Abstract
Alzheimer’s disease (AD), a progressive dementia, is one of the world’s most dangerous and debilitating diseases. Clinical trial results of amyloid-β (Aβ) and tau regulators based on the pretext of straightforward amyloid and tau immunotherapy were disappointing. There are currently no effective strategies for slowing the progression of AD. Further understanding of the mechanisms underlying AD and the development of novel therapeutic options are critical. Neurogenesis is impaired in AD, which contributes to memory deficits. Transplanted neural stem cells (NSCs) can regenerate degraded cholinergic neurons, and new neurons derived from NSCs can form synaptic connections with neighboring neurons. In theory, employing NSCs to replace and restore damaged cholinergic neurons and brain connections may offer new treatment options for AD. However there remain barriers to surmount before NSC-based therapy can be used clinically. The objective of this article is to describe recent advances in the treatment of AD models and clinical trials involving NSCs. In addition, we discuss the challenges and prospects associated with cell transplant therapy for AD.
BACKGROUND
Alzheimer’s disease (AD), characterized by progressive dementia, is one of the most life-threatening and burdensome illnesses [1]. Based on the age of onset, AD can be divided into two distinct groups: cases occurring after age 65 are classified as late-onset AD, whereas cases occurring before age 65 are classified as early-onset AD and account for less than 5% of all cases. The majority of cases are characterized by sporadic onset, while 1%–2% of cases are inherited in an autosomal dominant manner [2]. Although countless research has been conducted on AD pathogenesis, there remains a lack of effective disease-modifying therapies [3, 4]. Recently approved pharmacological treatments include the N-methyl-D-aspartate receptor antagonist memantine and cholinesterase inhibitors, which merely relieve associated symptoms [2, 5–7]. Current promising pharmaceutical treatments include anti-inflammatory, anti-amyloid, and immunomodulator therapies [6]. In addition, monoclonal antibodies capable of serving as disease-modifying drugs are being developed and tested, such as bapinezumab, solanezumab, crenezumab, and aducanumab [2]. Among these drugs, only aducanumab, which targets conformation-specific amyloid-β (Aβ) aggregates, has been approved by United States Food and Drug Administration (FDA) [8]. With recent data indicating that the global dementia population will triple by 2050 [6], further understanding of the mechanisms underlying AD and the development of novel therapeutic options are critical.
Tremendous investments have been made to inhibit processes underlying AD, although the results are mixed [1]. There are still no effective ways to curb AD progression [9]. Recently accumulated evidence suggests that stem cell transplantation may aid in maintaining brain homeostasis and enhancing cognition, learning, and memory [10–16]. Transplanted neural stem cells (NSCs) have been demonstrated to regenerate damaged cholinergic neurons, and NSC-derived neurons can form synaptic connections with nearby neurons, suggesting a potential strategy to slow the progression of AD [10]. Simultaneously, neurotrophic support, immunomodulation, and anti-inflammatory effects play a critical role in hindering AD progression [10]. Here, we evaluate recent research on NSC transplantation in AD mouse models and address the obstacles and potential benefits of cell transplant therapy for AD.
PATHOGENESIS AND PATHOPHYSIOLOGY OF AD
The pathophysiology of AD is still a mystery. The amyloid cascade hypothesis has been challenged because it provides no explanation for the silent period of AD [17]. Although the pathological criteria for AD diagnosis remain the formation of extracellular Aβ plaques and intracellular tau accumulation [2], clinical trial results for regulators of Aβ and tau under the guise of straightforward amyloid and tau immunotherapy were disappointing [18–22]. Accordingly, some scholars believe that the focus on Aβ peptide may be inappropriate [1]. Indeed, recent evidence indicates that the real cause of AD may occur upstream of Aβ and tau proteinopathies [17].
Like Aβ and tau, neuroinflammation plays a crucial role in AD pathogenesis [23, 24]. In an intriguing study [25], an analysis of gene expression profiles in the brains of healthy individuals and patients with AD of a variety of ages was carried out. This research found that the rise in expression of immune- or inflammation-related genes with age was considerably greater in patients with AD compared with healthy individuals. The brain becomes more prone to inflammatory processes as it ages, which may explain why ageing is the major driver of AD [17, 26].
Some scientists hold the view that ageing and age-related neurodegeneration, including AD, are characterized by the accumulation of damaged mitochondria [27, 28]. Indeed, mitochondrial dysfunction is central to the pathophysiology of AD. Specifically, mitochondrial dysfunction, such as dysfunction of mitochondrial axonal transport and mutations in mitochondrial DNA, plays imperative roles in the pathogenesis of AD that may result in oxidative stress, ATP depletion, and synaptic dysfunction [29, 30]. Mitophagy enhancement is a possible therapeutic option for AD-related tau hyperphosphorylation because it can increase microglial phagocytosis and decrease neuroinflammation [28].
Because amyloid accumulation may result from vascular defects, the vascular hypothesis has been proposed, which posits that breakdown of the blood-brain barrier (BBB) results in neurotoxic serum protein accumulation and synaptic dysfunction, ultimately leading to defects in Aβ and tau clearance [17, 31]. Numerous risk loci for AD have been identified by genome-wide association studies (GWAS) [32]. However, even after many large-scale GWAS, the apolipoprotein E (
In addition, abnormalities in neuronal cholesterol homeostasis and fatty acid metabolism seem to contribute to the induction of AD. Oxidized cholesterol is widely acknowledged as one of the main triggers of AD [39, 40]. Cholesterol 24-hydroxylase and cholesterol 27-hydroxylase (encoded by
Collectively, the results described above reveal that many pathophysiological processes, including neuroinflammation, abnormal microglia activation, metabolic failure, oxidative stress, and sustained cholesterol-associated neuronal distress, may be potential targets for intervention [18].
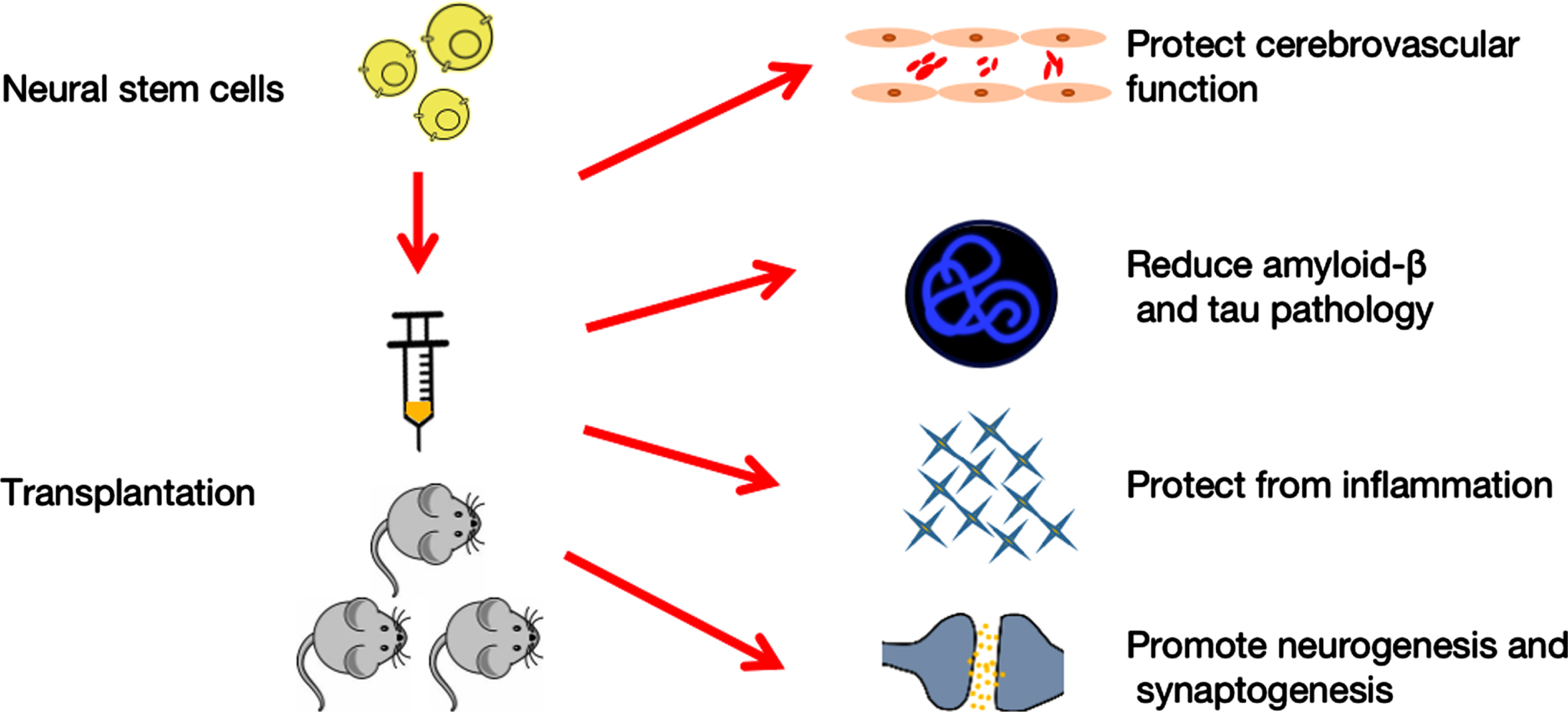
Neural stem cells therapy for Alzheimer’s disease. Transplantation of neural stem cells protects cerebrovascular function, decreases amyloid-β and tau pathology, decreases inflammation, and enhances neurogenesis and synaptogenesis, all of which affect behavioral performance.
Accumulated evidence has confirmed that AD is a complex disease with different interacting phases. Basic scientists divide AD into biochemical, cellular, and clinical phases [17]. Before cognitive symptoms are observed, alterations in cells drive the insidious progression of illness in the cellular phase [6, 17]. Irreversible and significant synaptic and neuronal loss occur during the preclinical phase [41]. Once homeostasis of the cellular phase is broken, the clinical phase is initiated [17]. Preclinical research is continually generating fresh information about parts of the intricate AD jigsaw, and analysis of this data may show patterns of pharmacological interactions rather than single prospective therapeutic targets [3].
NEURAL STEM CELL THERAPY FOR AD
Currently approved pharmacological treatments merely relieve associated symptoms, with a distinct lack of disease-modifying therapies. The disappointing results of Aβ-targeting therapies suggest the inadequacy of single-directed approaches for treatment of AD, while treatments addressing multiple mechanisms should be promoted. Recently, stem cell transplantation has received growing attention for the treatment of neurodegenerative diseases. Such therapies provide the possibility to target multiple mechanisms, potentially including the repair of damaged synapses, modulation inflammation, and neuroprotection through neurotrophic secretion. At present, the most often used stem cell types for the treatment of neurodegenerative illnesses are NSCs, mesenchymal stromal cells (MSCs), and induced pluripotent stem cells (iPSCs) [10]. In particular, NSC transplantation is a potential therapy for AD with tremendous therapeutic potential, such as protection of cerebral capillaries, attenuation of Aβ and tau pathologies, reduction of inflammation, and strengthening of neurogenesis. In addition, NSCs have demonstrated efficacy in treating various preclinical neurodegenerative models and the capacity to develop into central nervous system (CNS)-relevant cell types, despite the ongoing controversy over which kind of stem cell should be used to treat patients suffering from CNS injury [42]. Indeed, NSCs have been transplanted into patients with amyotrophic lateral sclerosis and Parkinson’s disease, and the results were promising [43, 44]. NSCs are ideal candidates for cell transplantation because they have a definite destiny and are relatively similar to functional neuronal types throughout their differentiation.
Cholinergic system impairment is expected to have a significant role in the cognitive and functional difficulties associated with AD [45]. In theory, employing NSCs to replace and restore damaged cholinergic neurons and brain connections may provide a new treatment option for patients with AD.
EFFECTS ON CEREBROVASCULAR FUNCTION
Several investigations have identified relationships between cerebrovascular dysfunction and a variety of AD features [46–48]. However, there is still debate about whether BBB malfunction is merely a consequence of inflammation and Aβ, or whether it is the first pathophysiological step leading to AD [46, 49]. Aβ can be cleared from the CNS into the circulatory system via the BBB and blood-cerebrospinal fluid barrier [46]. Thus, proper function of the BBB and cerebrovasculature is critical for the clearance of Aβ [17, 46]. Moreover, the protection of BBB functioning may promote Aβ clearance to curb AD progression [46]. The underlying vasculoprotective mechanism could be triggered by epidermal growth factor receptor (EGFR) secretion from transplanted genetically modified NSCs [46]. In addition, NSCs have the ability to reduce cerebral amyloid angiopathy and protect cerebral capillaries, especially if they are modified to use neprilysin (NEP) or vascular endothelial growth factor (VEGF) [46].
ATTENUATING AMYLOID-β AND TAU PATHOLOGY
The reduction in Aβ burden following NSC transplantation could be related to lower β-secretase concentrations [50], as well as a much higher presence of NEP [46]. Other pathways could include inducing microglial recruitment and activation, as well as triggering of immune responses to facilitate efficient Aβ phagocytosis and clearance from the CNS [46]. Although research has demonstrated that transplanted human NSCs (hNSCs) in the lateral ventricles of mice can effectively reduce levels of tau phosphorylation [51], the underlying mechanism remains unclear [46]. Neurotrophic and degradation enzymes produced by transplanted NSCs could prevent additional cognitive decline as the disease advances. In fact, clearance of Aβ may only have a minimal impact on global improvements because the environment generally remains conducive for Aβ production and aggregation [52].
ATTENUATING INFLAMMATION
Age-related changes in low-grade chronic inflammation may assist the neurodegenerative process in AD [53]. Transplantation of NSCs for AD appears to have an anti-inflammatory effect in preclinical studies [46]. Transplanted NSCs have been shown to lower pro-inflammatory cytokines including interleukin 1 (IL-1), IL-6, tumor necrosis factor α, and prostaglandin E2, as well as molecular markers associated with a pro-inflammatory environment like Toll-like receptor 4 (TLR-4) [54]. However, decreased inflammation seems to have nothing to do with Aβ levels [46]. Despite reduced inflammation and enhanced cognitive function, Aβ levels remained unchanged following NSC therapy [54]. However, unlike the former study, other research showed that mouse-derived NSCs can switch microglia states from pro-inflammatory to anti-inflammatory, resulting in bidirectional regulation of inflammatory cytokine levels [55].
EFFECTS ON NEUROGENESIS AND SYNAPTOGENESIS
The hippocampus has an imperative role in adult neurogenesis [56] and is one of the most affected areas in AD [57]. In the adult hippocampus, NSCs residing in the subgranular zone (SGZ) of the dentate gyrus (DG) can continually generate new neurons. The surviving cells later integrate into existing neuronal circuits. Abnormal neurogenesis in the adult hippocampus is an early important event in the progression of AD [58]. Dysfunctional neurogenesis may aggravate neuronal vulnerability to AD and lead to cognitive impairment [3]. There are still debates about the neurogenesis in humans. Some believe, for instance, that human hippocampal neurogenesis declines significantly from childhood to adulthood, reaching undetectable levels in adults [59]. Others maintain that human neurogenesis persists throughout the aging process [60]. Even in the face of controversial studies on human neurogenesis, the ability to stimulate NSCs to generate new neurons is a promising prospect for neuroreplacement therapy in AD. Multiple molecular players can attune adult hippocampal neurogenesis (AHN), mainly presenilin (PS1),
Recently accumulated evidence indicates a critical role of NSCs in neurogenesis and synaptogenesis [46]. Multiple gene products are synthesized by NSCs, including brain-derived neurotrophic factor (BDNF) and VEGF, which have paracrine effects that may attenuate AD-related neuronal loss and improve cognition [46]. Blurton-Jones et al. used triple-transgenic mice (3xTg-AD) to investigate the effect of NSC transplantation on AD-related cognitive dysfunction [62]. Their findings showed that instead of reducing Aβ and tau pathology, improved cognition resulted from a significant increase in hippocampal synaptic density mediated by BDNF [62]. Thus, both neurotrophin and BDNF released by transplanted NSCs increased synapse density and reversed cognitive impairments [62]. This study, however, did not investigate the effects of transplanted NSCs on AHN, which may benefit from increased axonal outgrowth or synaptic recovery.
NEURAL STEM CELL TRANSPLANTATION IN MOUSE MODELS
Although an increasing number of researchers are investigating the involvement of pathogenic proteins in the development of AD using animal models, the physiological relevance of these models to humans is debatable because they have not yet fully recapitulated human AD [52, 63–65]. Multiple limitations exist in many of these models, such as late onset of pathology, diverse genetic backgrounds, breeding issues, gender disparities in pathology, and substantial variability in Aβ levels [66]. As a result, exact characterizations of the favorable benefits of NSC transplantation are difficult [52]. The development of a transgenic murine model that overcomes the majority of these difficulties is critical. To mimic the microenvironment of patients with AD, 3xTg, Tg2576, APP/PS1, and 5xFAD mice are frequently employed [52] (Table 1).
Various mouse models to mimic AD
In 2003, Oddo et al. developed the 3xTg as a triple-transgenic AD animal model [67]. The model has APP Swedish, MAPT P301 L, and PSEN1 M146 V transgenes associated with familial Alzheimer’s disease (FAD). As the first transgenic AD model to display Aβ aggregation and neurofibrillary tangles derived from hyperphosphorylated tau protein, the 3xTg mouse model represents a breakthrough in the field of AD research. In contrast to the 3xTg model, Tg2576 mice express exclusively human APP Swedish. These mutations result in a substantial rise in the amount of Aβ generated, allowing mice to exhibit progressive pathogenic protein buildup and behavioral impairments despite the absence of neurofibrillary tangles or severe neuronal loss [52]. The APP/PS1 mouse model is one of the most widely used AD rodent models. Several human APP genes, including the Swedish mutation and
Here, we reviewed recent studies related to transplantation of NSCs in AD mouse models and summarized the details of transplantation, intervention targets, and effects (Table 2).
Neural stem cells transplantation in mice models
Aβ, amyloid β; GFP, green fluorescent protein; MWM, Morris water maze; NOR, novel object recognition; ChAT, cholinergic acetyl transferase; hiNPCs, human induced neural progenitor/stem cells; BDNF, Brain-derived neurotrophic factor; NM, not mentioned.
The most popular test used to evaluate the efficiency of transplantation was the Morris water maze. The task is conducted in a 1-m diameter circular pool filled with opaque water [42]. Mice are trained to swim to an invisible platform submerged 1.5 cm beneath the water’s surface.
The most common region for transplantation is the hippocampus. One to six months after transplantation, researchers attempted to evaluate the efficacy of transplantations. Although interventions targeting Aβ and tau seem promising for the treatment of AD, the results of studies shown in Table 2 are mixed. Indeed, there are a number of studies that show cognitive or behavioral improvements without changes in Aβ or tau [42, 69–72]. There could be several reasons for this phenomenon. Neurotrophic and degradation enzymes, such as NEP, matrix metalloproteinases, and insulin-degrading enzyme produced by transplanted NSCs, may aid in the clearance of Aβ or tau [55, 73–75], thereby halting or ameliorating disease progression. However, because the fundamental environment continues to be favorable for Aβ generation and aggregation, the ultimate improvements in cognition are modest [52, 72]. Moreover, it is widely recognized that AD is a multifactorial disease requiring a comprehensive treatment strategy, indicating that treatment for one of the causes is frequently inadequate.
Additionally, researchers have debated the optimal phase of disease for stem cell transplantation. Kim et al. [55] established that administering stem cell therapy at the optimal time is critical for achieving maximum therapeutic effects. In their study, NSCs were transplanted into 12-month-old and 15-month-old Tg2576 mice, and the outcomes were noteworthy. The group injected at 12 months demonstrated significant improvements in both cognitive impairments and neuropathological characteristics. In contrast, the group injected at 15 months demonstrated no significant improvement in cognitive dysfunction or Aβ neuropathology. They concluded that it was too late to intervene in disease progression once the symptoms of memory loss became apparent. Indeed, because AD takes so long to develop, it is crucial to prioritize treatment interventions based on the disease progression in each patient. It remains a major challenge in the treatment of AD to provide the correct medication to the correct patient at the correct time. Future research should focus on when and how to intervene in the AD process, as well as identification of early markers indicative of the onset of AD pathogenesis.
Numerous studies have established the critical role of NSCs in cognitive improvements associated with AD. However, prior studies [42, 76] focused exclusively on short-term effects. Marsh et al. examined long-term transplantation of human NSCs using 5xFAD mice and conducted behavioral testing and histological examination 5 months after transplantation [72]. The final analysis revealed no proof of improved cognition, change in BDNF levels, or enhancement of synaptic density. Their research emphasized the critical nature of rigorously evaluating the efficacy and safety of NSC transplantation in appropriate long-term models. Contrary to expectations, a similar experiment conducted 3 years later yielded the opposite findings [75]. The same 5xFAD mouse model was employed by Zhang et al. to investigate the therapeutic potential of human induced neural progenitor/stem cells (hiNPCs) for AD [75]. Unexpectedly, they found that the grafted hiNPCs successfully differentiated into neurons with long-term survival and generated graft-host synaptic connections that ultimately strengthened and restored the host hippocampal neural networks. As a result, AD mice implanted with hiNPCs had increased synaptic plasticity and cognitive abilities. These radically divergent outcomes may be explained by differences in the source of stem cells used in these two studies. Indeed, it is reasonable that reprogrammed somatic cells, such as hiNPCs, have a stronger capacity for brain differentiation [52].
CHALLENGES OF NSC-BASED THERAPY IN AD
Although stem cell treatments have shown efficacy in animal models, various human studies in AD patients have had unfavorable outcomes [77]. To date, there have been no clinical trials involving NSCs the treatment of AD. It remains crucial to accelerate stem cell-based translational research for AD [78]. When it comes to cell transplantation, NSCs should be the best choice because they have a clear fate and are relatively similar to functional neuronal types throughout their differentiation. Because NSCs are capable of differentiating into astrocytes, it is vital to explore how to guarantee preferential development of NSCs into neurons. Moreover, there are restricted cell sources for NSCs, which exhibit variations in differentiation plasticity [45]. Identifying the optimal technique for stem cell transplantation is likewise a formidable obstacle. The proper transplantation technique can increase the number of transplanted cells that survive and function. Accordingly, future clinical trials must investigate transplantation methods, such as intravenous injection, intracerebral transplantation, and the use of biological materials. In addition, investigations attempting to elucidate the function of NSCs in AD must address concerns such as adequate differentiation, isolation, cell dose, time frame, immune rejection, environment, and location of transplantation, all of which need further study [45, 80] (Fig. 2).
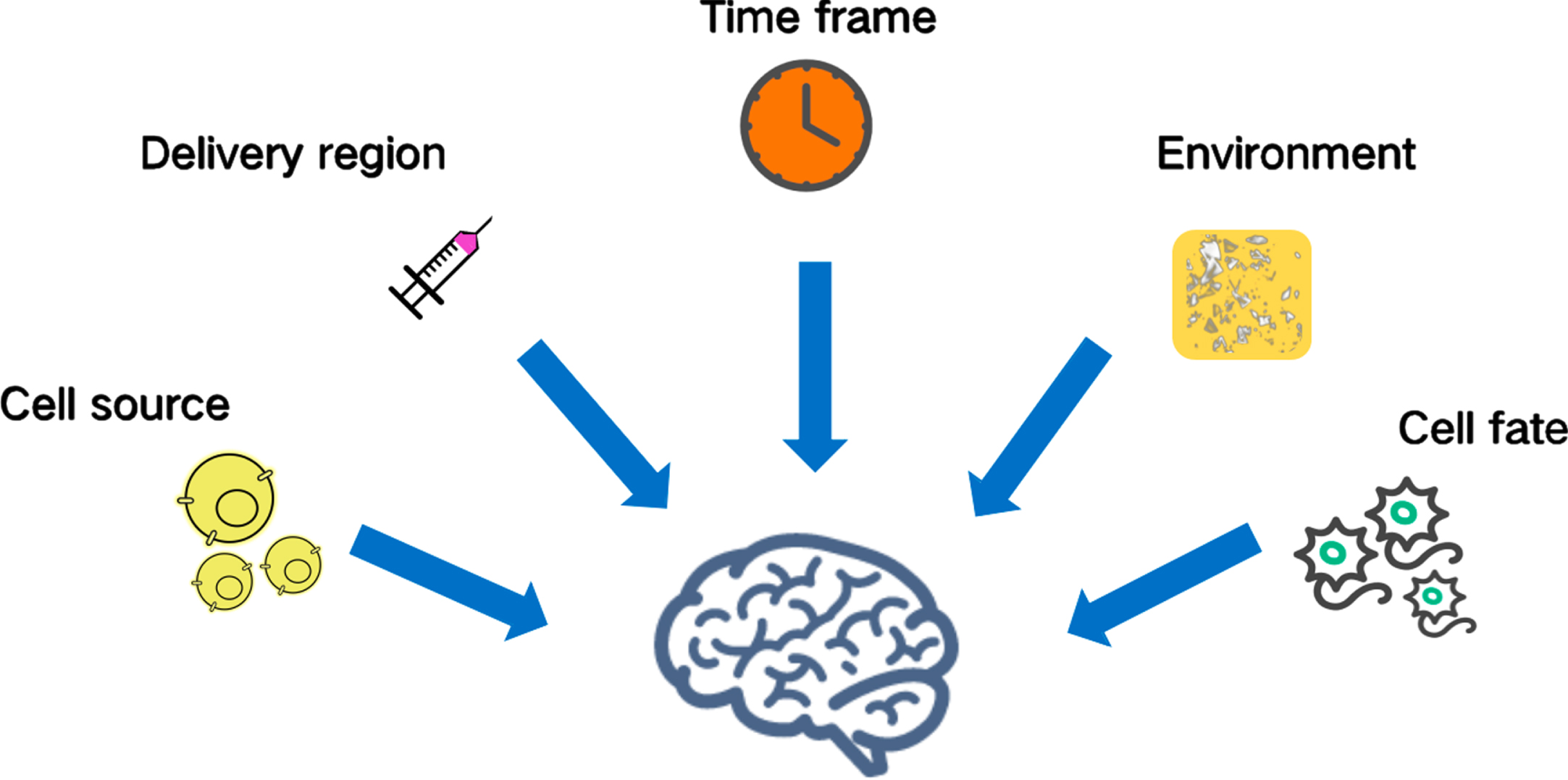
Challenges of neural stem cell-based therapy in Alzheimer’s disease. The source, adequate differentiation, isolation, cell dose, delivery region, time frame, immune rejection, and location of transplantation are all challenges requiring further study.
SOURCE OF NSCs
Preclinical research on human and mouse NSCs has been conducted to determine the effectiveness of NSC treatment for neurodegenerative diseases. No conclusion on the optimal type of NSCs can be made at present because no clinical trials have evaluated NSCs for the treatment of AD. However, we can provide a list of probable sources that might be used in AD. hNSCs may be produced from embryonic stem cell (ESC) lines or human fetal cadaver brain tissues containing neurogenic areas (like the SVZ) [81]. The safety of ESC-derived NSCs for transplantation has been established [81, 82], as they do not develop tumors when implanted into normal nude animals. However, when ESCs are implanted as donor cells, immunological incompatibility issues must be addressed. Although iPSC-based treatments will have fewer immunological problems, the fact that FAD patients may lack healthy somatic cells that can be used for reprogramming is a concern [83]. iPSCs are a type of pluripotent stem cells, meaning they can differentiate into all cell types. However, incomplete differentiation may cause the failure of transplantation. In addition, the potential risk for tumorigenesis remains a major challenge for iPSC-based clinical trials. Accordingly, high levels of quality control are needed before transplantation to avoid possible uncontrolled differentiation or proliferation within patient tissue. In summary, many characteristics, including safety, immunological response in the host environment, markers to characterize hNPCs, accessibility, and operability must be dealt with to effectively translate NSC-based treatments to clinical investigations.
REGIONS OF TRANSPLANTATION
Because AD affects numerous brain areas, including the parietal lobe, temporal lobe, sections of the cingulate gyrus, and frontal cortex, it is difficult to establish the best target for transplantation [83]. As summarized in Table 2, the most common region for transplantation is the hippocampus. Adult neurogenesis occurs in the DG of the hippocampus, which is critical to learning and memory [57, 84]. Although the basal forebrain and hippocampus are thought to be the best areas for cell transplantation, these regions are also prone to the most microenvironmental modifications [45]. Because of the altered microenvironment, transplanted NSCs may be unable to flourish in these locations. Although increasing the number of transplanted cells or repeating injections may solve the propagation problem, scientists continue to express reservations about the safety and invasiveness of repeated parenchymal injections performed using a stereotactic technique [85]. Consequently, numerous initiatives have been taken to develop promising techniques for minimizing treatment-related injuries [85]. Kim et al. conducted a phase I clinical trial in nine patients with AD to determine the safety and dose-limiting toxicity of MSCs generated from human umbilical cord blood [85]. To minimize transplantation-related harm, researchers implanted an Ommaya reservoir into the lateral ventricle to distribute the MSCs. The results indicated that this strategy was practicable, safe, and well tolerated.
TIMING OF TRANSPLANTATION
Because AD is an age-related disease, the timing of NSC transplantation is also critical. Prior animal studies have established that optimal timing of transplantation is critical for achieving the maximum therapeutic effect [55, 72]. When cognitive impairment and Aβ neuropathology exist, stem cell transplantation may be ineffective [55]. However, once the chronic course of AD is stopped or disrupted, the cognitive function of patients will improve or no longer deteriorate. Patients enrolled in future clinical trials may meet criteria for having a significant amyloid burden or neuronal degeneration, as determined by brain magnetic resonance imaging or positron emission tomography [85]. Because AHN is essential to the progression of AD, it may be possible to use it as an early indicator of AD to determine the optimal intervention window.
ENVIRONMENT
The environment into which NSCs are be transplanted is quite significant. Quiescent NSCs have a low metabolic rate and are sensitive to their surrounding signaling environment [86]. A variety of stressors, including Aβ or inflammatory molecules, can force neurons in the SVZ to leave or remain quiescent, leading to neuronal death rather than division [87]. Inflammation is thought to drive AD pathogenesis because it increases the Aβ burden [88]. The neuroinflammation in AD is considered to be mediated by the immune system [89], in which abnormal microglial activation is thought to play a crucial role [90]. Thus, controlling the level of inflammation or providing a harmonious environment for transplanted NSCs in AD brains might be beneficial to their neural differentiation.
FATE OF TRANSPLANTED NSCs
Even if we overcome all the previously mentioned obstacles, an uncontrollable cell fate would jeopardize the entire effort. To investigate the differentiation and migration of stem cells, immunofluorescence and confocal microscopy are two main technologies. Recent research indicates that modulating the Notch signaling system makes it feasible to precisely control the process and proportion of NSCs that develop into neural functional cells [91]. In addition to their therapeutic effectiveness, monitoring the interactions of implanted NSCs and their reactivity to the tissue environment all crucial considerations.
In addition to differentiating into CNS cells, stem cells can migrate beyond the transplantation region and form synapses with local CNS cells. Relevant synaptic markers, such as synaptophysin, synapsin, and growth-associated protein-43 (GAP-43) are frequently tested to evaluate synaptic integrity. Indeed, many of the experiments in Table 2 observed that transplantation of NSCs could improve cognition. However, the mechanism for this modulation is currently unclear. Moreover, whether transplanted NSCs could join the correct circuits and perform correctly requires more research. Given the incomplete understanding of AD pathophysiology, it remains debatable whether to direct NSC differentiation in only a single direction. Although recent research has established the significance of nanotechnology in monitoring and guided differentiation of NSC transplantation [5, 92–94], the safety of this technology in the human body requires additional investigation. In addition, transplanted cells may exist in the patient for more than 10 years, whereas the majority of current stem cell transplantation studies in mice are observed for only 3–6 months. Potential risks, such as incomplete differentiation of stem cells, might require longer observation periods in future research.
CONCLUSIONS AND FUTURE PROSPECT
Growing numbers of studies employing animal models are examining the role of pathogenic proteins in the development of AD. At present, none of the available animal models has been able to properly imitate human AD. As a result, it is difficult to exactly quantify the positive effects of NSCs in patients with AD. Although stem cell therapies have been demonstrated as effective in animal models, several human research studies on AD patients have had negative results. Thus, additional research and development of better animal or cell models should be encouraged.
It has been shown repeatedly that transplanting of NSCs for AD has some benefits, although the drawbacks and risks cannot be ignored. Ethical issues, immune rejection, genetic mutations, and the potential risk of tumorigenesis are all issues that need to be urgently addressed. Accordingly, no clinical studies employing NSCs in the treatment of AD patients have been conducted and more work must be done before NSC-based therapy can be used to benefit patients. Important factors include the cell source, cell dose, mode of dissemination, and timing of transplantation.
Prior animal research has shown that the appropriate timing of cell transplantation is crucial for attaining the maximal therapeutic impact. Current treatment studies are almost certainly predicated on early detection and intervention to avert future severe synapse and neuronal loss. Because AD is a complicated condition, it needs multiple interventions. Thus, combining NSC transplantation with already authorized medicines may pave a promising road for future treatment of AD.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the National Key Research and Development Program of China (2018YFA0108602, 2021YFE0114300), the National Natural Science Foundation of China (82170799, 82171475) and the CAMS Initiative for Innovative Medicine (2021-1-I2M-019).
