Abstract
Background:
The tau protein phosphorylated at Thr181 (p-tau181) in cerebrospinal fluid and blood is a sensitive biomarker for Alzheimer’s disease (AD). Increased p-tau181 levels correlate well with amyloid-β (Aβ) pathology and precede neurofibrillary tangle formation in the early stage of AD; however, the relationship between p-tau181 and Aβ-mediated pathology is less well understood. We recently reported that p-tau181 represents axonal abnormalities in mice with Aβ pathology (
Objective:
The main purpose of this study is to differentiate neuronal subtype(s) and elucidate damage associated with p-tau181-positive axons by immunohistochemical analysis of
Methods:
Colocalization between p-tau181 and (1) unmyelinated axons positive for vesicular acetylcholine transporter or norepinephrine transporter and (2) myelinated axons positive for vesicular glutamate transporter, vesicular GABA transporter, or parvalbumin in the brains of 24-month-old
Results:
Unmyelinated axons of cholinergic or noradrenergic neurons did not overlap with p-tau181. By contrast, p-tau181 signals colocalized with myelinated axons of parvalbumin-positive GABAergic interneurons but not of glutamatergic neurons. Interestingly, the density of unmyelinated axons was significantly decreased in
Conclusion:
This study demonstrates that p-tau181 signals colocalize with axons of parvalbumin-positive GABAergic interneurons with disrupted myelin sheaths in the brains of a mouse model of Aβ pathology.
INTRODUCTION
Phospho-tau proteins have been reported as promising fluid biomarkers for Alzheimer’s disease (AD). Increases in the levels of tau phosphorylated at Thr181 (p-tau181), Thr217 (p-tau217), and Thr231 (p-tau231) in cerebrospinal fluid (CSF) and plasma correlate well with amyloid-positron emission tomography (PET) positivity [1, 2] and detect AD during the preclinical period with high accuracy [3–8]. Moreover, a study in a mouse model of familial AD has shown that extracellular amyloid-β (Aβ) pathology is sufficient to increase p-tau181 and p-tau217 levels in CSF in the absence of tau tangles [9]. However, relationships between these p-tau proteins and Aβ-mediated brain pathology are less well understood.
By utilizing the
Widespread brain regions and several neurotransmitter systems are affected in AD brains. For example, aberrant neurotransmission and neurodegeneration in the cholinergic system are observed in the early stage of AD [11, 12] and have been a therapeutic target for improving clinical symptoms [13, 14]. The nucleus basalis of Meynert (NBM) is a major source of cortical cholinergic innervation with long and thin unmyelinated axons [15]. In the prodromal stage of AD, reduced cholinergic projections and tau pathology have been observed [16–20]. Monoaminergic neurons, including the noradrenergic system, are also affected in the early stage of AD, which is thought to cause cognitive deficits. Noradrenergic neurons in the locus coeruleus extend their unmyelinated axons throughout the cortex [21–24] and are reported to degenerate and develop tau pathology in the early stage of AD [25–29]. These reports suggest that p-tau181 signals might be localized to unmyelinated axons.
In addition to unmyelinated neurons in subcortical and brainstem regions, excitatory-inhibitory imbalance in myelinated neurons in the cortex has been linked to cognitive decline in early AD [30]. During AD progression, neuronal hyperactivity is hypothesized to be induced by Aβ pathology, followed by neuronal loss. And altered GABAergic transmission [31], especially fast-spiking parvalbumin-expressing interneurons, is suggested to play a role in this network dysfunction [32, 33]. Several studies have also shown that glutamatergic and GABAergic synapses are damaged by Aβ accumulation in the brains of AD patients [34–37]. These reports suggest that p-tau181 signals might localize to myelinated glutamatergic and/or GABAergic axons.
To elucidate from which neuronal subtype(s) p-tau181-positive axons originate, and the type of axonal damage induced by Aβ pathology, we utilized an
MATERIALS AND METHODS
Animals
Experiments were performed using 6- or 24-month-old male C57BL/6J and
Tissue preparation
All animals were deeply anesthetized by intraperitoneal administration of a combination of medetomidine (0.3 mg/kg), midazolam (4.0 mg/kg), and butorphanol (5.0 mg/kg), and whole brains were collected and processed for cryosectioning, as previously described [10].
Immunohistochemistry
Immunohistochemical staining was performed as described previously [10]. The primary and secondary antibodies used in this study were listed in Supplementary Table 1. For nuclear staining, 4′,6-diamidino-2-phenylindole (DAPI) or propidium iodide (PI) were used in an appropriate combination. For detection of Aβ amyloidosis, 1-fluoro-2,5-bis (3-carboxy-4-hydroxystyryl) benzene (FSB) was used.
Image acquisition and analysis
As described previously [10, 40], images were acquired using either a LSM700 or LSM780 confocal laser-scanning microscope (Carl Zeiss, Germany) fitted with 20× or 63× objectives. All image processing and analyses were performed with Fiji software using appropriate plugin.
Measurement of coverage with vesicular acetylcholine transporter (VAChT)- and myelin basic protein (MBP)-positive fibers
Procedures for measuring coverage with the fibers from the acquired images were described previously [10, 40]. We captured images using a LSM700 confocal microscopy with a 20× objective, and 10μm Z-stacks (0.85μm interval between images) were reconstructed with a maximum intensity projection. Immunoreactivities for VAChT or MBP were defined as numbers of signal-positive pixels within an entire image, which were determined by thresholding with an automated method (Li’s method for VAChT and Otsu’s method for MBP).
Counting of choline acetyltransferase (ChAT)-positive cell in the NBM
For counting number of ChAT-positive cells in the NBM, we captured images using a LSM780 confocal microscopy with a 20× objective and reconstructed 25μm Z-stacks (2.88μm interval between images) with a maximum intensity projection. We chose the coronal sections including the NBM evenly through rostral to caudal portions (– 0.34 to – 0.94 mm from bregma) and counted ChAT-positive cell number according to the procedure previously described [10, 40].
Measurement of immunoreactivity for p-tau181, vesicular glutamate transporter 1 (VGLUT1) and vesicular GABA transporter (VGAT)
Procedures for measuring immunoreactivity from the acquired images were described previously [10, 40]. For acquisition of immunofluorescence for p-tau181, VGLUT1, and VGAT, we captured images using a LSM700 confocal microscopy with a 20× objective, and reconstructed 10μm Z-stacks (0.85μm interval between images) with a maximum intensity projection. Fluorescent intensity from each signal was evaluated and expressed as relative percentage to WT mice.
Statistical analysis
All statistical analyses were conducted using GraphPad Prism 9 (GraphPad software) as previously described [10]. One-way ANOVA followed by the Tukey’s
RESULTS
p-tau181-positive axons do not overlap with unmyelinated cholinergic or noradrenergic axons in mouse brains
We recently reported that p-tau181 signals were localized to neuronal axons in WT mice [10], and that these structures were disrupted in
The
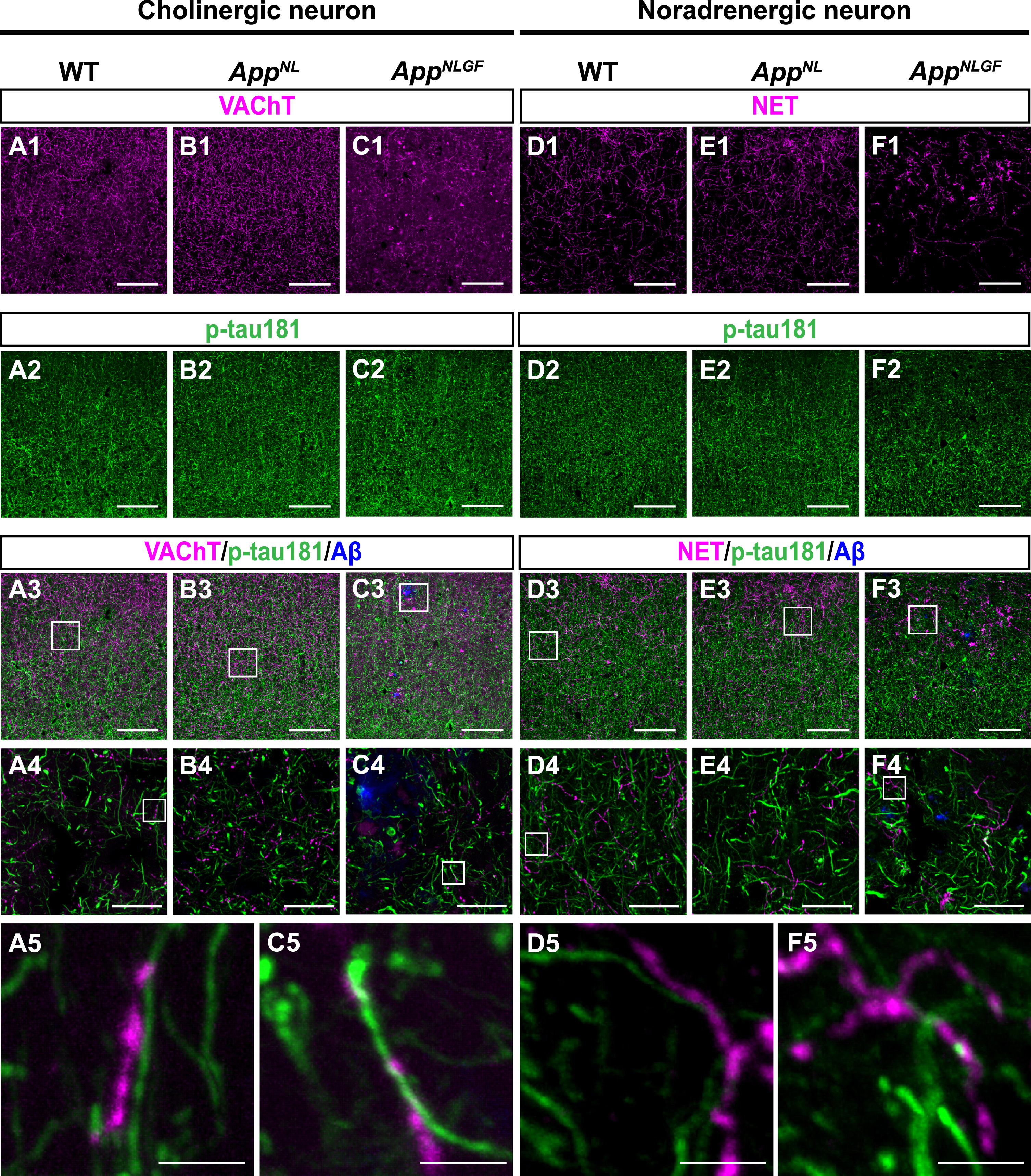
p-tau181-positive axons do not overlap with unmyelinated cholinergic or noradrenergic axons in mouse brains. Representative images of the cortex from frozen coronal brain sections immunostained with antibodies against VAChT (A1– C1; magenta in A3– C3), NET (D1– F1; magenta in D3– F3), and p-tau181 (A2– F2; green in A3– F3). FSB was used for detecting Aβ plaques (blue in A3– F3). Scale bars, 100μm. (A4– F4) Higher magnification of framed regions indicated in A3– F3. Scale bars, 20μm. (A5– F5) Higher magnification of framed regions indicated in A4 – F4. Scale bars, 2.5μm.
Taken together, these results indicate that p-tau181 signals are not localized to unmyelinated cholinergic or noradrenergic axons in mouse brains.
The density of cholinergic axons, but not p-tau181 signals, is significantly reduced in the cortex of App
NLGF
mice
We next analyzed and compared how cholinergic axons and p-tau181-positive axons were affected in the brains of
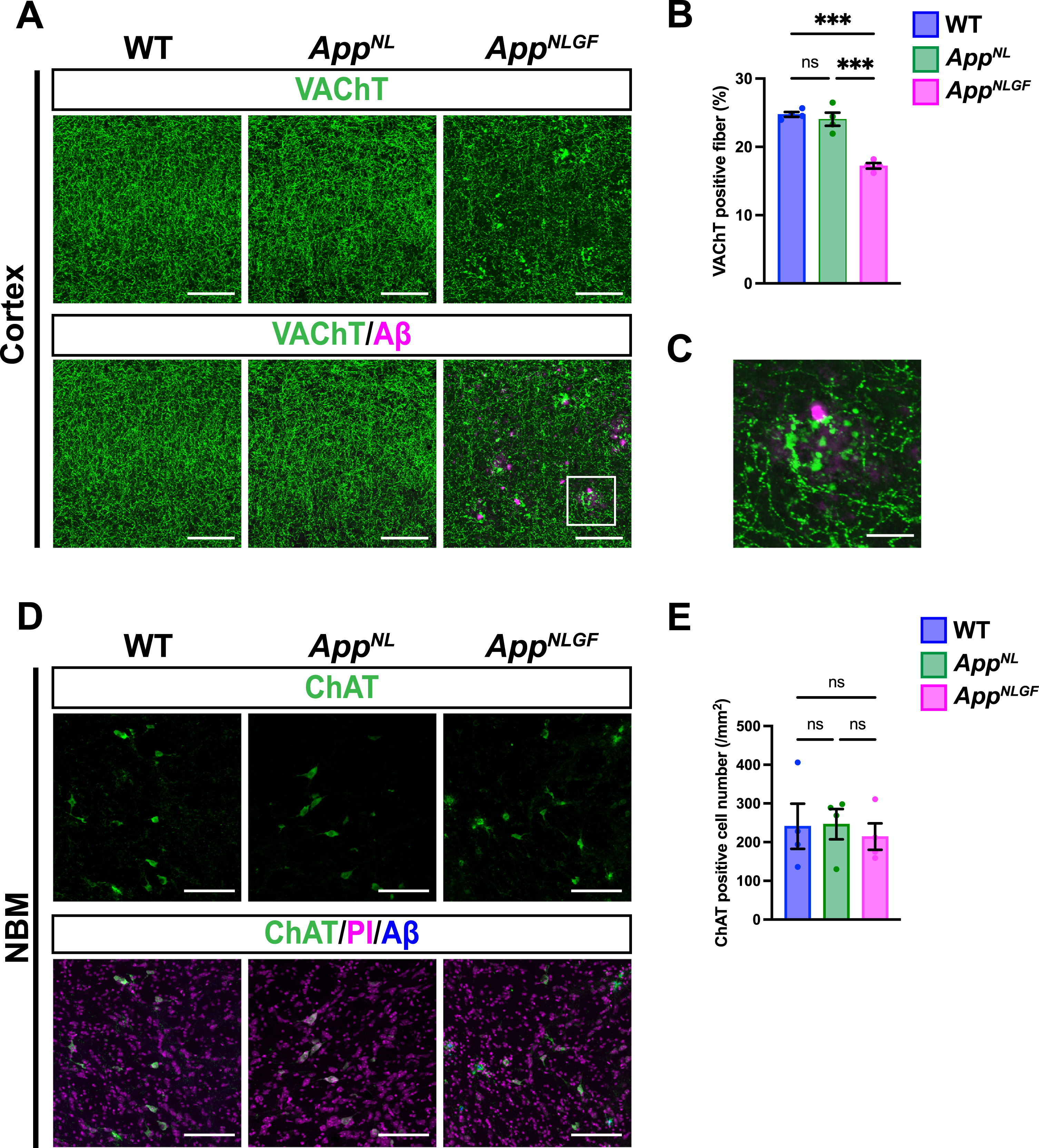
The density of cholinergic axons is significantly reduced in the cortex of
We next stained brain sections with an antibody against p-tau181, and the signal intensity was quantified (Fig. 3A, B). Although p-tau181-positive axons showed axonal dystrophy around Aβ plaques (Fig. 3C, higher magnification of framed region in 3A), the density of p-tau181-positive axons was not significantly reduced in the cortex of
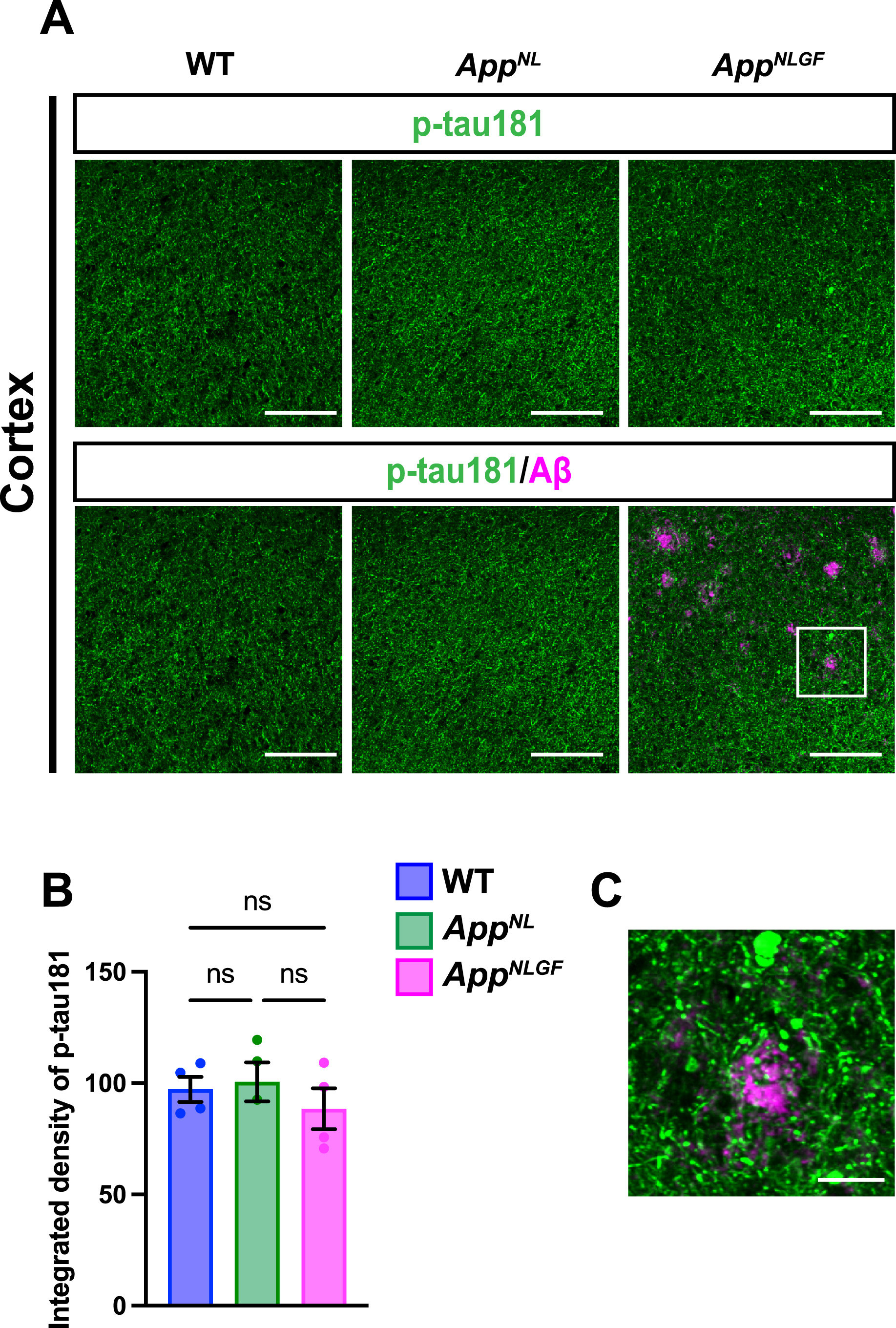
The density of p-tau181 signals is not significantly reduced in the cortex of
Myelin sheaths of p-tau181-positive axons are disrupted in App
NLGF
mice
As p-tau181 signals did not overlap with unmyelinated axons, we next examined whether p-tau181 signals are localized to myelinated axons. Brain sections from 24-month-old
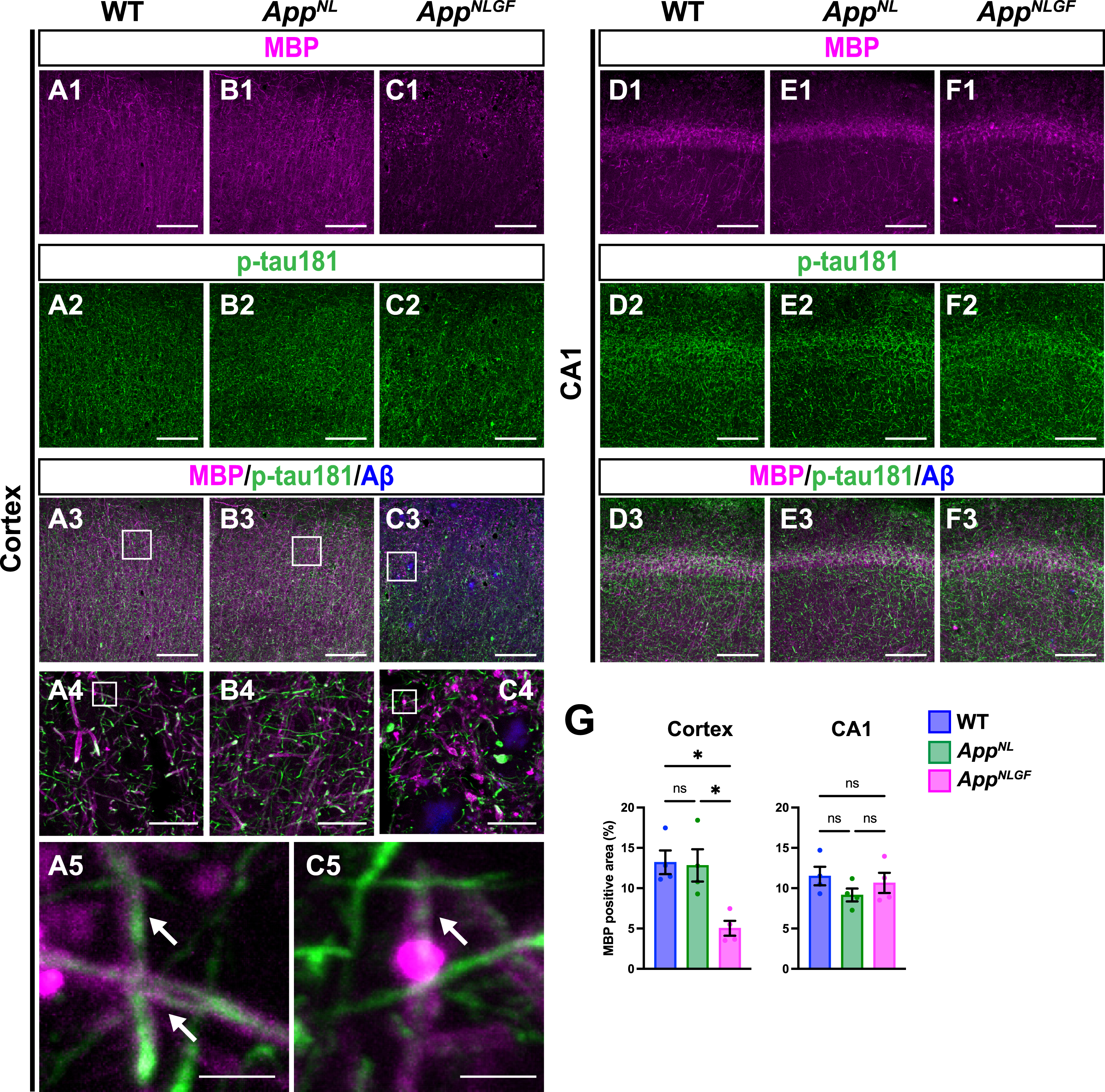
Myelin sheaths of p-tau181-positive axons are disrupted in
Of particular interest, coverage with MBP-positive myelin sheaths was significantly reduced in the cortex of
In contrast to the cortex, myelin sheaths were not significantly affected in the hippocampus of
Taken together, these results indicate that p-tau181 signals are localized to cortical myelinated axons whose myelin sheaths are significantly disrupted by Aβ pathology.
p-tau181-positive axons do not overlap with glutamatergic axons in mouse brains
As axons of glutamatergic excitatory neurons and GABAergic interneurons are both highly myelinated by oligodendrocytes [44], we first examined whether p-tau181 signals overlapped with axons of glutamatergic neurons. Brain sections of the cortices of 24-month-old
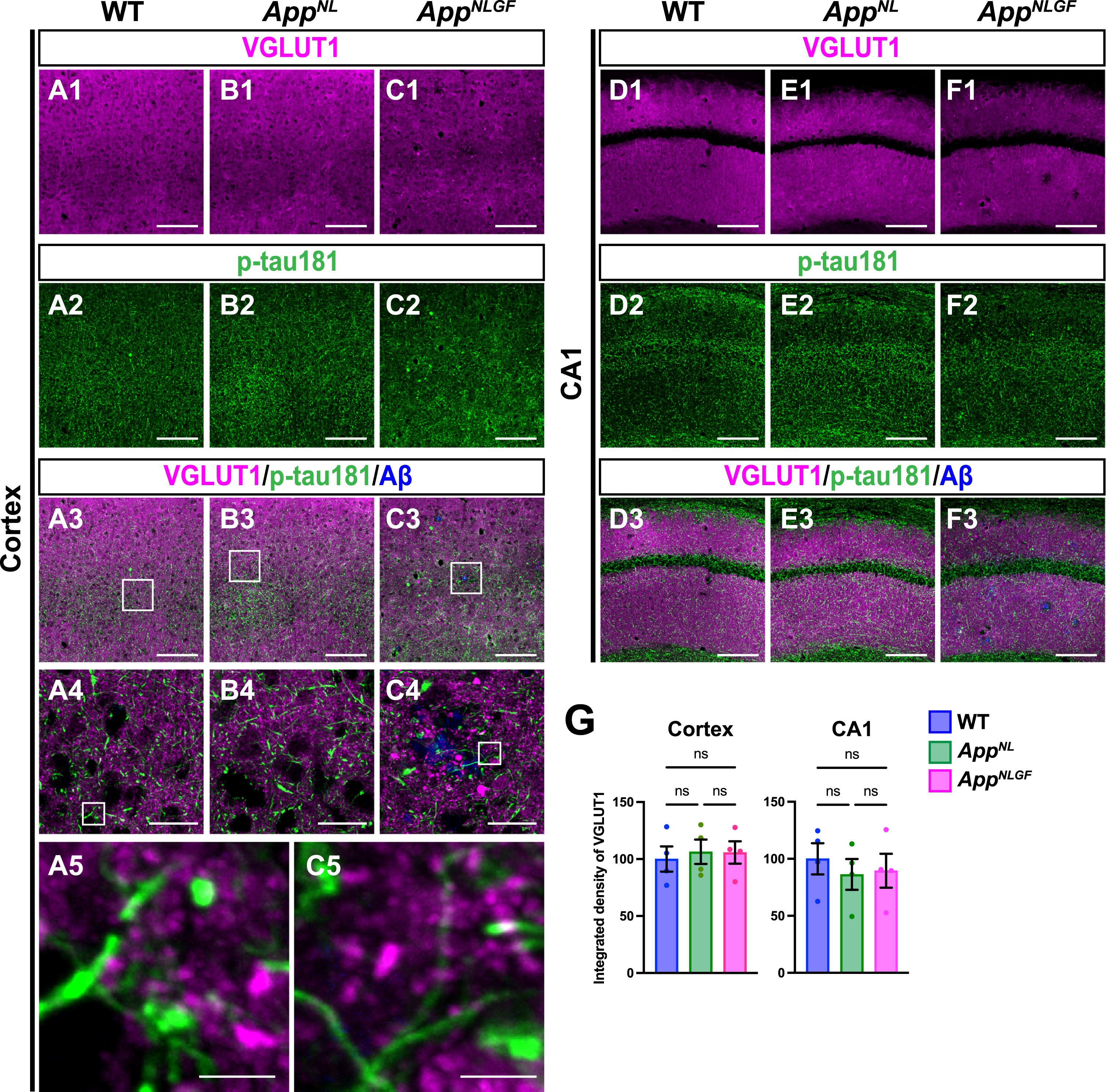
p-tau181-positive axons do not overlap with glutamatergic axons in mouse brains. Representative images of the cortex (A1– C3) and hippocampal CA1 subfield (D1– F3) from frozen coronal brain sections immunostained with antibodies against VGLUT1 (magenta) and p-tau181 (green). FSB was used for detecting Aβ plaques (blue). Scale bars, 100μm. (A4– C4) Higher magnification of framed regions indicated in A3– C3. Scale bars, 20μm. (A5 and C5) Higher magnification of framed regions indicated in A4 and C4. Scale bars, 2.5μm. (G) VGLUT1 immunoreactivity in the cortex or hippocampal CA1 subfield was quantified and expressed as relative percentage to WT.
We also analyzed the density of glutamatergic axons in
p-tau181-positive axons overlap with GABAergic axons in mouse brains
We next examined whether p-tau181-positive axons overlap with GABAergic interneurons. Brain sections of the cortices of 24-month-old
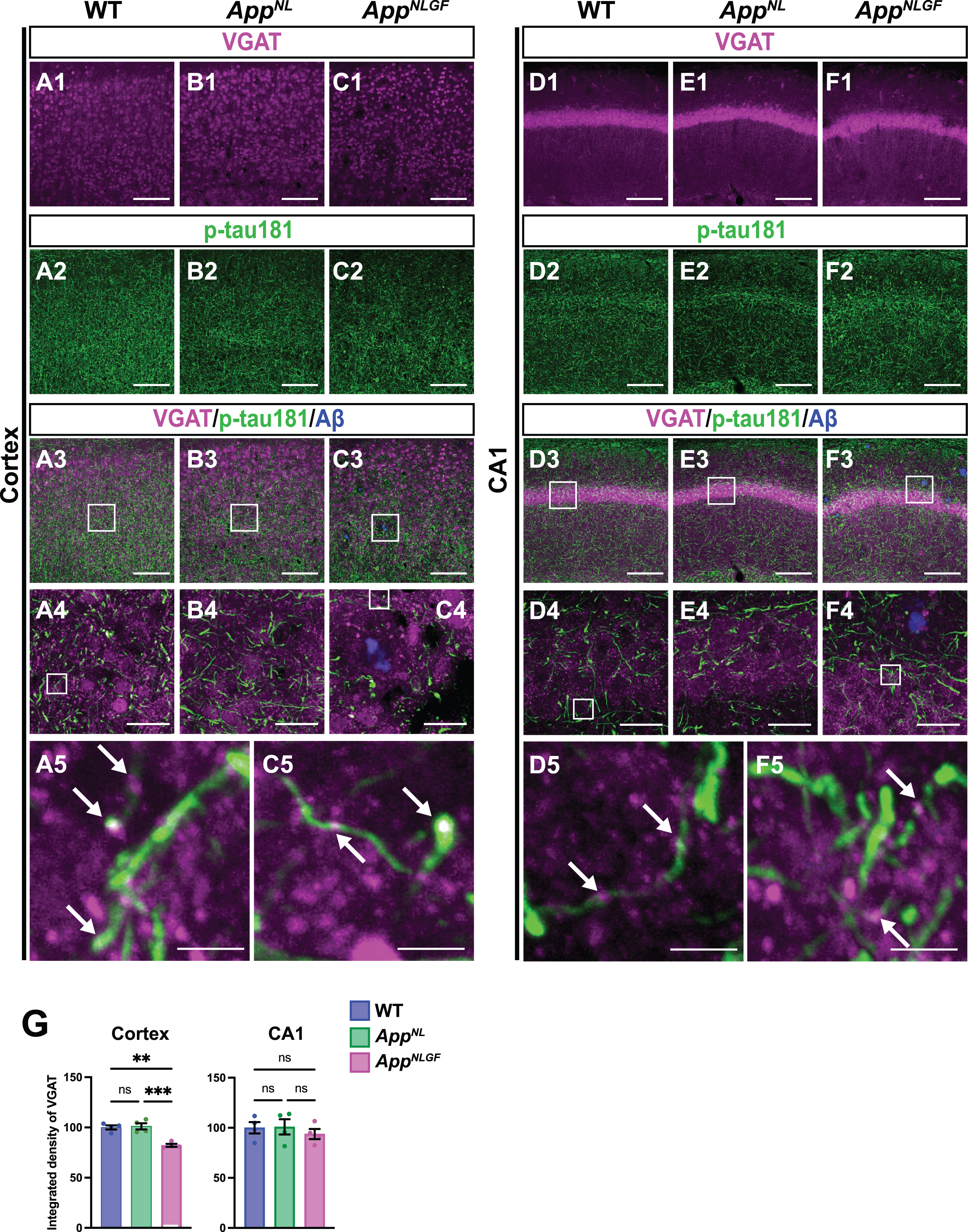
p-tau181-positive axons overlap with GABAergic axons in mouse brains. Representative images of the cortex (A1– C3) and hippocampal CA1 subfield (D1– F3) from frozen coronal brain sections immunostained with antibodies against VGAT (magenta) and p-tau181 (green). FSB was used for detecting Aβ plaques (blue). Scale bars, 100μm. (A4– C4 and D4– F4) Higher magnification of framed regions indicated in A3– C3 and D3– F3, respectively. Scale bars, 20μm. (A5, C5, D5, F5) Higher magnification of framed regions indicated in A4, C4, D4, and F4, respectively. White arrows indicate colocalization of VGAT and p-tau181 signals. Scale bars, 2.5μm. (G) VGAT immunoreactivity in the cortex or hippocampal CA1 subfield was quantified and expressed as relative percentage to WT.
We also analyzed the density of GABAergic axons in
Taken altogether, these results suggest that axons of GABAergic interneurons may be more affected than those from glutamatergic excitatory neurons in the brains with Aβ pathology.
p-tau181-positive axons overlap with parvalbumin-expressing axons in mouse brains
To further validate the above results, we examined whether p-tau181-positive axons overlap with axons of parvalbumin-expressing interneurons, which comprise nearly half of the interneuron population in the cortex [48, 49]. Brain sections of the cortices of 24-month-old
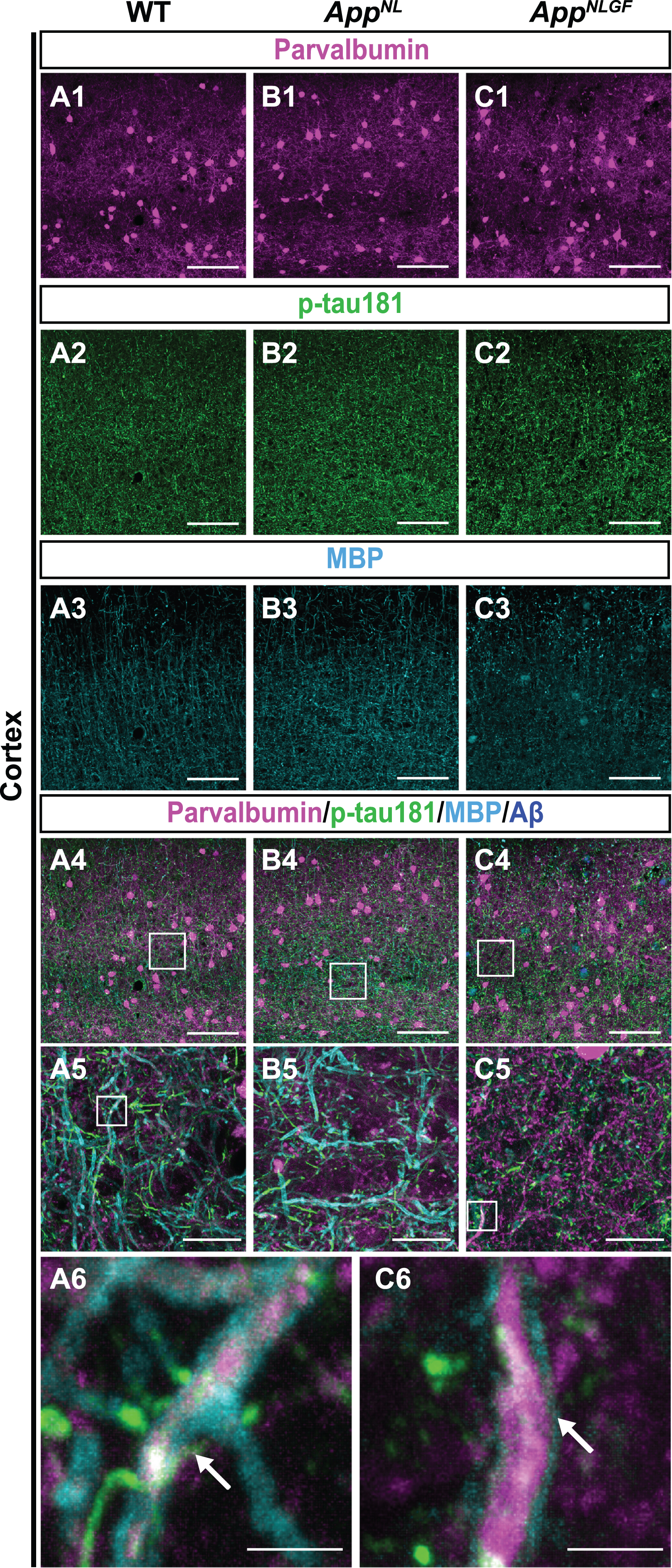
p-tau181-positive axons overlap with parvalbumin-expressing axons in mouse brains. (A1– C4) Representative images of the cortex from frozen coronal brain sections immunostained with antibodies against parvalbumin (magenta), p-tau181 (green), and MBP (cyan). FSB was used for detecting Aβ plaques (blue). Scale bars, 100μm. (A5– C5) Higher magnification of framed regions indicated in A4– C4. Scale bars, 20μm. (A6, C6) Higher magnification of framed regions indicated in A5 and C5. White arrows indicate MBP signals enwrapped with the p-tau181-positive axons of parvalbumin-expressing interneurons. Scale bars, 2.5μm.
Taken together, these results indicate that p-tau181-positive signals are localized to parvalbumin-expressing GABAergic interneurons in mouse brains, and that the myelin sheaths of these interneurons are significantly disrupted in the
DISCUSSION
p-tau181 in CSF is an established biomarker for AD [6, 50, 51], and recent studies demonstrate that increased levels of p-tau181 in plasma as well as CSF correlate with Aβ pathology in the preclinical and early stages of AD [1, 2]. Using
Excitatory-inhibitory imbalance may cause abnormal network activity and influence neuronal oscillations important for cognitive functions in early AD [52–54]. Neurophysiological studies in humans have shown that neuronal hyperactivity is associated with amyloid-PET positivity [55], suggesting that aberrant excitatory responses are induced by Aβ accumulation [56]. Several studies indicate that functional impairments of inhibitory GABAergic interneurons, especially fast-spiking parvalbumin-expressing neurons, underlie network hyperactivity in early AD [57] as inhibitory interneurons play critical roles in synchronizing neuronal activity and generating neuronal oscillations [58, 59]. Of particular interest, studies using resting-state magnetoencephalography and electroencephalography showed that lower Aβ42 levels and higher p-tau181 levels in CSF were associated with cortical network disruption in early AD [60, 61]. Our data demonstrate that p-tau181 is associated with axonal abnormalities in parvalbumin-expressing GABAergic interneurons in Aβ pathology in mouse brains (Fig. 7). This suggests links among p-tau181, impaired inhibitory neurotransmission, and Aβ pathology in early AD.
Evidence from neuroimaging studies demonstrates that Aβ pathology correlates well with myelin damage in the early stage of AD [62, 63]. A previous study has reported a relationship between myelin water fraction (MWF), a surrogate of myelin content, and p-tau181/Aβ42 ratio in the CSF from preclinical AD patients, in which higher levels of p-tau181/Aβ42 resulted in an increased decline in MWF with age [63]. Also, elevated plasma p-tau181 has been associated with lower axonal density quantified by MWF in the occipital lobe [64]. These reports suggest that increased levels of fluid p-tau181 correlate with reduced myelin content in human brains.
Single-cell transcriptomic analyses have also revealed that oligodendrocytes show AD pathology-responsive transcriptional signatures, suggesting that disruption of myelination plays a key role in AD pathogenesis [65]. A recent study demonstrated that myelin on parvalbumin-expressing inhibitory interneurons is critical for feedforward inhibition of cortical circuits [66], and that myelin loss reduces the number of presynaptic terminals from these interneurons and alters neural oscillations [67]. Interestingly, a large fraction of the myelin present in the cortex enwraps mainly the axons of parvalbumin-expressing GABAergic interneurons in mouse brains [44, 68, 69]. Our results showed that myelin sheaths are significantly disrupted (Fig. 4), and that, interestingly, axons of GABAergic interneurons (Fig. 6), but not of glutamatergic neurons (Fig. 5), are significantly reduced in the cortex of
Another interesting finding from this study is that p-tau181 signals colocalize well with axons of myelinated interneurons (Fig. 6) but not those of unmyelinated cholinergic or noradrenergic neurons (Fig. 1). We recently reported that signals of p-tau217 and p-tau231, other fluid biomarkers for Aβ pathology in the preclinical stage of AD, colocalized with the postsynaptic marker of myelinated glutamatergic neurons but not of unmyelinated cholinergic or noradrenergic neurons [10]. These results suggest that currently used p-tau biomarkers may reflect synaptic and axonal abnormalities of myelinated neurons but not of unmyelinated neurons. Dysfunction and degeneration of unmyelinated cholinergic and monoaminergic neurons are associated with cognitive decline and psychological symptoms in the early stage of AD [72–75] and have been important therapeutic targets to improve clinical symptoms [11, 76, 77]. Thus, developing biomarkers that reflect abnormalities in unmyelinated subcortical neurons will be important for AD diagnosis and drug development.
This study has several limitations. For example, this study did not address whether p-tau181 was released from demyelinated axons of parvalbumin-positive GABAergic interneurons into the CSF or plasma in mouse models. Further study is required to investigate the correlation among p-tau181 levels in the CSF/plasma/interstitial fluid, Aβ pathology and disruption of myelinated structures of parvalbumin-positive GABAergic axons in the brains of
In conclusion, this study demonstrates that AD-related p-tau181 is localized to myelinated axons of parvalbumin-expressing GABAergic interneurons in mouse brains. This study also suggests that increased p-tau181 levels in the CSF and plasma may reflect loss of myelin sheaths and dysfunction of GABAergic interneurons upon Aβ accumulation. This is thought to underlie cognitive deficits in the preclinical and early stages of AD.
Footnotes
ACKNOWLEDGMENTS
We thank Dr. Takaomi Saido at RIKEN Center for Brain Science and Dr. Takashi Saito at Nagoya City University for providing
FUNDING
This study was supported by the Research Funding for Longevity Science from National Center for Geriatrics and Gerontology, Japan, Grant No. 21-13 to KMI and MS, JSPS KAKENHI Grant No. JP20H03571 to KMI, No. JP22H02963 to MS, No. JP21K17655 to YS and No. JP21J00767 to YH.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
All data generated or analyzed during this study are included in this published article.
