as premorbid ID), with IQ scores ranging from the 30 s to 70 s [11–13]. The majority of adults with DS (75–80%) have mild or moderate ID; however, 2–5% of individuals with DS have an IQ in the borderline to normal IQ range and 15–25% have severe to profound ID [11–13]. In part, heterogeneity in premorbid ID is tied to type of trisomy 21, with mosaicism (2–3% of DS cases) associated with a higher IQ than the full (96% of DS cases) or translocation (1–2% of DS cases) types [14, 15]; however, other factors such as co-occurring medical conditions are also linked to IQ variability [16]. It is not known if differences in premorbid ID alter the trajectory of AD in DS. These effects could operate through biological mechanisms related to brain reserve, which is the idea that neural resources that contribute to differences in premorbid ID level may alter the ability to compensate for early AD pathology [17]. For example, in non-DS populations, higher (versus lower) intracranial volume appears to protect against cognitive decline in the presence of early AD pathology [18, 19]. Alternatively, variability in premorbid ID may impact the trajectory of AD in DS through cognitive reserve, or the concept that those with higher premorbid intelligence can better recruit alternate neural networks or more efficiently use existing networks to cope with early AD-related pathology [20]. Indeed, there is evidence outside of DS that adults with higher education have more pronounced levels of AD pathology before showing the clinical AD symptomology relative to adults with lower education, and thus maintained cognitive functioning for a longer period of time in the presence of early pathology [18, 22]. In line with these findings, it is possible that adults with DS with a higher premorbid ID level would have a later age of onset of clinical AD symptomology, despite a similar time course of AD pathology, relative to those with a lower premorbid ID level.
To date, only a handful of large-sized studies have compared the trajectory of AD by premorbid ID level in DS [4, 24]. These studies were limited in that they only stratified by mild versus moderate ID (excluded those with severe/profound ID), examined differences on only one or two cognitive measures, and analyses did not control for type of trisomy (full, mosaic, or translation) or apolipoprotein E (
The current study drew on a large cross-sectional cohort of adults with DS to compare the estimated age-trajectory of AD biomarkers of PET Aβ and tau PET and cognitive decline (across four instruments) between three premorbid ID levels (mild, moderate, and severe/profound) in models controlling trisomy type and
METHODS
Participants
Analyses involved cognitive data on adults with DS, a subset of whom also had brain imaging data. Data was collected at baseline from eight sites involved in the Alzheimer’s Biomarkers Consortium-Down Syndrome (ABC-DS). Study inclusion criteria included aged 25 years or older, karyotyping testing confirming DS, the absence of conditions contraindicative for brain imaging scans (e.g., pregnant or metal in the body), and the absence of non-AD untreated medical or psychiatric conditions that altered cognitive functioning. In addition, for the current analyses, a documented IQ or mental age score prior to any AD-related concerns was required. Adults with DS who had a clinical AD status of ‘unable to determine’ were also removed from analyses. Internal Review Boards approved the study, and consent and assent were obtained prior to participation.
Procedures
Data included in present analyses was part of a larger study protocol completed across multiple days. Participants completed a battery of directly-administered cognitive measures and a blood draw for karyotyping. Caregivers provided information on the adult with DS’s medical history and their cognitive, adaptive, and behavioral functioning. At four sites, PET and MRI imaging were conducted on the adults with DS. A physical and neurological exam were also completed at all sites with the adult with DS.
Measures
Socio-demographics
Date of birth was reported by the caregiver and used to calculate age in years. Karyotyping was used to evaluate
Premorbid ID level
Premorbid ID was defined as intellectual disability level prior to a clinical status of MCI-DS or AD dementia and was based on scores from the Stanford-Binet Intelligence Scales, Fifth Edition (SB5) [27], Abbreviated Battery or medical records documenting IQ using the Wechsler Adult Intelligence Scale [28], or Kaufman Brief Intelligence Test [29]. Premorbid ID level was coded as mild (1), moderate (2), or severe/profound (3) based on IQ standard scores (borderline: 70–85; mild: 50–69, moderate: 35–49, and severe/profound: <35) or age equivalent scores (borderline: >15 years; mild: 9–14 years, moderate: 4–8 years, and severe/profound: ≤3 years). For participants with a clinical status of MCI-DS or AD dementia, a prior IQ or mental age equivalent score, taken before AD-related concerns, wasused.
Clinical AD status
Clinical AD status was based on a case consensus process that involved at least three staff with expertise in AD in DS (including at least one psychologist and physician) who were blind to imaging/biofluid data. This process reviewed information from the current study visit and any prior visits from caregiver-reported and direct measures of cognitive functioning, adaptive behavior, and maladaptive behaviors, a neurological exam, medical and psychiatric history and recent life events [8]. Clinical AD statuses were: 0 = cognitively
Cognitive function
Four cognitive instruments were selected from a larger battery because they had assessed a cognitive domain shown in prior work [30–32] to decline early on in AD progression in DS.
MRI and PET
PET data were acquired using [11C] PiB for Aβ and [18F] AV-1451 for tau quantification, and MRI was used for anatomical reference. Scanning was performed on Siemens HR+, Siemens 4-ring Biograph mCT or GE Signa PET/MR (see [6] for details). Tracers were administered as 20–30 s bolus injections and saline flush. Images were acquired in 5-min frames and inspected and corrected for motion on a frame-by-frame basis. Time-averaged images were 50–70 min post injection for [11C] PiB and 80–100 min for [18F] AV-1451. Data were reconstructed using iterative methods and corrected for deadtime, attenuation, scatter, and radioactivedecay.
[11C] PiB and [18F] PET processing
[11C] PiB PET scans and associated T1 MRI were analyzed with the centiloid method [40] using SPM8 software. The 50–70-min PET images were registered to corresponding anatomical MR images. The MR scan was deformed to match the 152-subject template of the Montreal Neurological Institute (MN152) included with SPM8 and corresponding PET images were co-warped using the determined parameters. PiB radioactivity concentration was extracted for the centiloid standard global region and whole cerebellum, defined on the MNI152 template. Global SUVR was the ratio of tracer concentration in the global region to that of whole cerebellum. This tissue ratio was converted to centiloid values using linear+constant transformation specified for [11C] PiB [40].
The 80–100 min [18F] AV-1451 tau images were registered to T1 MRI. The next step was typically to parcellate the T1 MR images to determine regions of interest for the PET quantitation. However, the DS population presents analysis challenges due to anatomical differences between DS subjects and the neurotypical subjects used in the construction of FreeSurfer templates and also due to motion artifacts often seen with MR scans of DS subjects. Thus, an extended methodology (multi-template method) was employed. First, Subject T1 MR scans were processed through FreeSurfer 5.3 [41] resulting in each being parcellated into regions defined by the Desikan-Killiany (DK) atlas [41]. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Results were then inspected, and 12 high-quality scans were selected for use as templates, as part of the extended analysis. In most cases the FreeSurfer parcellations were edited to provide better conformation of the FS-based atlas to the template anatomy.
For each subject, each of the 12 templates was warped to the subject’s T1 MR using the Advanced Neuroimaging Tools (ANTs) software package [42, 43] resulting in 12 versions of the DK atlas in subject space. Each subject-space voxel was labeled with a final DK region name which was taken to be the region most often chosen by the 12 atlases. All results were accepted or rejected based on a visual rating of the final atlas adherence to subject MR anatomy. The object of this method is to produce high quality parcellations without the need for parcellation editing. In a few cases it was found that the previously performed direct application of FreeSurfer, i.e., not the multi-template method, along with previously performed editing, produced acceptable parcellations whereas the multi-template method did not. In such cases the direct FreeSurfer parcellations were used.
Radioactivity concentration of regions of interest of [18F] AV-1451 was expressed as SUVr and was determined using volume weighted average of tracer concentration within FS-based components reproducing the composite region defined by Jack et al. [44] divided by the cerebellar cortex concentration. The Aβ centiloid value and tau Mayo-composite SUVr were used to classify participants as Aβ±(threshold value 19) and tau±(threshold value 1.21) [44].
Statistical analysis
To examine variable distribution and potential outliers, boxplots, histograms, and descriptive statistics were used. In addition, Loess plots conducted in R 4.2.0 were used to visualize a smoother scatter plot of the relationship between age, cognitive performance, and AD biomarkers by premorbid ID group. We conducted general linear mixed models in SAS 9.4 to examine the associations between age and premorbid ID level, as well as their interactions, on cognitive functioning and AD biomarkers of PET Aβ and tau PET. A mixed model was conducted as site was included as a random effect; all other variables were added as fixed effects. The linear and non-linear effects of age and age by premorbid ID were explored in all models. Biological sex, trisomy type,
RESULTS
Preliminary analyses
Table 1 presents information on the sample socio-demographics. Of the 388 participants who met inclusion criteria for the larger ABC-DS study, 8 (2.1%) did not have an IQ or mental age score prior to AD-related concerns, and 19 (4.9%) had a clinical AD status of ‘unable to determine’. The remaining 361participants ranged in age from 25 to 81 years, with a mean of 45.19 years (
Participant characteristics and mean and standard deviation for study variables
M, mean; SD, standard deviation; ID, intellectual disability; CS, cognitively stable; MCI-DS, mild cognitive impairment-Down syndrome; mCRT, modified Cued Recall Test; DSMSE, Down Syndrome Mental Status Examination; DLD, The Dementia Questionnaire for People with Learning Disabilities. PET Aβ is in centiloids. Tau PET is in standard uptake value ratio.
Premorbid ID and cognitive functioning
Table 2 shows the results of the mixed general linear mixed models examining the effects of age and premorbid ID level, and their interaction, on cognitive performance in models that controlled for clinical AD status, biological sex, trisomy karyotype,
General linear mixed models of premorbid intellectual disability (ID) level on cognitive outcomes
ID, intellectual disability; CS, cognitively stable; MCI-DS, mild cognitive impairment-Down syndrome; AD, Alzheimer’s disease; DSMSE, Down Syndrome Mental Status Examination; DLD, The Dementia Questionnaire for People with Learning Disabilities. Age2 was not significantly related to Block Design (–0.045 [0.657]), mCRT (–0.012[(0.09]), DSMSE (0.003 [0.002]), DLD Social (0.033 [0.029]) or DLD Cognitive (0.002 [0.002]). Age2×Premorbid ID was not significantly related to Block Design (F = 0.087,
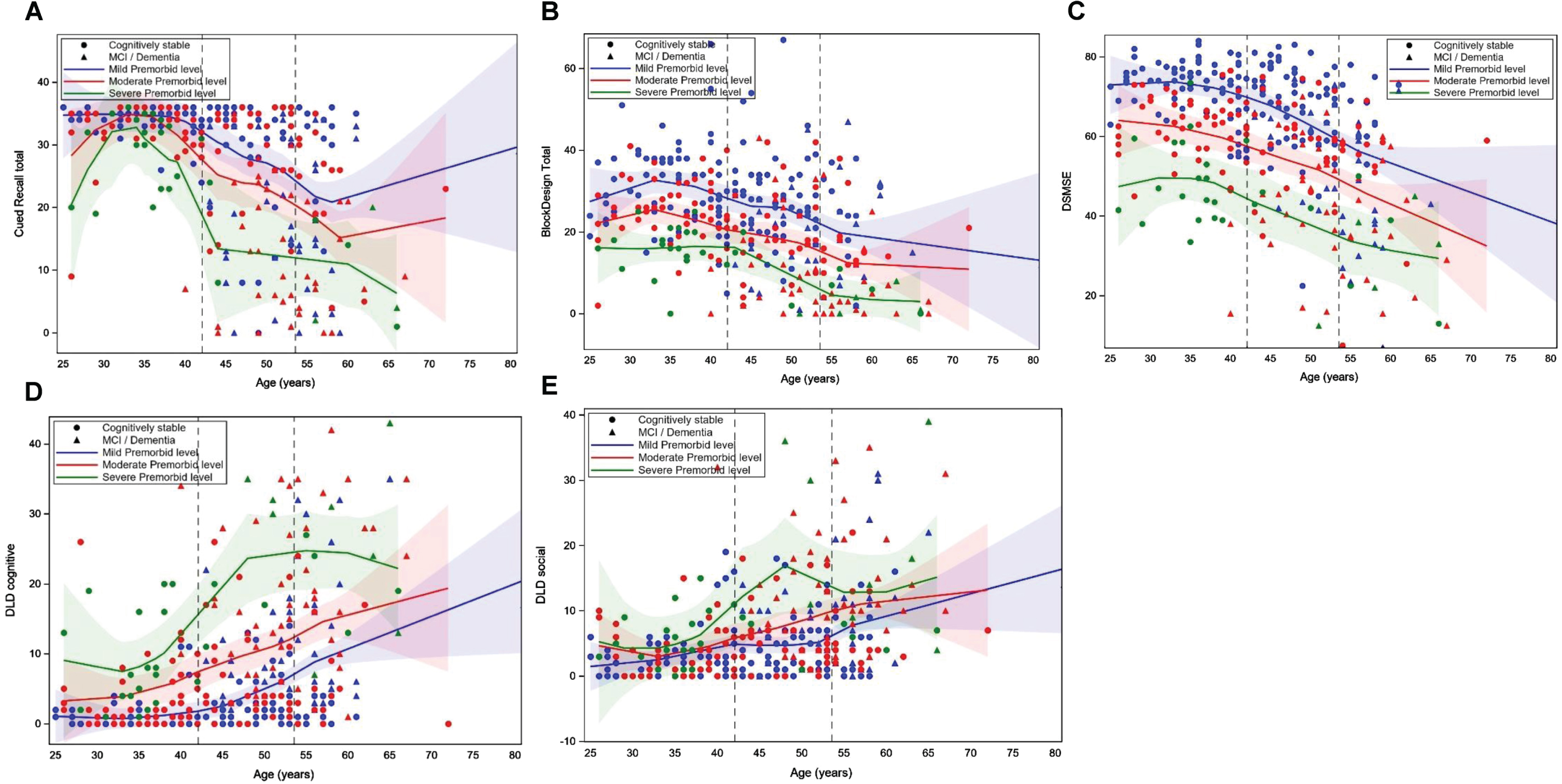
Loess scatterplots of the association between age and performance on the modified Cued Recall Test (A), Block Design Tasks (B), Down Syndrome Mental Status Exam (DSMSE) (C), Dementia Questionnaire for People with Learning Disorders (DLD) sum of cognitive score (D) and DLD sum of social score (E) by premorbid level of intellectual disability. The first dashed vertical line is the mean age of the cognitively stable group and the second line is the mean age of those with mild cognitive impairment (MCI) or dementia.
Premorbid ID and AD biomarkers
Table 3 shows the results of the general linear mixed model regressions examining the effect of age and premorbid ID level, and their interaction, on PET Aβ and tau PET in models controlling for the same set of control variables as above. The quadradic effect of age on cognitive performance was also tested. However, there was not a significant age2 or age2 by premorbid ID effect (see Table 2 footnote); thus, only the linear effect of age is included in final models. There was a significant positive effect of age on both PET Aβ (1.622 [
General linear mixed models of premorbid intellectual disability (ID) level on PET Biomarkers
ID, intellectual disability; CS, cognitively stable; MCI-DS, mild cognitive impairment-Down syndrome; Aβ, amyloid-beta assessed in centiloids. Age2 was not significantly related to Aβ (0.099 [0.067]) or Tau (0.003 [0.002]). Age2×Premorbid ID was not significantly related to Aβ (F = 0.134,
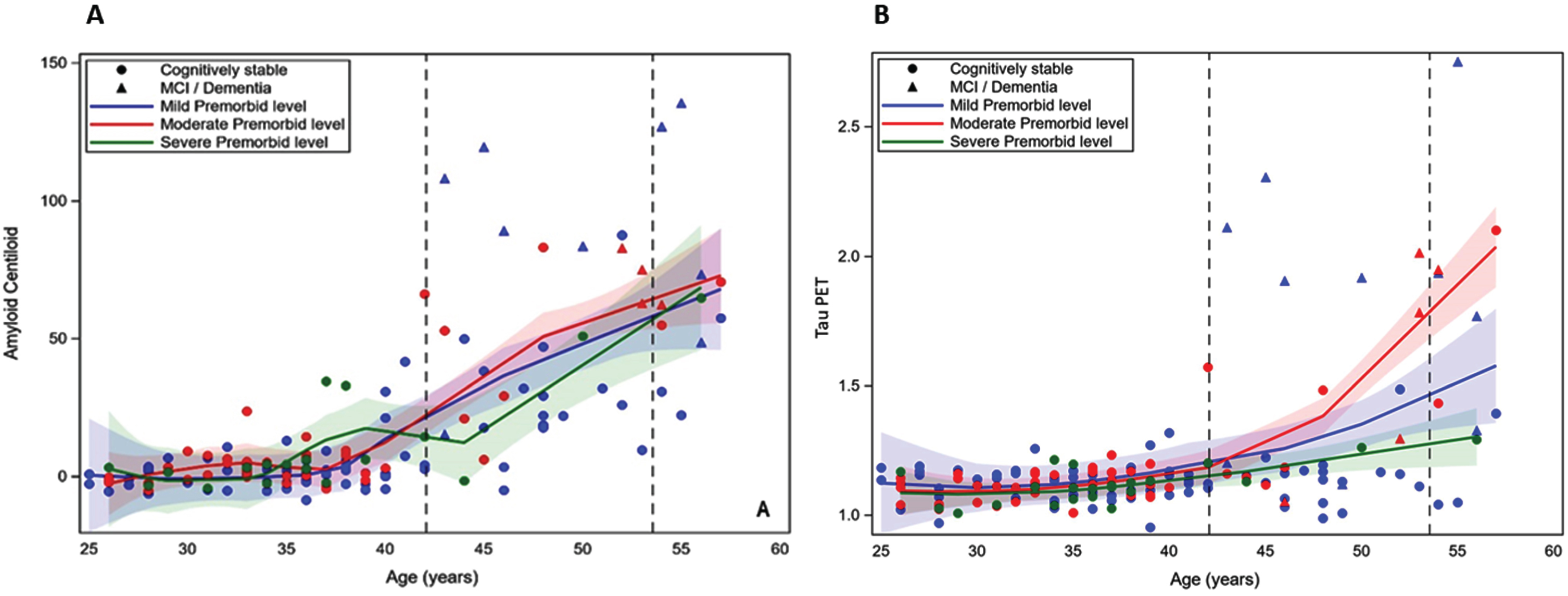
A and B. Loess scatterplots of the association between age and PET amyloid-beta (centiloids) and tau PET by premorbid level of intellectual disability. The first dashed vertical line is the mean age of the cognitively stable group and the second line is the mean age of those with mild cognitive impairment (MCI) or dementia.
Premorbid ID and MCI-DS and dementia
Table 4 displays the results of the general linear mixed models examining the interaction of premorbid ID level by clinical AD status (MCI-DS and AD dementia) on age in models controlling for biological sex, trisomy karyotype, APOE
General linear mixed models of premorbid intellectual disability (ID) level and clinical Alzheimer’s disease Status and their interaction on age
ID, intellectual disability; CS, cognitively stable; MCI-DS, mild cognitive impairment-Down syndrome; Aβ, amyloid-beta assessed in centiloids. *
DISCUSSION
As we prepare for AD clinical trials in DS, it is essential to understand whether phenotypic variability, including premorbid ID level, is associated with differences in AD progression. Findings from the present study provide the strongest evidence to-date that the timing of AD is similar across premorbid ID levels in DS. The age-trajectory of PET Aβ and tau PET was similar in adults with DS with mild, moderate, and severe/profound premorbid ID based on our cohort of adults with DS. Across all premorbid ID levels, PET Aβ increases occurred in the late 30 s, and tau PET increases occurred in the 40 s, which is a decade prior to the expected average age of transition to MCI-DS and AD dementia in DS, in line with studies on autosomal dominant AD [46] and previous studies on DS [2, 47]. The age-trajectory of early cognitive decline on tests of episodic memory, visuospatial ability, and overall dementia symptoms was also similar across premorbid ID groups. Despite the groups having different baseline performance levels, adults with mild, moderate, and severe/profound premorbid ID demonstrated a similar age trajectory of cognitive declines. The average age at time of study visit of adults with DS diagnosed with MCI-DS and AD dementia also did not differ by premorbid ID level, occurring in the early to mid-50 s, respectively. These mean ages are in line with prior work showing a mean age of AD dementia onset across studies of 53.8 years [10].
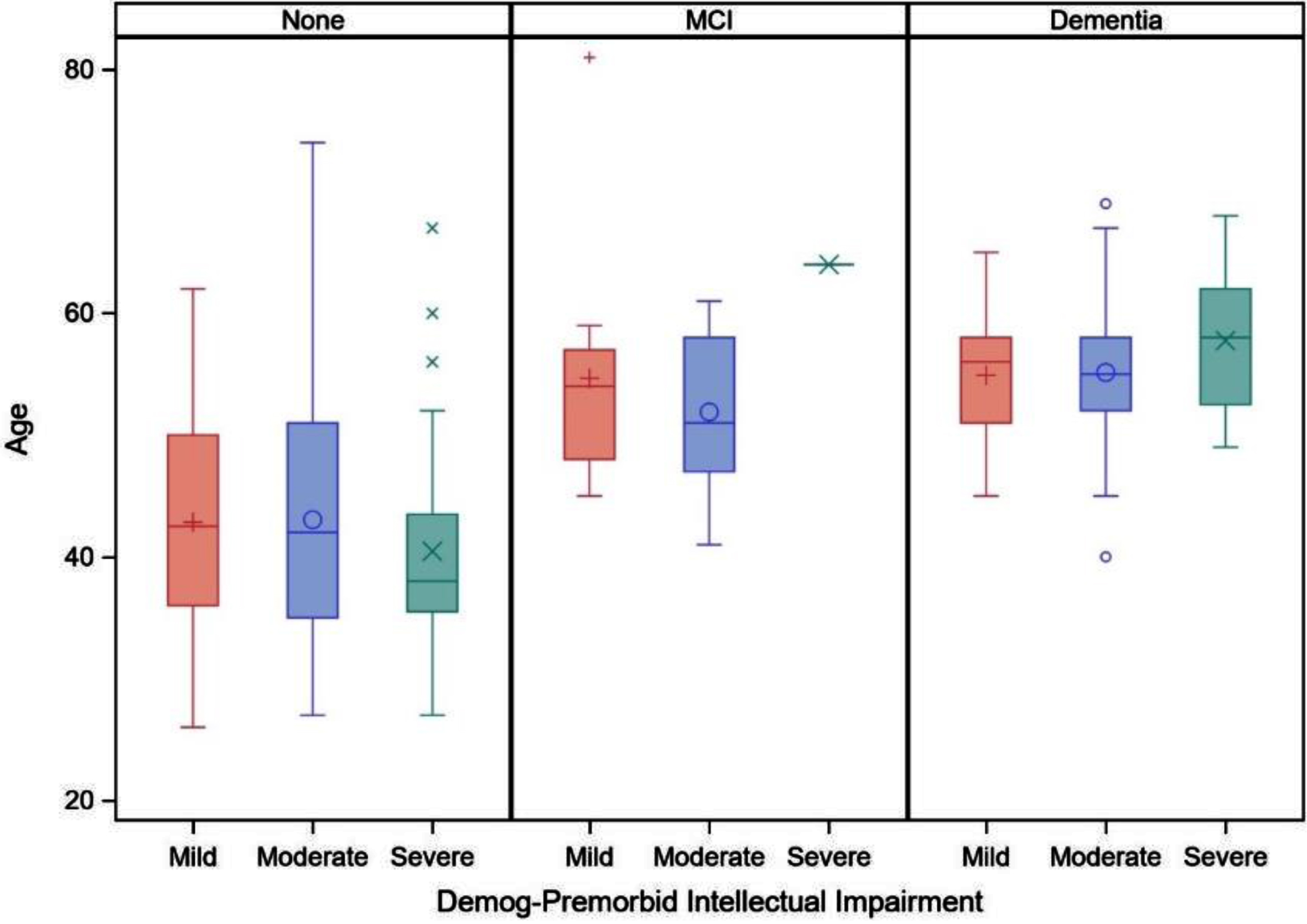
Boxplot of age on clinical status (cognitively stable [none], mild cognitive impairment [MCI], and dementia) by premorbid level of intellectual disability (mild, moderate, or severe/profound).
These findings are aligned with those from another large cohort of adults with DS, which reported a similar rate of decline in AD-related cognitive functioning for adults with mild versus moderate premorbid ID using the CAMCOG-DS and mCRT [24]. These findings are also in line with studies reporting no difference in age of dementia onset based on premorbid ID level in samples of people with ID due to etiologies other than DS [48]. Overall, these findings suggest that the time course of AD is comparable across premorbid ID levels in DS. Thus, AD clinical trials should not exclude individuals with DS based on premorbid ID level; rather, projected age trajectories of AD progression such provide sufficient inclusion criteria for participation in clinical trials. While premorbid ID level may not confer cognitive or brain reserve effects in people with DS— given that the time course of AD progression did not vary based on premorbid ID level— it is still possible that brain or cognitive reserve effects occur in this population. Indeed, there is some evidence that engagement in cognitively-stimulating leisure activities and employment complexity, which vary widely even among those with the same level of ID, are associated with less decline in cognitive functioning with age among individuals with DS [49, 50].
Results should be interpreted with consideration of study strengths and limitations. In terms of strengths, the study leveraged a large cohort of adults with DS. The focus was on cognitive measures with adequate testing properties across premorbid ID levels, and, thus, floor effects were limited. However, it is still possible that these floor effects, especially on the mCRT, obscured accurate detection of age-trajectories of decline for the severe/profound premorbid ID group, as has been reported by other groups [24]. In terms of other limitations, modeling of the timing of AD progression was based on cross-sectional data. Future within-person longitudinal data is needed to validate estimated trajectories in AD biomarkers and cognitive decline and better understand within-person declines. While the mean age of participants in the MCI-DS and dementia status groups at the time of the study visit did not differ by premorbid ID level, it is still possible that the age of initial transition to MCI-DS or dementia does. The sample included a relatively small number of individuals with severe/profound premorbid ID, and the inclusion criteria necessary for PET imaging restricted the participation of adults with DS with certain medical conditions. As a result, our sample may reflect healthier and higher functioning folks within each premorbid ID level relative to the broader DS population. The sample was also primarily white and non-Hispanic, and future work should make concerted efforts to recruit diverse samples. In the current study, clinical AD status groups (MCI-DS and AD dementia) focused on the presence of cognitive and/or functional decline from a baseline level. Behavioral changes often occurred alongside of the cognitive and functional changes, and thus potential differences in behavioral AD symptoms by premorbid ID level should be examined in future studies. Finally, our visualizations of age-trajectories suggest possible increases in cognitive performance and/or practice effects on some measures (e.g., mCRT) in early adulthood (20 s and early 30 s), which should be evaluated in the future.
Overall, the study adds to the characterization of the time course of AD pathology and clinical symptomology in DS. Understanding this progression across the varying phenotypic presentation of DS is essential for anticipated AD clinical trials in the DS population. The present study provides robust evidence of a similar time course in AD progression across mild, moderate, and severe/profound premorbid ID levels in DS. Importantly, these findings lay the groundwork for the inclusion of individuals with DS with a variety of IQ levels in clinical AD trials. Finally, while evidence suggests that the timing of AD progression does not differ by premorbid ID level, it is still possible that lifestyle factors such as employment or engagement in cognitively stimulating leisure activities, which vary widely among individuals with DS, effects AD progression which may not necessarily align with premorbid ID level among individuals with DS.
Footnotes
ACKNOWLEDGMENTS
The authors thank the adults with Down syndrome volunteering as participates in this study for their invaluable contributions to this work, along with their service providers and families. We also thank the broader ABC-DS research team.
FUNDING
Data collection and sharing for this project was supported by The Alzheimer Disease Biomarker Consortium on Down Syndrome (ABC-DS). Funding was received from the National Institute on Aging and the Eunice Kennedy Shriver National Institute of Child Health and Human Development (UO1 AG051406, UO1 AG051412, R01AG070028, P50HD105353, and T32HD007489).
CONFLICT OF INTEREST
GE Healthcare holds a license agreement with the University of Pittsburgh for the [11C]PiB PET technology involved in this study. William Klunk is a co-inventor of [11C]PiB and has financial interest in this license agreement. GE Healthcare did not provide financial support for this study nor did it have a role in designing the study or interpreting the results. Elizabeth Head is an Editorial Board Member of this journal but was not involved in the peer-review process now had access to any information regarding its peer-review.
