Abstract
Background:
The metabolomic and proteomic basis of mild cognitive impairment (MCI) and Alzheimer’s disease (AD) is poorly understood and the relationships between systemic abnormalities in metabolism and AD/AMCI pathogenesis are unclear.
Objective:
The aim of the study was to compare the metabolomic and proteomic signature of saliva from cognitively normal and patients diagnosed with MCI or AD, to identify specific cellular pathways altered with the progression of the disease.
Methods:
We analyzed 80 saliva samples from individuals with MCI or AD as well as age- and gender-matched healthy controls. Saliva proteomic and metabolomic analyses were conducted utilizing mass spectrometry methods and data combined using pathway analysis.
Results:
We found significant alterations in multiple cellular pathways, demonstrating that at the omics level, disease progression impacts numerous cellular processes. Multivariate statistics using SIMCA showed that partial least squares-data analysis could be used to provide separation of the three groups.
Conclusion:
This study found significant changes in metabolites and proteins from multiple cellular pathways in saliva. These changes were associated with AD, demonstrating that this approach might prove useful to identify new biomarkers based upon integration of multi-omics parameters.
INTRODUCTION
Alzheimer’s disease (AD) is a neurodegenerative disease characterized clinically by gradual loss of memory, cognitive decline, and multiple behavioral changes [1, 2] and is the most common form of dementia. The incidence of AD is projected to increase by 300%over the next thirty years, mainly due to the growing aging population in many countries worldwide [3], and this disease has therefore become a global concern in terms of social and economic impact [4–6]. In the United States alone, an estimated 5.8 million people more than 65 years of age are suffering from AD dementia [7]. In clinical practice, patients are not usually diagnosed with AD until the disease has progressed to an advanced stage of cognitive impairment, and there are limited opportunities for reversing disease progression. However, in earlier stages of the disease, often characterized by mild cognitive impairment (MCI), there is substantially more scope to intervene and modify risk factors in order to reduce the risk of cognitive decline and dementia. Those individuals who are identified with MCI have a higher risk of developing AD with advancing age, with approximately 14–18%of MCI patients over 70 years progressing to AD annually [8, 9]. Currently, the ability to detect the early stages of AD and its progression is minimal, making it difficult to determine the optimal clinical therapy. Therefore, a minimally invasive approach is needed to identify preclinical individuals with AD and MCI so that early strategies can be put in place to prevent or reduce the risk of developing clinical symptoms.
Methods for the investigation of AD are often invasive, expensive, and cannot adequately identify biomarkers [10–12]. If successful population-based screening for AD is to be put in place, this will require readily available, minimally invasive, and inexpensive methods for a robust, low-cost diagnostic test with high specificity and sensitivity. Previously, we used cells from the buccal mucosa as a minimally invasive strategy to identify potential biomarkers for AD [13–15]. However, saliva is increasingly gaining attention as a body fluid for the search of biomarkers of health and disease [16]. Recent developments in saliva proteomics have enabled the identification of saliva proteins, and indeed one study demonstrated approximately 40%of the blood proteins currently used in blood-based diagnostics were present in saliva [17].
It is not known precisely how AD is initiated nor what controls the progression pathway. While there have been many different hypotheses as to the cause of AD, the exact triggers and mechanisms remain elusive and are challenging to verify. Recent developments in proteomic and metabolomic methods in disease models of AD have provided valuable insights into the molecular basis of AD [18, 19]. Targeted metabolomics in saliva has also revealed new biomarkers for AD [20]. Furthermore, combined metabolomic and proteomic profiling of saliva may reveal dysregulation of cellular functions that contribute to the disease status of cells or may contribute to the pathology and clinical presentation in AD. Therefore, the aim of the study was to compare the metabolomic and proteomic signature of saliva from cognitively normal and dementia patients diagnosed with MCI and AD, to identify specific cellular pathways altered with the progression of the disease.
METHODS
Patient samples
This study was approved by the local Human Research Ethics Committees, CSIRO Ref 09/11, Ramsay Healthcare Ethics Committee and South Australian Health HREC. All methods were carried out in accordance with the approved guidelines, and all participants provided written informed consent before participating in the study. Diagnosis of MCI or AD was conducted by clinicians (Drs. Hecker, Faunt, Johns, and Maddison) based on the criteria outlined by the National Institute of Neurological and Communicative Disorders and the Stroke-Alzheimer’s Disease and Related Disorders Association (NINCDS-AD&DA) [21] and by using recommendations from the National Institute on Aging-Alzheimer’s Association (NI-AAA) workgroups on diagnostic guidelines for MCI [22] using clinical criteria. The diagnosis of dementia due to AD was made in the clinic setting and was consistent with “probable AD” criteria described in the guidelines paper from the NI-AAA [23]. Data reported in this study are from a total of 80 participants from the South Australian Neurodegenerative Disease (SAND) cohort, including: 1) the cognitively normal/healthy control (C) group (
Blood and saliva collection
Blood was collected and processed within 3 h of collection. The RNAPro•SAL ™ (Oasis Diagnostics Corporation®, Vancouver, WA, USA) saliva collection system was used, which provides for rapid salivary metabolite and protein collection and recovery, cellular removal, and biomarker stability at ambient temperature. All samples were stored at –80°C until analysis.
Biochemical measurements and statistics
Plasma vitamin B12, folate, vitamin D, homocysteine, cholesterol, triglyceride, HDL-cholesterol, and LDL-cholesterol were measured by a clinical laboratory service; SA Pathology (Adelaide, South Australia, Australia). C-reactive protein (CRP) was measured using commercial enzymatic kits (Beckman Coulter Inc, Brea, CA, USA) on a Beckman AU480 clinical analyzer (Beckman Coulter Inc, Brea, CA, USA) with CV = 2.36%.
Apolipoprotein E genotyping
Chemicals
All chemicals used were MS grade or higher, purchased from Sigma-Aldrich (Castle Hill, NSW, Australia) unless specified otherwise. Acrylamide and Bradford assay reagents were purchased from BioRad Laboratories (Gladesville, NSW, Australia). LC Proteomics FlexMix Calibration Solution and Retention Time Calibration Mixture were purchased from Thermo Scientific (Woolloongabba, QLD, Australia). GC Retention Time Calibration standard and isotopically labelled standards were purchased from Cambridge Isotope Laboratories (Tewksbury, MA, USA).
Untargeted metabolomics
Saliva samples were prepared following a modification of a previous method [25]. Briefly, aliquots of saliva (100μL) were processed using internal standards (d27-myristic acid and D-Glucose 13C6 and L-Glutamine-amide-15N. Dried extracts were derivatized before analysis on the GC-MS system. GC-MS analysis was performed on an Agilent 6890B gas chromatograph (GC) oven coupled to a 5977B mass spectrometer (MS) detector (Agilent Technologies, Mulgrave, VIC, Australia) fitted with an MPS autosampler. Total ion chromatogram (TIC) mass spectra were acquired within a range of 50–650
Untargeted proteomics
Saliva samples were prepared following a modification of a previous method [26]. Saliva protein was precipitated by cold acetone then air-dried before re-dissolving in 8 M urea. An aliquot of protein (5μg) was reduced and alkylated. Proteins were then trypsin digested and incubated at 37°C overnight. The digestion was stopped with 1μL of 10%(
Protein data analysis
Protein Discoverer 2.2 (Thermos Scientific) and Sequest HT search engine were used to identify peptides/proteins and quantify the relative abundance of proteins. The spectrum data was searched against the UniProt
Chemometric analysis and salivary metabolome and proteome integration
The metabolomics and proteomics data, after batch-effect adjustment and log transformation, were analyzed using multivariate data analysis software SIMCA (version 16, Sartorius Stedim Biotech, Umeå, Sweden) and MetaboAnalyst 4.0 [27]. The Gene Ontology Resource and Enrichr were used for Enrichment analysis. The cut-off level for significant metabolites was a signal-to-noise (
RESULTS
The gender, age, Mini–Mental State Examination (MMSE),
Age and gender, MMSE scores and various blood measurements between groups
*Bonferroni
The metabolomics and proteomics analysis yielded 173 common metabolites and 923 common proteins across the three saliva sample groups collected [age-matched control, control (
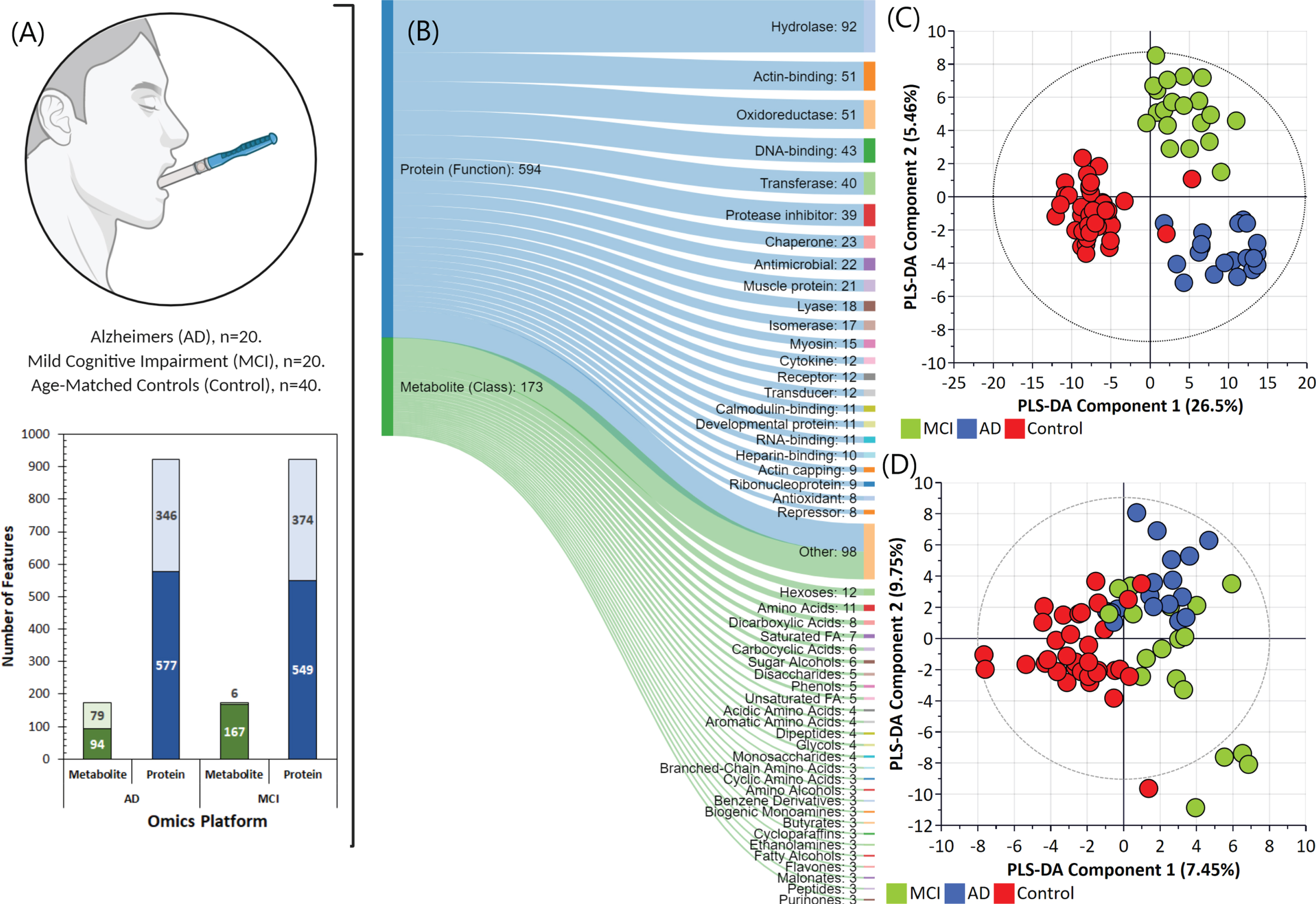
Omics analysis data overview. A) Saliva sample group summary and the number of metabolite and protein features identified and found to be statistically significant in the Alzheimer’s disease (AD) and mild cognitive impairment (MCI) groups compared to the control group. B) Overview of proteins and metabolites classified based on chemical ontologies and structural similarity and function. Partial least squares-data analysis (PLS-DA) score scatter plots for the (C) proteomics and (D) metabolomics datasets.
In order to explore variations between these sample groups, the metabolomic and proteomic data were first log transformed, normalized and analyzed by multivariate statistics using SIMCA. Figures 1C and 1D illustrate the partial least squares-data analysis (PLS-DA) score scatter plots for the proteomics and metabolomics datasets, respectively. As evident in Fig. 1C, the proteomics data alone can be used to provide separation of the three groups. In the AD group, 130 proteins were significantly changed in abundance, whilst 155 proteins were changed in abundance in the MCI group compared with controls (Log2fold change≥1.5-fold, ≤-1.5-fold, adjusted
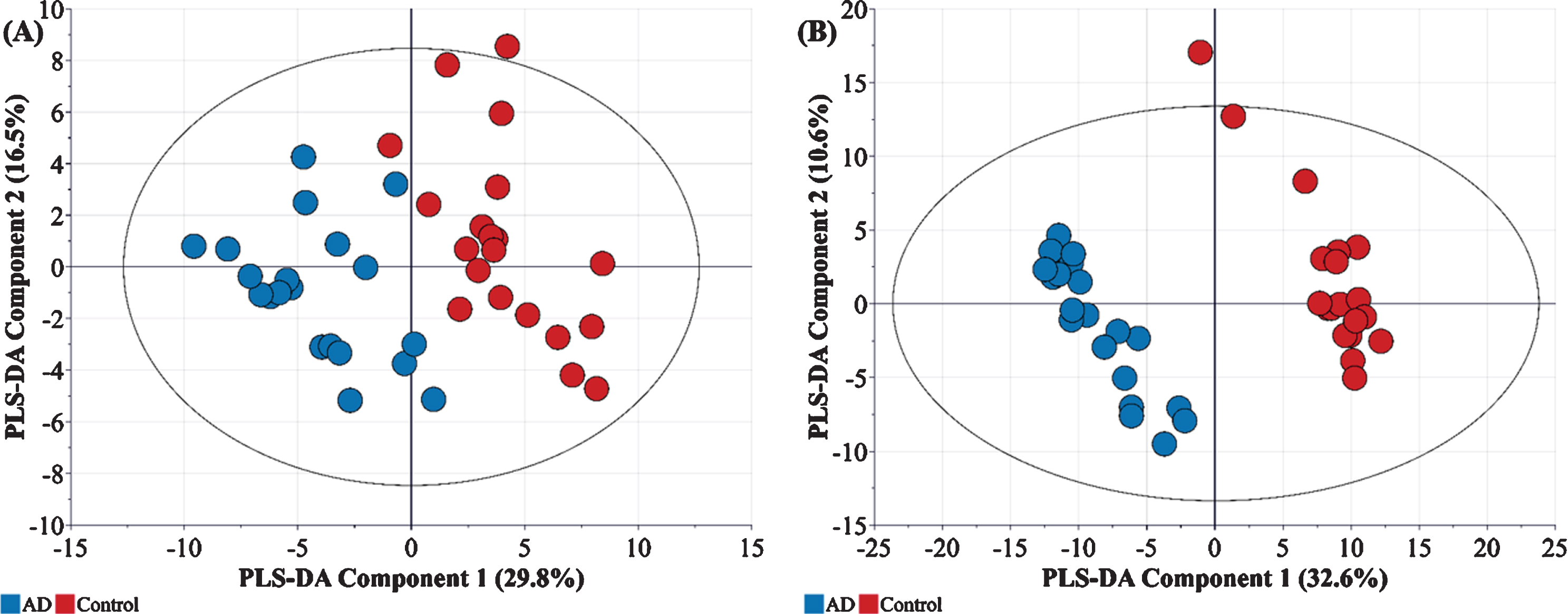
A) Metabolic (R2X = 0.464, R2Y = 802, Q2 = 703) and B) Proteomic (R2X = 0.462, R2Y = 0.996, Q2 = 0.973) profiles of AD samples (
The integration of metabolome and proteome profiles of AD and MCI though the ‘Joint pathway analysis’ tool indicated a presence of 69 metabolic pathways of which 15 were observed to be significantly different (FDR
Significant pathways expressed in saliva from AD and MCI (compared with controls)
The joint pathway analysis indicated that several major pathways, including the β-alanine pathway, citric acid cycle, cysteine and methionine metabolism, and phenylalanine metabolism showed significant increases in the AD group when compared to the MCI group (Fig. 3).
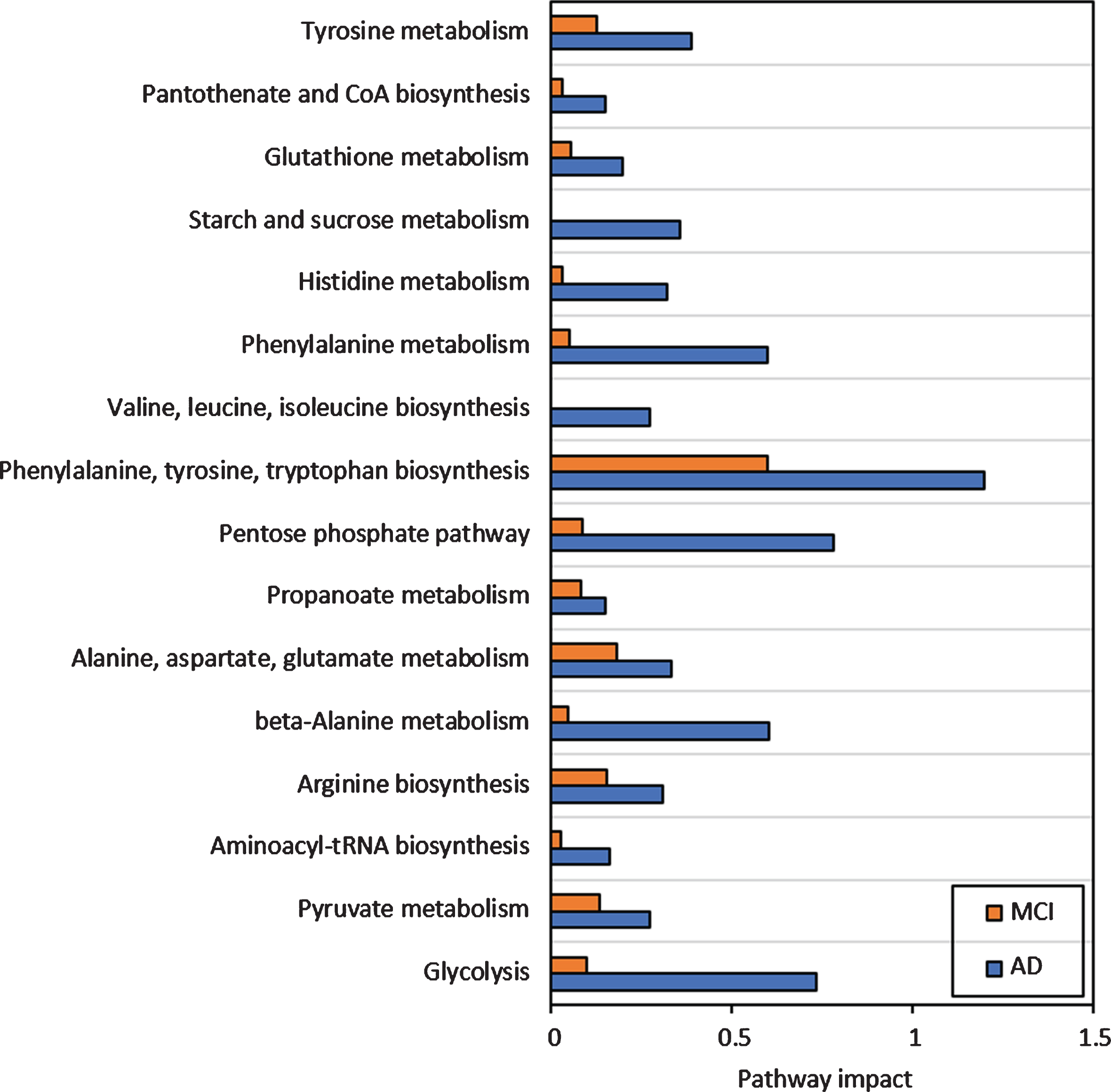
Impact of major pathways associated with the MCI and AD conditions compared with the control group.
One of the prominent observations was the accumulation of cholesterol in the saliva of patients with AD (Log2Fold change (FC) = +4.57), in contrast to depletion in the MCI (Log2FC = –1.62). Additionally, for arginine metabolism, a higher accumulation of aspartate, putrescine and ornithine was observed in the AD group compared with controls, which was associated with lower aldehyde dehydrogenase 3 (Uniprot ID P51648) (Log2FC = –2.08) with respect to MCI (Log2FC = –1.32). In tyrosine and phenylalanine metabolism, both tyrosine and phenylalanine accumulation were observed in the AD group but not in the control group. Significant accumulation of downstream products of tyrosine metabolism were further observed in the MCI group only. These included hydroxyphenyl lactate, tyrosol, and tyramine (Fig. 4A). Metabolomic-proteomic integration indicated that such accumulation of the downstream products of tyrosine metabolism was likely due to downregulated aldehyde dehydrogenase family enzyme activities in MCI and AD groups. It is known that the members of this family (P51648, P43353, P30838) catalyze numerous reactions in glycolysis and tyrosine and phenylalanine associated pathways (Fig. 4A). Energy pathways such as glycolysis, oxidative phosphorylation and citrate cycle are the most crucial metabolic pathways in eukaryotic cells due to their role in energy generation.
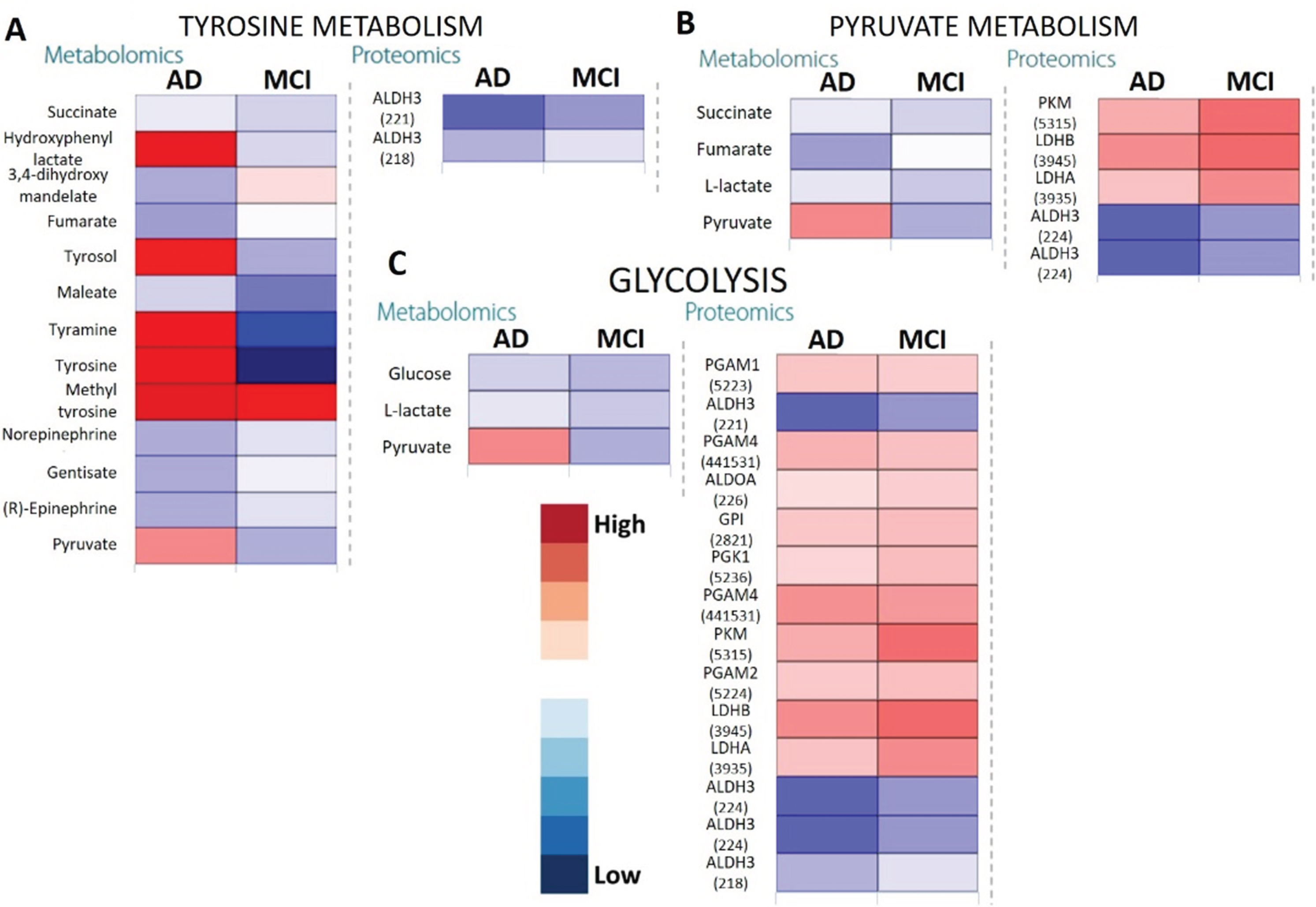
Heatmaps showing metabolomic and proteomic expressions during (A) Tyrosine metabolism, (B) Pyruvate metabolism, and (C) Glycolysis in the salivary matrix of AD and MCI patients with respect to control representatives (non-AD/MCI patients). The metabolites and proteins used in the multi-omics integration were selected based on the fold changes (Log2FC≥1.00 or Log2FC≤–1.00) with statistically significant differences False discovery rates (FDR≤0.05)). Note: The numerical IDs in parenthesis indicate Entrez Gene IDs of the proteins involved in these pathways.
Citric acid cycle showed three out of 42 elements that were differentially expressed only in the AD group (impact = 0.37, FDR = 0.0467) where it appeared to be somewhat upregulated. On the other hand, upstream pathways such as glycolysis and pyruvate metabolism showed higher impact (Fig. 3) especially in AD. This was confirmed by the upregulated pyruvate and downregulated citrate cycle metabolites such as citrate, succinate, and fumarate (Fig. 4) along with increased expression of glycolysis proteins, especially for the AD group as compared with controls. The downregulated succinate and fumarate, combined with low aldehyde dehydrogenase activities in AD also indicated the rerouting of citrate from citrate cycle [29, 30], causing a further upregulation of glycolysis, resulting in pyruvate upregulation in AD (Fig. 4C). This indicated an increased proteasome age activity, causing downregulation of oxidative phosphorylation and fragmenting the citrate cycle and rerouting it toward other pathways [31]. This could be further gauged from the increased antigen processing and presentation, especially through the outputs of antigen-processing cells and leukocyte transendothelial migration (Fig. 5A, B). It appeared that metabolites and proteins contributing to PD also increased throughout the transition of MCI to AD (Fig. 5C).
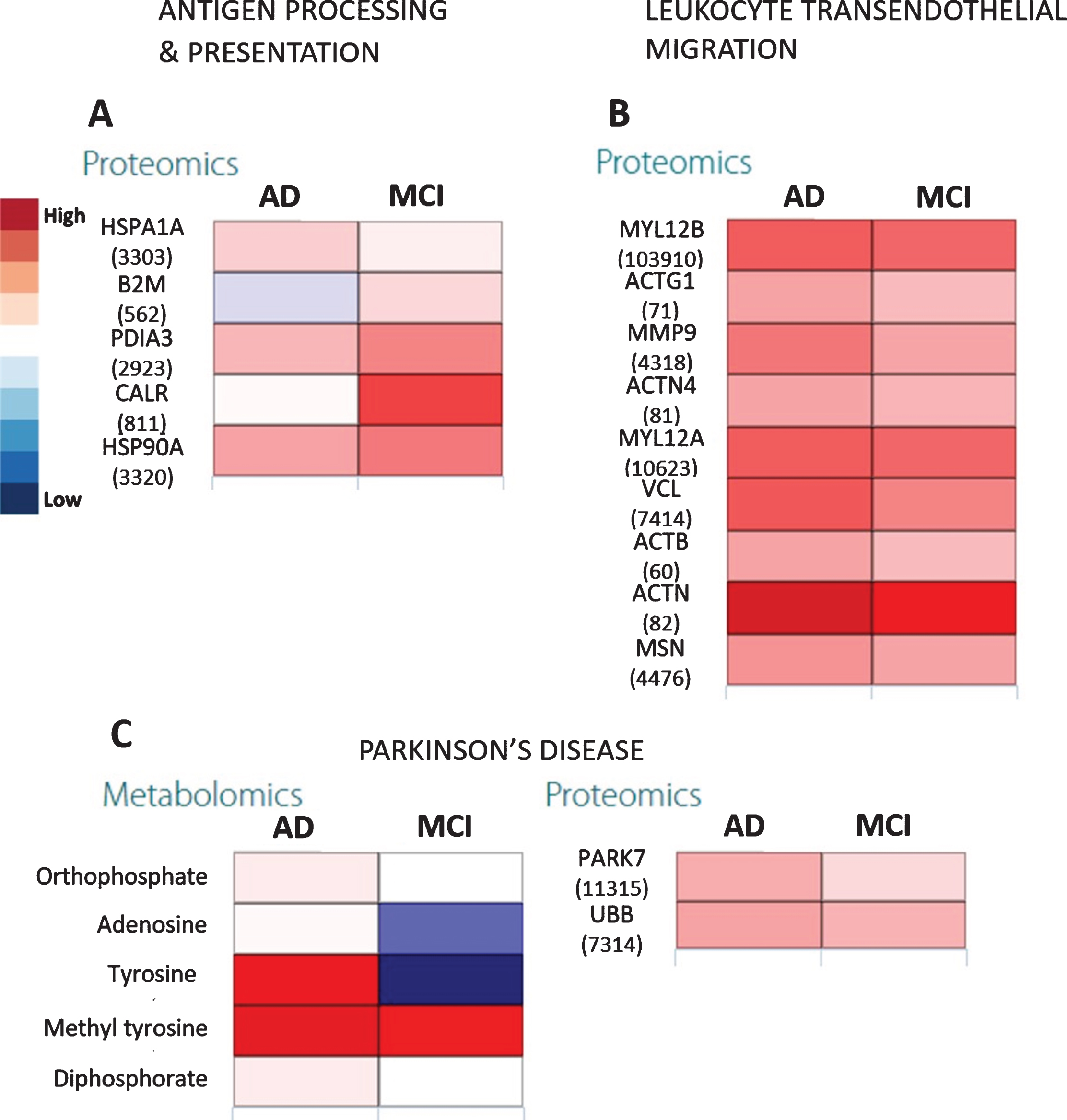
The heatmaps show proteomic expressions of (A) antigen processing and presentation and (B) leukocyte transendothelial migration in salivary matrix of AD and MCI patients with respect to their age- and gender-matched controls. C) Heatmaps showing proteomic expression during Parkinson’s disease in salivary matrix of AD and MCI patients with respect to controls. The metabolites and proteins used in the multi-omics integration were selected based on the fold changes (Log2FC≥1.00 or Log2FC≤–1.00) with statistically significant differences (False discovery rates (FDR≤0.05)). Note: The numerical IDs in parenthesis indicate Entrez Gene IDs of the proteins involved in these pathways.
DISCUSSION
Cognitive impairment detection at an early stage is vital, as the disease causes irreversible degeneration of cognitive capabilities in affected individuals. The likelihood of MCI transitioning to dementia annually lies between 14–18%[8, 9] and therefore, it is vital to identify and characterize the biomarkers associated with this transition to enable early diagnosis. Recent untargeted metabolomic analysis has shown the increased levels of glutamate, ornithine, cytidine and sphinganine-1-phosphate [32], and galactose and acetone [33] as probable biomarkers for the transition from MCI to AD. However, the presence of these metabolites as biomarkers of MCI to AD transition is doubtful and variable results exist [34, 35]. Additionally, elevated tau and phosphorylated tau proteins in cerebrospinal fluid have been reported to be indicative of an increased risk of MCI progression to AD [36]. However, salivary tau proteins have shown no correlation with AD in a recent study [37], indicating a necessity to search for more reliable biomarkers, and preferably from non-invasive biofluids such as saliva. To determine potential non-invasive biomarkers for AD, we carried out saliva proteomic and metabolomic analyses utilizing mass spectrometry methods. We found significant alterations in metabolites and proteins from multiple cellular pathways, demonstrating that at the omics level, disease progression impacts numerous cellular processes. These include energy production, cell/organelle structure, signal transduction, mitochondrial function, cell cycle progression, and proteasome function. Therefore, this approach could be used to identify new biomarkers with the results based upon integration of multi-omics parameters for better prediction and diagnostics.
Microglial and astrocyte cells have been shown to produce considerable levels of pro-inflammatory proteins that cross-migrate from cerebral matrix to blood [37]. Prior studies observed changes in serum with increased phenylalanine and tyrosine concentrations. In fact, a high phenylalanine/tyrosine ratio was shown to be a good indicator of AD, caused by the downregulated phenylalanine-4-hydroxylase activity which could prevent stress response metabolites such as L-DOPA and epinephrine [32, 39]. In our current study, phenylalanine and tyrosine accumulation was observed in the AD group, and this correlated with the downregulation of aldehyde dehydrogenase. This enzyme plays a critical role in detoxification and elimination of reactive oxygen species. A recent study with ALDH2 knockout mice has indicated a mutation of this protein in AD patients showing
An upregulated arginine metabolic pathway has been recently shown in AD, especially leading to an increase in ornithine levels through the urea cycle [32]. Our results were in line with this observation, albeit with slight differences as there was a moderate increase in arginine metabolism intermediates in MCI and AD compared to controls. However, a minor reduction of arginase 1 activity was observed when comparing the MCI to AD. Arginine metabolism is important for synthesizing polyamines, which act as agonists for neurotransmitter binding N-methyl-D-aspartate (NMDA) receptors [19]. Indeed, inhibition of arginase activity reversed amyloid-driven neuronal dysfunction and AD symptoms in animal models of AD [43].
Critical risk factors for AD such as hypercholesterolemia and increased serum saturated fatty acids are critical risk factors for AD [44] with gradually increasing levels of cholesterol associated with AD progression for instance [45]. We did not find significant changes in plasma total cholesterol in the MCI or AD groups compared with the control group. However, we observed decreased saliva cholesterol levels in MCI, while levels significantly increased in AD when compared with the control group. Additionally, the expression of steroid hormone biosynthesis enzymes appeared to be disrupted in the transition from MCI to AD, possibly preventing cholesterol degradation and subsequently accelerating AD progression. The study’s observations were in line with the previously shown association with AD of genes involved in sterol regulatory proteins and ATP-binding proteins [46].
One of the peculiar observations from our study was the expression of PD biomarkers in the MCI to AD transition (e.g., Parkinson’s disease protein 7; PARK7). Although PD is considered distinct from AD, recent pathological studies have shown that some similarities exist between the transition from MCI to AD and PD. These include downregulated NR4A1 gene expression which was similar in PD and AD [47]. We intend to conduct future studies that directly compare salivary metabolites and proteins that compare PD with AD, and these results may show the correlations and distinct differences for PD and AD pathologies and may shed additional light on their relationship.
In a study investigating serum metabolomic profiling in patients with AD, a total of eleven components were chosen as biomarker metabolites between AD and control groups, and these metabolites were closely related to seven biological pathways: arginine and proline metabolism, phenylalanine metabolism, β-alanine metabolism, primary bile acid synthesis, glutathione metabolism, starch and sucrose metabolism, and steroid hormone biosynthesis. These serum data may be useful for further understanding AD pathogenesis [39] and our saliva data which demonstrates similar pathway changes thus could provide a simpler non-invasive approach. It is important to note that two MCI and 13 AD participants were taking cholinesterase inhibitors medications; however, these medications are unlikely to affect the significantly impacted pathways revealed in our study. Overall, we found that biomarkers previously reported in cerebral, blood, and liver studies, were also observed in saliva. Additionally, the integrated proteomic-metabolomics approach displayed a greater model predictability for MCI to AD transition at the early stages of neurodegeneration (Fig. 6). This is the first pilot study identifying changes and abundance of saliva proteins and metabolites using an omics approach for the assessment of developing AD risk. Saliva has an additional advantage that it can be self-collected multiple times in a stress-free manner. As such, these practical aspects support the logical step to utilize saliva in the search for early AD biomarkers. Furthermore, this study provides further evidence that MCI and AD pathology is not restricted to neuronal cells but involves substantial changes in peripheral tissues.
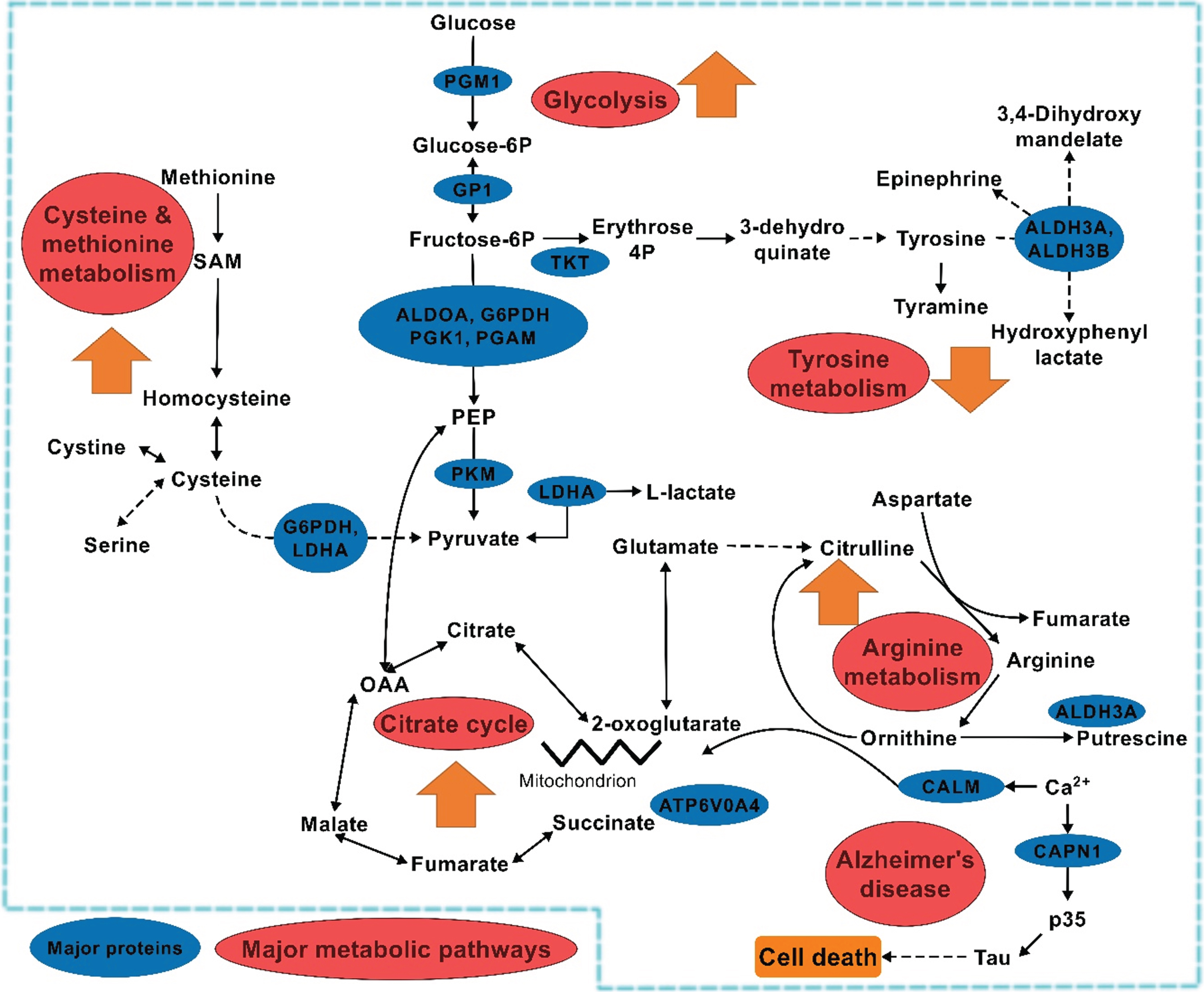
General metabolic behavior in saliva during MCI to AD transition. Note: The  indicates individual pathway behavior during the transition. Zigzag indicates break in the pathway.
indicates individual pathway behavior during the transition. Zigzag indicates break in the pathway.
Footnotes
ACKNOWLEDGMENTS
We thank all the volunteers who participated in this study as well as the nursing and office staff at the recruitment sites; the Calvary Rehabilitation Hospital, Modbury Hospital (Specialist Ambulatory Rehabilitation Centre) and South Australian Health and Medical Research Institute (CSIRO/SAHMRI) in Adelaide, South Australia. This study was supported by a CSIRO initiative; the Probing Biosystems - Future Science Platform research funding scheme.
