Abstract
Background:
Sleep disturbances are frequent in Alzheimer’s disease (AD).
Objective:
To summarize the impact of sleep disturbances on AD patients and their caregivers and the effects of currently available sleep therapies.
Methods:
Published studies (January 1985–March 2020) assessing the burden associated with insomnia/sleep disturbances in the AD population and insomnia treatment effects were identified by searching PubMed, Embase, and Cochrane Library and screened against inclusion criteria.
Results:
58 studies assessing patient and caregiver burden, institutionalization, and insomnia treatments in AD patients with sleep disturbances were identified. Sleep disturbances were associated with worse cognition, functional ability, and behavioral and neuropsychological functioning. Health status and quality of life of both patients and caregivers were reduced in the presence of sleep disturbances. Sleep disturbances were also associated with institutionalization. Although significant associations between sleep problems and clinical outcomes were apparent, there was generally no control for other influencing factors (e.g., cognitive status). Bright light and behavioral therapies as well as drugs showed some promise in AD patients, but studies were primarily small and limited data were available, particularly in regard to the effect on associated clinical burden.
Conclusion:
Sleep disturbances are a significant problem for AD patients and caregivers, associated with behavioral and psychological problems and cognitive decline. However, they remain poorly characterized and under-researched. As the global population is aging and AD is on thes rise, data from larger, prospective trials are required to fully understand the clinical correlates of sleep disturbances and the impact insomnia treatments can have.
Keywords
INTRODUCTION
Alzheimer’s disease (AD) is a progressive neurodegenerative disease, characterized by continuing cognitive decline and memory loss [1]; it accounts for 60% to 80% of dementia cases in adults > 65 years of age. With an aging global population, the AD population is expected to triple over the next 40 years, to over 13 million cases [2].
AD is associated with significant healthcare costs and resource utilization, particularly when patients require admission to care facilities [3]. The behavioral symptoms of dementia, including disturbed sleep, nighttime awakenings and wanderings place a substantial burden on family and caregivers, and lead to the early institutionalization of AD patients [2, 4].
Insomnia is characterized by difficulty initiating or maintaining sleep among individuals with adequate opportunity to sleep and is associated with daytime consequences, such as an inability to perform daily activities [5]. Insomnia symptoms and sleep disturbances (collectively referred to in this manuscript as sleep problems) increase with age and are common in AD patients, with a prevalence of > 50% [6 –8]. AD patients also experience more severe symptoms of insomnia and sleep disturbances, such as increased sleep latency and reduced sleep maintenance, as well as progressive deterioration and instability of circadian rhythms, compared to older adults without AD [9, 10]. Moreover, insomnia is also a risk factor for AD [11], and a bidirectional relationship is thought to exist between poor sleep and AD pathology [12].
Despite the high prevalence and substantial disease burden of insomnia in AD patients, including negative impacts of insomnia symptoms on mental and physical health, quality of life (QoL), and functional ability, treatment options for this patient population are limited. Benzodiazepine receptor agonists are associated with an increased risk of falls, fractures, and clinically significant impairments in balance and cognition upon awakening [13]. The American Geriatrics Society strongly recommends against the use of benzodiazepines and related non-benzodiazepines in elderly patients [14]. Moreover, current non-pharmacological treatments such as cognitive behavioral therapy for insomnia (CBT-I) may not be effective as AD patients may be unwilling or unable to participate in therapy [5]. Due to the limitations of non-pharmacological and pharmacological treatments for the management of insomnia in AD patients there is a significant unmet need. Currently, one medication (suvorexant) has been specifically approved for the treatment of insomnia in AD patients by the US Food and Drug Administration (FDA).
This literature review aims to summarize the clinical, economic, and QoL impact of insomnia and sleep disturbances on patients and their caregivers, as well as the abilities of currently available therapies to treat sleep problems and reduce their impacts in this population.
METHODS
Search strategy
We searched Publisher Medline (PubMed), Excerpta Medica Database (Embase), and Cochrane Library for articles published between January 1985, and March 2020, using combinations of keywords and Medical Subject Headings (MeSH) terms pertaining to AD, insomnia/sleep disturbances, disease and caregiver burden, healthcare resource utilization (HCRU), treatment patterns, and guidelines (Supplementary Tables 1–9). Reference lists of included publications were reviewed to identify relevant publications not captured through the database searches.
Criteria for study inclusion/exclusion
Eligible studies and reviews met the following inclusion criteria: 1) AD patients with insomnia, or sleep disorders, disturbances or dysfunctions (specific sleep disorder/disturbance not reported); 2) disease burden (QoL, functional status, cognitive functioning, behavioral symptoms, and impact on daily living) and caregiver burden outcomes, HCRU, treatment patterns and guidelines; 3) peer-reviewed; 4) English language. Publications were excluded if they included only sleep disorders other than insomnia such as hypersomnia, parasomnia, excessive daytime sleepiness, and obstructive sleep apnea. Where studies were published as both research articles and conference abstracts, the latter were excluded. Full PICOS criteria are reported in Supplementary Table 10).
Data extraction and synthesis
Data extraction was performed by one reviewer and cross-checked by a second reviewer using standardized forms to ensure accuracy. Data from the extracted publications were qualitatively synthesized to summarize the findings of the literature review.
RESULTS
The search identified 4,860 articles, from which 311 were taken forward to full text review. After screening the full texts against the eligibility criteria, 161 publications were included (123 primary studies and 38 reviews). Eight more publications were included through an additional review of the reference lists (Fig. 1). The majority of publications were excluded for not reporting data for AD patients with insomnia/sleep disturbances.
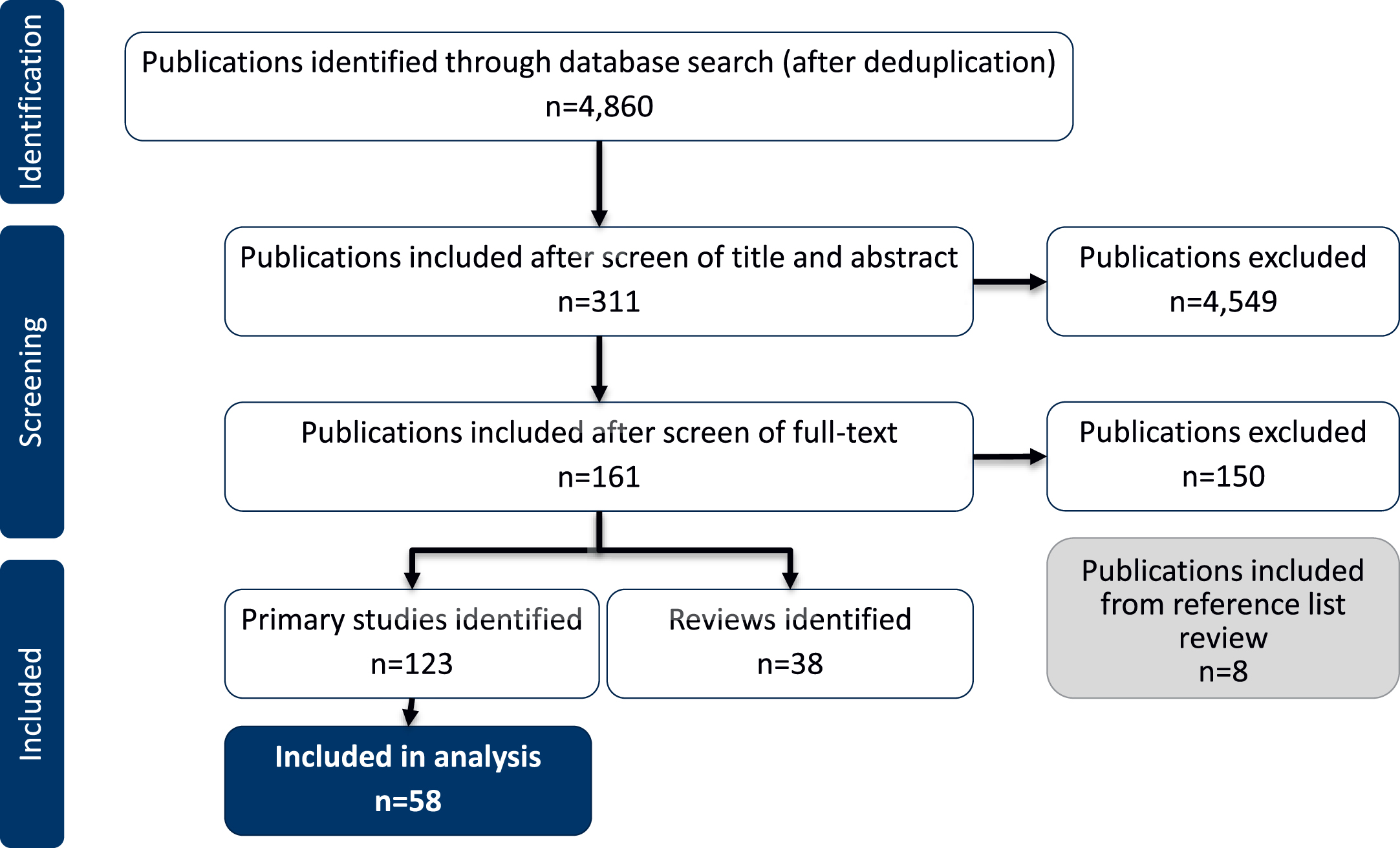
Study flow diagram.
Only studies characterizing the clinical and caregiver burden of sleep disturbances in patients reported to have AD (including probable or possible AD) were included in this review (58 studies). Clinical burden included the impact of sleep disturbances on patients’ health and wellbeing. Studies reporting prevalence and incidence data for sleep disturbances in AD patients and those reporting data for mild cognitive impairment (MCI) were not included in this review (65 studies). To avoid duplication, only primary studies are included in the results. Relevant reviews identified have been included in the Discussion.
The majority of included studiesw reported data for non-specific sleep disturbances in AD, with fewer evaluating insomnia specifically. Table 1 summarizes the included studies and sleep problem definitions.
Included studies and definitions used for cognitive impairment and sleep problems
AD, Alzheimer’s disease; AIS, Athens Insomnia Scale; BEHAVE-AD, Behavioral Pathology in Alzheimer’s Disease; CUSPAD, Columbia University Scale for Psychopathology in Alzheimer’s Disease; DLB, Dementia with Lewy bodies; DSM-5, Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; ESS, Epworth Sleepiness Scale; ICD, International Classification of Diseases; MCI, Mild Cognitive Impairment; MOUSEPAD, Manchester and Oxford Scale for Psychopathological Assessment in Dementia; NPI, Neuropsychiatric Inventory; NR, not reported; PSG, polysomnography; PSQI, Pittsburgh Sleep Quality Index; SDI, Sleep Disorders Inventory; SDSQ, sleep diary-derived sleep quality measure; UK, United Kingdom; US, United States; VaD, vascular dementia.
Clinical burden of sleep disturbances in AD and impact on QoL
Sleep disturbances were not consistently characterized across studies. Many relied on the Neuropsychiatric Inventory (NPI), which has a single item to screen for the presence of sleep disturbed behaviors, that incorporates symptoms of awakening the caregiver at night, waking early in the morning, and daytime napping, and rates the frequency, severity, and distress [4 , 72]. An important caveat is that there is insufficient clinical description of the nature of the sleep problems in most studies, and there is not a standardized tool for assessing sleep in AD patients. Despite this limitation, sleep problems in AD patients were associated with impairments across several domains (Table 2). No studies specifically assessed the clinical burden of insomnia in AD.
Studies reporting association between sleep disturbances and clinical burden in AD patients
AD, Alzheimer’s disease; ADCS-ADL, Alzheimer’s Disease Cooperative Study-Activities of Daily Living; ADL, Activities of Daily Living; AES-I, Informant-rated Apathy Evaluation Scale; B-ADL, Bayer Activities of Daily Living; Ba-ADL, The Barthel Activities of Daily Living Index; BDRS, Blessed Dementia Rating Scale; BEHAVE-AD, Behavioral Pathology in Alzheimer’s Disease; CAMCOG, Cambridge Examination for Mental Disorders of the Elderly; CDR, Clinical Dementia Rating; CSDD, Cornell Scale for Depression in Dementia; ESS, Epworth Sleepiness Scale; HADS, Hospital Anxiety and Depression Scale; IADL, Instrumental Activities of Daily Living; K-BNT, Korean version of the Boston Naming Test; MCI, mild cognitive impairment; MHCS, Mental Health Component Score; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; NPI, Neuropsychiatric Inventory; NR, not reported; PHCS, Physical Health Component Score; QoL, quality of life; QoL-AD, Quality of Life in Alzheimer’s Disease; RAVLT-IR, Rey Auditory Verbal Learning Test; RCFT, Rey-Osterrieth Complex Figure Test; RMBPC, Revised Memory and Behavior Problems Checklist; SI-ADL, Seoul-Instrumental Activities of Daily Living Scale; SNSB, Seoul Neuropsychological Screening Battery; STAI Y-1, State Trait Anxiety Inventory for Adults; TMT-B, Trail-making test; TST, total sleep time; WASO, wake after sleep onset.
Cross-sectional studies found significant negative correlations between Mini-Mental State Examination (MMSE) scores and sleep disturbances, suggesting that sleep disturbances are associated with worse cognition [29, 71]. Similar associations were reported for patients’ functional impairment [4 , 71]. Associations between sleep disturbances and decreased cognitive functioning were reported in five of the nine studies identified (Table 2) [23 , 71]. Similarly, associations between sleep disturbances and daily functioning were reported in 10 of the 14 studies identified (Table 2) [4 , 71]. Even studies that did not find an association between cognition or functional status and sleep problems (e.g., de Oliveira et al., and Moran et al.) reported sleep problems associated with behavioral disturbances, as defined by the NPI or BEHAVE-AD [25, 54]. Moran et al. found no significant cognitive or functional impairment differences between AD patients with and without sleep disturbances, but behavioral disturbances were significantly associated with sleep disturbance [54]. Two studies reported that longer sleep duration was associated with cognitive/functional impairment [23, 43]. In general, however, most studies have reported that poor sleep impacts both cognition and functional impairment.
Behavioral symptoms and neuropsychiatric functioning of AD patients are also worse among those with sleep disturbances [25 , 71], particularly depression and anxiety [25 , 71]. Several studies also reported significant associations between poor sleep and apathy in AD patients [55, 71]. Mulin et al. found that those with AD and apathy had reduced daytime activity levels and poorer nocturnal sleep quality [55]. Increased sleep and decreased circadian stability have also been associated with apathy in AD patients [25, 43]. Moran et al. found that sleep disturbances were associated with aggressiveness and greater global ratings for behavioral problems in AD patients [54]. Other studies reporting clinical impacts of sleep disturbances are summarized in Table 2.
Sleep problems are also associated with reduced QoL in AD patients [44 , 51]. Leng et al. reported that sleep disturbances, poor subjective sleep quality, and shorter sleep duration were all associated with reduced mental, physical, or total QoL scores [44]. In line with this, greater actigraphically-defined sleep efficiency has been associated with better physical QoL scores in AD patients [51].
Longitudinal studies suggest that sleep disturbances in AD may predict worsening clinical symptoms. Yin et al. found significantly lower MMSE and Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL) scores after five years in those reporting sleep disturbances, despite comparable scores in good versus poor sleepers at baseline [70]. They also reported a higher cumulative incidence of psychotic symptoms and sundowning syndrome among those with sleep disturbances after five years [70]. Escudero et al. also reported that sleep problems were predictive of cognitive and functional decline in AD patients (Montreal Cognitive Assessment [MoCA]) [28].
Caregiver burden and institutionalization of patients
Impact of insomnia/sleep disturbances on caregiver burden
Caregiver burden associated with sleep problems in AD patients was overall found to be increased (20 of 21 studies; Table 3). Only one study reporting caregiver burden outcomes included patients with a sleep disturbance defined as insomnia (30.4% with clinically diagnosed insomnia) [56]. Caregivers were typically spouses or adult children/grandchildren, and frequently aged over 50 years, with a mean reported caregiver age of 60–80 years. Ownby et al. found sleep disturbances in AD patients were associated with greater caregiver depression, but cognitive decline was not controlled for in the study [58]. To this point, Kazui et al. reported a significant increase in sleep disturbance-associated distress among caregivers of patients with more severe AD [38]. The type of sleep problem is also a factor; McCurry et al. reported that nighttime awakenings, but not excessive sleeping, were most disturbing to caregivers and associated with greater memory impairment and worse functional status in patients [47].
Studies reporting on the association between patient with AD and sleep disturbances and caregiver burden
*High observed rates of drug use including cholinesterase inhibitors, antipsychotics, and antidepressants among patients. For studies using the NPI to assess caregiver distress, only the sleep disturbance item was considered for this review. AChEI,acetylcholinesterase inhibitor; AD, Alzheimer’s disease; BIC-11, Burden Index of Caregivers-11; BPSD, behavioral and psychological symptoms of dementia; CDR, Clinical Dementia Rating; CES-D, Center for Epidemiological Studies Depression Scale; CSQ, Caregiver Sleep Questionnaire; DLB, Dementia with Lewy bodies; F, female; FTLD, frontotemporal lobar degeneration; GHQ, General Health Questionnaire; GSS, Gilleard’s Strain Scale; HAMA, Hamilton Anxiety Scale; HDRS, Hamilton Depression Rating Scale; M, male; NITE-AD, Nighttime Insomnia Treatment and Education for Alzheimer’s Disease; NPI, Neuropsychiatric Inventory; NR, not reported; n.s, not significant; PHQ-9, Patient Health Questionnaire-9; PSS, Perceived Stress Scale; PSQI, Pittsburgh Sleep Quality Index; QoL, quality of life; RC, regression model coefficient; RCT, randomized controlled trial; RMBPC, Revised Memory and Behavior Problem Checklist; SCB, Screen for Caregiver Burden; SD, standard deviation; SDI, Sleep Disorders Inventory; SF-12v2, 12-item Short Form Health Survey version; SF-36, 36-Item Short Form Health Survey; VaD, vascular dementia; ZBI,Zarit’s Burden Interview.
The literature overwhelmingly suggested that sleep disturbances and disruptive nighttime behaviors (awakenings and wanderings) in AD patients are associated with considerable distress, poor health status and QoL, and reduced sleep in caregivers [4 , 67]. Interestingly, objective caregiver burden has also been associated with mis-perceived poor sleep quality in patients by caregivers [51]. Only one identified study reported that sleep disturbances were not associated with caregiver burden; de Oliveira et al. reported no significant difference between the burden in caregivers of patients with satisfactory versus unsatisfactory sleep [25].
Sleep disturbances in AD patients are also associated with caregiver depression, anxiety, and mood [51 , 70], and have a negative impact on caregivers’ overall QoL [31, 56]. Caregivers’ sleep worsened in the presence of patients’ sleep disturbances [51, 56]; Okuda et al. found sleep disturbances in AD patients correlated significantly with higher caregiver PSQI scores (worse sleep quality) [56].
Several studies investigated the impact of specific sleep characteristics and behaviors on caregiver burden. Nighttime behaviors (awakenings and wanderings) had the largest impact on caregiver burden (four studies) [4 , 47]. Notably, those with anxiety were twice as likely to waken their caregiver versus those without, and heightened anxiety was reported among patients taking sedative or hypnotic, antianxiety, antipsychotic or antidepressant medications, suggesting that the presence of anxiety led to the prescription of these medications [49].
Institutionalization of AD patients with sleep disturbances
Insomnia (two studies) [17, 18] and sleep disturbances (one study) [70] are key factors influencing the institutionalization of AD patients. In a five-year study, more AD patients with sleep disturbances (caregiver report of≥2 nighttime awakenings) lived in special dementia care units than those without sleep disturbances [70]. Baek et al. reported that a greater proportion of AD patients with insomnia were admitted to a long-term care facility and had longer durations of admission than those without insomnia [17]. Insomnia was also independently associated with admission to a nursing home after discharge from a dementia unit [18]. Importantly, other factors influence institutionalization, such as the availability of home services [18].
Thus insomnia/sleep disturbances may both accelerate the rate of deterioration of AD patients and lead to institutionalization. Interestingly, a 2006 study by McCurry et al. reported that misperception of patient sleep by caregivers in comparison to actigraphy was associated with greater objective caregiver burden [51]. Caregivers play a key role in the reporting and management of sleep disturbances in AD patients, and their perception of patients’ sleep disturbance likely influences patient management, including the decision to admit AD patients into care facilities.
Treatment of sleep disturbances in Alzheimer’s disease
Few insomnia treatment studies have been conducted in AD patients. We identified only 23 interventional studies and the majority were small (14 studies with≤50 subjects), highlighting the need for further studies to assess treatment efficacy and patient and caregiver outcomes in this population.
Non-pharmacological treatments
Of eight studies investigating non-pharmacological treatments, some found improvements not only in patients’ sleep, but also other patient and caregiver outcomes (Table 4). Lee et al. reported improvement in subjective, but not objective, sleep (PSQI) in AD patients with insomnia treated with timed blue light, as well as improvements in behavioral disturbances and caregiver distress [42]. Ancoli-Israel et al. reported that daytime bright light exposure increased nocturnal sleep consolidation [16]. In contrast, some studies did not find light therapy effective in improving sleep overall [24, 27]. Of these studies, however, Dowling et al. found that people with the most impaired rest-activity rhythm responded to light therapy [27], and van Someren et al.’s 1999 reanalysis of Colenda et al.’s 1997 study found that light therapy improved interdaily rhythm stability [24, 73].
Impact of non-pharmacological treatments on sleep and clinical outcomes in AD patients
*Post-test outcomes controlled for baseline age, gender, depression, comorbidity limitations, MMSE and sleep apnea scores. ADL, Activities of Daily Living; ESS, Epworth Sleepiness Scale; GDS, Global Deterioration Scale; GHQ28, Global Health Quality of Life; IADL, Instrumental Activities of Daily Living; MMSE, Mini-Mental State Examination; NITE-AD, Nighttime Insomnia Treatment and Education for Alzheimer’s Disease; NPI, Neuropsychiatric inventory; PSQI, Pittsburgh Sleep Quality Index; STAI Y-1, State Trait-Anxiety Inventory; TST, total sleep time: WASO, wake after sleep onset.
Acupressure treatment was assessed in one study and improved sleep in elderly institutionalized AD patients with insomnia, but had no effect on cognition (MMSE) or functional status (ADL) [64].
McCurry et al. reported that an approach combining sleep hygiene, walking, and light exposure (NITE-AD) reduced the frequency and duration of nighttime awakenings, and lowered daytime sleepiness (Epworth Sleepiness Scale [ESS]) and levels of patient depression (RMBPC) versus controls [50]. They also reported improvements in patients’ actigraphically-measured total wake time with NITE-AD versus controls; similar results were reported for the groups receiving light and walking therapies [52]. However, caregiver-reported subjective outcome measures (Sleep Disorders Inventory [SDI]) did not replicate these results, but caregiver reports are not always consistent with actigraphy [52]. Unfortunately, improvements in sleep with any interventions were not sustained at six months, likely due to reduced adherence over time [52]. Indeed, in the latter NITE-AD study, patients adhering to recommendations≥4 days/week had significantly better sleep outcomes [52]. These findings highlight a key challenge with non-pharmacological treatment, which may not always be feasible in patients with AD and other comorbidities due to inability or lack of willingness to partake. Notably, active training and support for caregivers led to a greater success in implementing good sleep hygiene practices in AD patients [48].
Pharmacological treatments
Only 15 studies investigated the effects of pharmacological sleep treatments in AD patients (Table 5). Although several treatments showed promise in improving clinical and caregiver symptoms, inconsistent clinical results were also apparent and there are insufficient data supporting many commonly used treatments.
Pharmacological treatments: impact on sleep and clinical outcomes in AD patients
AD, Alzheimer’s disease; ADCS-ADL, Alzheimer’s Disease Cooperative Study-Activities of Daily Living; ADAS-Cog, Alzheimer’s Disease Assessment Scale – Cognition; AE, adverse event; AIS, Athens Insomnia Scale; bid: twice a day; CASI, Cognitive Abilities Screening Instrument; CDR, Clinical Dementia Rating; CGI, Clinical Global Impression; ESS, Epworth Sleepiness Scale; FAST, Functional Assessment Tool for AD; HAMA, Hamilton Anxiety Scale; HAMD, Hamilton Depressive Scale; LS, least squares; MMSE, Mini-Mental State Examination; NPI, Neuropsychiatric Inventory; NR, not reported; PSG, polysomnography; PSQI, Pittsburgh Sleep Quality Index; TST, total sleep time; WASO, wake after sleep onset.
Benzodiazepines and non-benzodiazepine ‘z-drugs’
A small study (N = 7) by McCarten et al. investigated the efficacy of the benzodiazepine triazolam (0.125 mg at bedtime) in patients with AD but failed to detect significant effects on sleep parameters versus placebo (Table 5) [46]. Yin et al. reported improvements in sleep quality (PSQI) and daytime sleepiness (ESS) with the non-benzodiazepine zolpidem tartrate, versus no treatment in a 5-year study; no change was seen in MMSE, ADCS-ADL, or NPI scores with zolpidem tartrate after five years versus no treatment, suggesting no significant impact on cognitive decline or neuropsychiatric behaviors, but no safety outcomes were reported [70].
Melatonin/melatonin receptor agonists
Two studies found melatonin to be ineffective at improving sleep, agitation scores, or circadian rhythm parameters in AD patients based on actigraphy [30, 65]. Conversely, Brusco et al. reported significant improvement in sleep quality and decreased sundowning with melatonin 9 mg, but this was an open label study with only 14 patients [20]. Similarly, Wade et al. reported significantly improved sleep efficiency among AD patients with insomnia taking prolonged-release melatonin in a double-blind, placebo-controlled study after 24 weeks, but PSQI scores were not significantly different between groups [69]. Cognition was also improved with melatonin versus placebo (for MMSE and AD Assessment Scale-Cognition scores), as well as self-care and activities of daily living scores (IADL) [69].
Antidepressants
A small study by Camargos et al. found that trazodone 50 mg increased sleep duration and efficiency in AD patients versus placebo, was well-tolerated, and did not increase daytime sleepiness [21]. Petrescu et al. also reported that patients taking trazodone (50–200 mg) had more total sleep time in both patient- and nurse-reported measures, although it was associated with more side effects than quetiapine [60]. Interestingly, trazodone may have a possible neuroprotective effect in AD patients with insomnia; a retrospective study by La et al. reported that among a group of older adults, including some with AD, those not using trazodone declined 2.4-fold faster than trazodone users (median dose 50 mg) across an average of 3.75 years based on MMSE results [40]. Notably, while trazodone is an antidepressant, a dose of 50 mg is consistent with use for insomnia, rather than depressive disorders. A small study assessing the antidepressant mirtazapine reported no effects on sleep outcomes in AD patients (Table 5) [62].
Antipsychotics
A five-year study by Yin et al. reported improved daytime (ESS) and nighttime (PSQI) sleep symptoms with low-dose risperidone versus zolpidem tartrate, melatonin, or no drug treatment in AD patients with sleep disturbances [70]. Risperidone also alleviated neuropsychiatric symptoms (NPI) versus no treatment and did not significantly affect cognition or functional autonomy versus zolpidem, melatonin, or no treatment, suggesting no acceleration in mental deterioration [70]. Risperidone was also associated with a significant improvement in caregiver mood and sleep and a lower expectation that a patient would be institutionalized versus the melatonin and no treatment groups, implying improved outlook; institutionalization of risperidone-treated patients was significantly lower than in patients not receiving sleep medication [70]. However, this was not a randomized trial, and treatment group assignment was based on the preference of the spousal caregiver. In a nursing home-based study, Meguro et al. reported that low-dose risperidone was effective at increasing patient sleep and decreasing daytime wandering [53]. Importantly, patients did not exhibit daytime oversleeping or side effects which would have prevented them participating in daily activities [53].
Orexin receptor antagonists
Orexin receptor antagonists have emerged as a new generation of insomnia treatments. In a polysomnography study of AD patients with insomnia, Herring et al. reported that suvorexant significantly increased total sleep time versus placebo, and decreased waking after sleep onset, with no worsening of cognitive (MMSE) or psychomotor tests (digit symbol) [35]. Currently, suvorexant is the only pharmacotherapy specifically approved for the treatment of insomnia in AD patients, although other drugs in this class are also being tested in the AD population.
DISCUSSION
This literature review demonstrates that sleep disturbances in AD patients are associated with greater cognitive decline, poorer functional ability, and reduced QoL. Furthermore, behavioral symptoms and neuropsychiatric functioning of AD patients, including depression, apathy, psychotic symptoms, and sundowning syndrome, are more frequent and severe among those with sleep disturbances. Sleep disturbances and associated disruptive nocturnal behaviors in AD patients also result in considerable caregiver burden, including increased distress, depression, and sleep problems, and decreased QoL. Sleep disturbances are a major factor influencing the institutionalization of AD patients, likely through their impact on caregiver burden. Previous reviews have also identified nighttime awakenings and troublesome behaviors (e.g., wandering) as the most burdensome symptoms and key motivating factors for institutionalization [1, 74].
In addition, sleep problems have been associated with increased risk for development and progression of AD [9 , 70]. Recent meta-analyses found that patients with insomnia had a 1.51-fold higher risk (95% CI: 1.06–2.14) of developing AD [11], and that patients with sleep problems had a 1.55 (95% CI: 1.25–1.93) times higher risk of AD and a 1.65 (95% CI: 1.45–1.86) and 3.78 (95% CI: 2.27–6.30) times higher risk of cognitive impairment and preclinical AD, than people with no sleep problems, respectively [9].
However, evidence of the independent association of sleep problems and clinical outcomes is generally lacking in the literature. Caregiver burden is likely to be multifactorial and impacted by variables beyond patients’ sleep symptoms, including the severity of patients’ AD. Multiple patient and caregiver factors influence the institutionalization of AD patients, not only the overall clinical status of AD patients, but also the health, functionality, and economic status of their caregivers.
Nevertheless, insomnia and other sleep disturbances are recognized factors contributing to increased burden in AD patients, and the effective management and treatment of sleep symptoms may be helpful in improving clinical outcomes and delaying institutionalization. However, sleep problems are not only poorly characterized in this population, but there are also very few studies evaluating the effects of sleep treatments specifically in AD patients. Non-pharmacological sleep treatments, such as behavioral and light therapy, are typically used as first-line approaches in patients with insomnia and sleep disturbances, despite a lack of consistent data and standardized recommendations in this population [1, 75]. However, access to non-pharmacological sleep treatments is limited due to availability and cost restraints [5]. The limited research for non-pharmacological sleep treatments reflects the obstacles faced with initiating practices in clinical care, particularly in low-resource environments, such as the cost, time taken to train carers, limited equipment, and lack of clear guidelines to deliver treatment [76]. For patients with dementia and in long term care, patients may also be unable or unwilling to partake in such therapies, preventing successful implementation [5]. Implementing approaches that actively include input from care staff, residents and their families may help overcome the challenges associated with non-pharmacological treatments in long-term care [76]. Practices that aim to improve the daily routines and quality of the sleeping environment for AD patients in care homes (such as the LOCK program, a specific frontline staff huddling program which enables staff to:
Many currently available pharmacological treatments for insomnia are associated with a risk of further cognitive impairment, falls and fractures, and next-day residual effects in the elderly and therefore should be carefully evaluated prior to being prescribed. Studies investigating insomnia therapies in elderly patients have largely excluded patients with cognitive impairment or dementia. As a result, there remains uncertainty about the balance of benefits and risks associated with common sleep treatments when used in AD [78].
Few studies evaluating the use of sleep medications in AD patients were identified and were generally small. While treatments such as trazodone [21 , 60], risperidone [53, 70], and suvorexant [35] showed some promise in improving clinical and caregiver symptoms, further data from larger, prospective trials are required. Previous reviews have drawn similar conclusions, highlighting the scarcity and contradictory nature of current evidence [1, 79].
Benzodiazepines are also frequently used in AD patients [79, 80], but a systematic literature review by Defrancesco et al. (2015) reported no conclusive evidence for the improvements in sleep quality in AD patients taking benzodiazepines [81]. Moreover, benzodiazepines are associated with side effects including daytime sleepiness, worsened insomnia, confusion, and amnesia. Though non-benzodiazepine ‘z-drugs’ may be associated with fewer side effects than benzodiazepines [1 , 82], adverse events are still apparent. The American Geriatric Society recommends that both benzodiazepines and non-benzodiazepines be avoided in the elderly [14].
This review found mixed results between studies evaluating melatonin, which echoes findings from previous reviews [80 , 84]. A 2016 Cochrane review concluded that melatonin was unlikely to benefit AD patients with sleep problems [85]. Conversely, the combination of melatonin and bright-light therapy has been shown to have beneficial results in the nocturnal sleep of AD patients [1, 10], suggesting a multifaceted approach targeting circadian rhythm stabilization may enhance the outcomes of sleep treatments in AD, which is not surprising given that AD patients tend to have both sleep and circadian rhythm disturbances.
The use of antipsychotics in the treatment of sleep and other behavioral disturbances in AD has been extensively discussed [1 , 87]. While they may be useful in the treatment of sleep disturbances and to alleviate neuropsychiatric symptoms [70], they are not without risks. While no significant adverse events were reported in the studies included in this review, antipsychotics can cause residual sedation, confusion, and falls, and have metabolic and cardiovascular side effects [1 , 89]. Antipsychotics also carry a black-box warning from the FDA for increased death in the elderly with dementia.
Suvorexant, currently the only approved pharmacotherapy for insomnia in AD, has demonstrated improved sleep and was well-tolerated in AD patients [35]. Suvorexant has also been shown to improve sleep maintenance and onset and was well-tolerated in elderly patients with insomnia [90]. Lemborexant, another orexin receptor antagonist, has also demonstrated promising results in improving sleep without significant adverse events in elderly patients [91]. Although not the focal population for this review, lemborexant has shown beneficial results in nighttime and daytime symptoms with no worsening in cognitive functioning in AD patients with Irregular Sleep-Wake Rhythm Disorder [92]. However, at present, the suvorexant trials in elderly patients and those with AD have reported far more conclusive results, based on objective PSG measures [35, 90]. While orexin receptor antagonists have been highlighted as promising options for elderly patients and those with dementia across the literature, data so far are limited [1 , 93].
The paucity of studies evaluating therapies in this population highlights the importance of further research in people with or at risk for AD, as there is insufficient evidence that treatments suitable for non-demented elderly persons will have an advantageous benefit-to-risk ratio in AD patients [14, 93]. Importantly many current studies do not adequately categorize sleep problems in AD patients which may include insomnia and circadian rhythm disorders. Successful treatment likely requires addressing both disorders which may have impacted the results in studies assessing the treatment of insomnia alone. As studies reporting data for sleep disorders other than insomnia, such as obstructive sleep apnea, were excluded from this analysis, this review has not discussed the burden of sleep disorders existing comorbidly with insomnia in AD patients. However, as the presence of comorbid sleep disorders may influence the clinical presentation and burden of symptoms, the impact of available sleep treatments in these patients should be further considered.
Limitations
There are several limitations to this review, with the most significant being that publications reported on sleep disturbances and insomnia without clear and consistent definitions. Sleep in AD is characterized by increased insomnia and sleep fragmentation, as well as circadian rhythm abnormalities, including deterioration and instability of rhythms, with delayed sleep phase and daytime napping. AD patients also have an increased rate of other sleep disorders that further contribute to sleep disturbance, including sleep apnea, sleep movement disorders (e.g., restless legs), and parasomnias such as REM sleep behavior disorder [94 –97]. Furthermore, medications commonly given to AD patients including many antidepressants, cholinesterase inhibitors, and memantine can cause or worsen insomnia. In general, the studies covered in this review did not comprehensively assess and enumerate the specific sleep and rhythm disturbances in participants, evaluate them for other sleep disorders, or control for medication effects. As a result, it is likely that sleep problems and sleep disorders other than insomnia were captured, particularly in studies that used non-specific definitions for sleep disturbance. For example, the NPI question used to assess sleep disturbance in many of the studies simply asks, “Does the patient awaken you during the night, rise too early in the morning, or take excessive naps during the day?”; it combines symptoms of insomnia with excessive sleepiness, and the questionnaire does not include screening for other sleep disorders.
Studies in this review generally did not control for other factors that could have influenced the clinical burden of disturbed sleep, most notably the degree of cognitive impairment and presence of comorbidities such as depression, agitation, and other behavioral disturbances commonly reported in AD patients. Thus, any associations, or lack thereof, may be obscured by the worse sleep and greater impairment that patients with severe AD inherently have.
Finally, only publications reporting specifically on AD patients were included and thus this review may have omitted data from studies on the burden of insomnia in AD patients which reported a population more broadly defined as dementia. Finally, the review and synthesis of the identified publications included only a qualitative review of the data and no further analyses were undertaken.
Conclusions
Insomnia/sleep disturbances confer a significant burden on both AD patients and caregivers and are an important risk factor for the institutionalization of patients. However, sleep and rhythm disturbances remain poorly characterized by clinicians and under-researched in this population, and no data were identified on the economic burden they may confer. Additional research is needed to fully quantify these clinical and economic burdens, especially given the large population at risk for AD. Furthermore, there is a paucity of data on effective treatments for insomnia in AD patients and the impact of treatment on clinical burden in patients and their caregivers.
The aging global population will increase the number of individuals at risk of developing AD. Sleep disturbances, particularly insomnia, appear to exacerbate this risk, and most currently available pharmacotherapies for insomnia have not been studied sufficiently in AD patients; treatment guidelines advise against the use of many pharmacotherapies in older adults. Due to the impact of insomnia/sleep disturbances on AD risk and progression, and the associated patient and caregiver burden, there is a need to further evaluate the burden of disease in AD patients, investigate therapies targeted to this population, and determine whether treating insomnia and other sleep disorders may decrease the risk of developing AD and/or slow its progression.
Footnotes
ACKNOWLEDGMENTS
Medical writing assistance, under the direction of the authors, was provided by Ashley Enstone, Patrick Lavelle, and Amy Sears of Adelphi Values PROVE™, in accordance with Good Publication Practice (GPP3) guidelines. This assistance was funded by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA (MSD). Christopher Lines, PhD, and Alan Lipschitz, MD of MSD assisted with editing of the paper.
