Abstract
Background:
Hearing loss is highly prevalent in older adults, particularly among those living with dementia and residing in long-term care homes (LTCHs). Sensory declines can have deleterious effects on functioning and contribute to frailty, but the hearing needs of residents are often unrecognized or unaddressed.
Objective:
To identify valid and reliable screening measures that are effective for the identification of hearing loss and are suitable for use by nursing staff providing care to residents with dementia in LTCHs.
Methods:
Electronic databases (Embase, Medline, PsycINFO, CENTRAL, and CINAHL) were searched using comprehensive search strategies, and a stepwise approach based on Arksey & O’Malley’s scoping review and appraisal process was followed.
Results:
There were 193 scientific papers included in the review. Pure-tone audiometry was the most frequently reported measure to test hearing in older adults living with dementia. However, measures including self- or other-reports and questionnaires, review of medical records, otoscopy, and the whisper test were found to be most suitable for use by nurses working with older adults living with dementia in LTCHs.
Conclusion:
Although frequently used, the suitability of pure-tone audiometry for use by nursing staff in LTCHs is limited, as standardized audiometry presents challenges for many residents, and specific training is needed to successfully adapt test administration procedures and interpret results. The whisper test was considered to be more suitable for use by staff in LTCH; however, it yields a limited characterization of hearing loss. There remains an urgent need to develop new approaches to screen hearing in LTCHs.
Keywords
INTRODUCTION
Hearing impairment, cognitive decline, and multi-morbidity
Hearing impairment is one of the most prevalent chronic conditions affecting older adults [1–4], and is a leading contributor to disability globally [5]. The prevalence of hearing impairment increases markedly with age. Using criteria based on pure-tone audiometric detection thresholds, approximately half of older adults have clinically significant hearing loss by 75 years of age, with more than 80% affected by 80 years of age [6–8]. Age-related changes in audiometric thresholds can result from damage to the sensory cells and/or from metabolic changes in the inner ear [9]. Neural changes can also occur throughout the auditory system, from synaptopathy in the inner ear [10] to reduced brain volumes at higher cortical levels [11].
Significant associations between age-related declines in hearing and cognition have been found across several cognitive domains [12, 13]. For those with typical age-related declines in auditory and cognitive processing, listening may be relatively easy in favorable communication situations, such as when attention is focused on conversation with a familiar person in a quiet place with no distractions. However, it is often difficult to understand speech in less favorable everyday communication situations, when background noise, group interaction, and multi-tasking demands often co-occur (for a review, see Pichora-Fuller et al. [14]). Amplifying sounds can help, but this does not fully overcome the difficulties that older adults experience in complex listening situations. Notably, even when words have been correctly heard, they may not be remembered as well when listening occurs in noisy compared to quiet conditions, due to increased cognitive demands during listening [15].
The connections between auditory and cognitive functioning are disrupted further as hearing thresholds worsen. The severity of hearing loss has been directly associated with cognitive decline in older adults [16]; this association can begin even when audiometric thresholds are still be within the clinically normal range [17]. The risk of dementia increases as the degree of threshold hearing loss worsens [18]. Furthermore, the long-term effects of auditory decline are implicated in changes at a neural level [19], with evidence suggesting that central auditory dysfunction, as measured on dichotic speech tests [20, 21], may be a prodromal symptom of Alzheimer’s disease [22]. Indeed, at the population level, the estimated risk for dementia is reportedly greater for hearing loss than for all other potentially modifiable risk factors at mid-life [23].
With population aging and life expectancy increasing, the prevalence of hearing loss, dementia, and many other chronic health conditions is rising. Multi-morbidity, defined as two or more chronic conditions in one individual [24, 25], affects the majority of adults aged 65 and older [24, 26], with its prevalence predicted to double over the next 20 years [27]. Hearing loss has the potential to contribute to and interact with the multiple comorbidities that increase the risk of dementia [16] and can curtail healthy life expectancy when combined with neurological disorders [28]. Importantly, factors that contribute to frailty, including dementia [29], falls [30], and poor psychosocial health [31], as well as poor physical health and impaired activities of daily living [32], have all been associated with hearing loss. These factors are further compounded when declines in hearing, especially in combination with cognitive decline, make communication and social interaction challenging, which can lead to isolation and depression in older adults [33–35]. Individuals living with multi-morbidity are at greater risk of becoming care dependent, and adults with dementia are among those at highest risk of long-term care dependency [26, 36].
Crucially, managing hearing loss could be a valuable part of care for older adults, especially those living with dementia, multi-morbidities, or frailty [37]. The 2019 World Health Organization guidelines for Integrated Care of Older People (ICOPE) [38] has identified hearing as one of the key capacity domains for functioning. In particular, sensory and cognitive impairments are very common, and managing hearing loss could be beneficial for those living with dementia, including residents of long-term care homes (LTCHs).
LTCHs, also referred to as nursing homes, provide a combination of housing, medical, and personal care services for people who need ongoing daily assistance over extended periods of time. Research conducted across five LTCH settings in the United States found that over 80% of residents aged 60 years and older had at least mild hearing loss, with approximately 50% having moderate-to-severe hearing impairment that would render speech largely inaudible [39, 40]. Across studies conducted in Australia and Canada, more than half of LTCH residents have been found to be living with dementia, with behavioral symptoms of dementia found in more than 70% of residents [41–43]). Not surprisingly, with the average age of LTCH residents in Ontario, Canada, found to be 87 years, some degree of cognitive impairment has been identified in 94% of residents, with almost one third living with both cognitive impairment and dual sensory (hearing and vision) impairments that have compounding functional consequences [42]. Still, very low rates (<10%) of hearing aid use have been reported [42]. Moreover, LTCH environments are often challenging for residents experiencing cognitive and sensory loss, even more so during the recent COVID-19 pandemic, due to mandated measures for infection prevention and control, such as distancing, mask wearing, and reliance on technology over in-person social and communicative interactions.
Hearing screening
Hearing screening, to identify the presence of hearing loss or monitor for changes in hearing, is a necessary step in managing hearing problems so that functioning in everyday life can be optimized. The World Report on Hearing proposes universal hearing screening for older adults [44]. In some countries, existing practice standards advise that adults be screened for hearing loss as needed or requested, and with increasing frequency after the age of 50 years, particularly in those who are vulnerable to risk factors associated with hearing loss [45]. In addition, hearing screening has been recommended for those who are being assessed for dementia [46]. Depending on the results of hearing screening, various recommendations can be made to improve communication functioning, with identification of a previously undiagnosed hearing loss or changes in hearing typically triggering referral for a full audiologic assessment and rehabilitation [45].
The methods used to test hearing will depend on the specific purpose of testing, the setting, and the qualifications of the tester. Assessing the degree and type of hearing loss, and planning rehabilitation, may involve otoscopy to inspect the ear canal and ear drum for problems such as impacted cerumen (wax) or infection, physiological tests to examine different levels of the auditory system from the ear drum to the cortex, behavioral tests to evaluate different auditory abilities, and questionnaires to measure self-reported or other-reported communication functioning and socio-emotional experiences of and reactions to hearing problems. The most common behavioral test is pure-tone audiometry, which is used clinically to measure hearing thresholds (an individual’s ability to detect simple pure tones). It is also common to use behavioral tests to measure supra-threshold auditory abilities such as an individual’s ability to understand words presented in quiet or in noise, using either one ear at a time (monaural tests), or using both ears, with either the same (diotic tests) or different (dichotic tests) information presented to the ears. As healthcare professionals qualified to measure hearing using a wide range of basic and specialized tests, audiologists have the training and expertise to modify the administration of tests if adaptations are needed for individuals who are unable to complete standard testing procedures. Given their advanced training and specialized clinical settings, audiologists can usually obtain reliable test-retest hearing assessment results for those living with mild levels of dementia [47].
People living with dementia are among those for whom non-standard testing methods may be needed [48–50]. Although audiometry can be successfully administered in audiology clinics to test hearing in people living with dementia, its suitability for use by non-audiologists to screen people living with dementia is limited by the need for specialized expertise to adapt test procedures for many of these individuals. Even though evidence-based practice guidelines devised for front-line nursing staff in care facilities recommend the use of otoscopy and pure-tone audiometry [51, 52], it is important to complement these with staff- and self-reporting tools to evaluate a resident’s level of activity and participation in the LTCH [39]. Yet, the shortage of regulated nurses in most LTCHs and a lack of training in the use of these tools likely precludes the implementation of these recommendations [53], while referral for ear and hearing care following screening may not be prioritized or regularly implemented.
In light of mounting evidence implicating hearing loss in neurocognitive decline, there is an imperative to develop and evaluate approaches to screen hearing that are suitable for administration by non-audiologists working with older adults who are living with dementia [54, 55]. Simple, low-tech measures, such as the whisper [56] and finger rub tests [57], are commonly used by primary care physicians and neurologists who recognize the need to screen for hearing impairment in older adults who are at risk [58–61]. Although the complexity and progressive nature of dementia-related symptoms necessitates the involvement and collaboration of many health professionals [62, 63], audiologists are seldom included as members of inter-professional teams and the presence or severity of hearing loss is an aspect of the individual’s impairment profile that is often under-estimated by the other health specialists within the circle of care [50, 65].
In addition to the consequences of hearing loss for everyday functioning, hearing loss is also important to consider because it can affect other assessments of individuals who may have comorbid conditions. Importantly, hearing loss can negatively affect performance on the most frequently used screening tests for cognition, such that tests administered verbally can result in an over-estimation of the presence or level of cognitive impairment when sensory impairments are not considered [66, 67]. The presence and severity of hearing impairment has not only been found to be under-estimated by the specialists tasked with the assessment and diagnosis of cognitive disorders [64, 65], but also by the health professionals responsible for the care of older adults living with dementia, many of whom may have multiple comorbidities [50].
It has been recommended that hearing be assessed when residents are first admitted to an institutional residence, as well as on an ongoing basis to monitor for change [48]. In institutional care settings around the world, the standardized Resident Assessment Instrument (RAI) Minimum Data Set (MDS) is the most commonly used assessment tool (http://www.interrai.org). The RAI-MDS is designed to collect data across many domains regarding residents’ strengths, needs, and potential risks for the purposes of guiding care planning and monitoring [68]. According to the RAI-MDS protocol, a resident should be assessed upon admission and re-evaluated on a quarterly and at least annual basis, as well as upon detection of a significant change in a resident’s health or functional status [69]. The information gathered for the assessment may be based on records of audiologic assessments or hearing aid use noted during chart review, observation of the resident’s communication abilities, or ratings of hearing abilities by the resident or key informants such as family members or care providers. RAI-MDS ratings of hearing ability correlate significantly with the results of pure-tone audiometry [70], and other common screening measures, such as the Hearing Handicap Inventory for the Elderly (HHIE-S) [71]. Still, hearing impairment in residents is often not identified using the MDS [70, 72].
In general, estimates of the prevalence of hearing loss in older adults are higher when behavioral tests are used compared to when self-report measures are used to identify hearing loss [73–75]. When records of audiologic assessments are not available during chart review, the MDS relies heavily on self-report or other-report of a resident’s hearing loss and hearing aid use, with under-reporting of hearing problems leading to lack of referral of residents with hearing loss for audiologic assessment and rehabilitation [76]. Consequently, there are reductions in the quality of life and provision of care for those whose hearing loss has not been addressed [77–80].
The assessment and management of hearing needs in persons living with moderate to severe dementia can add even more challenges. Health professionals working in LTCHs may observe the communication difficulties or withdrawal of a resident, but attribute the source of these difficulties to cognitive impairment without recognizing the contribution of hearing impairment [41, 72]. Nursing and other care professionals have reported challenges in distinguishing between the symptoms of dementia and hearing impairment insofar as both affect communication [41, 81], and they have expressed interest in accessing further education, as well as learning better ways to screen hearing and how to use the results to better tailor the provision of care [81].
Compared to residents with cognitive impairment alone, those who have combined cognitive and hearing impairments are more likely to have communication problems, as well as reduced social interactions and participation (e.g., in group activities). These problems are even greater in residents who have impairments in hearing, vision, and cognition [42]. Increased awareness of sensory problems, their co-occurrence with cognitive decline and increased functional burden, and how these can be alleviated, could provide key information to the planning and provision of care and accommodations within LTCH environments. More effective hearing screening could also result in more effective management of hearing problems of residents living in LTCHs, to improve communication functioning, social interaction, and overall well-being.
The present study
The purpose of the present study was to conduct a scoping review of the literature to determine which hearing screening methods have been used to assess people who are living with dementia. A second step was to then identify which of these screening methods are reliable and valid tests that would be suitable for use by nurses working in LTCHs. As part of a larger mixed-methods investigation [82], our aim was to find suitable methods for screening hearing to identify residents in need of referral to a hearing health care professional. Screening and referral were assumed to be prerequisites for an evidence-informed approach to care for residents in LTCHs that would result in appropriate interventions for hearing loss and improved quality of nursing care, which would facilitate more effective communicative interactions, improved functioning, and greater engagement in daily activities for residents. Better management of hearing problems could also potentially help mitigate declines.
MATERIALS AND METHODS
Following a study protocol and scoping review methodology previously described [82, 83], a stepwise approach based on Arksey & O’Malley’s scoping review process was undertaken [84]. A scoping review methodology was used to allow for an extensive investigation of the existing literature to identify the range and degree of research activity that could answer a broad research question, while also identifying any gaps in the current evidence base [84]. An assessment of the methodological rigor and quality of the included studies, formally associated with a systematic review, was not conducted; however, the literature search and reporting of results was systematically carried out [85].
Identifying the research question
The question that the research sought to answer was which hearing screening measures and approaches have been used effectively to identify hearing loss in older adults with dementia living in LTCHs [82]. We aimed to provide an overview of the measures used to screen the hearing of persons living with dementia as reported in the literature, and to summarize the reported reliability, validity, and sensitivity of these measures. Screening measures for hearing loss were defined as tools, tests, or instruments used in the preliminary evaluation of hearing ability, without necessarily measuring the degree of hearing loss or diagnosing the nature of the impairment.
Search terms were formulated, trialed, and revised by an Information Specialist at the Toronto Rehabilitation Institute-University Health Network, before then being incorporated into search strategies appropriate to each database (Supplementary Material). Keywords to search for the concepts of hearing screening, long-term care settings, and dementia in the literature included, but were not limited to: hearing, auditory, audiologic or dual sensory testing, screening, assessment, or evaluation; persons with hearing disorders/impairment, or deaf-blind disorders; long-term care, nursing home, or homes for the aged; dementia, Alzheimer’s/neurodegenerative disease, or cognitive disorders; aged, elderly, geriatric, older, or senescent.
Identifying relevant studies
Four databases (Embase, Medline, PsycINFO, CINAHL) and the Cochrane Central Register of Controlled Trials (CENTRAL) were searched using formulated strategies (Supplementary Material), with searches limited to research published between January 1995 and January 2020; further updates were not undertaken after the onset of the COVID-19 pandemic. No limitations were placed on the type of research or the language in which it was reported, so that peer reviewed as well as non-peer reviewed publications were captured, including conference proceedings and academic dissertations. Research reported in a language other than English was interpreted by multi-lingual members of the study team or by use of translation software (Google Translate). Specifically, six reports in French, German, and Spanish were reviewed by members of the team with proficiency in these languages; whereas reports in Dutch, Portuguese, Italian, Mandarin, or other were translated by use of online tools with only the relevant study information judged for eligibility, and then extracted in five that were included.
Study selection
Reference management software (EndNote, Clarivate Analytics) was used to collate the resulting citations and remove duplicate results. Citations were exported into a spreadsheet format. The title and abstract of each paper were screened by two independent reviewers, who coded citations for inclusion or exclusion in accordance with agreed criteria (Table 1). Any discrepancies were discussed between the reviewers, and if consensus could not be reached on the study’s inclusion or exclusion, disagreements were arbitrated by a third reviewer. Studies reporting on the screening for hearing loss in participants or clinical groups aged 65 years or older with a diagnosis of dementia or cognitive impairment were included in the review.
Procedure and exclusion codes for review: The raters were instructed to exclude a study using the first exclusion code that applied from the following criteria and to flag if they were uncertain about excluding a study. All studies not excluded were included
Charting the data
After a full review of articles, data from the final set of accepted studies were extracted by two reviewers to populate a formulated spreadsheet, including the following: author(s), year of publication, country, type of testing environment (e.g., clinic, research laboratory, LTC facility, or other), study design, sampling method, participant demographics (age, gender, dementia type, and disease severity), name of hearing measure, auditory ability assessed, reported psychometric properties (validity, reliability, sensitivity, and specificity), as well as any details pertaining to the sensibility of tests. Sensibility was specified as the feasibility and acceptability by users and administrators of the test in the environmental and patient context of test administration [86, 87]. Details relevant to the measure’s sensibility included any adaptations to test administration for the clinical population, the number of participants who could not complete the test, and any specific training or qualifications required for test implementation or interpretation [82].
Summarizing and reporting the results
Incorporating an optional component of Arksey and O’Malley’s scoping review methodology, accredited practitioners with expertise in hearing and dementia care were also consulted during the review process [84]. As part of this second step of appraisal, two audiologists, an otologist, and a neuropsychologist, who all had expertise in hearing functioning in older adults experiencing cognitive impairments, reviewed and rated the suitability of the extracted tests for their use by nurses in LTCHs to screen hearing in residents with dementia. Two experts reviewed the selected studies to identify the hearing tests that were used and to categorize them in accordance with the domain of ear and hearing status evaluated. Finally, all four experts appraised the tests for their potential suitability. In judging suitability, the reviewers considered pre-determined factors that included the availability and cost of required test equipment, the level of specialized training needed for test administration and interpretation, whether the test procedures fell within the scope of practice of nurses, and the test’s suitability for testing residents in LTCH settings. Any discrepancies in the judgements of suitability were explored through face-to-face discussion and then reconciled among the four experts.
RESULTS
Data extraction
Five databases were searched on the 13 January 2020, yielding 2,333 identified publications. After duplicates were removed, 301 papers selected for full-text review were assessed for eligibility in the final scoping report. A further 108 studies were excluded, resulting in 193 papers describing research in older adults with dementia and/or cognitive impairment, which contained tests that potentially could be used by nurses to screen hearing in this population (see PRISMA flow chart in Fig. 1).
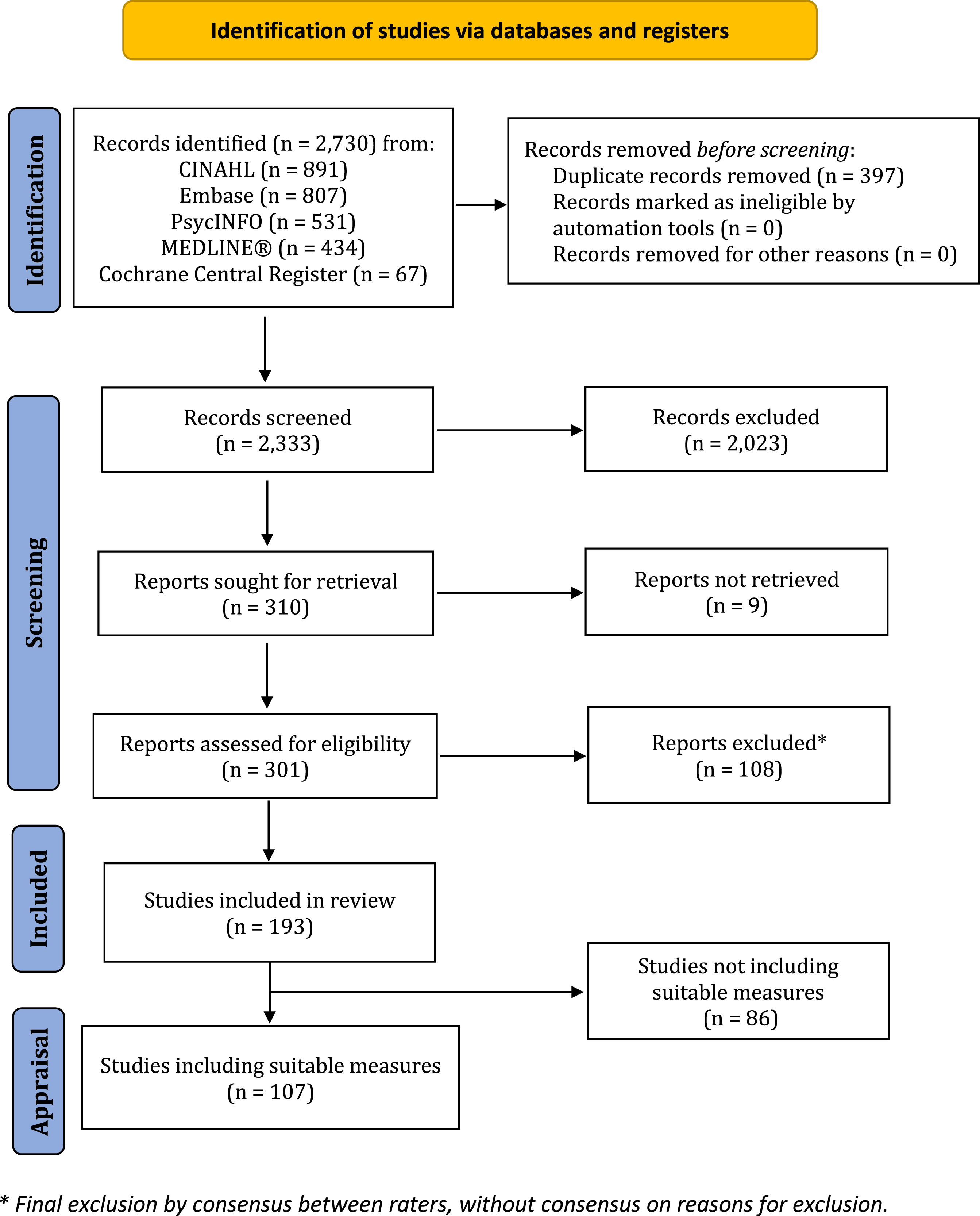
PRISMA 2020 Flow Diagram [291].
Cognitive status of participants
A mixture of dementia types, unspecified dementia or cognitive impairment indicative of dementia was most frequently reported, across almost half the reviewed studies (n = 86), but the most common diagnosis among participant groups was dementia of the Alzheimer’s type (n = 59). In cases where disease severity was reported, studies mainly included participant groups with between mild to severe (n = 51) or mild to moderate cognitive impairment (n = 41). Approximately 10% of the studies reported testing the hearing of individuals with mild dementia alone (n = 20), and fewer reported on the testing of only participants who were moderately (n = 4) or severely cognitively impaired participants (n = 2); whereas most research reports did not describe the severity of dementia of their participants (n = 71).
Study test environments
Of those studies reporting on the testing environment, a clinical setting was the most frequently reported (71 studies). For 42 studies, hearing was evaluated within the LTCH setting, and an additional 15 studies were conducted across two or three settings, which included LTCHs, private residences, and community centers. A further 15 studies involved measures administered in participants’ private residences alone, while 18 studies were conducted exclusively in a research environment, and 30 studies did not specify the environment in which assessment data was collected.
Data analysis
The tests extracted from the review were categorized by the expert reviewers to indicate which aspect of ear and hearing status was being tested (e.g., pure-tone audiometry versus speech understanding), as well as differences among measurement approaches (e.g., self-report, or behavioral or physiological test), or whether the measures involved the examination of anatomical structures (e.g., presence of cerumen) or functional aspects of hearing (e.g., other-report measure). Although the majority of studies (n = 121) reported relying on only one method of screening, more than a third of the studies (n = 72) described the use of more than one method to evaluate the hearing of participants. Overall, 87 different screening measures were identified and grouped by category, while two further measurement approaches were not described sufficiently for categorization.
Expert appraisal
More than one third (n = 33) of the identified tests or measurement approaches were judged to be suitable for use by nurses to screen for hearing loss in older adults living with dementia (see Table 2). Of the 56 tests deemed not to be suitable, many were tests that are commonly part of a clinical hearing assessment battery, but these tests were deemed unsuitable for use by nurses to screen residents with dementia in LTCH, most often due to the requirements of specialized or costly equipment and/or training. Excluded for these reasons were specialized behavioral tests of higher-level auditory processing (n = 20), physiological tests such as auditory evoked potentials (n = 5), middle ear testing (n = 4), and otoacoustic emissions (n = 2). Notably, although they are common audiologic measures, pure-tone audiometry (n = 3) and speech testing (n = 14) were deemed unsuitable for use by nurses in LTCHs due to the need for specialized equipment and training for their administration and interpretation. Assessment by use of a tuning fork (n = 1), and questionnaires focused solely on hearing aid use or without the inclusion of questions relevant to hearing ability (n = 5) were also deemed unsuitable for determining if a person had a hearing loss. Two more measurement approaches lacked sufficient detail for evaluation. The identified measures were deemed to be unsuitable for the aforementioned reasons, but none were eliminated because they were beyond the scope of practice of nurses.
Hearing tests deemed suitable for screening older adults living with dementia in LTCH, with citations listed in columns corresponding to the reported setting of test administration. Citations are highlighted with an asterisk if instances of sensibility were reported (i.e., cases in which test administrated was amended, participants excluded, or training for test administrators referenced)
From the total of 193 studies in the review, 107 studies used at least one test considered to be suitable [42, 88–190], whereas 86 research articles did not describe implementing any of the measures deemed suitable [8, 191–274]. All suitable tests conducted in studies in which their testing environment was described (n = 71 studies) are shown in Table 2; an additional seven studies cited the use of suitable tests but without specification of the testing environment [89, 186–190].
Of all tests identified, whether they were deemed to be suitable or unsuitable for use by nurses in LTCH to test residents living with dementia, the frequency with which specific psychometric properties were described across all included studies were: reliability (n = 23 studies), validity (n = 10), and sensitivity and/or specificity (n = 9), with multiple test properties reported for some tests. The investigation of the psychometrics properties of tests were most often reported for pure-tone audiometry (n = 11), speech testing (n = 4), auditory attention tests (n = 4), self-report measures (n = 2), or other-report measures (n = 2). Test-retest reliability was the most often reported test property. However, studies also reported on the unreliability of testing, including that of pure-tone audiometry and self-report measures [123]. Some studies also reported that where reliable results were obtained, repeated instructions were required to overcome challenges in communication in order to obtain valid responses [274]. Across studies, sometimes one study reported on the validity of a measure, such as the RAI MDS questionnaire [171], but findings were not always consistent with those of other studies [72].
Notably, the administration of hearing measures described in the papers also included considerations for the sensibility of the testing approach. Across all studies, there were 161 reports of the test administration requiring either adaptations to standard administration procedures, exclusion of participants with moderate or severe dementia due to inability to complete the test reliably, or requirements for specific training of the test administrator. Again, the reported sensibility of test administration most frequently involved pure-tone audiometry (35 studies), self-report or other-report measures (n = 41), auditory evoked potentials (n = 9), speech testing (n = 8), central auditory processing (n = 8), middle ear testing (n = 7), as well as the whisper test (n = 7). In the case of the whisper test, sensibility was considered most often in terms of the test administrator’s qualifications, whereby it was noted that the test was conducted by nursing or geriatric medical professionals. Based on the criteria for suitability, the screening tests shown in Table 2 were suggested as a starting point for staff working in nursing homes.
Measures that were judged to be suitable for use by nurses with older adults living with dementia were mostly cited in studies conducted in LTCH, nursing home, private residential or clinical care settings, and were also described as allowing for some adaptation when used with participants living with dementia (studies highlighted with an asterisk in Table 2). For suitable measures of self- and other-reports, as well as evaluation of a person’s ear health and audiologic history, the adaptability of these measures and the inclusion of family and carer information may be seen as critical factors for their successful administration. Not all questionnaires or report measures addressed the respondent’s hearing alone, but they included questions that would be suitable to screen for hearing loss in older adults living with dementia; for example, the American Speech-Language-Hearing Association Functional Assessment of Communication Skills for Adults (ASHA FACS) [190], Camberwell Assessment of Need for the Elderly (CANE) [275], Multi-Dimensional Dementia Assessment Scale (MDDAS) [276], and the Resident Assessment Instruments [277]. The screening approaches judged to be unsuitable for use by nurses with older adults living with dementia were more likely to be have been implemented in clinical or research environments, with adaptations for use more often requiring the expertise of specialist clinicians and researchers.
DISCUSSION
This scoping review sought to answer the question of which hearing screening measures and approaches have been effectively used to identify hearing loss in older adults living with dementia, with a view to finding reliable and valid tests that would be suitable for use by nurses working in LTCHs. The identification of residents who have hearing problems is critical for determining the need for referral to a specialist or for accommodating accessibility needs (e.g., use of assistive technology or communication strategies), and for informing person-centered care. The identification of residents who have hearing loss is necessary so that appropriate accommodations are implemented to better support successful communication, meaningful engagement in everyday activities, quality of care, and to thereby delay or reduce the risks of further cognitive decline, such as social isolation in LTCHs. As there is a heightened need for suitable and validated screening measures for the identification of hearing loss in persons living with dementia that can be used across LTCH settings by nursing professionals, the focus of this discussion will center on the tools which could be suitable for use in this setting by these staff.
Suitable measures for the screening of hearing in adults living with dementia
The review found that a wide range of measurement approaches have been used to measure aspects of hearing and auditory functioning in individuals living with dementia, including subjective measures (based on self- or other-reports), behavioral measures (requiring a patient response to tones or speech), and physiological measures (examining the ear canal for cerumen [wax], immittance to assess middle ear function, otoacoustic emissions to evaluate inner ear status, or evoked response potentials to examine auditory brain responses). Pure-tone audiometry was the most frequently reported hearing test.
The consulted team of accredited healthcare professionals with expertise in the assessment of older adults living with hearing loss and dementia identified a subset of the hearing screening measures extracted in this scoping review that were deemed to be suitable for nurses to use in order to screen the hearing of residents living with dementia in LTCHs. Suitable measures included measurement approaches involving otoscopy to determine the need for cerumen management, in addition to the use of interview questions, collecting relevant hearing and ear disease history from medical charts, and using questionnaires such as the Nursing Home Hearing Handicap Index for the patient and the carer [278], the Hearing Handicap Inventory for the Elderly [279], as well as questionnaires and inventories to screen for tinnitus and hyperacusis. The whisper test was found to be the most suitable behavioral measure. The suitability of these measures was based on procedures falling within the scope of practice of nurses with no prohibitive requirements for specialized or costly equipment or extensive training.
The review indicated that a noteworthy proportion of adults with cognitive impairment were unable to complete standard test protocols or required the tester to modify the procedures used to administer tests. Sensibility issues were noted for 54 tests. In many studies conducted across various settings, patients or residents living with severe or very severe dementia were excluded from the outset if measures involved verbal reporting or other behavioral responses [122, 197]. In one study [246], despite audiometric testing being led by certified and experienced audiologists, the majority of 307 participating nursing home residents were unable to perform standardized pure-tone or speech audiometry, with over half (54%) not completing reliable audiologic assessments due to dementia or “dementia-characterized behaviors”. Among participants whose hearing was successfully screened using pure-tone audiometry, the majority were found to have hearing problems, but those identified as having problems were older and performed more poorly on cognitive measures compared to those without hearing problems [140]. Overall, a large proportion of studies relied on the expertise of audiologists or geriatricians to implement hearing assessments with appropriate adaptations to test procedures for individuals experiencing more severe cognitive impairment. These findings bring to light the importance of the testing environment (i.e., a quiet location), as well as the training required to successfully administer screening tests with knowledge of how to adapt standard procedures to the cognitive capacities and abilities of the individuals being screened. Thus, almost two-thirds of hearing tests identified in the present review were not found to be suitable for nurses to administer in LTCHs.
Suitable measures for the screening of residents in LTCH settings
The testing environments reported in the studies included various clinical settings (memory clinics, primary care centers, and geriatric departments of hospitals), as well as research laboratories and clinics, and private residences. However, less than a quarter of the studies included in the review focused on testing residents in LTCH settings. We were most interested in the studies involving testing by nursing professionals in LTCHs, as both hearing loss and cognitive impairments are highly prevalent in residents living in LTCHs, and because residents with complex multi-morbidities must first be identified as requiring an evaluation of their hearing before being referred to the relevant audiologic or medical specialist. Finding a suitable testing space that is quiet and without distractions in LTCHs can be a challenge.
The requisite identification and referral for clinical assessment outside of the LTCH, however, may be lacking and may not be practical. Meanwhile, combined hearing and cognitive impairments will significantly affect the communication functioning and social interactions of residents and their overall quality of care. In LTCHs, the most frequently cited hearing screening approaches used by nurses were those that relied on self- and/or other-report measures. These measures could be simple yes/no questions about hearing problems and hearing aid use, observations made during a clinical interview, or the use of standardized questionnaires, review of medical chart data, or nursing report forms. As part of a comprehensive assessment system used in LTCHs, the interRAI Long Term Care Facilities (interRAI LTCF) and RAI-MDS have been designed to gather relevant clinical information across many functional domains to guide the treatment and care planning of older adults who may experience complex and multiple chronic conditions [280]. The interRAI LTCF, along with the RAI-MDS 2.0 and 3.0 were cited in 36% of the studies conducted in this care setting. However, the use of the interRAI LTCF as a screening tool has also been found to have some limitations. A series of reports by the investigators of the Services and Health for Elderly in Long TERm Care (SHELTER) study investigated sensory impairments among residents of 59 nursing homes in eight European countries based on the interRAI LTCF. A striking finding of these reports was that residents with “extremely severe cognitive impairment” were excluded due to inaccuracies in their assessments. Their exclusion highlights that, despite the interRAI LTCF being the most widely used screening measure in LTCHs worldwide, and even though it could include information provided by other professionals or clinical records, older adults with more severe impairments are at increased risk of not being identified as having hearing loss or needing referral to the relevant healthcare specialist [160, 161].
Other approaches that were used by nurses in the LTCH setting were reviews of medical records and charts, nursing assessment forms and questionnaires (e.g., Nursing Home Hearing Handicap Index for Carer; [278]), self- or other-reports, and observation of hearing difficulties. The use of self-report measures is a concern insofar as such measures typically under-estimate the presence of hearing loss that would be detected using behavioral tests such as audiometry [173]. In order to address inaccuracies in documented hearing impairments or discrepancies between self- and other-reports, additional education on observable indicators of hearing loss, as well as reporting biases, is warranted for healthcare professionals working with this patient population.
Additional approaches to hearing screening included some physical measures that could be used by nurses, such as screening otoscopy (5 studies), and behavioral approaches such as the whisper test (n = 6). Studies reported on the use of a whisper test across clinical, residential, and LTCH settings, without requiring any adaptations of the test procedure itself [94, 148]. By testing the participants’ ability to hear the examiner’s whispered voice from a distance of 60 cm, and by dichotomizing results as the presence or absence of hearing difficulties, the researchers found that a third of older adults presented with uncorrected hearing impairment. Testing was often conducted by “geriatric-trained” investigators; however, in one study, research nurses were trained to achieve satisfactory accuracy and inter-rater reliability in recognizing hearing loss as observed in response to speech and whispered voice during an interview [175]. Along with otoscopic examination, which already falls within the scope of nursing practice (for those who have completed a certification course), the whisper test has potential utility for nurses, once additional training has been provided. Nonetheless, these screening procedures are limited insofar as they do not provide information about the degree of hearing loss and cannot be used to quantify the degree of change in an individual’s hearingstatus.
Several of the studies highlighted the role that nurses could play in conducting otoscopic evaluation to determine if ear care for residents is needed to remove impacted cerumen from the ear. Importantly, removal of cerumen occluding the ear canal to restore sound transmission to the eardrum has many benefits; it can improve hearing in more than 10% of cases [121], as well as increasing the accuracy of audiometric testing, and enabling devices such as hearing aids to perform properly. In one study, two thirds of LTCH residents assessed by otoscopy had impacted cerumen, but this condition would not have been discovered from responses to self-report measures alone [173]. In a study in which 15 residents were assessed by an experienced audiologist and student audiologists, one third of residents presented with wax occlusion, with three of these five residents having bilateral occlusion [172]. In another study, 52% of participating residents required removal of cerumen before their hearing could be evaluated accurately and, when comparing the results of audiometric assessment by an experienced audiologist with staff ratings on the RAI-MDS, 44% of residents had unidentified or underestimated hearing loss [70]. These results spotlight the need to physically examine the ear canal for impacted cerumen as a precondition to further assessment, as well as the need for training of the responsible healthcare professionals to undertake cerumen management. While nurses were reported to conduct otoscopy, referral to a specialist was typically made for cerumen removal [113]; however, in many jurisdictions, cerumen management is within the scope of practice for nurses who have completed certified training[51].
The administration of test measures by nurses in the LTCHs was predominantly reported in studies that used interRAI LTCF and RAI-MDS to gather data on residents’ hearing status, with the exception of a cross-sectional survey of English residential homes by Mann and colleagues [175]. Across 17 homes, research nurses assessed residents’ hearing ability by observing responses to “speech and whisper”, with 40% of residents, the majority of whom had dementia, presenting with hearing difficulties. It was also found that staff recognition, responsiveness and quality of care provided to residents with sensory impairments, such as the updating of care plans and provision of aids, influenced the residents’ experience of depression, with appropriate interventions associated with lower levels of depression in these residents [175]. This study indicates that the informal assessment of hearing by use of whispered voice can be implemented by nurses to successfully identify residents with dementia who are experiencing hearing loss. It also supports the development of the nurse’s role in resident assessment, so that suitable and adaptable approaches can be used to screen residents for hearing loss, with the consideration of the resident’s individual needs and capabilities, at a time and place that will also reduce any additional burden and thereby maximize the reliability of results. These are key factors which nursing professionals are precisely qualified to determine.
Identified limitations and priorities for future research
The present review found that the studies conducted in LTCH settings most frequently used either self- or other-report measures, or a combination of these measures with otoscopy and behavioral measures (e.g., the whisper test) to screen for hearing loss in residents living with dementia. There have been recent efforts to establish the reliability of the most common audiometric measures for use with adults living with dementia [47, 49]. Although these approaches may be successful for screening hearing in adults with mild or even moderate cognitive impairment, the most often reported concern for the sensibility of test administration was the severity of cognitive impairment in older adults with dementia. In particular, reliable and feasible methods to screen hearing in those with severe dementia are still needed for nurses to use in a less controlled and more ecological manner, as would be required in the LTCH setting. The review did reveal an evident need for increased involvement of healthcare professionals working in LTCHs to identify residents with dementia who could benefit from appropriate referral and rehabilitation for hearingloss.
There is growing consensus on the need to identify and appropriately address hearing loss in older adults [23, 44]. Calls have been made for increased cross-disciplinary education of health and social care professionals, along with evidence-informed policy improvements, to address the need for increased awareness of sensory problems and better collaboration among care providers to adequately support individuals living with combined sensory and cognitive impairments [281]. With advances in digital and remote technologies [44], previous methods that relied upon specialized and costly equipment and training may become more accessible, ecological, and adaptable to the LTCH environment.
There are limitations to the current study, principally, that the team of experts who appraised the suitability of hearing instruments for use by nurses did not include the expertise of nursing professionals. Following the endorsement of specialists in both sensory and cognitive aging [282], a consequent study was conducted using the screening approaches judged to be suitable for use by nurses to screen for hearing loss in residents with dementia in a LTCH setting in order to test their feasibility and reliability, and with the results forthcoming.
CONCLUSION
While many tools and strategies have been used to measure hearing in older adults with cognitive impairments, most studies have been conducted in clinics or laboratories rather than in LTCH settings. In addition, most studies of hearing screening that have taken place in LTCHs have been conducted by specialists with specific training, and the experience to make necessary adaptations to test administration, not all of which is easily transferable to nursing staff. Identifying reliable, valid, and feasible hearing screening tools for nurses is a necessary step in this quest to develop new approaches and implement informed policies to identify hearing loss in residents living with dementia in LTCHs by the most appropriate, responsive, and practicable means, so that the sensory and communicative needs of affected residents can also be addressed.
Footnotes
ACKNOWLEDGMENTS
The database search was conducted by Jessica Babineau, an Information Specialist at the Toronto Rehabilitation Institute-University Health Network. We thank Dawn Guthrie, Jonathan Jarry, Xochil Argueta-Warden, Maxime Grossi, Morgan Hempinstall, Karine Elalouf, Huiwen Goy, and Swaathi Balaji for their contributions to this research. This work was financially supported by the Alzheimer Society Research Programme (ASRP), Alzheimer Society of Canada; grant number RG 16-08. Dr. McGilton is supported by the Walter and Maria Schroeder Institute for Brain Innovation & Recovery. The sponsor had no role in conducting the study or preparing this manuscript. Dr. Wittich is supported by an FQRS Junior 2 chercheur boursier career award (# 281454).
