Abstract
The brain changes of Alzheimer’s disease and other degenerative dementias begin long before cognitive dysfunction develops, and in people with subtle cognitive complaints, clinicians often struggle to predict who will develop dementia. The public increasingly sees benefits to accessing dementia risk evidence (DRE) such as biomarkers, predictive algorithms, and genetic information, particularly as this information moves from research to demonstrated usefulness in guiding diagnosis and clinical management. For example, the knowledge that one has high levels of amyloid in the brain may lead one to seek amyloid reducing medications, plan for disability, or engage in health promoting behaviors to fight cognitive decline. Researchers often hesitate to share DRE data, either because they are insufficiently validated or reliable for use in individuals, or there are concerns about assuring responsible use and ensuring adequate understanding of potential problems when one’s biomarker status is known. Concerns include warning people receiving DRE about situations in which they might be compelled to disclose their risk status potentially leading to discrimination or stigma. The Advisory Group on Risk Evidence Education for Dementia (AGREEDementia) welcomes all concerned with how best to share and use DRE. Supporting understanding in clinicians, stakeholders, and people with or at risk for dementia and clearly delineating risks, benefits, and gaps in knowledge is vital. This brief overview describes elements that made this group effective as a model for other health conditions where there is interest in unfettered collaboration to discuss diagnostic uncertainty and the appropriate use and communication of health-related risk information.
THE IMPORTANCE OF CONSULTATION: AGREEDementia
As various forms of dementia risk evidence (DRE), such as biomarkers for brain amyloid, demonstrate clinical utility and transition beyond research use only, the public increasingly wants access to their personal DRE. Participants in dementia research want their test results [1]. When researchers and clinicians hesitate in responding to requests for this personal information, requestors feel frustrated and patronized by barriers designed to protect them from potential harm. A person potentially facing a progressive degenerative disease may feel time pressure to know if they have or will develop dementia. Informed, autonomous decision making around appropriate access to DRE in people with or at risk for dementia is vital and depends on promoting understanding in all, including patients, research participants, and clinicians, by clearly delineating evidence of benefits, risks, and gaps in knowledge. In contrast to open discussion and consultation around best practices, publications are slower and content is often filtered by fear of misstatement and subjective judgments of whether questions are worthy of being published. To address the need for agile, informed, and responsive communication between all users of DRE, the Advisory Group on Risk Evidence Education for Dementia (AGREEDementia.org) was convened initially by Nina Silverberg of the National Institute on Aging (NIA) and originally was comprised largely of members of NIA supported Alzheimer’s Disease Centers. Over time the group evolved into an independent body that sought to be widely inclusive and to promote open dialogue. Such inclusiveness has been demonstrated by the attraction of interested centers from Brazil, Germany, and the United Kingdom. The aims are 1) to evaluate the current evidence on putative predictors of dementia risk, 2) to guide whether, when, and how to disclose DRE, and 3) to consult to support ethical and appropriate use of and communication about biomarkers and other dementia risk-related information. Here we describe elements that support the group’s effectiveness as a guide to future working groups with similar aims.
GROUP COMPOSITION
As of the beginning of 2022 there were 122 members, although the level of activity of individual people has varied. The majority (82%) of people were from academia, broadly including clinical researchers, students, research staff, genetic counselors, and healthcare lawyers. The remaining 18% were people living with or at risk for dementia, care partners, United States government staff (National Institutes of Health, Food and Drug Administration) or were representatives from advocacy groups (e.g., the Lewy Body Dementia Association, the Alzheimer’s Association, the Association for Frontotemporal Degeneration, and Cohen Veterans Bioscience).
A strength of the group is that it draws individuals from vastly different disciplines and perspectives with a universal interest in how DRE is developed and communicated. AGREEDementia has become a forum for early career investigators and those new to Alzheimer’s Disease and Related Disorders work to seek mentorship and networking in addition to specific resources. Participants learn about AGREEDementia through a variety of means. The bulk of members came initially from the US NIA funded Alzheimer’s centers. Members also come from the Alzheimer’s Association International Conference meetings and workgroups. Members tell their students, staff, and colleagues. The
GROUP FORMAT
One critical aspect that adds value to the group is the hybrid structure of a meeting that everyone attends (hereafter referred to as the All Hands meeting), and smaller working subgroups that enable discussion and development of work products. In the first half of the All Hands meeting, members hear announcements and identify key concerns of general interest, as well as updates from each of the working groups. Having a time when all members hear these updates enables them to collaborate with other subgroups if there is a potential synergy. In the second half of the All Hands meeting, a speaker presents relevant topics. These presentations ensure that the members have a shared foundational knowledge, thus making advice and guidance more informed. Where possible, lecturers from other fields that addressed similar problems are recruited, for example pharmacogenetics of depression [2] or genetics of movement disorders [3]. Most workgroup meetings are open to all members; however, there have been some exceptions when workgroups requested privacy to develop ideas or voice concerns. For example, a group dedicated to enhancing cultural sensitivity of research staff and outreach workers requested privacy from their supervisors. In sum, with these few exceptions, the group is structured so that all members can attend all monthly groups including 1) All Hands where members can address the entire group and 2) their home working group where they are responsible for a project. All Hands meetings and minutes update members on all workgroup activities so that members can also attend or join any other additional workgroups as much or as little as they wish.
Having both an internal workspace and an externally facing website has enhanced operations. Initially, potential new members felt overwhelmed and confused by the multiple meetings and we (typically AR) needed to meet with each new potential member individually to explain that there were two types of meetings involved in participation: the All Hands meeting and a workgroup of their choosing. Integration of new members improved with a public website that explains the group structure (All Hands, Workgroups) and the aims of the different workgroups they can choose to join. Informal collaboration has been facilitated with a separate, online, private workspace for group members. This space enables sharing of articles, contact information, personal skills and knowledge, and drafts, without fear that work products under development will be misused.
RATIONALE UNDERLYING THE STRUCTURE OF THE SUBGROUPS
Working groups enable more free and in-depth discussion and the structure of the six working subgroups powerfully supports the mission of AGREEDementia. Figure 1 lists the working groups and broad general missions. To facilitate group interactions, the schedule for all meetings are listed in the All Hands agenda. This information enables members to attend meetings of a subgroup based on something they heard at the All Hands call.
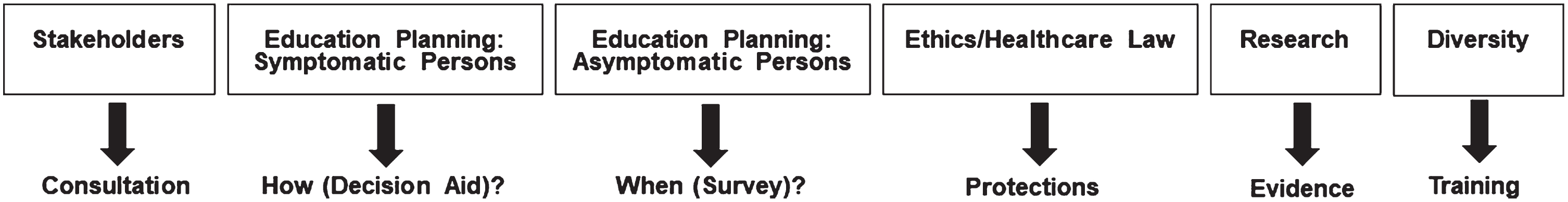
Subgroup structure and missions.
Stakeholders
This group consists of people living with dementia, their care partners, research participants, individuals with elevated risk for dementia (based on genetic or Alzheimer’s disease (AD) biomarkers), and members from advocacy organizations for people with dementia and care partners. One research participant and co-chair of the group has authored a book on the impact of learning
Education planning: People with symptoms
One critical distinction impacting how DRE is used is whether or not the stakeholders have symptoms of dementia. Hence, these workgroups are separated. In general, for people living with symptoms of dementia, when DRE is communicated it is integrated with differential diagnosis (e.g., biomarkers may be used to determine the etiology of cognitive impairment) and care management. There may also be evidence of disability, and diagnosis may guide treatment and/or planning for future decline. Furthermore, in people with symptoms, DRE is typically more predictive of progression than in asymptomatic individuals. The focus of this subgroup is thus more often to provide guidance about how to communicate DRE when the DRE is used appropriately, and hence this group has studied [8] and created decision tools to assist people in deciding whether or not to learn their personal DRE, such as amyloid PET scan results. This group discusses in this forum the appropriate uses of a blood amyloid measure [9]. Additional relevant tools are/will be housed on NIA’s Alzheimer’s & Dementia Outreach, Recruitment & Engagement Resources (ADORE) [10].
Education planning: People who are asymptomatic
In contrast, for people who are asymptomatic, an indicator of an elevated risk of dementia may be less predictive than for symptomatic individuals. Hence, sharing DRE is rarely part of routine clinical care, and much of the discussion focused on communication is in research settings. In this setting, discussions around the benefits and risks of sharing DRE should include warnings around the risk of stigma and discrimination since there are circumstances when the stakeholder may be compelled to disclose their status [11]. The focus of this group is more typically
Research evidence
Ultimately the strength of evidence for the clinical utility of DRE, such as for the prediction of dementia, is important for guiding what is shared, and hence there is a dedicated subgroup to evaluate the studies and evidence. This Research/Analytic subgroup focuses on types of DRE that are used in research, and potentially may be included in clinical settings at some point in the future (genetics, neuroimaging, cerebrospinal fluid biomarkers, and plasma biomarkers). Early discussions focused on difficulties with the precision and interpretation of
Ethics and healthcare law
The landscape of ethical concerns in communicating dementia diagnosis are well summarized elsewhere [18]; however, here are some issues identified. With respect to group dynamics, supporting stakeholder autonomy and choice including respect for wishes to know or not know DRE is important in addressing disagreements around best practices, particularly in balancing individual differences in personal decisions surrounding beneficence and non-maleficence. Specifically, supporting knowledge of risks, protections, and benefits of knowing and using DRE and how they can be balanced has been an important theme. Several members in AGREEDementia have studied the risk of psychological distress [19] and stigma [20] in learning of increased risk for dementia. Building education around the risks/benefits and supporting choice in direct to consumer (DTC) testing for
Diversity
Issues of justice are critically important for this workgroup since social determinants of health and disparities are related to biomarkers [31]. Studies that include diverse cohorts are critical for understanding how best to use DRE (e.g., [32]). Whereas initially this group focused on education and mentorship of trainees and young investigators, it became clear that an urgent weakness was training research staff in sensitive outreach to diverse cohorts. This group is developing a publicly available lecture series designed to better train coordinators about issues of cultural sensitivity and diversity, such that we may better enroll and retain underrepresented or underserved communities in clinical research.
NEXT STEPS TOWARD IMPLEMENTATION
Discussing best practices and supporting two way communication
The science in this field is moving quickly and education around a broad framework of general principles of appropriate use and effective care will support more rapid understanding and communication. Education must stress that clinically valid conclusions from DRE typically must be interpreted in a larger context of a comprehensive evaluation, information often absent in research projects or direct to consumer products. Treatable contributors to cognitive dysfunction must not be dismissed by attributing all dysfunction to AD dementia on the basis of amyloid brain positivity or other DRE. It is unclear how to explain these interpretive limitations of DRE to the public to foster understanding and a sense of feeling respected. Groups with inclusive membership can support effective two-way communication to shape messages and that reach larger communities.
Immediate and sustained impact
A positive impact would mean that stakeholders and all who care for people with or at risk for dementia would feel comfortable with decisions and methods of communicating and using DRE. The impact of the group on the field will depend in part on what it produces in collaborative papers, products (e.g., webinars, decision tools) and the degree to which members give credit and acknowledge its contributions to their accomplishments. One urgent need is increasing involvement of historically underrepresented groups in research and policy. At present we informally estimate only about 5% of the AGREEDementia members to be from underrepresented groups and we are working to increase this representation. Because the group is relatively new and completely volunteer based, decisions around leadership fall on whoever is willing to work (e.g., set agendas, lead meetings, organize speakers) and the burden is minimal relative to the opportunities made possible for collaborations. Aside from a small contribution from the Stanford Alzheimer’s Disease Center to support the external website, there are minimal fiscal needs. Collaborations with the NIA publicize valuable resources. If there were a decision to seek financial support to grow efforts, there are various alternatives. One model of financial support could be through grants, another model of support could be through requesting paid membership, and an alternative approach is through professional organizations that provide infrastructure through paid membership such as Alzheimer’s Association International Society to Advance Alzheimer’s Research and Treatment (ISTAART).
Outreach to promote understanding
Broad outreach beyond research centers supports generalizability to clinics and this issue is vital. Most of what is known about people’s reactions to learning about DRE comes from research in carefully selected cohorts and much needs to be learned about how best to integrate with diverse patients in clinics. For example, patients may have comorbid mental health issues that may exacerbate their distress. Whereas more extensively discussed elsewhere in this issue [9], interpreting and using biofluid based biomarker data, particularly with comorbid conditions, is often not straightforward [33]. Many studies have few nonwhites and there are differences across groups in biomarker levels and genetics (e.g., [34–36]). Some economically disadvantaged populations are also at disproportionately elevated vascular risk [37, 38] likely in part due to social determinants of health [31]. These issues have implications for how DRE is used and explained.
Understanding limitations of DRE
A careful understanding of measures and relationships between measures is needed. For example, Schindler suggests that brain amyloid levels in cognitively normal individuals are correlated with the age at dementia onset, but because amyloid accumulation plateaus around the time of symptom onset, amyloid levels are not well correlated with dementia severity [34]. Blood biomarkers offer tremendous opportunities but issues related to implementation are critical achieving that potential [32].
Biomarkers, genetics, therapy, and future clinical practice
Aducanumab demonstrated how therapeutic innovations involving biomarkers and genetics introduce new ethical complexities. Whereas sharing
Summary
Effective use of DRE can transform dementia diagnosis and care; however, defining responsible practice and respectful use is complex. Supporting effective collaboration between stakeholders and the community developing and supporting use of DRE requires education around the nuances of interpreting these results while the science progresses rapidly. It requires collaboration among people with diverse expertise such as researchers, ethicists, legal scholars, educators, clinicians, and people living with and at risk for dementia to provide education. AGREEDementia represents an open, welcoming, group that bridges the gap in communication between small, academic, working groups and the general public who struggle to have their voices heard. The group’s hybrid structure in which there is one overall meeting that broadcasts opportunities for synergy and education (All Hands meetings), and smaller, accomplishment-focused, working groups, is effective but difficult to understand without an explanatory, public website. The public website also broadcasts accomplishments and external, educational opportunities while an internal website supports collaboration. Ultimately, ongoing collaboration will be supported through mutual respect for the deeply personal needs of individuals struggling with the risk and effects of dementia.
Footnotes
ACKNOWLEDGMENTS
This research was supported in part by the Stanford Alzheimer’s Disease Research Center (P30AG066515) and the War Related Illness and Injury Study Center. Multiple other grants from NIA support other authors and their centers including R01AG068206,P30AG066509, P30AG062421, P30AG072931, K01AG057796, U24AG041689 and R56AG069130. FFO receives support from The State of São Paulo Research Foundation (FAPESP), grant #2015/10109-5. ACR, JAL and LT receive support from the Office of Research and Development of the Department of Veterans Affairs (VISN 21 MIRECC, RX003152).
The views expressed in this manuscript are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs, the National Institute on Aging (NIA), or any of the organizations of which they are members or from which they receive support. In addition to the authors listed, Nina Silverberg, Grayson Donley, Jessica Harper of NIA contributed intellectually and administratively. Robyn Shapiro has provided legal consultation. Rebecca Edelmayer and other members of the Alzheimer’s Association and Angela Taylor of the Lewy Body Dementia Association supported continued engagement and outreach to new members.
None of the authors identified relevant COI but most received federal funding, most from the NIA but some from other NIH institutes (NINDS, NIMH) and some from the Office of Research and Development of the Department of Veterans Affairs including the Mental Illness Research Education and Clinical Center (MIRECC). These authors include ACR, DB, NAC, LC, SSD, JG, CEG, JAL, MM, RLN, MWP, ARF, AR, MR, DS, CGS, LT, JT, EMW, SW, LSW, DTZ. In addition, some authors receive funding from the Alzheimer’s Association (JJA, JPC, JDG, NTA). JWA is an unpaid consultant of Memtrax. JPC also receives funding from the Doris Duke Charitable Foundation and serves on a medical advisory board for Humana Healthcare. JLH receives funding from Eisai/Biogen, Lilly, and Biohaven. JDG received support from BrightFocus Foundation, Eli Lilly, Genentech, Biogen, Eisa and also consults with SiteRx. JHL consulted with Genentech and Biogen. FFO received support from FAPESP –The State of São Paulo Research Foundation. SES has analyzed data provided at no cost by C2N Diagnostics to Washington University, but she has not received any personal compensation from any for-profit organizations.
