Abstract
Background:
Monoclonal antibodies (mAbs) are currently among the most investigated targets for potential disease-modifying therapies in Alzheimer’s disease (AD).
Objective:
Our objectives were to identify all registered trials investigating mAbs in MCI due to AD or AD at any stage, retrieve available published and unpublished data from all registered trials, and analyze data on safety and efficacy outcomes.
Methods:
A systematic search of all registered trials on ClinicalTrials.gov and EUCT was performed. Available results were searched on both platforms and on PubMed, ISI Web of Knowledge, and The Cochrane Library.
Results:
Overall, 101 studies were identified on 27 mAbs. Results were available for 50 trials investigating 12 mAbs. For 18 trials, data were available from both published and unpublished sources, for 21 trials only from published sources, and for 11 trials only from unpublished sources. Meta-analyses of amyloid-related imaging abnormalities (ARIA) events showed overall risk ratios of 10.65 for ARIA-E and of 1.75 for ARIA-H. The meta-analysis of PET-SUVR showed an overall significant effect of mAbs in reducing amyloid (SMD –0.88), but when considering clinical efficacy, data on CDR-SB showed that treated patients had a statistically significant but clinically non-relevant lower worsening (MD –0.15).
Conclusion:
Our results suggest that the risk-benefit profile of mAbs remains unclear. Research should focus on clarifying the effect of amyloid on cognitive decline, providing data on treatment response rate, and accounting for minimal clinically important difference. Research on mAbs should also investigate the possible long-term impact of ARIA events, including potential factors predicting their onset.
Keywords
INTRODUCTION
The last Alzheimer’s disease (AD) drug development pipeline showed that disease-modifying therapies (DMT) are currently the most frequently investigated agents, being 82.5% of the total number of considered agents. Among the drugs considered as DMTs, 16 (15.4%) are anti-amyloid beta (anti-Aβ) monoclonal antibodies (mAbs), and 11 (10.6%) are anti-tau mAbs [1].
Research on anti-Aβ agents has been ongoing for about 15 years [2], though the lack of clinical benefits has prevented the marketing authorization except for the approval of Biogen’s aducanumab so far only by the Food and Drug Administration (FDA) [3]. The main issue when considering amyloid-targeted agents is the lack of evidence supporting the association between amyloid load and cognitive outcomes, with studies reporting only an indirect link between amyloid plaques and cognitive decline [4, 5].
Another relevant issue related to the use of anti-Aβ mAbs in patients with AD was that a higher risk of developing vasogenic cerebral edema and cerebral micro-hemorrhages was observed in participants treated with mAbs. These abnormalities were first identified through magnetic resonance imaging (MRI) during trials testing one of the first investigated mAbs, bapineuzumab. In 2011, a working group that was specifically created to investigate these events, renamed this type of vasogenic edemas and micro-hemorrhages as amyloid-related imaging abnormalities (ARIA), and specifically ARIA-E (vasogenic edema) and ARIA-H (micro-hemorrhage) [6]. An increased risk of both ARIA-E and ARIA-H was also observed in patients treated with other anti-Aβ mAbs, even if with heterogeneous risk ratios due to some differences in the pharmacodynamics of each mAb. These differences are the result of mAbs having been produced with the objective of overcoming the safety and efficacy issues observed when testing preceding mAbs [7].
During the last few years, six systematic reviews (SR) and meta-analyses have been published investigating anti-amyloid agents, including mAbs, for the treatment of AD or mild cognitive impairment (MCI) [8–13]. The 5 SRs included 11 to 17 randomized controlled trials (RCTs) each, published up to 2020, and all reported a statistically significant, but clinically non-relevant, improvement in cognitive and neurobiological outcomes (i.e., amyloid-positron emission tomography (PET) and cerebrospinal fluid (CSF) p181-tau), along with a significantly higher risk of ARIA events [8–13]. However, most of these SRs only included published studies [8, 11] and considered widely heterogeneous molecules, such as active and passive immunization [9, 10] and all anti-amyloid agents [12]. Moreover, one SR only included phase III studies [11], and another only included phase II and III studies [10], with the latter also including only studies enrolling participants with mild to moderate AD. When reporting outcomes, most of these SRs considered only Alzheimer’s Disease Assessment Scale–Cognitive Subscale and Mini-Mental State Examination as clinical outcomes, leaving out the Clinical Dementia Rating scale-Sum of Boxes (CDR-SB) which is clinically considered as the only scale reporting both cognitive and functional traits.
Therefore, considering the limitations of these SR, our objectives were to: 1) identify all registered trials investigating mAbs for the treatment of MCI due to AD or AD at any stage; 2) retrieve all available data, both published and unpublished, from registered trials; 3) cumulatively analyze all available data on both safety outcomes, focusing on ARIA-E and ARIA-H, and efficacy outcomes, specifically considering the CDR-SB score and cerebral amyloid burden measured by amyloid-PET.
METHODS
This SR was performed following the Cochrane handbook for systematic reviews [14] and reported based on the PRISMA statement [15]. We checked for the originality of our SR on PROSPERO, PubMed, ISI Web of Knowledge (WoK), and The Cochrane Library Databases (CLD). The SR protocol was registered on PROSPERO (CRD42021259855).
Search strategy and selection criteria
We initially performed a structured search (Supplementary File 1) in the two main registration databases, ClinicalTrials.gov (CT) and the European Clinical Trials Register (EUCT). No restrictions were applied for status, study design, study phase, date of publication, or language. We selected only studies that investigated any type of mAb in participants with a clinical diagnosis of AD or MCI. A list of all registered trials, and a list of all investigated mAbs were created. Available information on all the identified trials was searched both on the registration databases in the form of files with trial results and on the literature databases in the form of published studies. Based on the list of active principles obtained from CT and EUCT, a search string was defined (Supplementary File 1) and used on PubMed, ISI-WoK, and The CLD. No restrictions were applied for date of publication, study design, or language. A list of all trials with available information was defined, and details on the source of data were specified.
Study selection
Studies were initially selected based on their titles and abstracts, duplicates were removed, and full texts of all selected studies were gathered for independent assessment. Further potentially relevant articles were also retrieved from the references of identified studies. Selected studies were applied predefined eligibility criteria. All trials reporting safety and efficacy data on any type of mAbs for the treatment of subjects with a diagnosis of MCI due to AD or AD at any stage were included. Studies enrolling healthy participants or participants with any diagnosis other than MCI or AD at any stage, and all studies investigating any drug other than mAbs were excluded. In case of studies enrolling both healthy participants and patients with AD, only data on patients with AD were considered. Case reports, case series, non-systematic or narrative reviews, letters, commentaries, and editorials were also excluded. SRs were only considered to check for references and consistency of results.
Study selection, and data extraction and evaluation were performed by four independent reviewers (AA, EL, PP, VZ). Disagreements were resolved by discussion, consensus, or a third researcher. All available data up to July 7, 2021, were gathered and analyzed.
Data extraction and quality assessment
A list of all registered studies investigating mAbs for the treatment of AD or MCI was defined. For each trial, information on trial identification number, phase, and status were gathered. Trials were classified as completed, terminated, and ongoing, with ongoing defined as including any status indicating a non-completed study (e.g., active, enrolling, etc.).
For all studies for which information were avai-lable, the source of data was recorded (i.e., unpublished results retrieved from registration databases, published studies). Data were extracted using standardized forms including year of publication, characteristics of the included population, type of mAb, and results for each considered outcome. In case of information available from multiple sources, data were compared and, in case on inconsistencies, the most recent source was selected. Supplementary material and further additional reference material were also retrieved, and, in case of partial or incomplete data, the missing values, where possible, were calculated (e.g., graphs).
Published RCTs meeting inclusion criteria were qualitatively assessed using the Cochrane Risk of Bias (RoB) tool [14]. The RoB tool was used to classify the risk of bias as ‘Low’, ‘High’, or ‘Unclear’. Other potential biases and/or methodological flaws were also considered. Only published studies were applied the RoB tool.
Data synthesis and analysis
Results were summarized in a narrative form. Meta-analyses of available data were performed for both safety and efficacy outcomes using the software Review Manager version 5.4.1. Data were stratified for type of mAb and organized in chronological order from the least to the most recently developed mAb, to account for heterogeneity and to identify possible evolutions over time in the safety or efficacy profile.
For safety outcomes, two meta-analyses of the frequency of adverse events (AE) and severe AEs (SAE) were performed, along with two specific meta-analyses on the reported frequency of ARIA-E and ARIA-H in all the studies for which data were available. Only events defined as ARIA within each study were included in the analyses, and data were stratified for way of administration in case of mAbs having more than 1 of them. To analyze the possible weight of potential misclassification due to the heterogeneity in the definition of ARIA across studies, we also performed a sensitivity analysis including all events classified as micro-hemorrhages, superficial siderosis, hemosiderin depositions, vasogenic edemas, etc. reported in the studies. For studies that provided data on APOE ɛ4 status, a further subgroup analysis was performed on the frequency of ARIA events according to APOE carrier status.
For efficacy outcomes, data on the mean change from baseline in CDR-SB score and the PET standardized uptake value ratio (SUVR) were meta-analyzed. We considered the CDR-SB scale as a main clinical endpoint as it includes both cognitive and functional aspects, while we considered PET-SUVR as a main endpoint for its accuracy in asse-ssing brain amyloid burden. As data on these continuous variables were provided per dose-group compared to a single placebo group, participants in the placebo group were subdivided as equally as possible, to avoid their being over-represented in the meta-analyses, leaving the group mean and SDs unchanged. This method is only partially effective in overcoming the unit-of-analysis error, but at least allowed to approximately investigate potential differences across intervention arms [14]. Meta-analyses were performed using a random effect model, and data for categorical variables were presented as risk ratios (RR) with their 95% confidence intervals (CI), while data for continuous variable were presented standardized mean differences (SMD or mean differences (MD) with their 95% CIs.
RESULTS
Searches on CT and EUCT yielded 3,881 and 687 records respectively. After screening, 97 protocols were included referring to RCT testing mAbs in subjects with MCI or AD. Of these, 56 were registered only on CT, 1 was registered only on EUCT, while 40 were registered on both platforms.
Bibliographic searches on literature databases yielded 9,116 records. After a first screening, 58 records were selected. Of these, 25 were further excluded, as they did not meet the inclusion criteria. Overall, 34 articles were included, referring to 11 mAbs [16-49]. Four out of these 34 studies did not report any NCT/EUCT code therefore they were considered as having no trial identification code [20, 45]. The flow diagram of included studies is reported in Fig. 1. This led to a list of 101 trials either completed/terminated or still ongoing (97 from registration databases and 4 from literature alone), that were identified either using their NCT or EUCT code or using the “no code available” label.
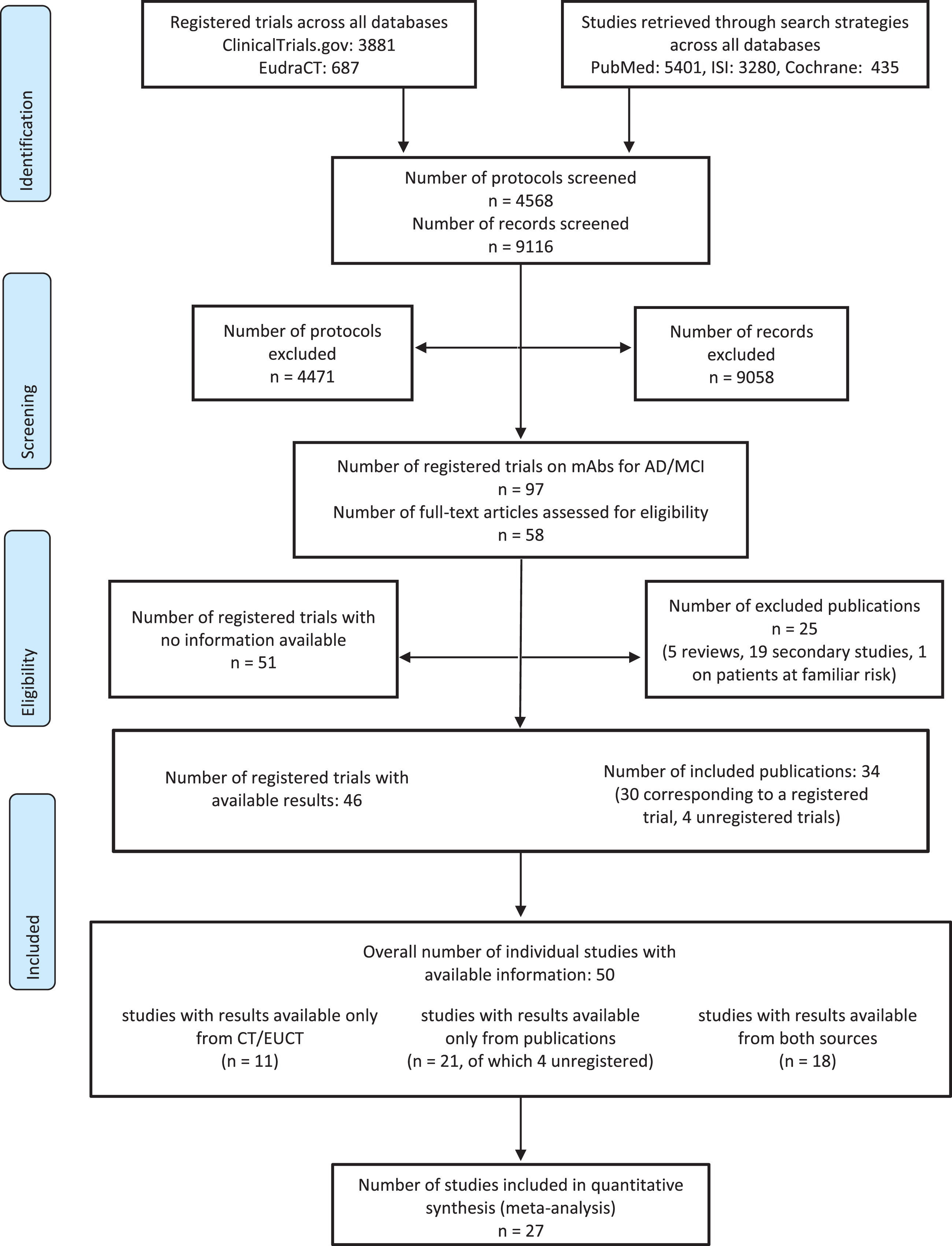
Flow diagram of identified published and unpublished studies.
A total of 75 out of 101 studies were classified as either completed (n = 56) or terminated (n = 19), investigating 22 mAbs. Considering both published and unpublished data, results were available for 50 trials, all classified as completed/terminated except for 2 studies classified as still ongoing, but with published safety and efficacy data [32, 34] (Table 1). All the unregistered studies were considered as completed. For 18 trials data were available from both published studies and registration databases [16, 47–49] for 21 trials data were available only from published studies [17, 44–47], and for 11 trials data were available only from unpublished sources (Supplementary Table 1).
Summary of the main characteristics and results of the RCT for which data were available
AD, Alzheimer’s disease; APOE, apolipoprotein E, ARIA-E, amyloid-related imaging abnormalities-vasogenic edema; ARIA-H, amyloid-related imaging abnormalities-micro-hemorrhage; BAPI, Bapineuzumab; CDR-SB, Clinical Dementia Rating scale-Sum of Boxes; CREN, Crenezumab; CSF, cerebrospinal fluid, CT, computerized tomography; EU, European Union; FCSRT, Free and Cued Selective Reminding Test; FP, first posted; FU, follow up; IV, intravenous; IWG, International Working Group; LS, least squares; MAD, multiple ascending dose; MCI, mild cognitive impairment; mITT, modified intention-to-treat; MMSE, Mini-Mental State Examination; MRI, magnetic resonance imaging; NIA-AA, National Institute on Aging-Alzheimer’s Association; NINCDS-ADRDA, National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association; OLE, open label extension; PET, positron emission tomography; PL, placebo; PLA, placebo part A; PLB, placebo part B; RCT, randomized controlled trial; SAD, single ascending dose; SC, subcutaneous; SD, standard deviation; SE, standard error; SUVR, standardized uptake value ratio; T, treatment; T-HD, treatment-high dose; T-IV, treatment-intravenous; T-LD, treatment-low dose; T-SC, treatment-subcutaneous; W, White; J, Japanese. †data partially calculated based on available graphs; §Only data on events defined as ARIA within each study are included in the table, data on all events not classified as ARIA are included in the sensitivity analyses; ∫Aduhelm product information was not considered as “unpublished source” as it was not provided by the registration databases; however, some data were retrieved from this source to carry out meta-analyses; **p<0.05 for the change from baseline within treatment group; *In the EXPEDITION 1 and EXPEDITION 2, the study reported that the composite SUVr did not change significantly in the solanezumab or the placebo groups in both studies; #No measure of dispersion available.
Overall, the 101 registered trials investigated 27 mAbs. Most of them targeted different forms of the Aβ peptide (n = 17), 8 targeted different forms of tau, 1 targeted the microglial receptor TREM2, and 1 targeted SEMA-4D. Data were available only on 12 anti-Aβ mAbs, while no data were available on the remaining 15 mAbs.
Methodological quality of published studies
Methodological quality was assessed only for studies published in journal articles (Supplementary Figure 1). The overall quality of included studies was moderate to low. The main limitation of included studies was a lack of information on the randomization process and the procedures for allocation concealment and blinding. Moreover, most of the studies were phase I with limited sample sizes, and some were structured in sub-phases, or divided participants in subgroups, thus further limiting the sample size and making the study design more complex.
Most of the included studies reported only AEs and SAEs with frequencies≥5%, with some choosing higher cut-offs (e.g.,≥10%) or reporting only AEs or SAEs occurring in≥2 participants. In these cases, we adopted a more conservative approach, considering them as being at unclear or high risk of bias for selective reporting. This approach was chosen due to some mAbs having risen safety concerns due to the occurrence of specific AEs, such as ARIA. Thus, a more complete reporting would have been expected.
Safety
Results from the meta-analysis of AEs reported a marginally but significantly higher frequency of AEs in treated participants compared to placebo, with a RR of 1.04 (95% CI 1.02–1.06, I2 74%). However, data from the meta-analysis of SAEs did not show significant differences between groups (RR 1.02, 95% CI 0.96–1.09, I2 20%) (Supplementary Figures 2 and 3).
When considering ARIA events, a significant heterogeneity in the definition and reporting of ARIA was observed across studies. In some studies (n = 14), the information on the overall frequency of ARIA was missing, while 6 RCTs reported that no events of ARIA occurred during the study. In some cases, the criteria adopted for discriminating between what was considered as ARIA and what was considered as micro-hemorrhages, vasogenic edemas, etc. was unclear. This heterogeneity could be due to the evolution over time of the definition of ARIA as research on mAbs progressively increased [6]. To account for heterogeneity and over-time variations, data were stratified per mAb and presented in a chronological order. Moreover, a sensitivity analysis including events that were not defined as ARIA was also performed.
The meta-analysis of data on ARIA-E showed an overall 10.65 RR of ARIA-E for any mAb (Fig. 2), that slightly increased to 10.86 (95% CI 8.38–14.06) when performing the sensitivity analysis (Supplementary Figure 4). Ten RCTs reported that no events of ARIA-E occurred during the study period and therefore were not included in the forest plot. The meta-analysis of data on ARIA-H showed an overall RR of 1.75 (Fig. 3), that increased to 2.11 (95% CI 1.87–2.38) when performing the sensitivity analysis, which also allowed to include data for intravenous (IV) bapineuzumab (3.01, 95% CI 2.01–4.51) that were missing in the previous analysis, as in studies on bapineuzumab micro-hemorrhages and siderosis were not yet clearly identified and diagnosed, nor defined as ARIA-H (Supplementary Figure 5). Ten RCTs reported that no ARIA-H events were observed during the study period.
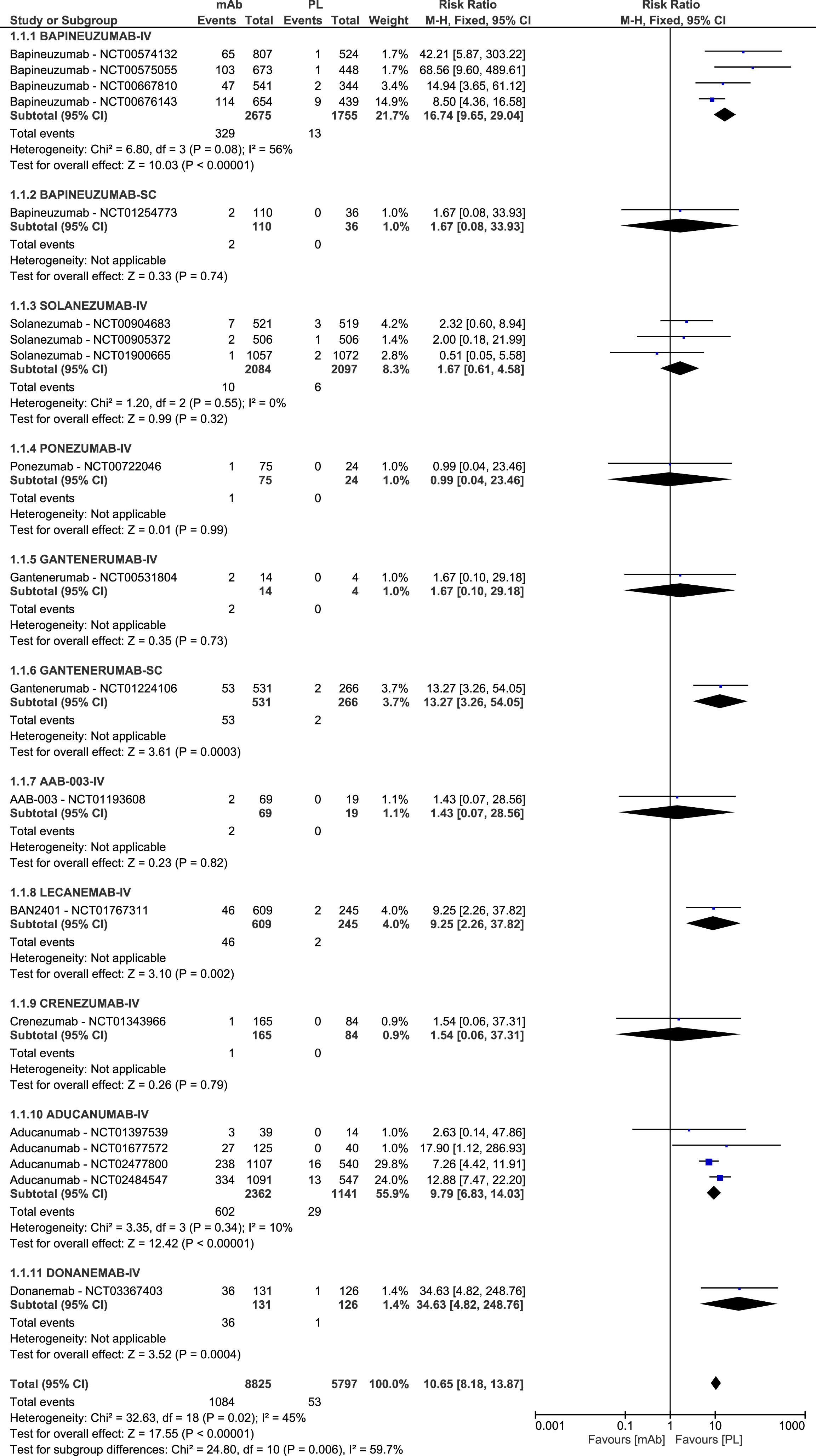
Forest plot of the meta-analysis of available data on the frequency of ARIA-E.
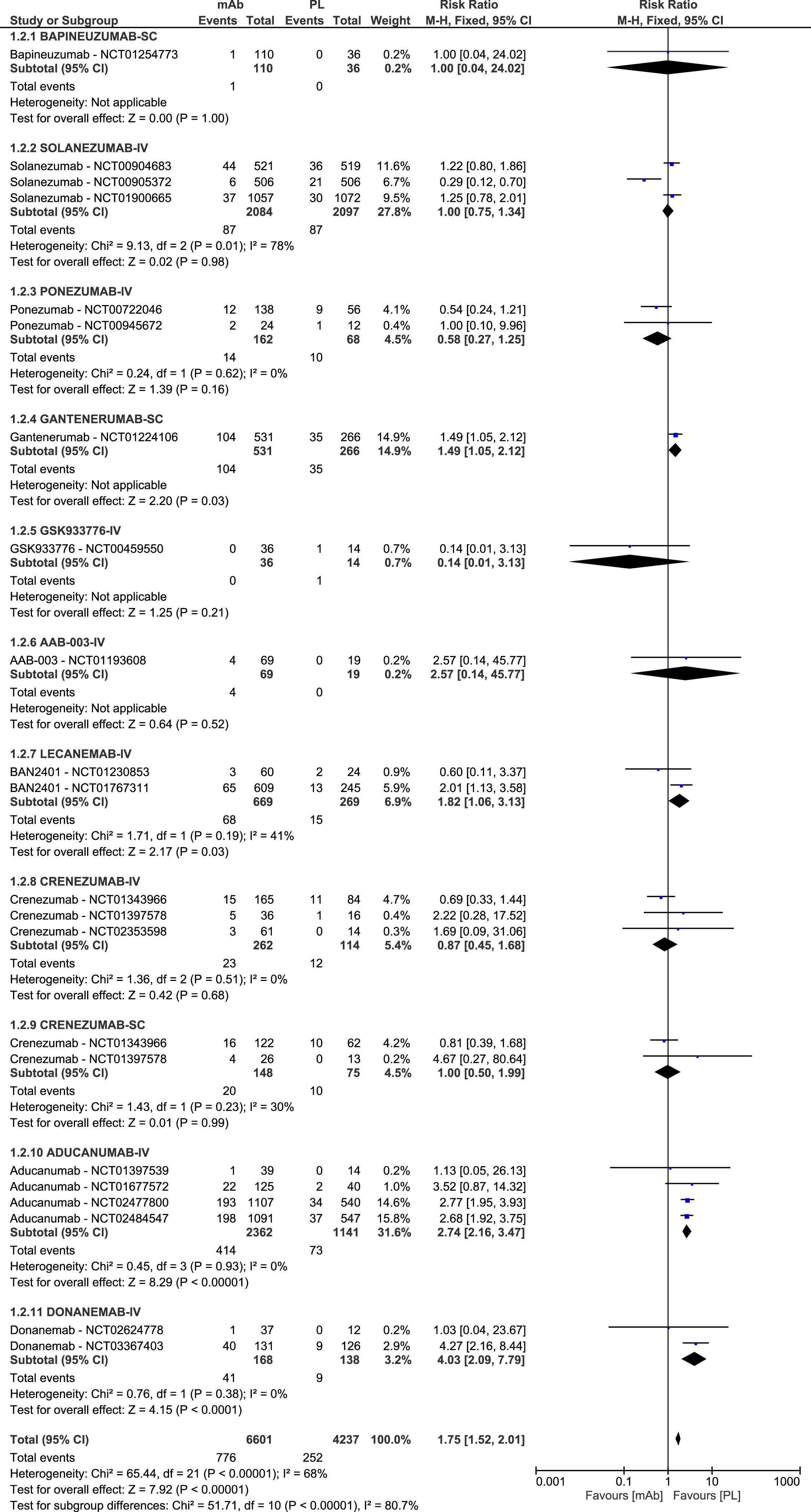
Forest plot of the meta-analysis of available data on the frequency of ARIA-H.
No evolutions over time in the frequency of both ARIA-E and ARIA-H were observed, with the most recent mAb, donanemab, showing the highest risk of both ARIA E (RR 34.63, 95% CI 4.82–248.76) and ARIA-H (RR 4.03, 95% CI 2.09–7.79), and two less recent mAbs, ponezumab and GSK933776, reporting the lowest risks of ARIA-E (RR 0.99, 95% CI 0.04–23.46) and ARIA-H (RR 0.14, 95% CI 0.01–3.13) respectively.
When considering mAbs by different ways of administration, no differences were observed in the frequency of ARIA-H between IV and subcutaneous (SC) crenezumab, while significantly less ARIA-E were observed in SC compared to IV bapineuzumab, and significantly more ARIA-E were observed in SC compared to IV gantenerumab. However, results on SC bapineuzumab were based only on 1 single relatively small phase II study, and results on IV gantenerumab only on 1 small phase I study.
When considering the potential effect of APOE status on the risk of ARIA, some differences were observed between carriers (APOE+) (ARIA-E RR 13.47, 95% CI 8.18–22.17, I2 43%; ARIA-H RR 1.50, 95% CI 1.05–2.15, I2 15%) and non-carriers (APOE-) (ARIA-E RR 12.10, 95% CI 6.00–24.41, I2 30%; ARIA-H RR 2.18, 95% CI 0.85–5.56, I2 0%). Data stratified per type of mAb showed a higher risk of ARIA-E in APOE+participants for aducanumab (APOE+: RR 7.83, 95% CI 1.10–55.85; APOE- RR 2.96, 95% CI 0.38–22.88), donanemab (APOE+: RR 30.32, 95% CI 4.23–217.25; APOE- RR 8.51, 95% CI 0.48–152.42), and SC gantenerumab (APOE+: RR 41.61, 95% CI 2.57–672.70; APOE- RR 3.02, 95% CI 0.69–13.16) (Supplementary Figure 6), and a higher risk of ARIA-H in APOE+participants for aducanumab (APOE+: RR 3.05, 95% CI 0.76–12.22; APOE- RR 1.85, 95% CI 0.22–15.35) (Supplementary Figure 7).
Efficacy
Data on change from baseline of PET-SUVR were available from 14 studies on 7 mAbs, while data on CDR-SB scores were available from 16 studies on 8 mAbs.
The meta-analysis of PET-SUVR levels showed an overall significant effect of mAbs compared to placebo in reducing the amyloid burden, with an SMD of –0.88 (95% CI –1.30 to –0.47; I2 95%). A reduction in the amyloid burden was observed in all treated groups compared to placebo, except in the group treated with IV crenezumab, who showed a lower decrease in the amyloid burden (SMD 0.36, 95% CI –0.40 to 1.12). A statistically significant difference between groups in the reduction of amyloid burden was observed for 4 mAbs, with the highest difference reported for donanemab (SMD of –2.56, 95% CI –2.91 to –2.21) (Fig. 4).
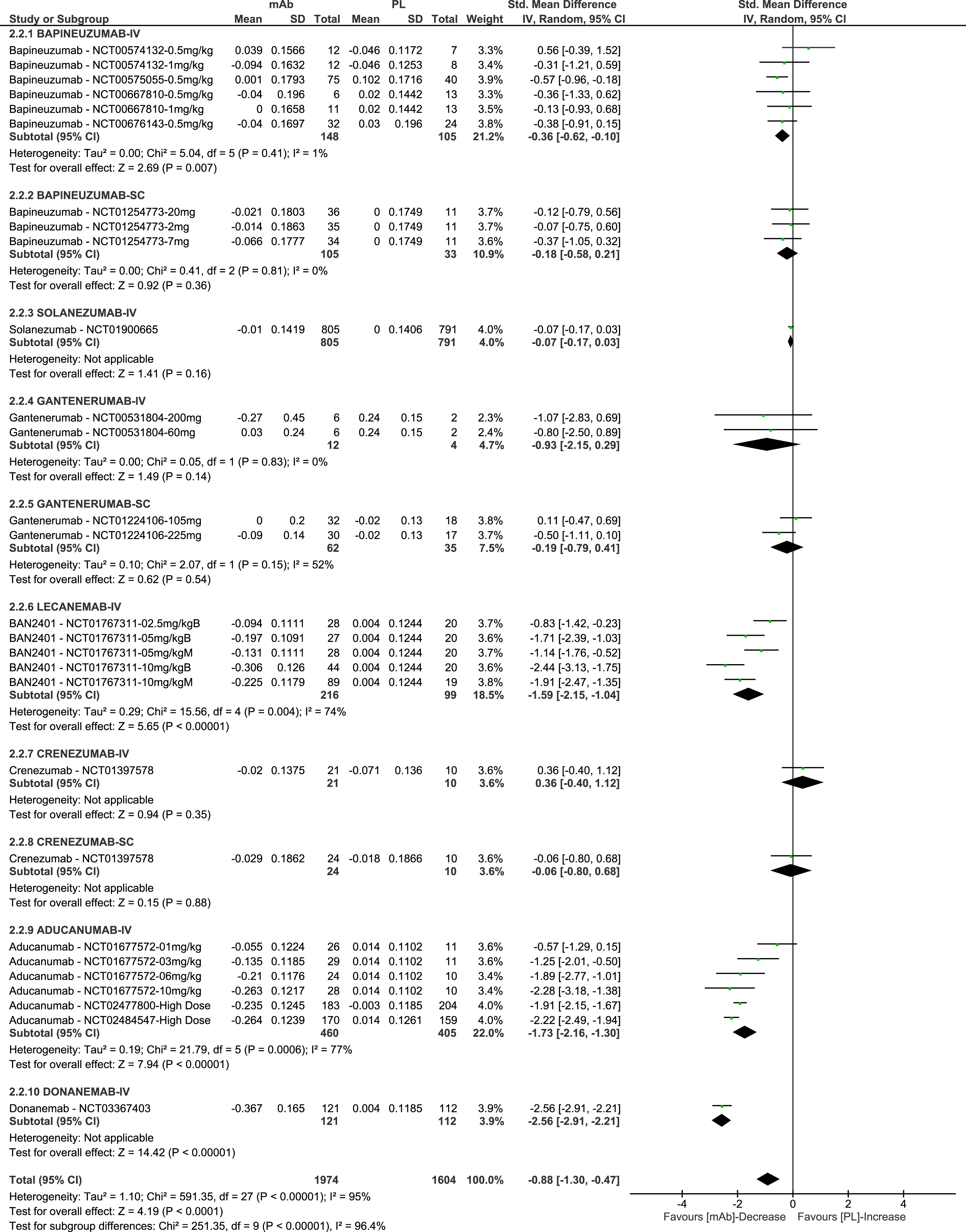
Forest plot of the meta-analysis of available data on change from baseline in PET-SUVR.
When considering clinical efficacy, almost half of the studies included in the meta-analysis were classified as terminated due to futility, and the CDR-SB scale was often considered as a secondary endpoint across all considered studies (Fig. 5). None of the included studies reported an improvement from baseline in the CDR-SB score, except for 1 small phase I study on AAB-003 that reported a very small improvement from baseline in the placebo group (–0.35, SD 1.935). Results from the meta-analysis of CDR-SB scores showed that patients treated with mAbs had a statistically significant lower worsening, with a MD –0.15 (95%CI –0.26 to –0.04); I2 2%. However, considering 1-2 points in the CDR-SB scale as a minimal clinically important difference [50], the observed differences between groups were not clinically relevant. No analyses per responder were reported.
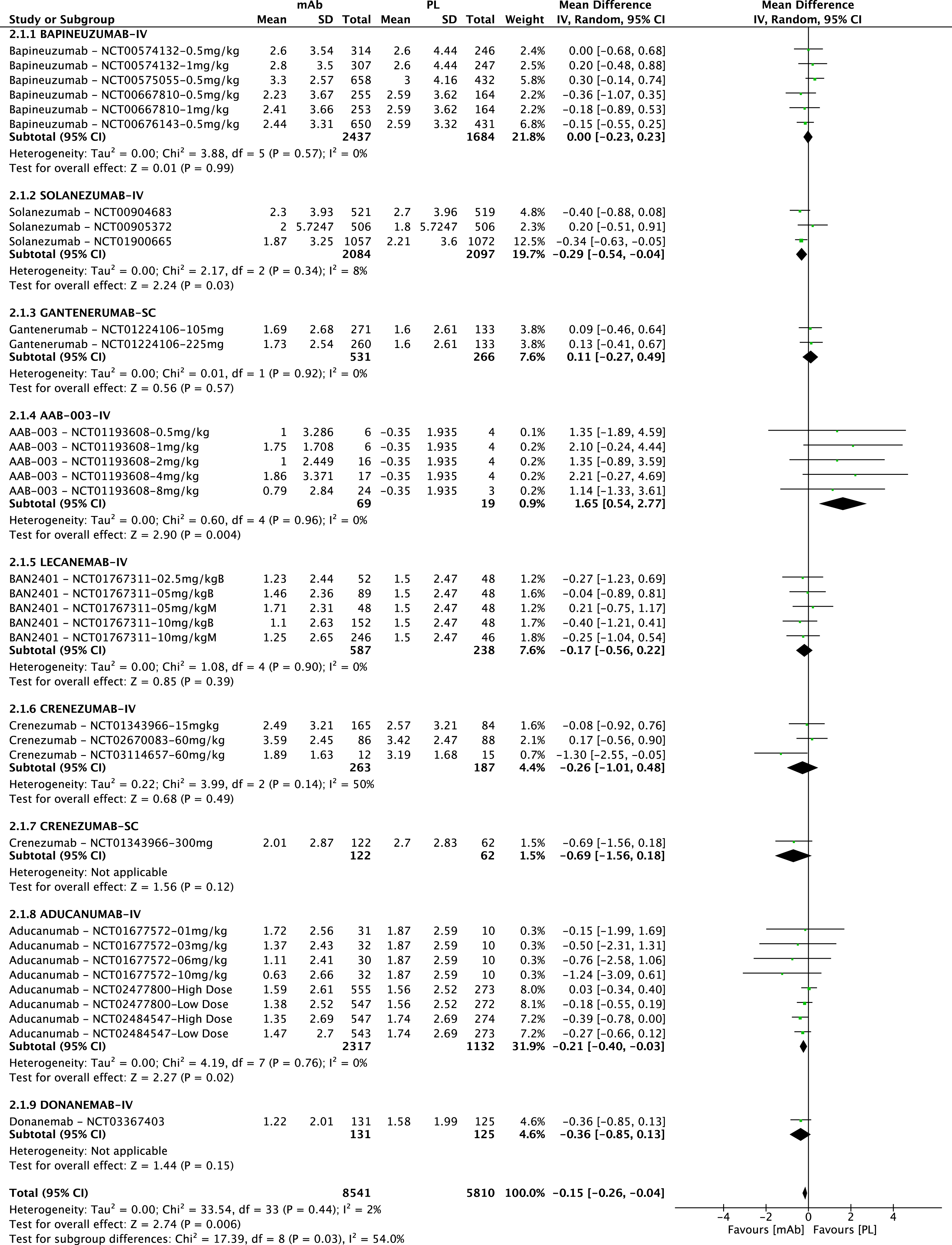
Forest plot of the meta-analysis of available data on change from baseline in the CDR-SB score.
When considering potential evolutions over time, as for PET-SUVR levels, the most recently developed mAbs seem to be more effective in decreasing the amyloid burden. Specifically, lecanemab, aducanumab, and donanemab, that are still investigated for the treatment of people with MCI due to AD or AD, seem to be the ones with the most significant results in terms of PET-SUVR levels. However, results from the CDR-SB scale do not show a similar profile, with all mAbs having substantially similar ranges of results.
DISCUSSION
Since 2005, 101 trials have been registered investigating mAbs in patients with MCI or AD. Of these, information is available from 50 trials enrolling nearly 18,000 participants. To our knowledge, this is the first SR reporting the number of trials with published results (n = 39) and unpublished results (n = 66) and documenting the number of trials with results only available from registration databases (n = 11) and those with no data available (n = 51). The rate of unpublished and unavailable results is clearly in contrast with the principles of open science and open data movement. Missing information both hinders the process of critical evaluation of acquired evidence and affects the possibility of raising hypotheses on new possible etiopathogenetic mechanisms in AD that could be investigated and tested as possible therapeutic targets [51]. Moreover, the lack of negative studies published in journal articles prevents the opportunity for a realistic discussion among the scientific community. Therefore, the US National Academy of Medicine recommended the creation of a culture of responsible sharing of data from clinical trials [52].
Our results on safety data showed an overall higher risk of both ARIA-E and ARIA-H in patients treated with mAbs compared to placebo. When analyzing data in a chronological order to account for a possible evolution of the safety profile over time, the analysis showed no variations over time. However, newer mAbs, such as donanemab, seemed to have a common profile showing an increase in the neurobiological efficacy despite an increase in the frequency of ARIA events. However, ARIA should be considered as treatment-related AEs, and, since they are extremely challenging to account for both in RCTs and in clinical practice, specific protocols should be defined, including criteria for identifying the type of subjects eligible for MRI, the timing of MRI controls, and the criteria for interrupting treatment. The accurate and timely identification of ARIA is, in fact, essential, as no data are available on their potential long-term consequences. A longitudinal study on families with dominantly inherited AD reported that the presence of either prevalent or incident cases of cerebral micro-hemorrhages predicted a faster decline of the CDR score [53]. This shows that, though a very small proportion of ARIA events were symptomatic, and most of them resolved within the duration of the study, the potential effect of ARIA on the natural history of the disease remains unclear. Conducting further and more targeted statistical analyses to explore whether specific variables can be useful to predict the risk of both overall and symptomatic ARIA, could also help in reducing the safety concerns related to these drugs.
To analyze the efficacy of mAbs, we considered PET-SUVR data, which are currently considered as one of the reference standard measures for assessing in vivo the cerebral amyloid burden, and the CDR-SB scale, which is currently the most widely used cognitive and functional measure in clinical practice. We chose amyloid-PET as it has been proven to have a high accuracy in detecting amyloid burden [54], with good inter-rater agreement [55], and no significant differences between tracers [56] and compared to CSF. A study investigating the diagnostic accuracy of CSF Aβ1–42 and amyloid-PET reported an overall 77% concordance and 23% discordance between the two measures [57], and another study investigating the accuracy of the CSF Aβ1–42/Aβ1–40 ratio and amyloid-PET reported a 65% and 88% concordance for MCI (n = 48) and for AD (n = 7) [58] respectively. Results from the meta-analyses of PET-SUVR data showed an overall significant effect of mAbs in reducing the amyloid burden, with the most recent mAbs having the highest effect. However, when considering results on clinical efficacy, data showed an overall statistically significant but clinically non-relevant lower worsening of the CDR-SB scores in patients treated with mAbs compared to placebo. All included studies reported results only in terms of means and standard deviations or standard errors, with none of them reporting data as response rates nor providing a definition of responders accounting for the minimum clinically important difference that, when considering the CDR-SB, is 1-2 points [50, 59]. The observed lack of clinical efficacy is in line with results from a recent meta-analysis on anti-amyloid treatments in AD reporting that lowering or removing amyloid plaques led to no substantial improvement in cognition [12]. Some postmortem studies also showed that in subjects who were treated with active immunization the removal of amyloid plaques, even though lasting for 14 years after treatment, failed to either stop or slow disease progression [60, 61]. Therefore, currently available evidence does not support a clear link between amyloid load and cognitive performance, suggesting that reducing the amyloid load might have low-to-no effect in improving cognitive performance or slowing cognitive decline [4].
On June 7, 2021, the FDA authorized the marketing of aducanumab (ADUHELM, Biogen), under accelerated approval procedure. Based on the summary of product characteristics, aducanumab “is indicated for the treatment of AD”, and “treatment with ADUHELM should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was initiated in clinical trials”. The approval was based on results from a surrogate endpoint, the reduction of amyloid plaques, that was considered as “reasonably likely” to predict a clinical effect [62, 63]. Furthermore, Elli Lilly, based on results from a phase II study [32], recently announced their intention to apply to FDA for an “accelerated approval” of donanemab (https://www.drugdiscoverytrends.com/lilly-pushing-for-accelerated-fda-approval-of-alzheimers-drug-donanemab/). Considering the mentio-ned evidence questioning the relation between amyloid burden and cognitive outcomes, the approval of aducanumab has raised a large-scale debate thr-oughout the scientific community [64, 65]. These events, in fact, seem to suggest some similarities with the approval procedures of some oncological drugs. A recent study reported that between 1992 and 2019 the FDA authorized 194 oncological drugs, all based on surrogate endpoints for a specific cancer type [67]. The study also reported that the use of surrogate endpoints increased from 2016 to 2019, underlining that, though surrogate endpoints can ease trial completion, they increase uncertainty in the actual benefit of marketed intervention. As highlighted in the study, the FDA steadily accepting surrogate endpoints that are not validated nor jus-tified by regulatory precedents, along with its unlikeliness to demand strict confirmation of any clinical benefit after market approval might have relevant consequences. When considering AD and mAbs, though the approval procedure of aducanumab required Biogen to provide additional data on clinical efficacy in the next 9 years, such extended period further increases uncertainty, underlining that currently available data are not robust enough to show a direct impact, that could be relevant to patients over time, of removing amyloid plaques on clinical outcomes. Moreover, the lack of data on the possible long-term effects of ARIA on brain and cognitive performances raises additional concerns on the risk-benefit profile for mAbs. On the same basis, in fact, the European Medicines Agency considered that the studies presented by Biogen “did not show that the medicine was sufficiently safe as images from brain scans of some patients showed abnormalities suggestive of swelling or bleeding, which could potentially cause harm” and that “it is not clear that the abnormalities can be properly monitored and managed in clinical practice” [67]. Furthermore, the European Medicines Agency “noted that although Aduhelm reduces amyloid beta in the brain, the link between this effect and clinical improvement had not been established”, and therefore the European Medicines Agency’s opinion was “that the benefits of Aduhelm did not outweigh its risks and it recommended refusing marketing authorisation” [67].
This SR aimed at systematically collecting all available information on trials on mAbs for MCI and AD, documenting the amount of published, unpublished, and unavailable data. A possible limitation to this SR is that we did not report data for all the outcome measures considered in the included trials. However, the PET-SUVR and CDR-SB scores are reported to be accurate tools to measure the amyloid load and the cognitive and functional performance of subjects with MCI or AD. Our purpose was also to attempt to provide an “historical” overview of research on this topic, thus focusing on the interpretation of data on mAbs over time rather than considering results only in a cumulative way regardless of whether mAbs were still under investigation or already discontinued. Though our SR has no regulatory purposes, our results strongly suggest that the risk-benefit profile on mAbs remains unclear.
Research on mAbs should be focused on clari-fying whether removing the amyloid burden affects the progression of cognitive decline, providing data also on treatment response rate, accounting for MCID. This is extremely relevant considering that the target population is shifting towards the earlier stages of the disease in an assumption that removing plaques in a relatively still intact brain can lead to a higher clinical effect. Research on these drugs should also be focused on determining the possible long-term impact of ARIA events, investigating potential factors predicting their onset, as the treatment with mAbs is still linked, even in the most recently developed ones, to a significantly higher risk of ARIA.
