Abstract
Background:
Dementia, mainly Alzheimer’s disease (AD) and vascular dementia (VaD), remains a global health challenge. Previous studies have demonstrated the benefits of acupuncture therapy (AT) in improving dementia. Nevertheless, the therapeutic targets and integrated biological mechanisms involved remain ambiguous.
Objective:
To identify therapeutic targets and biological mechanisms of AT in treating dementia by integrated analysis strategy.
Methods:
By the identification of differentially expressed genes (DEGs) of AD, VaD, and molecular targets of AT active components, the acupuncture therapeutic targets associated with the biological response to AD and VaD were extracted. Therapeutic targets-based functional enrichment analysis was conducted, and multiple networks were constructed. AT-therapeutic crucial targets were captured by weighted gene co-expression network analysis (WGCNA). The interactions between crucial targets with AT active components were verified by molecular docking.
Results:
Our results demonstrated that 132 and 76 acupuncture therapeutic targets were associated with AD and VaD. AT-therapeutic crucial targets including 58 for AD and 24 for VaD were captured by WGCNA, with 11 in shared, including
Conclusion:
The improvement of AD and VaD by AT was associated with modulation of synaptic function, immunity, inflammation, and apoptosis. Our study clarified the therapeutic targets of AT on dementia, providing valuable clues for complementing and combining pharmacotherapy.
INTRODUCTION
Dementia is a neurocognitive disorder with difficulties in memory, language, and behavior resulting in impairment of daily activities [1]. As the population growth and aging trend, the global dementia population is estimated to surge from 57.4 million cases in 2019 to 152.8 million by 2050, posing a staggering public health challenge [2]. Alzheimer’s disease (AD) and vascular dementia (VaD) are the predominant subtypes of dementia, which can co-occur as mixed dementia with overlapping risk factors and pathological basis [1]. Despite the indistinction between AD and VaD over clinical symptoms, the pathological characteristics of AD are commonly accompanied by neuritic plaques containing amyloid-
Although mainstream pharmacological interventions including acetylcholinesterase inhibitors and N-Methyl-D-Aspartic acid (NMDA) receptor antagonist have been administered for the management of dementia symptoms, and anti-inflammatory treatments for neuroinflammation have shown potential benefit [4, 7], considering the moderate efficacy, the range of potentially long-term adverse effects, and specific therapeutic medications that remain to be investigated, non-pharmaceutical interventions (NPIs) are also introduced into the palliative and preventive management of dementia, not only to assist pharmacotherapy, but also to reduce the underlying side effects of pharmacological strategy, thus promoting the health of dementia patients in a comprehensive manner [3, 8].
Acupuncture therapy (AT) is one of the widely used traditional medicine therapies in East Asia for more than 2,000 years [9], and the 2019 World Health Organization Global Report indicated that AT is currently the most common form of practice among diverse traditional and complementary medicines, as reported by 113 Member States [10, 11]. Manual acupuncture (MA) and electroacupuncture (EA) are the main types of AT. In MA, with needles inserted directly into acupoints through the skin, intense mechanical stimulation is triggered by manipulation; in EA, needles are electrified to generate dual mechanical and electrical stimulation to acupoints, and the therapeutic effects of acupuncture are exerted with adjustments of parameters including acupoint selection, stimulation method, and stimulation intensity [12, 13]. From 2010 to 2020, 133 guidelines worldwide included 433 AT recommendations, and statistics demonstrated that neurological disorders were among the most frequently recommended areas for AT application [14, 15]. Evidence maps and overview of systematic reviews indicated that the therapeutic scope of AT covers 77 diseases and conditions under 12 areas, and in particular, AT has moderate efficacy and certainty evidence for the improvement on the severity of dementia symptoms by VaD [11, 16]. Three retrospective cohort studies suggested that integrated measures coupled with AT were beneficial in reducing long-term dementia risk in patients, whether after traumatic brain injury or stroke, in comparison to conventional treatment [17–19]. Assessment of randomized controlled trials (RCTs) concerning efficacy and safety indicated that AT positively improves the cognitive function of dementia patients and demonstrated considerable therapeutic effects in combination with conventional medications compared to pharmacological interventions alone, which not only possessed substantial safety but also contributed additionally to overall health [20–22].
Current research evidence demonstrated that AT has diverse functions in modulation of synaptic plasticity, immunomodulation, anti-inflammation, and anti-apoptosis in response to a series of associated active components including acetylcholine (ACh), noradrenaline (NA), dopamine (DA), serotonin (5-hydroxytryptamine, 5-HT), gamma-aminobutyric acid (GABA), brain-derived neurotrophic factor (BDNF), and glial cell-derived neurotrophic factor (GDNF), thus exerting neuroprotective and cognitive regulatory effects [23–33]. However, the shared and mutually independent therapeutic molecular targets and the integrated biological mechanisms of AT on AD and VaD remain ambiguous. Therefore, by conducting an integrated analysis, we aimed to 1) systematically identify the shared and mutually independent therapeutic targets and crucial targets of AT in treating AD and VaD, 2) investigate the shared and mutually independent integrated biological mechanisms of AT on AD and VaD, and 3) explore target interactions and construct networks of AT on AD and VaD, in an effort to contribute to subsequent ongoing studies and to provide informative support for the future inclusion of AT into NPI management strategies for dementia (Fig. 1).
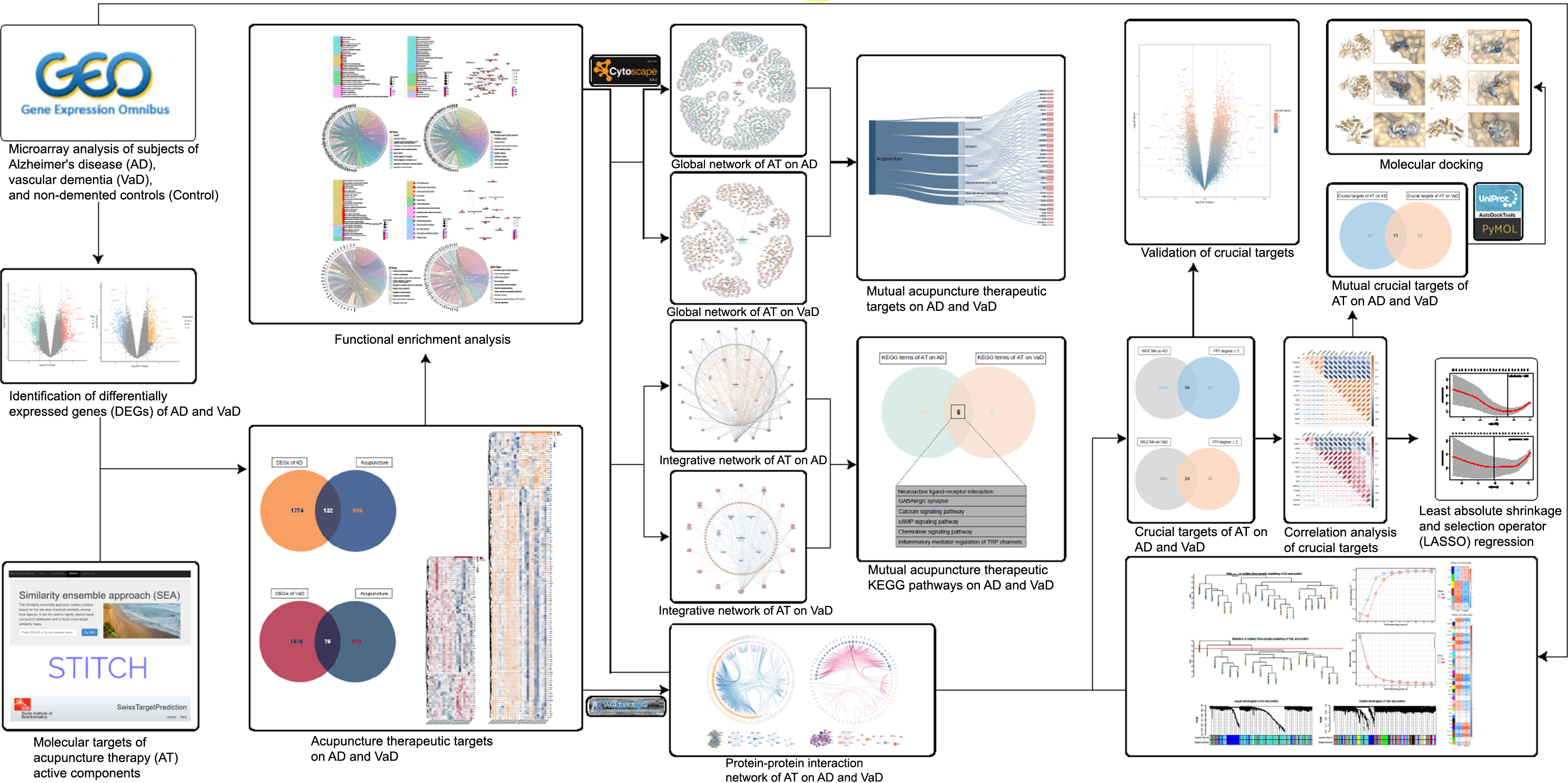
Workflow of this study. This figure demonstrates the process of identification of acupuncture therapeutic targets and multiple molecular mechanisms in treating dementia.
MATERIALS AND METHODS
Datasets search strategy
Datasets relevant to AD and VaD were retrieved from Gene Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/geo/). The inclusion criteria were defined as 1) the dataset needed to contain both AD, VaD, and matched controls to prevent batch differences, 2) the total sample size of controls combined with either AD or VaD was not less than 15, and 3) the organism of the dataset was Homo sapiens. The exclusion criteria were defined as 1) the dataset contained only matched controls with either AD or VaD, 2) the total sample size of controls combined with AD or VaD was less than 15, 3) the data were derived from non-Homo sapiens, 4) the information was incomplete, and 5) the subject had underlying health conditions or was intervened by any measures.
Data preprocessing
Background correction and normalization of the raw data files from the dataset were conducted with methods of normal– exponential convolution and quantile by “limma” R-package. Data quality evaluation was implemented using “arrayQualityMetrics” package, and probe sequence re-annotation was further conducted. After constructing matching index according to the obtained transcripts file Gencode v34, probe sequence file of the dataset was matched to the transcripts for binary alignment (BAM) file with “Rsubread” package, thus extracting information from the BAM file and obtaining matching data with “Rsamtools” package. Finally, normalized and re-annotated gene expression matrix was generated from the included dataset.
Identification of acupuncture therapeutic targets associated with AD and VaD
Dataset GSE122063 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc = GSE122063) contained gene expression profiles by microarray of frontal cortex obtained from individuals who died with AD (
Previous evidence supported the significance of the active components, mainly ACh, NA, DA, 5-HT, GABA, BDNF, and GDNF, in the pathology of dementia, being able to effectively respond to AT stimulation that could generate underlying biological effects through their downstream networks of molecular targets [23–33]. Therefore, the molecular targets of AT active components were extracted with Canonical SMILES (Simplified Molecular Input Line Entry System) from PubChem database (https://pubchem.ncbi.nlm.nih.gov/) through Similarity Ensemble Approach (SEA) [35] (https://sea.bkslab.org), STITCH5.0 [36] (http://stitch.embl.de), and SwissTargetPrediction [37] (http://www.swisstargetprediction.ch) databases. Finally, acupuncture therapeutic targets associated with the biological response to AD and VaD, referred to as ACAgenes and ACVgenes, were identified by extracting the overlap of molecular targets of AT active components and the DEGs of AD or VaD.
Functional enrichment analysis
Functional enrichment analysis of the ACAgenes or ACVgenes was conducted by “clusterProfiler” [38] and “org.Hs.eg.db” packages. The biological process (BP), cellular component (CC), and molecular function (MF) terms of Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) were obtained and ranked under a
Construction of visual networks
The Metascape [40] (https://metascape.org/) database was applied to evaluate the interaction and clustering of the ACAgenes or ACVgenes. After the classification and integration of the results, the protein-protein interaction (PPI) network was constructed with “ggraph” and “igraph” packages in accordance with the degree value, which defines the number of connections between a single node to other nodes, representing the interactive intensity among the nodes in the PPI network. The MCODE module of Cytoscape 3.8.2 (https://cytoscape.org) was utilized to perform the analysis and construct clustering sub-networks. Further, the global network and acupuncture-component-target-pathway integrative network were constructed by the categorization and integration of associated information. Finally, the mutual targets from ACAgenes and ACVgenes were displayed in Sankey diagram with “networkD3” package, and the mutual KEGG terms were presented in Venn diagram.
Weighted gene co-expression network analysis
Weighted gene co-expression network analysis of microarray data of AD, VaD, and non-demented subjects was performed with “WGCNA” [41] package. The genes with a top 10,000 median absolute deviation were preliminarily filtered, and outlier samples were removed.
The co-expression similarity sij was defined by calculating the Pearson’s correlation coefficient between the expression vector xi and xj from gene i and gene j:
Further, the adjacency matrix aij was calculated in the following method:
The value of the adjacency matrix aij representing the strength of network connectivity of gene i and gene j was derived by selecting a reasonable soft-thresholding power (
Subsequently, the modules were identified by hierarchical clustering of the weighting coefficient matrix to convert the adjacency matrix into a topological overlap matrix (TOM), which represents the overlap in shared adjacent genes:
The average linkage hierarchical clustering was conducted according to the TOM-based dissimilarity measure with a minimum gene dendrogram size of 30, and genes with similar expression profiles were grouped into identical gene modules using DynamicTreeCut algorithm. To further diminish the complexity of the network, modules with similarity above 0.75 were merged.
Finally, based on the association of each module with traits (AD or VaD), modules of significant correlations (
Validation of crucial targets
External datasets from the GEO database were applied as validation datasets, thus checking the robustness of the screened crucial targets. Limited by the current lack of external VaD-related datasets in the GEO database, we evaluated only the crucial targets of AT on AD. In the validation dataset GSE150696 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc = GSE150696), the genes involved in AD were identified from gene expression profiles by microarray obtained from individuals who died with AD (
Molecular docking
Molecular docking was conducted to examine the interaction between AT active components and the corresponding mutual crucial targets of AT on AD and VaD. The crucial targets were screened with UniProt [43] database (https://www.uniprot.org) to enroll targets that have original ligand with
Least absolute shrinkage and selection operator (LASSO) regression
LASSO regression was performed with “glmnet” [45] package to generate a series of penalty parameters lambda for the logistic model fitted by the crucial targets based on the binomial distribution traits. By conducting ten-fold cross-validation, the optimal lambda corresponding to the minimum mean of binomial deviance values was detected, thus identifying the core targets among the crucial targets that were more strongly associated with AD or VaD and of greater interest, whose regression coefficients that were not penalized to zero under the assurance of the optimal lambda.
RESULTS
Identification of acupuncture therapeutic targets associated with AD and VaD
By differential gene expression analysis, we identified 1,906 and 1,692 DEGs for AD and VaD, respectively. In comparison to controls, 805 of the DEGs for AD were upregulated and 1,101 were downregulated (Fig. 2A, Supplementary Table 1), while 844 of the DEGs for VaD were upregulated and 848 were downregulated (Fig. 2B, Supplementary Table 1).
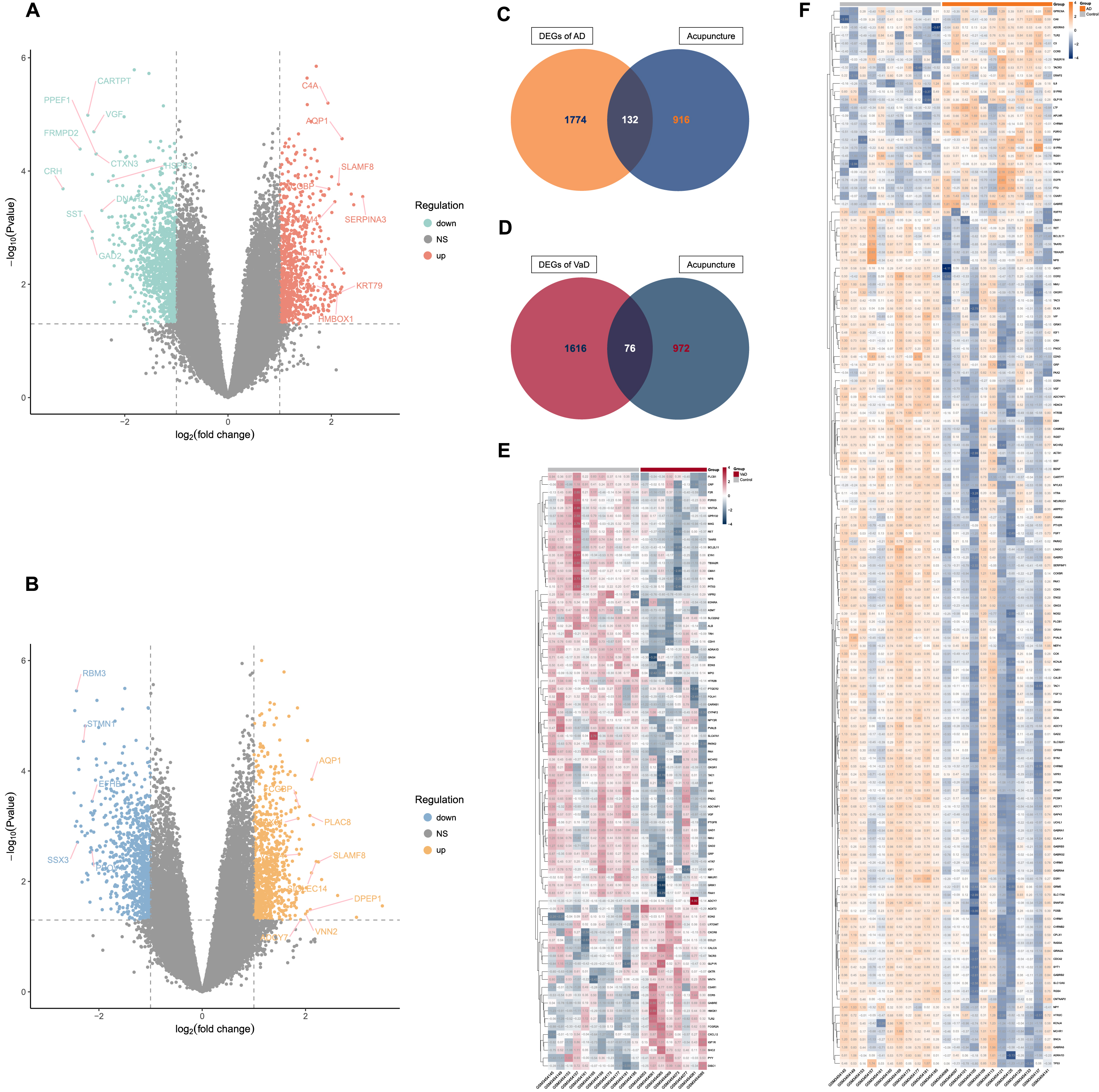
Identification of acupuncture therapeutic targets of Alzheimer’s disease (AD) and vascular dementia (VaD). A) Volcano plot that shows the differentially expressed genes (DEGs) between non-demented control group and AD group. B) Volcano plot that shows the DEGs between non-demented control group and VaD group. C) Venn diagram depicts the 132 overlapped acupuncture therapeutic targets on AD (ACAgenes) between the 1,906 DEGs and the 1,048 molecular targets of acupuncture therapy (AT) active components. D) Venn diagram depicts the 76 overlapped acupuncture therapeutic targets on VaD (ACVgenes) between the 1,692 DEGs and the 1,048 molecular targets of AT active components. E) Heatmap of the 76 ACVgenes in 19 samples. “VaD” group stands for vascular dementia, “Control” group stands for non-demented controls. F) Heatmap of the 132 ACAgenes in 23 samples. “AD” group stands for Alzheimer’s disease, “Control” group stands for non-demented controls.
Further, a total of 1,048 molecular targets of AT active components were collected from databases of SEA, STITCH5.0, and SwissTargetPrediction (Supplementary Table 2).
When the DEGs of AD and VaD were compared against the molecular targets of AT active components, 132 ACAgenes and 76 ACVgenes were retained respectively, which were acupuncture therapeutic targets associated with the biological response to AD and VaD (Fig. 2C-F, Supplementary Table 3).
Functional enrichment analysis of acupuncture therapeutic targets associated with AD and VaD
A total of 905 GO terms (Supplementary Table 4) including 749 BPs, 79 CCs, and 77 MFs were enriched and 35 KEGG terms (Supplementary Table 4) were identified from the 132 ACAgenes, and a total of 585 GO terms (Supplementary Table 4) including 530 BPs, 8 CCs, and 47 MFs were enriched and 14 KEGG terms (Supplementary Table 4) were identified from the 76 ACVgenes. The cluster tree plots systematically demonstrated the top 30 BP and KEGG terms (total 14 KEGG terms for VaD) of AT on AD and VaD (Fig. 3A, B, F, G). The complex interactions among KEGG terms were shown as enrichment map plots (Fig. 3C, H).
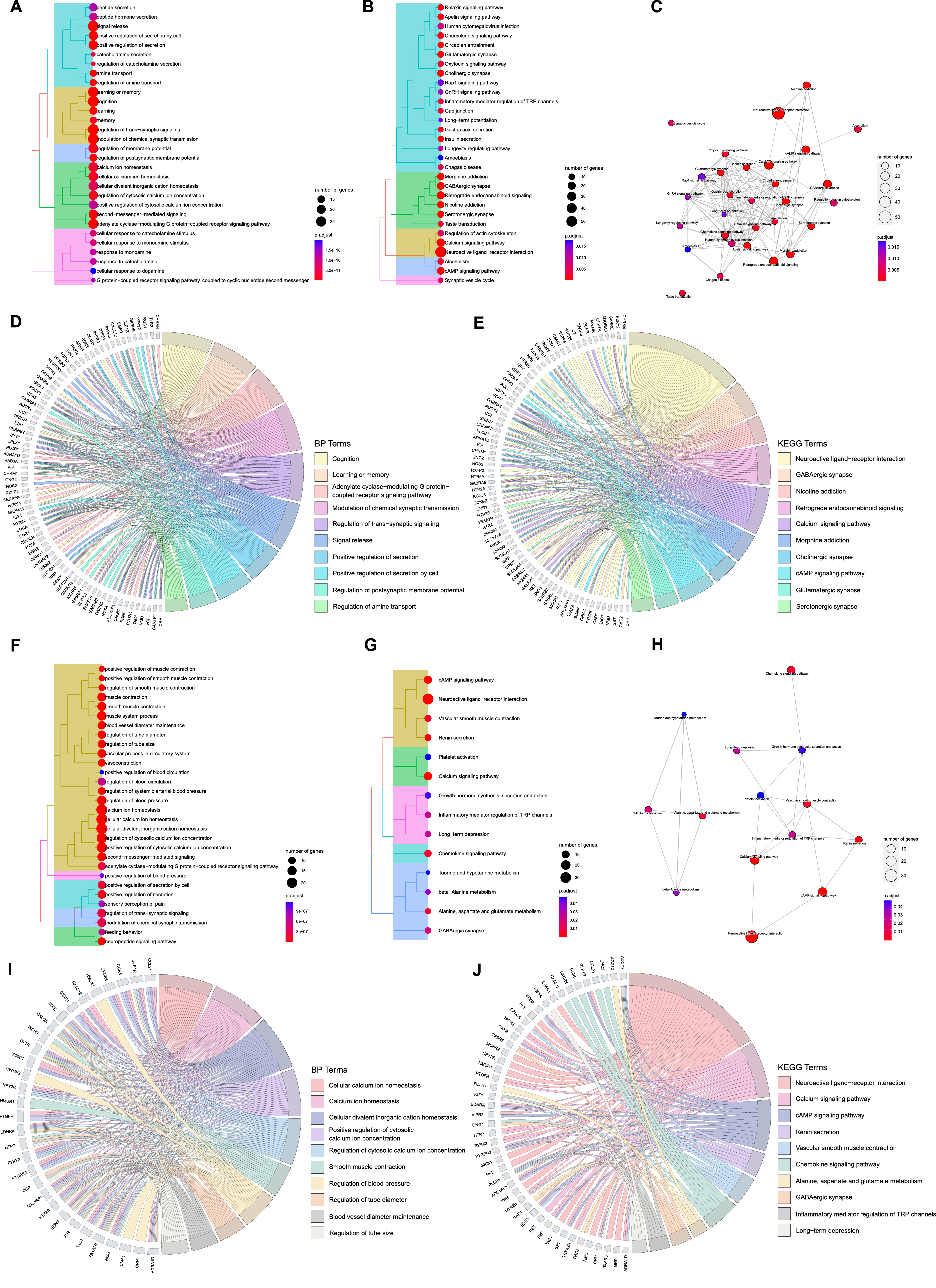
Functional enrichment analysis of acupuncture therapeutic targets associated with Alzheimer’s disease (AD) and vascular dementia (VaD). A) Tree plot of top 30 biological process (BP) terms of acupuncture therapy (AT) on AD (
Then, we focused on the more concerning top 10 terms of BP and KEGG with their corresponding targets, as revealed in chord plots (Fig. 3D, E, I, J). For AT treating AD, ACAgenes including
Among the top 10 terms in the results of KEGG analysis, we found that ACAgenes such as
Construction of visual networks based on acupuncture therapeutic targets associated with AD and VaD
The interactions among the differentially expressed 132 ACAgenes and 76 ACVgenes were revealed in the PPI network (Fig. 4A, B). With the successive increment of degree values, both the size of nodes enlarges and the density of edges enhances correspondingly, which leads to gradual enhancement of the interaction intensity on targets. As shown by the degree value ranking of 118 out of 132 ACAgenes,
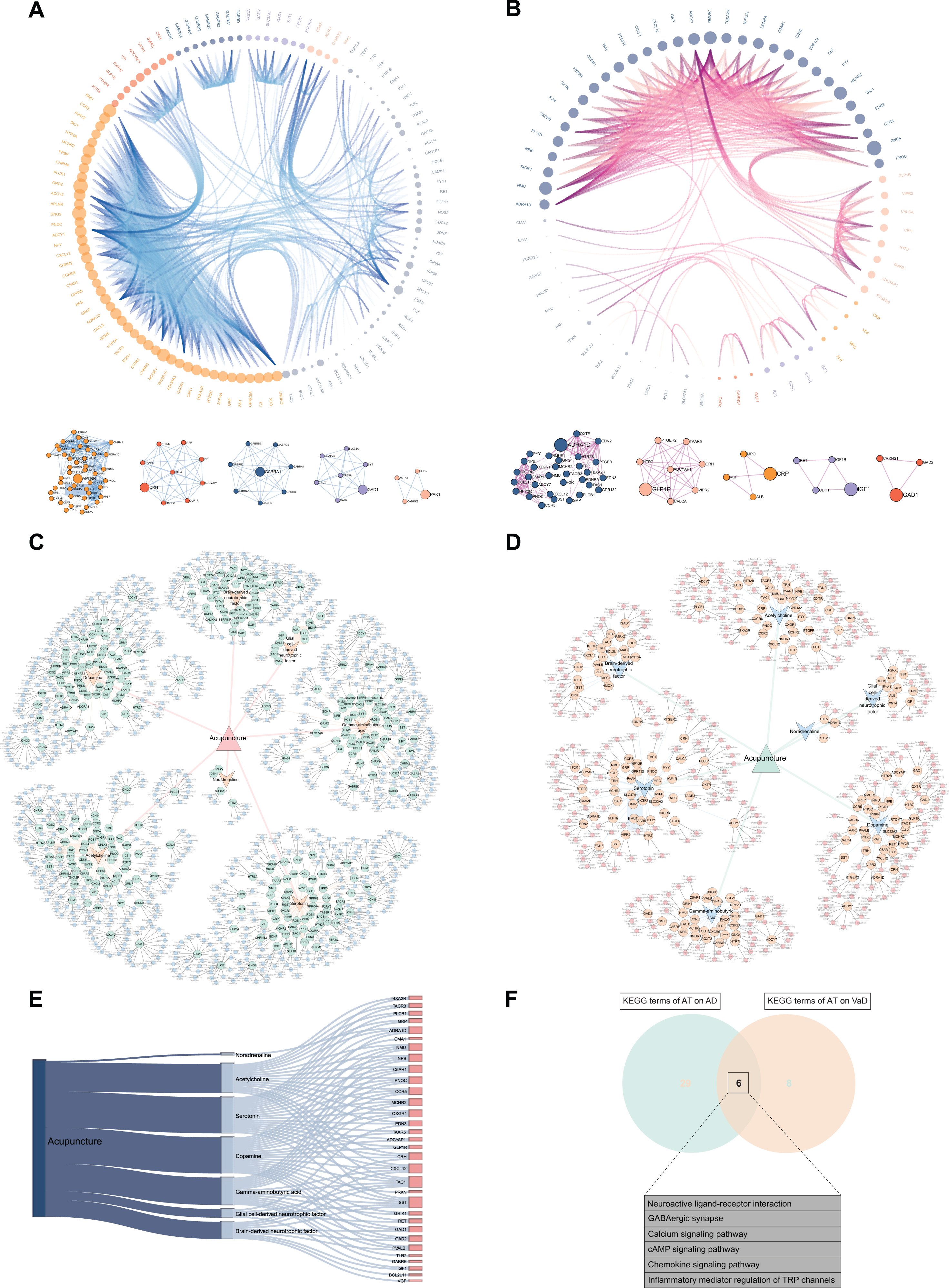
Construction of visual networks based on acupuncture therapeutic targets associated with Alzheimer’s disease (AD) and vascular dementia (VaD). A) PPI network and clustering sub-networks of acupuncture therapy (AT) on AD. In this network, the interaction of 118 targets in PPI network out of 132 acupuncture therapeutic targets on AD (ACAgenes) is displayed. According to the increasing degree value from 1.00 to 57.00, the size of nodes and the density of edges correspondingly increase, leading to gradual enhancement of the interaction intensity on targets, and 5 clustering networks driven by 5 hub genes are further identified. B) PPI network and clustering sub-networks of AT on VaD. In this network, the interaction of 66 targets in PPI network out of 76 acupuncture therapeutic targets on VaD (ACVgenes) is displayed. The degree values of targets increase from 1.00 to 38.00, and 5 clustering networks driven by 5 hub genes are further identified. C) Global network of AT on AD. In this network, the associations of the 7 acupuncture active components, 132 ACAgenes, and the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways related to each target are systematically displayed. D) Global network of AT on VaD. In this network, the associations of the 7 acupuncture active components, 76 ACVgenes, and the KEGG pathways related to each target are systematically displayed. E) Sankey diagram of the 32 mutual targets from ACAgenes and ACVgenes. F) Venn diagram depicts KEGG terms sharing of AT on AD and VaD.
Screening of crucial targets by performance of weighted gene co-expression network analysis
WGCNA was conducted to further screen out crucial targets for AT on AD and VaD. The genes with the top 10,000 median absolute deviations were filtered, followed by the removal of 0 and 2 outlier samples for the analysis of AD or VaD with controls respectively. Finally, 23 and 17 clustered samples were extracted to construct the respective matrices of AD or VaD with controls (Fig. 5A, B).
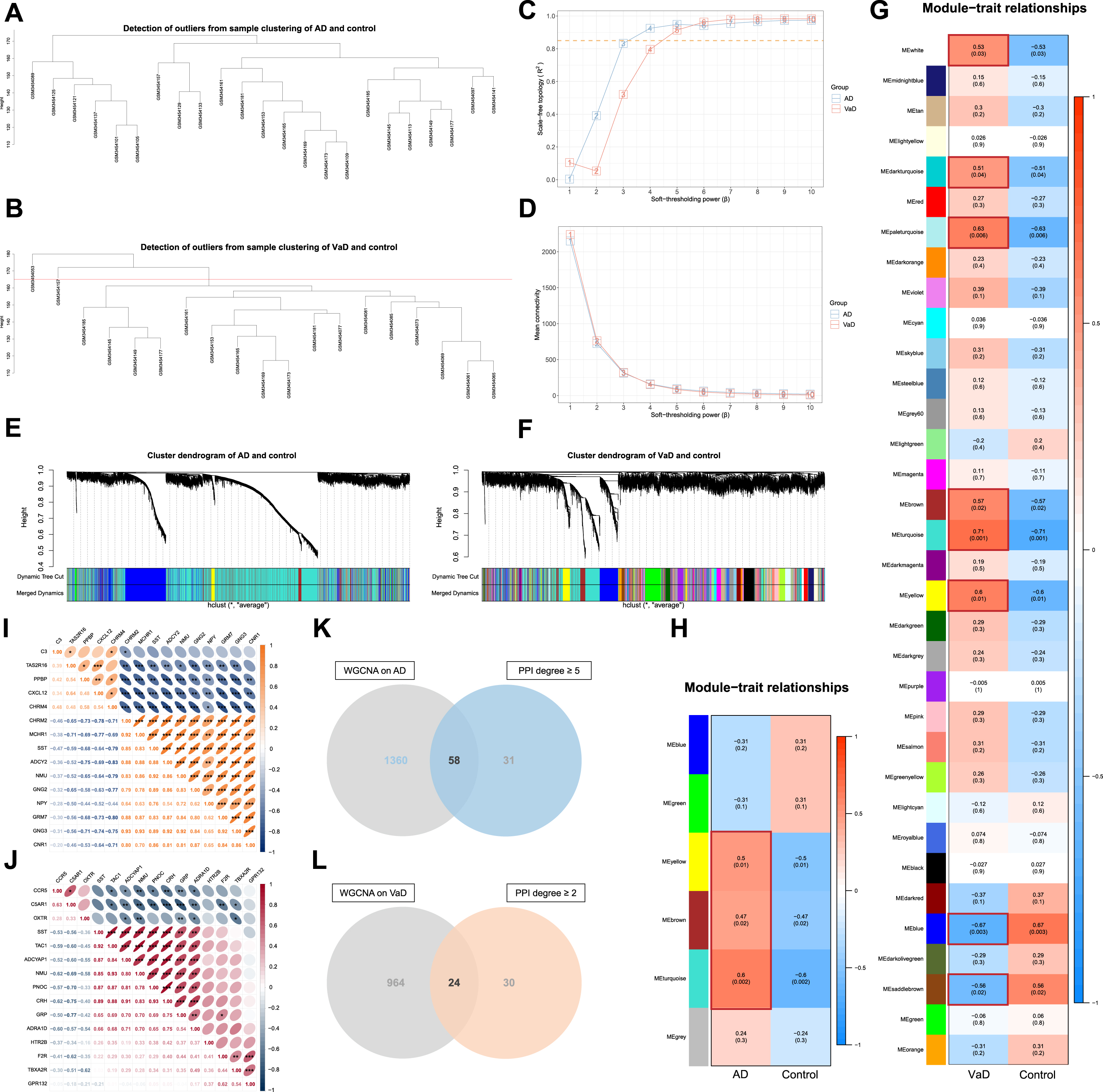
Screening of crucial targets by performance of weighted gene co-expression network analysis (WGCNA). A) Cluster analysis of samples from Alzheimer’s disease (AD) and controls to detect outliers. B) Cluster analysis of samples from vascular dementia (VaD) and controls to detect outliers. C) Determination of soft-thresholding power
Subsequently, each adjacency matrix aij was calculated from the soft-thresholding power (
Hierarchical clustering of average linkage was performed according to the dissimilarity that was measured based on the TOM with a minimum size of 30 for the gene dendrogram, and unclustered genes were grouped into grey modules (MEgrey). A total of 6 and 34 modules of AD and VaD were identified by DynamicTreeCut algorithm, and the modules required no merging due to the similarity less than 0.75 (Fig. 5E, F, Supplementary Figure 2A, B). The TOM network heatmaps were displayed by randomly selected genes (Supplementary Figure 2C, D).
Finally, modules of significant correlations (
Validation of screened crucial targets
The analysis of differential gene expression for the validation dataset indicated a total of 13,536 significant DEGs in AD compared to controls (
Molecular docking of AT active components with crucial targets
A total of 11 mutual crucial targets of AT on AD and VaD were extracted (Fig. 6A). including
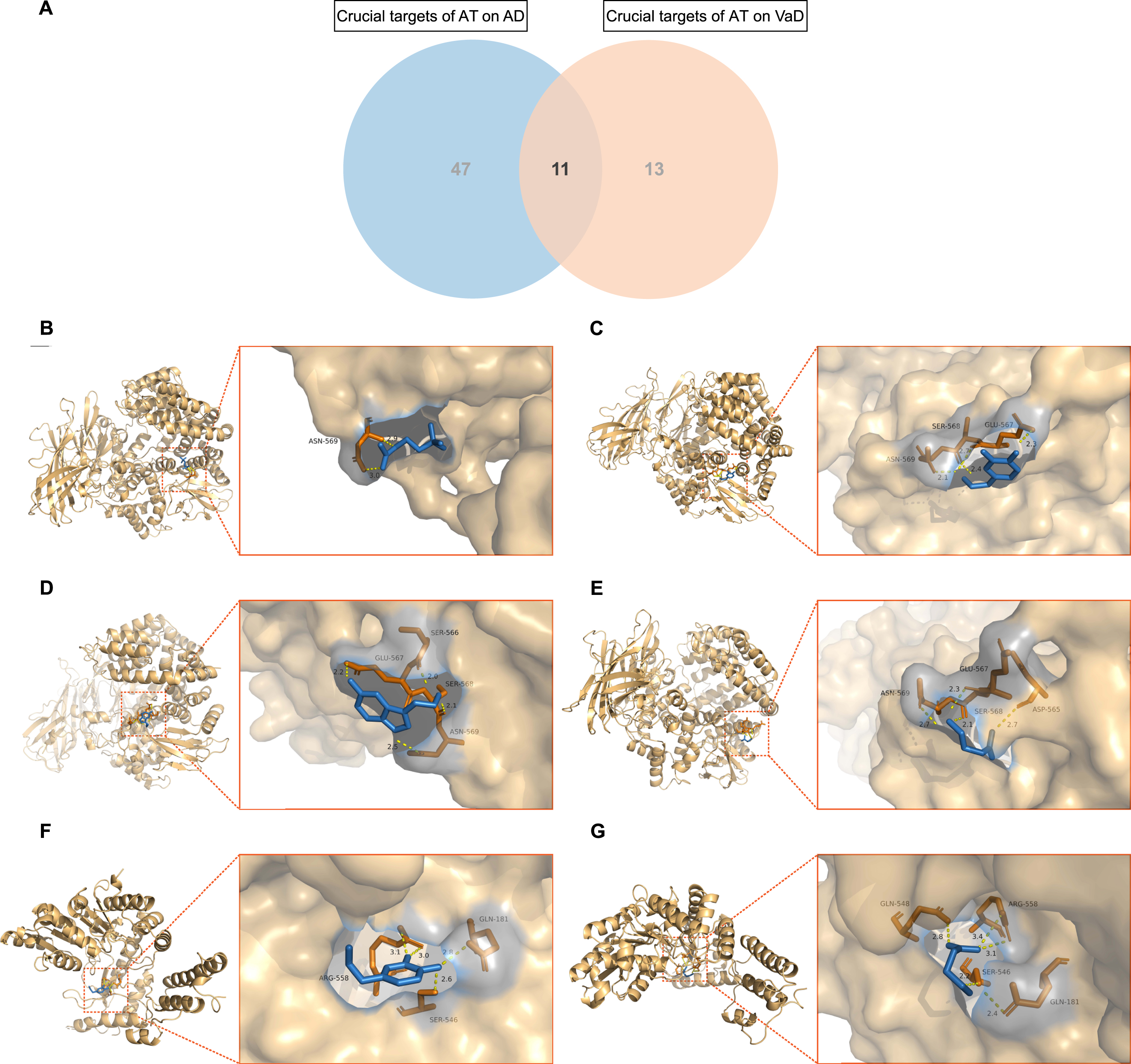
Molecular docking of acupuncture therapy (AT) active components with crucial targets. A) Venn diagram depicts the 11 mutual crucial targets of AT on Alzheimer’s disease (AD) and vascular dementia (VaD). B) Acetylcholine (ACh) formed 2 hydrogen bonds with amino acid residue ASN-569 of TAC1. C) Dopamine (DA) formed 4 hydrogen bonds with amino acid residues ASN-569, SER-568, and GLU-567 of TAC1. D) Serotonin (5-hydroxytryptamine, 5-HT) formed 4 hydrogen bonds with amino acid residues GLU-567, SER-566, SER-568, and ASN-569 of TAC1. E) Gamma-aminobutyric acid (GABA) formed 4 hydrogen bonds with amino acid residues ASN-569, GLU-567, SER-568, and ASP-565 of TAC1. F) DA formed 4 hydrogen bonds with amino acid residues ARG-558, GLN-181, and SER-546 of GAD2. G) GABA formed 5 hydrogen bonds with amino acid residues GLN-548, ARG-558, GLN-181, and SER-546 of GAD2.
Identification of core targets by LASSO
Machine learning algorithm of LASSO regression was used to identify the core targets for AT on AD and VaD from the series of crucial targets. The optimal penalty parameter lambda corresponding to the minimum mean of binomial deviance values by LASSO regression in AD or VaD was detected to retain groups of core targets, as 6 core targets including
DISCUSSION
Previous studies on AT for dementia have tended to be either based on the evaluation of efficacy in clinical patients or in-depth exploration of a single mechanism based on animal models, but systematic multi-target and multi-mechanism studies of AT on dementia patients remain limited. Here, for the translation significance of our findings, we started from the microarray of dementia patients via data mining, combined with the AT-associated active components identified in animal models, to reveal the therapeutic targets and biological integrated mechanisms of AT by bioinformatics and network topology approaches. Our results indicated that multiple genes and signaling pathways associated with the regulation of synaptic function, immunity, and inflammation are simultaneously and potentially involved in the shared or mutually independent biological processes of AT on AD and VaD, which depend on the initial participation of a series of active components.
In previous RCTs, AT has demonstrated benefits in enhancing cognition of patients with cognitive impairment, potentially contributing to preventing or delaying the onset of AD and VaD [46, 47], and assisting pharmacotherapy to jointly improve dementia symptoms and overall clinical status, serving as a safe and effective NPI [20–22]. The active components underlying the therapeutic mechanism of AT were revealed in a series of animal models, mainly involving neurotransmitters including ACh, NA, DA, 5-HT, GABA, and neurotrophic factors including BDNF and GDNF [23–33]. In our analysis, we first collected and integrated the downstream molecular targets of these AT active components through multiple databases and compared them with the DEGs of AD and VaD to screen for therapeutic targets on which AT has potential impact, the 132 ACAgenes and 76 ACVgenes.
Then a range of therapeutic mechanisms of AT were revealed via functional enrichment analysis on ACAgenes and ACVgenes, in which the KEGG terms shared including neuroactive ligand-receptor interaction, calcium (Ca2 +) signaling pathway, cAMP signaling pathway, chemokine signaling pathway, GABAergic synapse, and inflammatory mediator regulation of transient receptor potential (TRP) channels. Ca2 + signaling pathway is intimately involved in neurotransmitter release, synaptic plasticity, and other crucial neuronal functions through binding to calmodulin (CaM), triggering the activation and conformational changes of Ca2 + /calmodulin dependent protein kinase II (CaMKII) and CaMKIV, thus serving in synaptic strengthening and regulating the transcription of cAMP response element-binding protein, participating in memory formation [48]. In previous studies, physical stimulation generated by MA can be perceived by TRP vanilloid receptors 1 (TRPV1) to modulate Ca2 + permeability and transmit signals to nerve terminals via calcium wave propagation [49], while EA stimulation can be beneficial to considerably upregulating CaM1, CaMKII, and CaMKIV in the hippocampus and modulate cognitive functions [50]. In AD with excessive A
By further constructing PPI networks of ACAgenes and ACVgenes and performing WGCNA, the crucial targets of AT on AD or VaD in the global targets were captured. In particular, 11 shared crucial targets were identified, including
This study has several strengths. We conducted an integrated analysis of AT in treating AD and VaD to identify shared and mutually independent therapeutic targets and biological mechanisms to provide a comprehensive perspective on the potential therapeutic effects of AT. Moreover, we conducted a robust screening of therapeutic targets by constructing PPI networks in combination with WGCNA and LASSO regression, thus identifying a series of crucial and core targets of AT in treating AD and VaD, and performing targets validation to provide informative support for further studies. Nevertheless, this study has some limitations. First, we only investigated several crucial active components associated with the therapeutic effects of acupuncture, but other active components involved in acupuncture treatment remain to be explored by more high-quality evidence. Then, the databases that were used in this study are subject to continuous updates, so more functional or expression-related hits or targets remain to be supported by further studies. Moreover, relevant datasets with larger sample sizes are limited, and due to the majority of studies focusing on AD and the remaining limited datasets of VaD, we validated only the crucial targets of AT in treating AD. Additionally, the therapeutic effects of acupuncture are complicated by multidimensional parameters including acupoint selection, manipulation method, stimulation intensity, and session, and the heterogeneity in acupuncture prescriptions also induces variation. Thus a detailed classified analysis according to the characteristics of concrete acupuncture prescriptions is desirable.
In conclusion, AT, in response of diverse crucial active components (ACh, NA, DA, 5-HT, GABA, BDNF, GDNF) with multiple pathways, effectively modulates the downstream molecular targets network, exerts synaptic plasticity regulatory, immunomodulatory, anti-inflammatory, and anti-apoptotic functions, and alleviates the symptoms of dementia. As an effective and safe NPI, AT holds additional and considerable benefits in avoiding or mitigating the possible long-term side effects of pharmacological therapy, assisting the current management strategy, as well as improving the patients’ quality of life and relieving the pressure on the health systems.
DATA AVAILABILITY
The gene expression profiles by microarray that support the findings of this study are available in the datasets (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc = GSE122063) and (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc = GSE150696) from the Gene Expression Omnibus (GEO) database(https://www.ncbi.nlm.nih.gov/geo/). The molecular targets of acupuncture active components are available in the Similarity Ensemble Approach (https://sea.bkslab.org), STITCH 5.0 (http://stitch.embl.de),and SwissTargetPrediction (http://www.swisstargetprediction.ch). The information on protein-protein interaction is available from the Metascape (https://metascape.org/). The structures of targets can be freely downloaded from the UniProt database (https://www.uniprot.org). The Cytoscape 3.8.2 (https://cytoscape.org), AutoDockTools 1.5.6 (https://ccsb.scripps.edu/mgltools/), AutoDock Vina (https://vina.scripps.edu/), and PyMOL 2.4.0(https://github.com/schrodinger/pymol-open-source) are all open-source software that can be freely downloaded for academic use to construct and to visualize networks and molecular docking respectively. All other data are available within the article, Supporting Information is available from authors upon request.
Footnotes
ACKNOWLEDGMENTS
The authors gratefully acknowledge the developers involved and the researchers sharing the data.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
