Abstract
Background:
Depletion of blood-derived progenitor cells, including so called “early endothelial progenitor cells”, has been observed in individuals with early stage Alzheimer’s disease relative to matched older control subjects. These findings could implicate the loss of angiogenic support from hematopoietic progenitors or endothelial progenitors in cognitive dysfunction.
Objective:
To investigate links between progenitor cell proliferation and mild levels of cognitive dysfunction.
Methods:
We conducted
Results:
Fewer colony forming units were observed in samples from older adults with a Clinical Dementia Rating global score of 0.5 versus 0. Older adults whose samples developed fewer colony forming units exhibited worse performance on neuropsychological measures of memory, executive functioning, and language ability.
Conclusion:
These data suggest blood progenitors may represent a vascular resilience marker related to cognitive dysfunction in older adults.
INTRODUCTION
Vascular risk factors increase risk of age-related cognitive decline and dementia [1]. Although the evidence of vascular risk factors contributing to the risk of cognitive dysfunction in older adults is robust, research on vascular resilience factors is sparce. One potential vascular resilience factor involves progenitor cells that originate from bone marrow and other tissue compartments and are present in adult peripheral blood [2]. Vascular protective cells, termed endothelial progenitor cells (EPCs), are crucial in blood vessel and capillary formation and healing, and also have been shown to mediate neuronal repair mechanisms following brain injury [3]. Two types of time dependent EPCs exist, named accordingly as early EPCs and late EPCs [also called endothelial colony forming cells (ECFCs)] based on the timing of their appearance
Early EPCs, a heterogeneous combination of monocytes/macrophages [6] and T-lymphocytes [7], are also called “circulating angiogenic cells”, “myeloid angiogenic cells”, “proangiogenic cells”, “hematopoietic endothelial progenitor cells”, “CFU-Hill cells”, and “small EPCs” [8–10]. Early EPCs are thought to support brain health by homing to sites of neural or cerebrovascular injury and secreting angiogenic growth factors and cytokines (e.g., vascular endothelial growth factor; VEGF), thereby activating adjacent endothelial cells and promoting angiogenesis by regenerating mature endothelial cells and neurogenesis by supporting neural cell growth [11, 12]. Early EPCs appear to be further protective against neurovascular injury by secreting a number of neurotropic factors in addition to VEGF [13], facilitating neuronal tissue repair [14–16], and contributing to the neurogenic [17], neurovascular [18], and oligovascular niche [17–20]. Putative early EPCs are cultured from peripheral blood mononuclear cells (PBMCs), historically using
Evidence corroborating potentially protective effects of EPCs in brain injury and neurodegeneration comes from studies using cell surface EPCs markers (CD34+, CD133+, CD309+) and investigations into the EPC angiogenic secretome in individuals with MCI and/or Alzheimer’s disease (AD) [2]. Neurogenesis [30] and angiogenesis [31] are both impaired in AD, and circulating EPCs are accordingly depleted in AD [30, 31]. AD patients also present with reduced angiogenic activity of the EPC secretome [32]. More specifically, circulating CD34+ levels are reduced in early AD and related to AD neuropathology (i.e., amyloid-β levels) in the cerebrospinal fluid [33].
The present study aims to expand on prior work by investigating the link between
METHODS
Following participant informed consent and approval by the University of Southern California (USC) Institutional Review Board (IRB), the study protocol was implemented at the Vascular Senescence and Cognition (VaSC) Laboratory in the USC Department of Psychology.
Participants
Sixty-five community dwelling older adults (Mage = 70.94; SDage = 7.39), free of dementia or clinical stroke, underwent venipuncture, neuropsychological testing, and brain MRI. Participants were recruited from the community via university platforms, including the USC School of Gerontology Healthy Minds Program and the USC Alzheimer’s Disease Research Center, as well as additional recruitment from the local community surround the USC campus via word-of-mouth, flyers, and community outreach events. Inclusion criteria for participants consisted of being a minimum age of 55 years and independently living. Exclusion criteria included history of dementia, clinical stroke, learning disability, traumatic brain injury, or other systemic or neurological illness or treatment that may affect central nervous system functioning. Vascular risk factors were determined by clinical interview and objective measures, including blood pressure, body mass index (weight in kg/ height in m2), dyslipidemia, (i.e., history of elevated total cholesterol, low density lipoprotein cholesterol or triglycerides, or reduced high density cholesterol), diabetes (history of type I or II), history of cardiovascular disease (angina, intermittent claudication, myocardial infarction, stent placement, coronary artery bypass graft), or history of transient ischemic attack. Blood pressure was measured twice from each arm on two separate days, and systolic and diastolic pressures were averaged across these time points. Pulse pressure was calculated as difference between systolic and diastolic pressure, and mean arterial pressure was calculated as the sum of diastolic blood pressure and one-third of the pulse pressure.
Procedures
Blood draw
Fasting venipuncture was performed prior to neuropsychological testing during each participant’s first visit, with approximately 25 mL of blood being drawn into EDTA coated tubes. Samples were centrifuged and PBMCs were isolated from the plasma before performing cellular assays as described below.
CFU-Hill in vitro culture
CFU-Hill colonies were cultured from PBMCs to isolate putative early EPCs. PBMCs were isolated from anticoagulated whole blood by density gradient centrifugation in Histopaque-1077 (Sigma Aldrich). Cells were first seeded on 12-well fibronectin-coated plates (Day 0) at 2.5×106 cells/well in CFU-Hill Liquid Medium (Stem Cell Tech). Non-adherent cells were then collected and replated onto 24-well fibronectin-coated plates at 1.0×106 cells/well (Day 2). CFU colonies were next scored as a central core of round cells surrounded by radiating spindle-shaped endothelial-like cells (Day 5) (Fig. 1A, B). Following day 5 in culture, CFU-Hill colonies were counted, and mean colony number calculated.
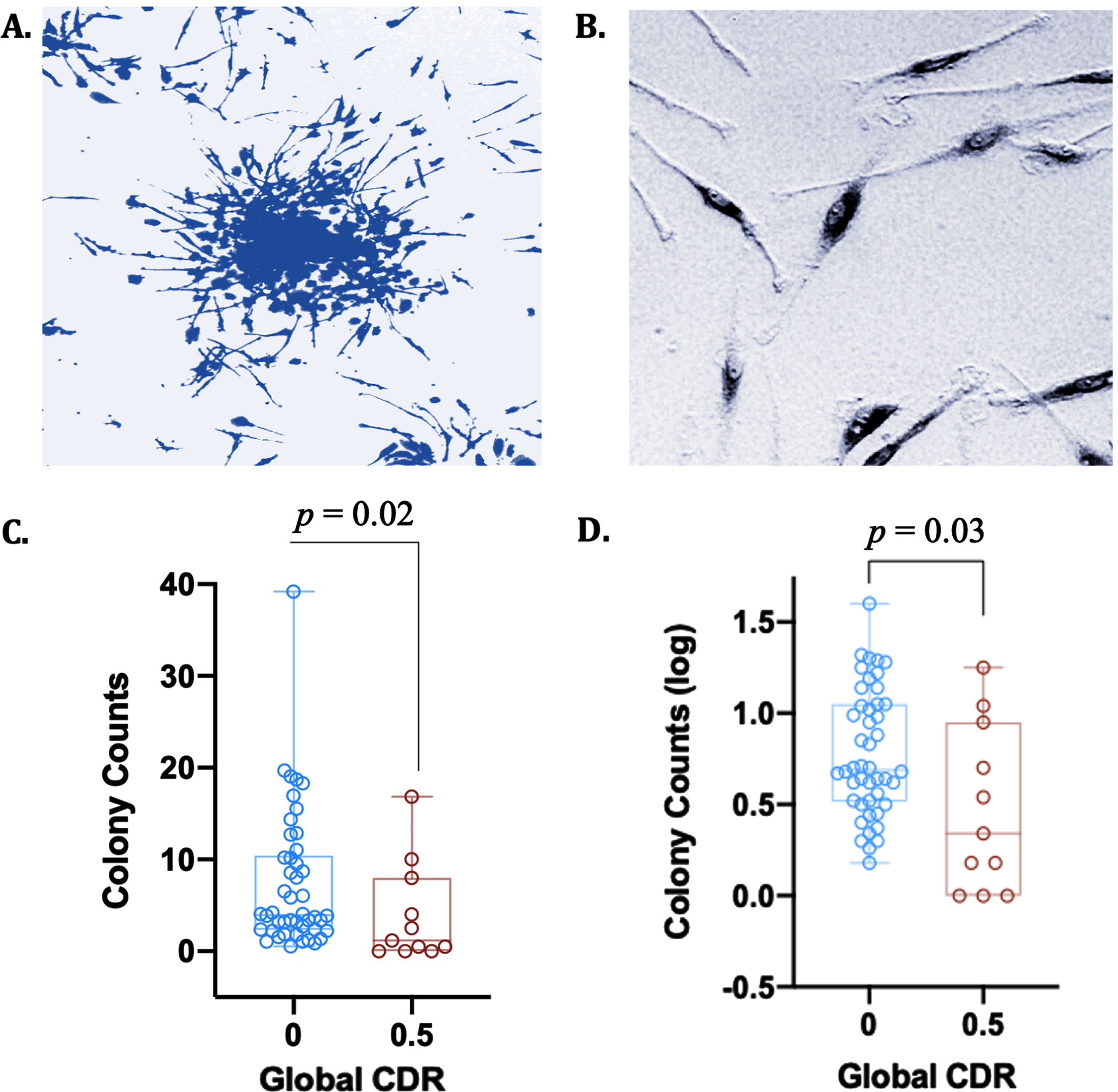
CFU-Hill colony counts are attenuated with cognitive impairment on the CDR. A) Giemsa stain of a CFU-Hill colony cultured from blood
APOE genotyping
Cognitive testing
The cognitive test battery in the present study consisted of several neuropsychological tests in each cognitive domain of interest, in addition to a global measure of cognitive and functional performance related to the deficits in AD and other dementias. Cognitive domains for this study included memory [Verbal memory: Wechsler Memory Scale-Revised (WMS-R) Logical memory I and II; Visual memory: WMS-R Visual Reproduction I and II], executive functioning (Delis-Kaplan Executive Function System (DKEFS) Trail Making Test B, FAS Fluency, Golden Stroop), attention (DKEFS Trail Making Test A, Digit Span Forward), and language (Naming: Multilingual Naming Test [MiNT], Fluency: Animal and Fruits & Vegetables Naming.
The Clinical Dementia Rating Scale (CDR) was used as a measure of cognitive and functional impairment indicating MCI and/or very mild dementia. The CDR is a 5-point scale characterizing cognitive and functional domains applicable to AD and similar dementias. Functional domains were based on a participant and informant semi-structured interview, while cognitive domains were evaluated by administering a participant and informant semi-structured interview and brief cognitive tests. A global CDR score was calculated by scoring each domain and totaling the CDR Sum of Boxes based on criteria detailed elsewhere [40]. A global score of zero indicates no clinically meaningful cognitive or functional impairment, while a global score of 0.5 indicates MCI and/or very mild dementia. While different criteria are used to diagnose MCI, several previous studies have used a global CDR of 0.5 as a diagnostic criterion [41–44].
Statistical analyses
Data were first examined for outliers and deviations from normality using skewness and kurtosis measures. Log10-transformation was used to normalize the distribution of putative EPC proliferation and Trails B scores. While the CDR 0 and 0.5 groups were unequal in sample size, heteroscedasticity was not an issue between groups. Due to the relatively equal variances in both groups, the one-way ANOVA models are valid despite unequal sample sizes.
Participant groups (CDR 0 versus CDR 0.5) were compared across demographic characteristics and vascular risk factors using independent sample
RESULTS
Demographics and vascular risk factors
Participants with indications of mild cognitive and/or functional impairment as measured by the CDR (i.e., CDR of 0.5) were significantly older than participants who had a CDR of 0 (
Demographics and clinical characteristics for the CDR 0 and 0.5 group
CFU-Hill colony proliferation and cognitive dysfunction
CFU-Hill colony log counts were significantly depleted in participants with a CDR of 0.5 (
Lower CFU-Hill colony counts predicted worse performances on tests of executive functioning, verbal memory, and language after controlling for age, sex, and education [(1) Animals:
CFU-Hill colony counts predict short-term verbal memory, executive functioning, and language
N = 65. All values are based on statistics from model 2 of the hierarchical multiple regression model (where model 1 predictors included age, sex, education, and model 2 predictors included age, sex, education, and CFU-Hill log counts). *
DISCUSSION
The present study findings indicate attenuated proliferation of blood-derived progenitors
Our observations of decreased
The cognitive domains related to progenitor cell proliferation in the present study are immediate memory, executive dysfunction and verbal fluency, all of which are implicated in aging and vascular disease [50] and have also been linked to MCI and AD dementia [51, 52]. Memory, executive and fluency deficits are commonly observed in the early stages of MCI and can be part of the dementia prodrome [52], suggesting attenuated progenitor proliferation may be related to dementia risk at an early stage. Importantly, our findings reveal a relationship between progenitor proliferation and cognition that is detectible even in patients with no history of stroke. These findings warrant further investigation into progenitor cell functioning as an early-stage vascular process related to cognitive decline. The present study also did not assess for AD or cerebrovascular pathological markers, thereby limiting conclusions regarding the role of progenitor cell proliferation in a specific pathophysiological process. It is possible that attenuation of progenitor proliferation reflects a nonspecific decrease in progenitor vitality, possibly indicating or even contributing to susceptibility to neurocognitive dysfunction.
While the mechanism contributing to the association between progenitor cell proliferation and age-related cognitive dysfunction is unknown, there are several potential explanations. One mechanism may involve cellular senescence in the aging brain, which has been shown to contribute to cognitive impairment and decline [53]. Endothelial cell senescence has recently emerged as a possible mechanism underlying cerebral microvascular pathologies. Specifically, single-cell RNA sequencing indicated that approximately 10% of cerebral microvascular endothelial cells were senescent in the aging mouse brain [54]. Applying single-cell RNA sequence technology to progenitor cells derived from human whole blood may help elucidate the degree to which senescent endothelial progenitor cells in particular are associated with cognitive dysfunction. Additionally, recent work suggests hematopoietic stem/progenitor cells (HSPCs) show diminished vascular reparative capacity and impaired mobilization following ischemic injury in aging mice. These deficits in HSPC function are reversible following the activation of factors that stimulate vascular repair (i.e., angiotensin-converting enzyme-2 and the Mas Receptor) [55]. These findings underscore the hypothesis that reduced vascular repair capacity of progenitor cells occurs in aging, which in turn may increase susceptibility to vascular injury and subsequent clinical consequences, including cognitive impairment. In addition to the cellular senescence occurring in the aging vasculature, several other cerebral metabolic changes are also co-occurring in the vasculature as a result of aging that may also lead to progenitor cell exhaustion, including alterations in pro-geronic factors (e.g., serum response factor, insulin-like growth factor-1 and VEGF-A), increased oxidative stress, changes in DNA metabolism, and lifestyle factors such as nutrition and dietary habits [56–58]. Future studies on progenitor cells and aging would benefit from including these cellular and lifestyle factors to delineate to what extent different cell types, factors in the vasculature, and lifestyle choices are associated with age-related cognitive dysfunction.
The cross-sectional design of this study represents a study limitation that precludes any interpretations regarding causality. Age is the greatest risk factor of MCI, vascular cognitive impairment and dementia, and AD, reflected in our samples as the CDR 0.5 group which was significantly older than the CDR 0 group. However, age was not related to progenitor cell proliferation, suggesting that depletion of progenitor cell colony counts in the CDR 0.5 group cannot be explained by solely demographic factors. With regards to other demographic factors, although the relative diversity of our sample with respect to African American representation is a study strength, the complexity of the sample racial makeup together with the relatively small sample size precluded inclusion of racial subgroups in most of the statistical analyses. However, there were no significant differences in racial makeup between cognitively unimpaired and impaired participants. Additional study limitations include the small sample size of the CDR 0.5 group, although it is noted that significant relationships between colony counts and neuropsychological measures were observed in the total sample. Longitudinal studies evaluating progenitor cell proliferation as a predictor of cognitive decline will provide crucial information regarding the potential value of progenitor cells as early indicators of cognitive decline. Lastly, the well-established CFU-Hill assay used in the present study does not definitively characterize EPC phenotype. True EPCs are challenging to isolate and thus define due to the lack of a specific molecular determinant or cell surface marker that is uniquely associated with this cell population. Thus, while CFU-Hill colonies do not specifically isolate EPCs, these colonies express several cell antigens consistent with an EPC phenotype (i.e., CD31, CD105, CD144, CD146, von Willebrand factor, and KDR/VEGF-R2) [59, 60]. Further studies of the interplay among progenitors, cognitive dysfunction, structural brain integrity, and disease-specific biomarkers are warranted to evaluate the potential role of progenitor cell proliferation as a resilience indicator with implications for dementia risk assessment and prevention. We did not include imaging markers in the present study as we did not have sufficient brain MRI data available for analysis; however, future studies should incorporate brain MRI to examine associations between progenitor cells, cognition, and brain integrity.
Footnotes
ACKNOWLEDGMENTS
We thank the study participants and staff at the University of Southern California and the University of California, Irvine for making this study possible.
FUNDING
This work was supported by National Institutes of Health grants (R01AG064228, R01AG060049, R01AG082073, P30AG066519, P01AG052350) and the Alzheimer’s Association (AARG-17-532905).
CONFLICT OF INTEREST
Daniel Nation is an Editorial Board Member of this journal but was not involved in the peer-review process. All other authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy restrictions.
