Abstract
Background:
Neuroinflammation is an innate immunological response of the central nervous system that may be induced by a brain insult and chronic neurodegenerative conditions. Recent research has shown that neuroinflammation may contribute to the initiation of Alzheimer’s disease (AD) pathogenesis and associated epileptogenesis.
Objective:
This systematic review aimed to investigate the available literature on the shared molecular mechanisms of neuroinflammation in AD and epilepsy.
Methods:
The search included in this systematic review was obtained from 5 established databases. A total of 2,760 articles were screened according to inclusion criteria. Articles related to the modulation of the inflammatory biomarkers commonly associated with the progression of AD and epilepsy in all populations were included in this review.
Results:
Only 7 articles met these criteria and were chosen for further analysis. Selected studies include both
Conclusion:
Neuroinflammation was directly associated with the advancement of AD and epilepsy in populations compared to those with either AD or epilepsy. However, more studies focusing on common inflammatory biomarkers are required to develop standardized monitoring guidelines to prevent the manifestation of epilepsy and delay the progression of AD in patients.
INTRODUCTION
Amyloid-β (Aβ) plaques, the key hallmark of Alzheimer’s disease (AD), were first reported in epileptic patients long before the acknowledgement of AD by Alois Alzheimer [1]. The relative risk of unprovoked seizure surges significantly up to 10-fold greater occurrence in late-onset AD in comparison to the general population of the same age [2, 3], and an 80% of seizure recurrence risk after a first seizure episode was reported in an elderly AD population [3]. The linkage between AD and epilepsy pathologies has since then remained obscure [2]. To date, pharmaceutical treatments available for AD and epilepsy are mainly targeting symptomatic control for the disease/disorder instead of curative treatment [4].
The most common form of dementia is AD, which is a neurodegenerative disease characterized by memory loss and progressive neurocognitive impairment [5]. The widely recognized hypothesis on the predominant cause of AD is due to the aberrance in the processing and polymerization of regularly soluble proteins. As a result, a misfolded protein with modified conformation is produced which could lead to abnormal or loss of neuronal functions [6, 7]. The key pathologies for AD are the presence of amyloid-β (Aβ) and hyperphosphorylated tau [4, 8]. In brief, amyloid pathogenesis is initiated with altered cleavage of an integral protein on the plasma membrane, amyloid-β protein precursor (AβPP), by β-secretases (BACE1) and γ-secretases to produce insoluble Aβ fibrils [9, 10]. Consecutively, Aβ oligomerizes, diffuses into the synaptic cleft and disrupts synaptic signaling. The polymerization of Aβ oligomers into insoluble amyloid fibrils leads to aggregation into plaques, leading to kinase activation, hyperphosphorylation of microtubule-associated tau protein, and the formation of intracellular neurofibrillary tangles (NFTs). Accordingly, the clinical characteristics of AD include the presence of extracellular aggregates of Aβ and NFTs constituted of hyperphosphorylated tau protein in the cortical and limbic regions of the brain [5]. On the other hand, epilepsy is one of the most prevailing neurologic conditions with an occurrence of one in every 2000 population [11]. Epilepsy is a condition defined as at least two unprovoked seizures developing more than 24 hours apart or one unprovoked seizure with a 60% probability of developing further seizures similar to the general recurrence risk after two unprovoked seizures over the next 10 years [12]. The manifestation of seizure is due to the disproportionate, hypersynchronous firing of neurons, which disrupts the excitatory and inhibitory imbalance in the cerebral cortex of the brain.
Recent studies have shown that non-convulsive epileptiform activity appears to be an under-reported comorbidity of AD [13]. Besides that, shared pathological similarities such as hippocampal atrophy, neuronal death, gliosis, neuroinflammation, and neuropsychiatric and cognitive comorbidities were also observed in AD and epilepsy patients [14]. Following this, several experimental studies have demonstrated a lowered threshold for seizures in AD models, suggesting neuronal hyperexcitability may contribute to the behavior deficit in AD [2]. Although the exact mechanisms of associating seizures with the onset and trajectory of AD pathogenesis remain unclear, numerous biomarkers and mechanisms have been proposed. Aβ and tau protein, GABAergic and glutamatergic alterations, the role of the noradrenergic nucleus Locus Coeruleus, and neuroinflammation are possible pathological mechanisms to underlie the increased risk of seizures in AD [15–17]. Among the proposed mechanisms, increasing evidence has found neuroinflammation was presented in both AD [2, 18] and epilepsy [16, 19], potentially linking the pathogenesis between these two conditions.
Neuroinflammation is triggered when there is a brain insult, such as traumatic brain injury, infections, or status epilepticus (SE) [20]. Neuroinflammation is described by the initiation of the central nervous system (CNS) immune response, mediated by astrocytes, microglia, endothelial cells of the blood-brain barrier (BBB), neurons, and peripheral immune cells extravasation in the CNS parenchyma [20]. For decades, AD has been tightly linked to neuroinflammation [21–23]. Aβ interacts directly with receptors on microglia and astrocytes including receptors for advanced glycation end products (RAGEs), Toll-like receptors (TLR), chemokine-like receptor 1, and the α6β1 integrin [24, 25], inducing an acute inflammatory response, and triggering the complement and inflammatory enzymes [2]. Consequently, the microglia and astrocytes are activated, releasing pro-inflammatory chemokines and cytokines [26]. Extracellular and intracellular Aβ and tangles produce intense toxicity that causes synaptic damage [2], increasing oxidative stress [27], which in turn causes the infiltration of microglial surrounding the plaques regions [24, 28]. Microglia can also activate T-mediated neurotoxic immune response as antigen-presenting cells and exacerbate neurodegeneration [28]. Persistent secretion of pro-inflammatory cytokines from reactive microglial may, in return, further promote amyloid deposition [29]. Thus, the unavailing inflammatory escalation response towards Aβ proteins predating the onset of cognitive declination in AD has been hypothesized [26].
In some instances, proinflammatory conditions such as autoimmune disorder, fever as well as the underlying neuroinflammation in AD, is a risk factor for seizures [2]. According to prior evidence from
The aforementioned findings have supported the role of neuroinflammation in promoting seizures and cognitive decline seen in AD. Both AD and epilepsy precipitate aberrant neuroinflammation; in which neuroinflammation is seen as a risk determinant for seizures and may exacerbate AD. However, a concise depiction of the responsible inflammatory biomarkers and the subsequent pathology that correlates with epilepsy and AD have not been summarized previously. Thus, this systematic review aimed to critically evaluate the currently available literature investigating the association of neuroinflammation in both AD and epilepsy, which could potentially serve as a guide in exploring alternative therapeutic strategies.
MATERIALS AND METHODS
Search strategy and studies identification
A literature search was conducted using databases namely PubMed, Ovid Medline, Scopus, and EMBASE. The search was limited to publications from the last 5 years, January 2018 to August 2022 for the retrieval of recent, relevant, and updated studies. The search terms “Neuroinflammation”, “Alzheimer’s Disease”, “Epilepsy”, “Seizure”, and “Cognitive Decline” were used to extract relevant articles from the databases. A title, abstract, and keyword search were performed using the search terms. The Boolean operator “AND” and “OR” was used to link the terms together on all databases. Articles were first screened through their titles and abstracts before proceeding with the full-text screening of relevant articles. Additionally, electronic searches were restricted to the availability of abstracts and studies published in the English language.
Study selection
The study selection for this systematic review is based on Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) protocol. Studies eligible for inclusion include peer-reviewed original research articles in
Data extraction, quality assessment, and data analysis
Key study data were extracted from each study. These include first author, study model, specimens used, measure outcome, and biomolecules studied. Systematic Review Centre for Laboratory Animal Experimentation Risk of Bias (SYRCLE RoB) tool was used to evaluate the quality and risk of bias of the animal interventional studies that were included in this review [35]. “Yes” indicates a low risk of bias and ‘No’ indicates a high risk of bias in this quality item whereas ‘Unclear” indicates insufficient data was provided for evaluating the degree of bias. The literature search, selection and quality analysis of the selected articles were performed by two independent researchers. Meta-analysis was not conducted in this review due to variability in statistical analysis utilized, methodologic diversity (e.g., study design, including procedures, measured outcome), and heterogeneity (e.g., type of specimens) of the selected studies.
RESULTS
Study characteristics
The initial systematic search and reference tracking yielded 2,760 articles (Fig. 1). Of these initial 2,760 articles, 288 duplicates were removed and 2,472 articles were included after screening of the abstracts. Most of these 2,440 articles were either conference abstracts, or review papers, or do not fulfil the study inclusion criteria. Hence, a total of 32 full-text articles were screened and 25 articles that do not measure the association of either AD, epilepsy, and seizure together with neuroinflammation were excluded. A total of 7 articles were finally used for the analyses and summary. The characteristics, findings, and quality of articles based on SYRCLE’s risk of bias checklist of each study (Supplementary Table 1) were summarized and tabulated in Tables 12, based on their experiment design. Although most of the included study has a high risk of selection bias, the overall quality remained unbiased. Out of the included studies, there were only one research of the included articles was conducted in
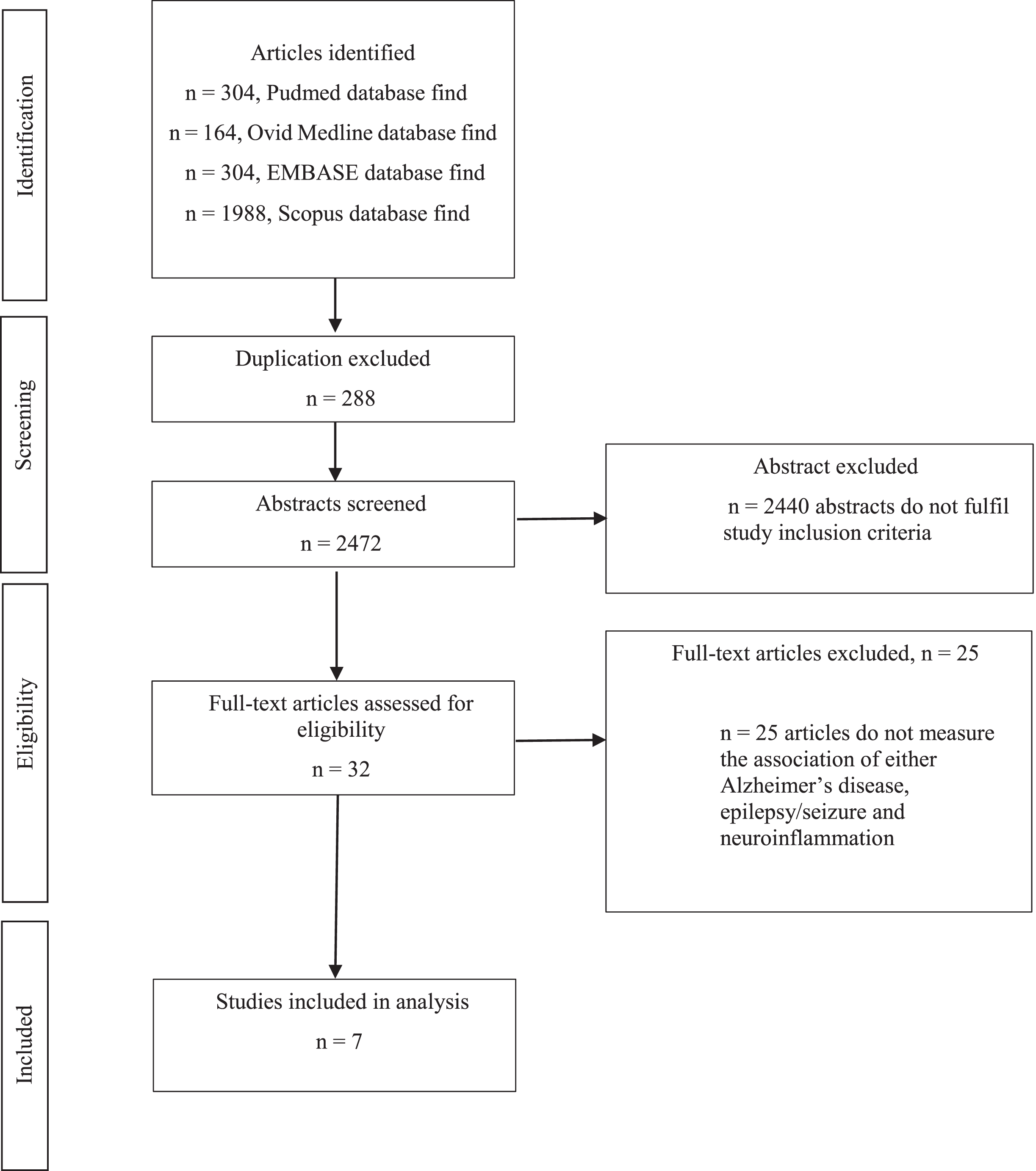
A systematic PRISMA flowchart for articles identification
AD, Alzheimer’s disease; CA, cornu ammonis; DG, dentate gyrus; GFAP, glial fibrillary acidic protein; sEPSPs, spontaneous excitatory postsynaptic potentials; a Detailed items presented in the Supplementary Material.
AD, Alzheimer’s disease; MTLE, Mesial temporal lobe epilepsy; GFAP, Glial fibrillary acidic protein; Iba1, Ionized calcium-binding adaptor molecule 1; IL, Interleukin; TNF-α, tumor necrosis factor-alpha; CCL 12, Chemokine (C-C motif) ligand 12; PHF, paired helical filaments; Cdk5, Cyclin-dependent kinase 5; APP, amyloid precursor protein; BACE1, Beta-secretase 1; ADAM10, A Disintegrin and Metalloproteinase 10; PS1, Presenilin-1; GSK-3β, Glycogen synthase kinase-3 beta; SE, Seizure epilepticus; IHK, Intrahippocampal injection of kainic acid; CA, Cornu Ammonis; DG, Dentate gyrus; CD68, Cluster of differentiation 68; TREM2, Triggering receptor expressed on myeloid cells 2; sIPSPs, Spontaneous inhibitory postsynaptic potentials; NPY, Neuropeptide Y; LEC, Lateral entorhinal cortex; Wnt, Canonical Wingless/integrated; PTZ, Pentylenetetrazole; ENT, Entorhinal cortex; AAV, Adeno-associated viral vector; hTau, Human Tau; WT, Wildtype; KO, Knock-out; PTX, Picrotoxin; a Detailed items presented in Supplementary Table 1.
Role of neuroinflammation in AD-associated epilepsy
The direct and indirect correlation between AD and seizure progression in association with neuroinflammation were extracted from the findings of the included studies. Figure 2 depicted the pathology of neuroinflammation in AD and AD-associated epileptogenesis in relation to microgliosis and astrogliosis and their downstream mechanisms. A significant reduction in the level of inflammatory markers IL-1β in mice was detected after an episode of SE in transgenic mice without AD; however, changes in the cytokines and chemokines such as TNF-α, IL-6, C-C motif chemokine ligand 2 (CCL2), and C-C motif chemokine ligand 12 (CCL12) were not significant in comparison in mice with and without AD [37]. On the contrary, one other study has found an increase in IL-1β levels in AD transgenic mice as AD progressed among the other cytokines (IL-1α, MIP2, RANTES, and KC) [42]. Moreover, overexpression in IL-18 cytokine has demonstrated an increased risk of seizures in AD, so as under-expression, thus a balance of inflammatory cytokine is critical in suppressing the development of seizures in AD. Although no significant differences were detected in IL-1β and TNF-α in both wildtype and transgenic mice with AD, the study investigating AD pathology and neuroinflammation demonstrated a decreasing trend of expression in IL-10 in both wildtype and transgenic mice with AD [38]. Other neuroinflammation makers include glial fibrillary acidic protein (GFAP), cluster of differentiation 68 (CD68) immunoreactivity, and ionized calcium-binding adaptor molecule 1 (IBA1). The increase in the aforementioned neuroinflammatory markers indicates higher activation of glial cells, leading to heighten inflammation response. Two studies using both AD and epileptic mice models displayed a raise in GFAP levels [36, 37]. A study investigating signaling dysregulation in canonical wingless/integrated (Wnt) associated with AD pathology and neuroinflammation in knock-in transgenic AD mouse model showed an increase of CD 68 during the latent phase of AD [39], similarly with another study utilizing Arctic Aβ mice AD model has also demonstrated an increase CD 68 staining and expression of triggering receptor expressed on myeloid cells 2 (TREM2), but not inflammatory cytokines during a seizure [38]. Correspondingly, an increase of microgliosis was seen collectively in the mice after seizurogenic administration in an epileptic model, however, a significantly lesser number of microglial and astroglial were observed in tau KO mice [40]. An AD mice model induced with neuronal activity showed to have increased IBA1 intensity which was associated with the worsening of AD hallmark [41].
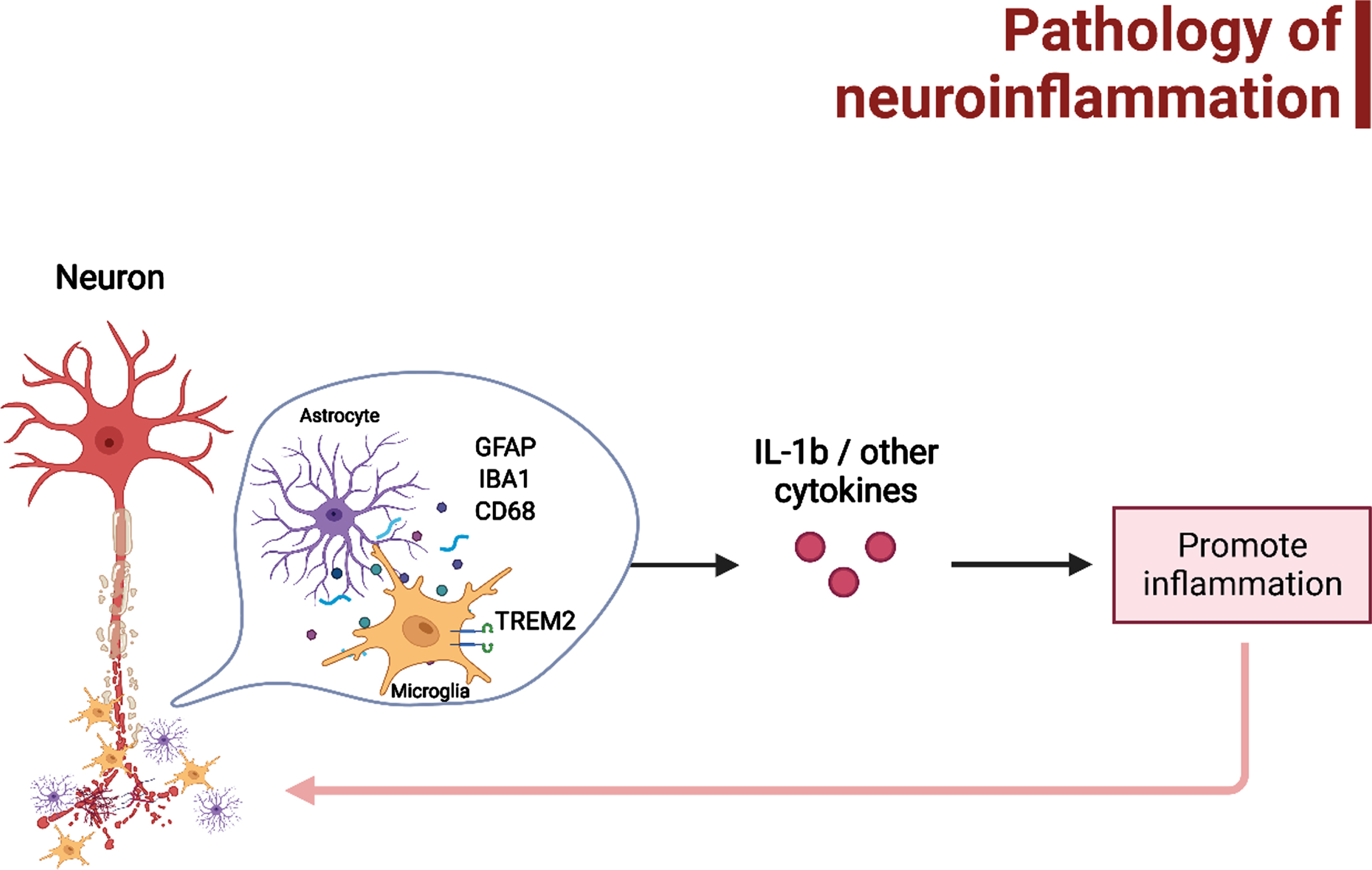
The possible pathology of neuroinflammation that may lead to AD and/or AD-associated epileptogenesis, which have been mainly related to microgliosis and astrogliosis. Figure was created in BioRender.com.
DISCUSSION
The evidence from the rodent studies has suggested a possible relation of neuroinflammation in the pathogenesis of AD and AD-associated epilepsy. During neuroinflammation, glial activation produces functional and morphological changes within the cells that affect the neural-glial and glial-glial interactions [43]. Such alterations may lead to synaptic communication impairment, imbalance in neurotransmitter homeostasis, and potentially axonal degeneration and neuronal death [44]. The neurodegeneration process in AD is defined by synapse dysfunction and neuronal loss [8]. Recent findings have supported the notion on the abnormal progressive accumulation of Aβ oligomers is related to synaptic pathology and impaired neurogenesis in AD. Aβ oligomers might cause a cascade of events that triggers hyperexcitability and neuroinflammation that promotes neurotoxic effects, leading to a pernicious cycle of the neurodegenerative process [5].
In response to brain insult, astrocytes become activated [45]. Astrocytic activation happens as a result of hypertrophy and upregulation of intermediate filaments, such as glial fibrillary acidic protein (GFAP). Various cytokines are shown to involve either during the initiation or modulation of reactive astrogliosis, including IL-1β, TNF-α, and transforming growth factor beta-1β (TGF-1β), that are expressed on astrocytes and their interaction stimulates an astrocytic reactive response. Astrocyte-derived cytokines such as IL-1β and TNF-α are involved in the promotion of neurotoxicity meanwhile TGF-1β is believed to be neuroprotective. On the other hand, approximately a quarter of the cells within the brain are microglia, that actively screen and modulate the CNS environment to maintain homeostasis and physiological requirements, as well as respond to pathological affairs via brain immune cells for innate immune response coordination [46]. Microglial as pathological sensors of brain insult will be activated immediately and undergo critical morphological changes. In the initial phase, activation of the microglial is deemed as neuroprotective by migrating to the site of the insult and stimulating inflammatory response for healing. However, altered morphology accompanied by more reactive or activated microglia, in conjunction with increased activated microglia markers and proinflammatory cytokine levels (for example, IL-1β and TNF-α) disrupts brain function and neuronal dysfunction [47, 48].
On the other hand, a decrease in tau and neuroinflammation has been strongly associated with lessening seizure severity, improved cognitive functions, and mortality in many epilepsy models [38, 49]. The range of cognitive and memory impairment in AD is similar to epilepsy, implicating a possible contribution of seizures in numerous cognitive and behavioral aspects of AD-related alterations [50]. Evidence from several studies has shown patients with AD suffer from acquiring and retaining memory effectively [51], which is consistent with the neuropathological and imaging findings that showed the vulnerability of the ENT and hippocampus in AD [52, 53], Correspondingly, seizures that affect the hippocampus, especially in temporal lobe epilepsy (TLE), have cumulative impacts on cognitive functions after recurrent seizures [50]. Circuit remodeling and alteration in the circuitry and molecular level of the hippocampus are possible mechanisms causing cognitive impairment in epilepsy and AD.
Applicability of targeting neuroinflammation as therapeutic strategy in AD and epilepsy
Recent literature has shown the presence of neuroinflammation in AD long before the deposition of amyloid plaques, which is also seen in some types of epilepsies, including TLE and SE [54, 55]. Microglial, astrocytes, and oligodendrocytes contribute to the regulation of neuroinflammation [56]. In AD, Aβ activates the glial and causes the release of several proinflammatory cytokines, leading to the manifestation of generalized neuroinflammation [2]. Consequently, this process would promote neurotoxic effects that may drive a vicious cycle of the neurodegenerative process due to heighten neuronal excitability. In both conditions, astrogliosis and microgliosis modify the glutamate-glutamine cycle [55]. The proinflammatory cytokines such as IL-6, IL-1β, and TNF-α that are released from microglial and astrocytes play a role in modulating the release of glutamate and altering its post-synaptic uptake, along with the signaling reduction of GABA [2, 55]. However, as the available literature that studied the stringent association of neuroinflammation on both AD and epilepsy is extremely limited, the effect of neuroinflammation represented in this systematic review may not reflect the true implication. In addition, the increase in neuronal hyperexcitability has also been postulated to increase the production of the Aβ via β- and γ-secretases stimulation, and promote the pathological expression of AD, causing a higher extent of neurodegeneration and cognitive deficit [54]. Furthermore, disruption in the BBB that happens in both AD and epilepsy was hypothesized as a consequence of the proinflammatory response [57]. The surge in toxic materials in the CNS due to the impaired BBB integrity that favors the perpetuation of a proinflammatory environment would further activate the microglia and proinflammatory cytokine release, thus, imbalance the excitatory-inhibitory system and pro-excitatory synaptic dysregulation [58]. As a result, this could potentially increase the occurrence of interictal epileptiform activity and also epileptic seizures [57]. The association and consequences of neuroinflammation in AD and epilepsy was shown in Fig. 3. As discussed, the aforementioned factors may depict a better correlation in the advancement of AD and epilepsy, but it is also important to acknowledge that the development of both conditions as one other comorbidity is an endless process and neuroinflammation is part of that process as an implication, and potentially a causal relationship.
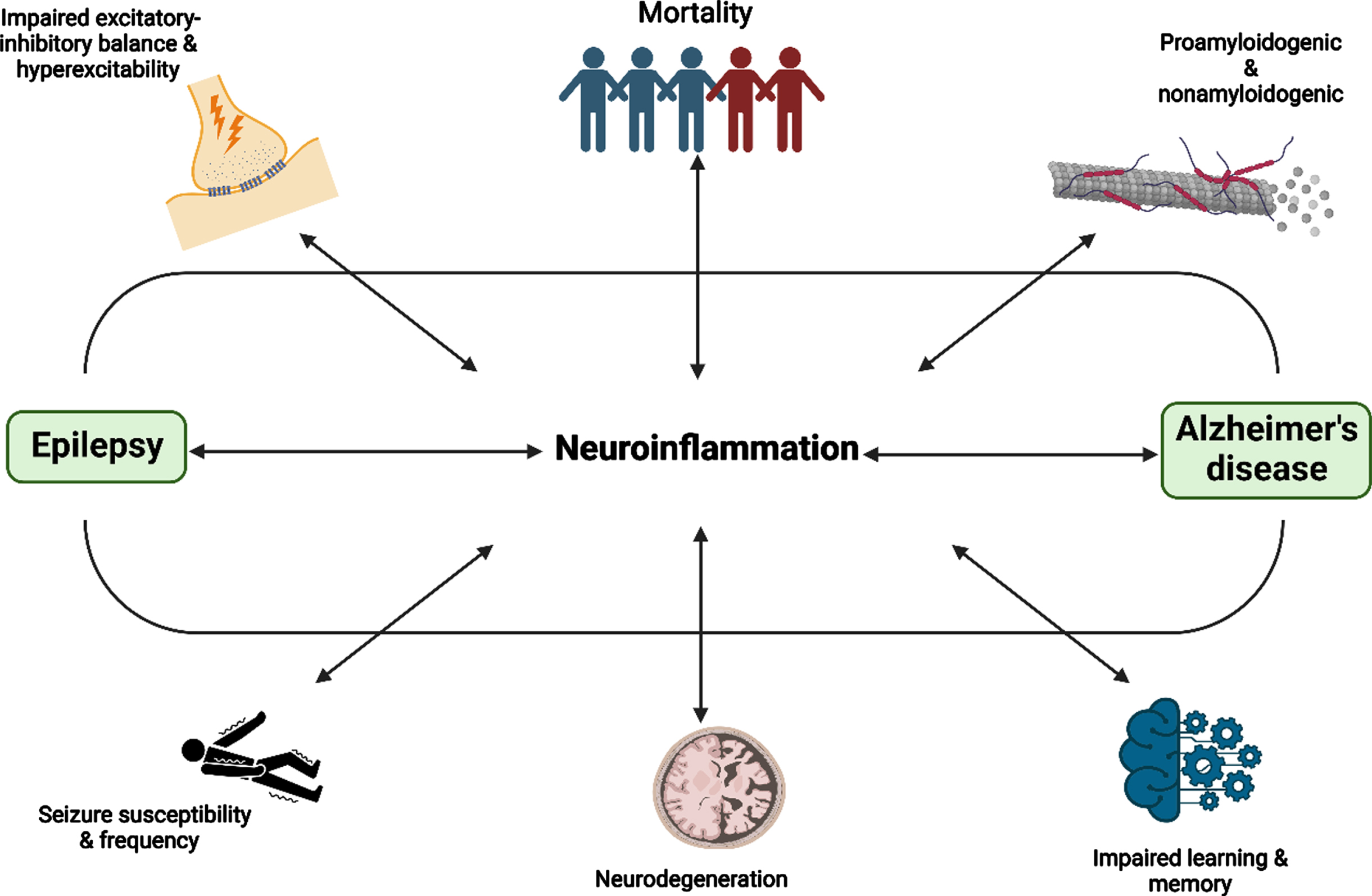
The association and consequences of neuroinflammation in AD and epilepsy. Figure was created in BioRender.com
More and more researchers are focusing on the effort in diminishing prolonged neuroinflammation in AD and epilepsy. In preclinical studies, therapeutic targets of neuroinflammation involving antibodies against inflammatory cytokine (TNF-α) and receptors (TREM2 and CD33) expressed in the microglia have shown favorable results in controlling neuroinflammation, and subsequently the progression of AD [18]. Meanwhile, inhibitors of TLR3 or TLR4 expressed by microglia [19] and anti-high motility group box-1 (HMGB-1) [59] in preclinical studies have also demonstrated a significant reduction in epileptic seizures by disrupting the inflammatory mechanisms. Inflammation-targeting small molecules in the management of AD and seizures were seen to produce positive neurologic outcomes due to their smaller size and low molecular weight that could penetrate the CNS domain [18, 19]. The therapeutic effects of some of the available nonsteroidal anti-inflammatory drugs (NSAID) have been postulated to target pathological hallmarks of AD and decrease the frequency of spontaneous seizures by interacting with inflammatory pathways [18, 19]. With this, the clinical benefits of anti-inflammatory treatment could be extrapolated as a potential therapeutic approach in patients with both AD and epilepsy, nonetheless, more studies are warranted as the aforementioned preclinical evidence focuses only on a condition, either AD or epilepsy, at a time.
Limitation and future direction
In this review, we have systematically summarized studies that evaluated the direct and indirect interaction of neuroinflammation between AD and seizure. The evidence presented in this systematic review can be used by researchers to enhance current understandings of the role of neuroinflammation in the progression of AD and epilepsy pathophysiology and to potentially identify and target inflammatory biomarkers as a new therapeutic approach for treatment. Moreover, the study result might be a good guide to employ and explore the other factors that may be involved in the trajectory of the conditions’ progression. However, the study models included in this systematic review solely utilized transgenic rodent models. Although transgenic models are useful in providing relevant information on the role neuroinflammation played in the pathophysiology of AD, such models do not accurately reflect the initiation and development of commonly seen sporadic AD in patients [60, 61]. Furthermore, the outcome measures were not standardized between the studies as various types of inflammatory markers and methods were used to assess the progression of AD and seizure. In addition, the studies that followed different types of study designs may also partially influence the conditions’ progressions and data presentation potentially causing a disparity in the context of the targeted inflammatory biomolecules.
Conclusion
This systematic review has summarized the evidence supporting the correlation between neuroinflammation in the progression of AD and epilepsy. The prospect of targeting neuroinflammation as a curative approach for both conditions may seem premature at this moment but noticeably promising. However, there is a need for more studies focusing on the common inflammatory biomarkers to develop standardized monitoring guidelines to prevent the manifestation of epilepsy in AD patients as well as AD initiation in epileptic patients.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgements to report.
FUNDING
The project was funded by the Ministry of Higher Education, Fundamental Research Grant Scheme (FRGS/1/2020/SKK0/MUSM/02/6).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
Data sharing is not applicable to this article as no datasets were generated or analyzed during this study.
