Abstract
Background:
The effect of regional brain amyloid-β (Aβ) pathology on specific cognitive functions is incompletely known.
Objective:
The relationship between Aβ and cognitive functions was investigated in this cross-sectional multicenter study of memory clinic patients.
Methods:
The participants were patients diagnosed with Alzheimer’s disease (AD,
Results:
Hierarchic multiple regression analyses were performed on each cognitive test as dependent variable, controlling for demographic characteristics and
Conclusion:
Five subcortical and cortical regions with Aβ pathology are differentially associated with cognitive functions and stages of disease in memory clinic patients.
Keywords
INTRODUCTION
A pathological hallmark of Alzheimer’s disease (AD) is the progressive accumulation of senile plaques in the brain containing amyloid-β (Aβ). The amount of pathological Aβ is possible to measure
A behavioral hallmark of AD is cognitive decline commonly evaluated by means of tests of memory and other cognitive domains. The first and most early cognitive change in typical AD is a decline in episodic memory during the preclinical stage of AD, when patients are not demented but symptomatic years ahead of the diagnosis of AD. Change of memory is followed by change in the executive and visuospatial functions in the prodromal stage closer to the diagnosis of AD, when multiple cognitive functions are affected without marked dysfunction in daily life, i.e., MCI. Finally, verbal functions become impaired in association with dysfunction in daily living in the early AD stage [15–17]. When a diagnosis is decided upon, the cognitive impairment encompasses several domains including memory and at least one other cognitive domain. In this way, it is possible to discern three stages of AD development: the single symptomatic stage many years ahead of a clinical diagnosis of AD, the prodromal stage years ahead of an AD diagnosis (impairment in multiple cognitive functions) and the early AD stage the diagnostic criteria are fulfilled, when there is clear dysfunction in daily living.
Most studies on the relationship between PiB PET and cognition have used global measures of neocortical PiB retention. In these studies, the association between PiB and cognition has been reported to be absent or weak [5, 19], while it seems to be significant in MCI [5, 21] and shows mixed results in healthy aging possibly due to sample characteristics and variability of methods utilized [5, 22]. Relatively few studies using PiB PET measures have examined the association between amyloid pathology in specific brain regions and tests in specific cognitive functions. When the relationship between specific regions and specific cognitive tests has been studied, marked associations have been reported for subcortical regions and episodic memory in heterogeneous samples of patients including AD, MCI, and healthy controls [8, 23].
In the present explorative study of brain-behavior associations in AD, the rationale was to focus on specific tests covering changes in AD in relation to regional brain PET Aβ pathology and possible modifying conditions. It is well known that there is an interaction between Aβ and the
The purpose of the study was to investigate the relationship between regional PET Aβ deposition in cortical and subcortical brain regions in each hemisphere, rather than global neocortical PiB, and the performance in AD-sensitive cognitive tests rather than global cognitive measures in patients with AD and MCI as well as healthy control (HC) individuals recruited from European memory clinics, i.e., three groups varying in level of cognitive performance. In addition, possible modifying conditions, education, and
METHODS
Participants
The participants in the European PiB PET study [5] were recruited from five European PET centers (Leuven/Belgium, London/UK, Munich/Germany, Stockholm/Sweden, Turku/Finland. All patients had been referred from primary to a memory clinic at each European site because they had symptoms of suspected dementia. Patients diagnosed with AD or MCI were eligible for the study. All patients had participated in research studies on PiB PET at each center. Patients were excluded if they had psychiatric symptoms, signs of abuse, head trauma, or other neurologic conditions. This study included 215 individuals. The controls were recruited among carers and relatives. Only those controls, who did not report any cognitive problems were included into the study. Details of inclusion/exclusion criteria and technical scanning information has been presented previously [5].
In the present study on PiB and cognition, 27 of the 215 individuals were excluded as they did not participate in any assessment of cognitive functions and 10 individuals completed less than 5 cognitive tests. In addition, three participants (2 AD and 1 MCI) were excluded as they performed extremely poor, see Statistical analyses). Accordingly, 175 individuals completed both a PET scan and an assessment of cognitive functions. Among these subjects, 83 patients were diagnosed with AD, 60 with MCI, and 32 were HC subjects.
Diagnosis
The diagnosis was decided at each site by experienced experts based on all clinical information. The AD diagnosis fulfilled the NINCDS-ADRDA criteria for AD [28] and the DSM-IV criteria for dementia [29]. The MCI diagnosis was based on the modified Petersen criteria for MCI [30]. The healthy status of HC was checked by in-house examination at each site.
Clinical examination
All patients had been referred to and examined for suspected dementia development according to local routines at each center. A detailed description of examination routines at each center has been presented in a previous publication [5]. Neuropsychological testing was used to measure cognitive function following the standard procedures at each center, see below. The
11C-PiB PET imaging
The synthesis of 11C-PiB was performed at each center according to good manufacturing practice. The PET examination was performed at each center following the routine procedure at each center. The imaging data were submitted to the Wolfson Molecular Imaging Centre, Manchester, UK, for central processing. The cerebellar region was used as reference. Twenty-three regions of interest (ROI) were anatomically defined, using the Montreal Neurological Institute atlas [31] and PiB values were normalized. All details of the whole PET procedure have been presented previously [5].
Neuropsychological assessment
Seven cognitive domains were covered by standard neuropsychological tests. The tests were administered and evaluated according to standard procedures [32] for each test. Global
All the neuropsychological raw scores except MMSE and TMTA and TMTB were standardized by means of transformation into z-scores using age-matched normative values according to test manuals of each test at the University Clinic of Cologne, Germany, the responsible site for test data. For the MMSE test, normative data reported by Crum and associates [34] were used. The TMTA and TMTB test scores (latencies) were transformed to z-scores according to a meta-study on these tests [35] after excluding abnormal values (latencies≥200 s for TMTA and latencies≥300 s for TMTB) in order to normalize the distributions. In addition, participants with extremely poor performance were excluded if the mean z-score across the seven tests exceeded
Statistical analyses
Multiple regression analyses (one for each cognitive domain) were conducted, with cognitive test as dependent variable using participants, who performed better than -5 in z-score on mean cognitive function across all seven tests. This criterion was chosen to exclude participants with extremely poor performance on specific tests. The independent variables years of education and
RESULTS
Comparison of groups
The demographic and clinical characteristics of the three groups are presented in Table 1. There were no significant differences between the three groups in age (
Demographic and clinical characteristics (MMSE and
PiB measures across 13 brain regions in AD, MCI, and HC
The PET PiB measures across 13 brain regions and two hemispheres in three groups of subjects (AD, MCI, and HC) are presented in Table 2 and visualized in Fig. 1. It should be noted that these PiB measures were not part of the diagnostic procedure. There was no difference in PiB between hemispheres in any region in the HC group (all ps > 0.1). However, in the MCI group, PiB was significantly larger in the right hemisphere (
The PET PiB measures across 13 regions and two hemispheres (within subjects) with age, gender, education, and
11C-PiB retention (
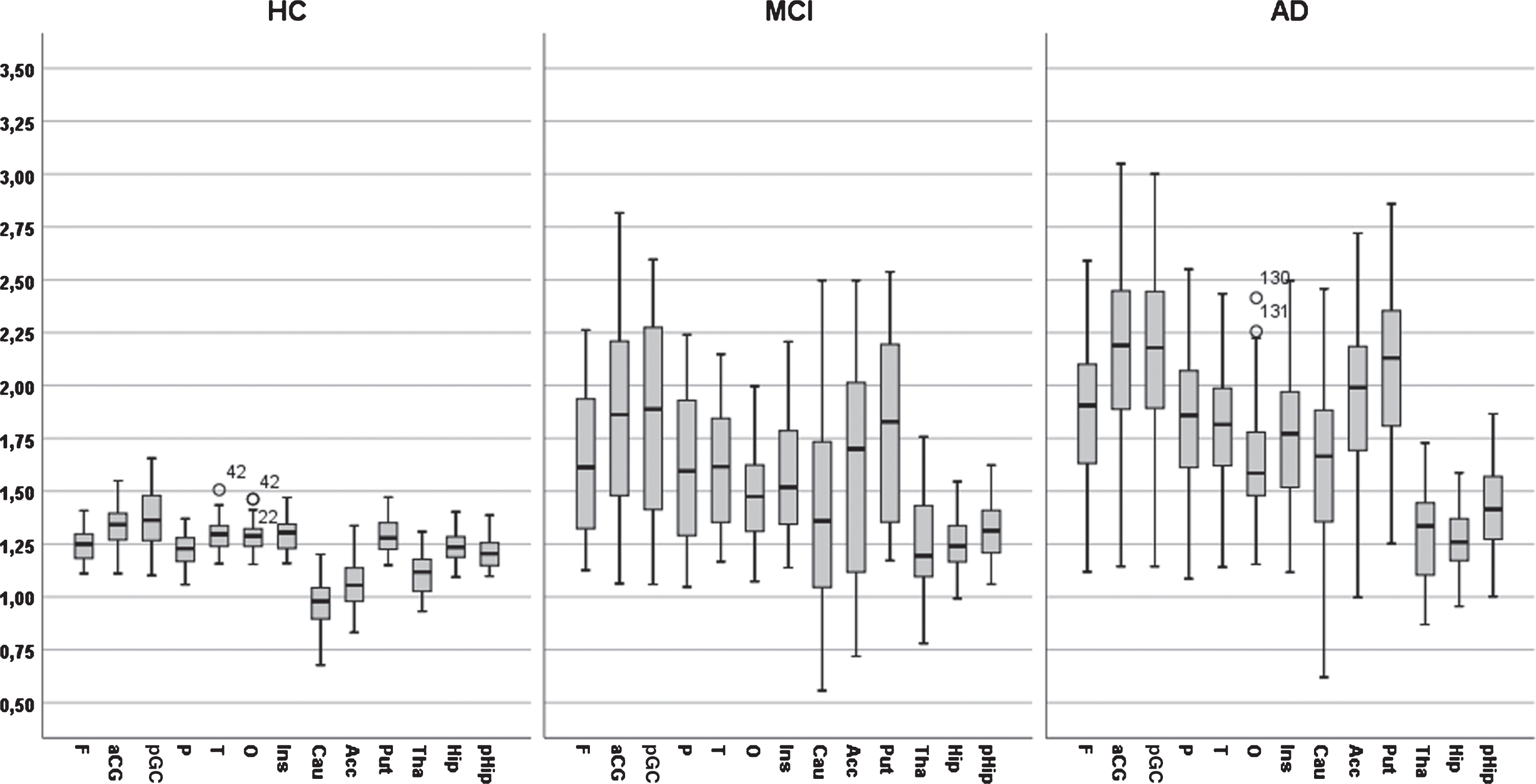
A bar graph of PIB value across 13 regions of interest for three diagnostic groups (AD, MCI, and Controls).
The interaction between diagnosis and ROI was due to a differential pattern of group differences across ROIs. Diagnostic groups differed markedly and significantly in 10 ROIs (frontal, parietal, occipital, temporal, anterior and posterior gyrus cinguli, caudate, putamen, and accumbens), significantly but less markedly in two ROIs (thalamus and parahippocampus) and not significantly in one ROI (hippocampus).
Test results across cognitive domains in AD, MCI, and HC
The neuropsychological results in z-scores (
Test results in z-score (Mean±SD) results on 7 neuropsychological tests in three groups of individuals: Alzheimer’s disease (AD), mild cognitive impairment (MCI), and healthy controls (HC)
Relationship between regional PiB measures and cognitive performance
For each cognitive domain, the relationship between the test and the predictors was computed using multiple regression analyses. In the first block of predictors, years of education, and number of
In Table 4, the outcome of the seven multiple regression analyses is presented. All analyses were significant (all ps < 0.001) and the multiple correlation coefficients varied from 0.42 for semantic fluency to 0.64 for word list delayed recall. The first step revealed that years of education were a positive predictor in all cognitive tests. It was significant in s three tests (verbal learning, verbal retention, and executive function). The presence of
Multiple regression analyses of cognitive test results in relation to predictors: education and
r, multiple correlation coefficient; edu, years of education;
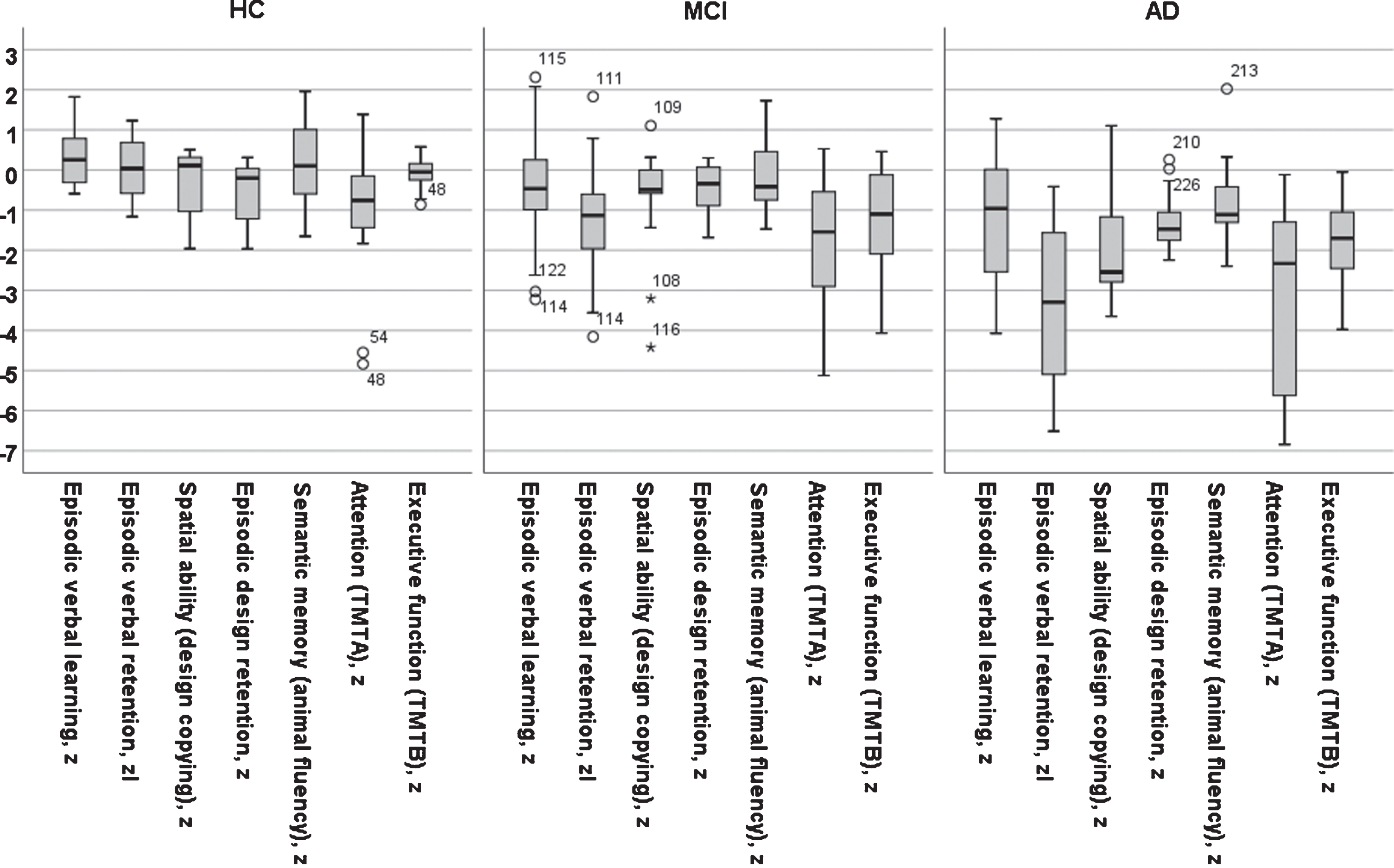
A bar graph of test results across five cognitive domains in z-score for three diagnostic groups (AD, MCI, and Controls).
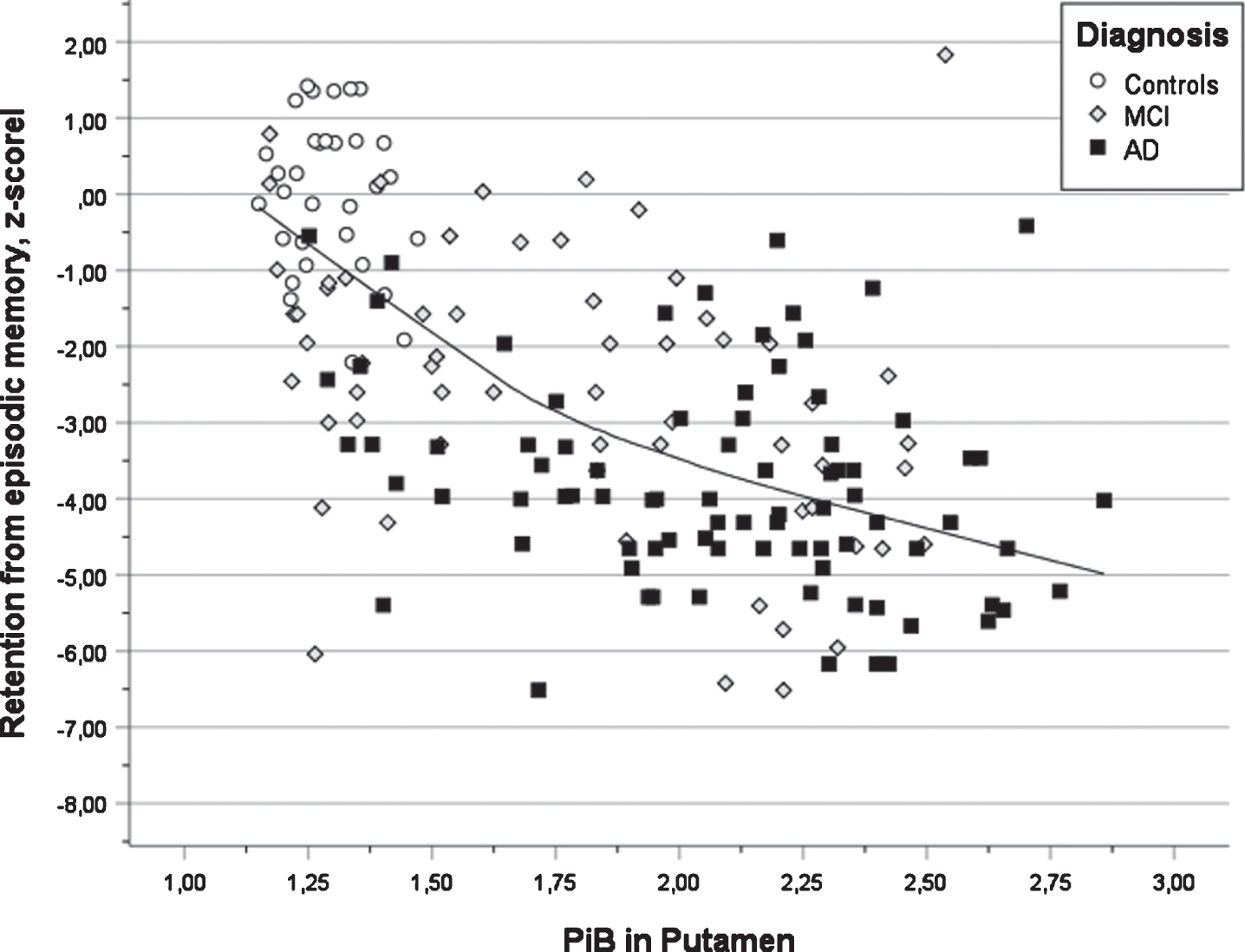
Scatter plot of test results in verbal learning versus PIB value in putamen for three diagnostic groups (AD, MCI, and Controls) also showing the regression line using local weighted regression.
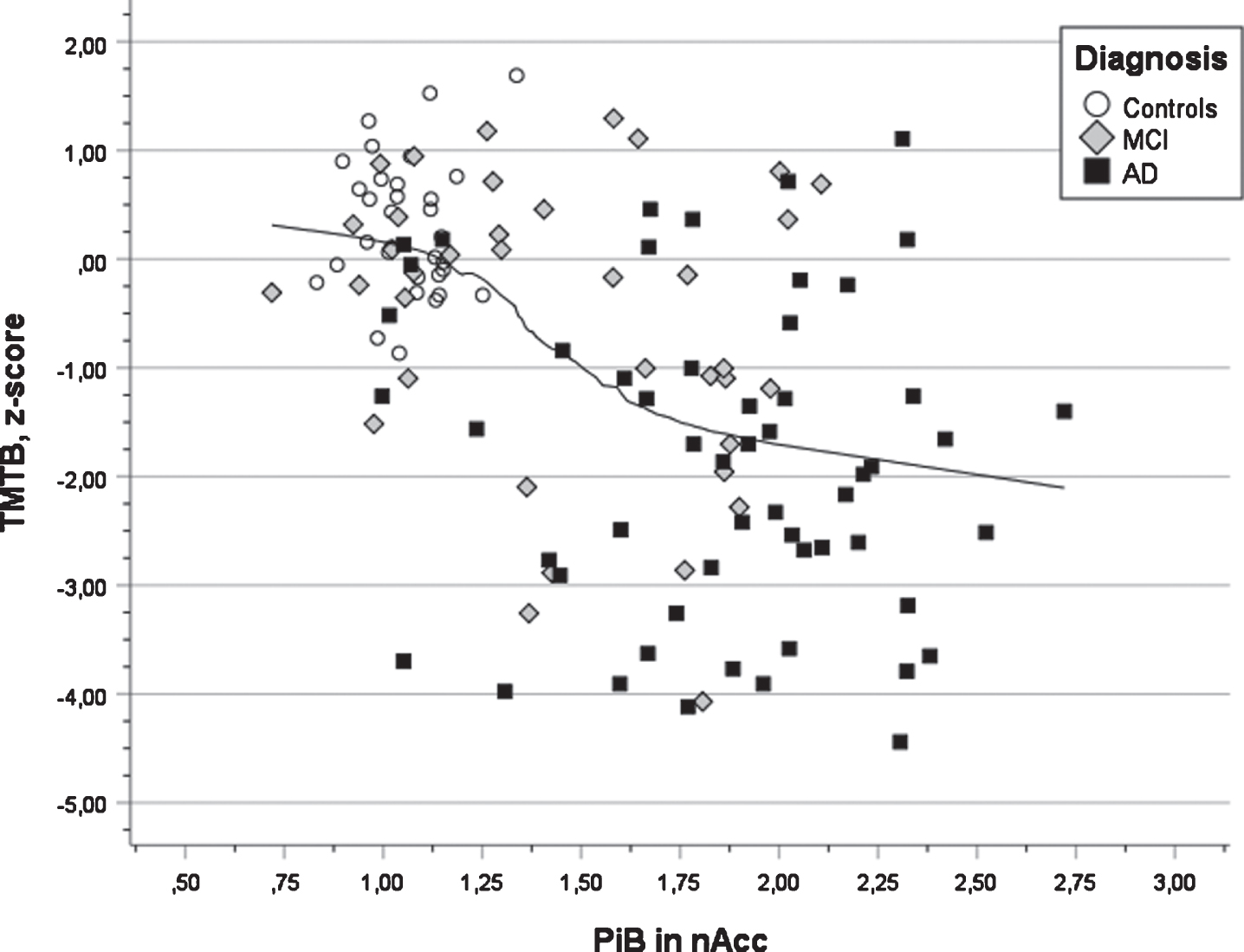
Scatter plot of test results in executive function (TMTB) versus PIB value in nucleus accumbens for three diagnostic groups (AD, MCI, and Controls) also showing the regression line using local weighted regression.
DISCUSSION
The present study investigated the
Five distinct brain-behavior relationship were obtained between performance in specific cognitive tests and Aβ pathology. Aβ pathology in the left putamen was associated with verbal learning and retention, Aβ pathology in the right gyrus cinguli was associated with visual search in the TMTA test (attention), and Aβ pathology in the left nucleus accumbens was associated with the TMTB test (executive function). Furthermore, significant brain-behavior relationships were obtained between Aβ brain pathology in the right occipital region and visuospatial tasks (copying and delayed reproduction of target design) as well as the left parietal region and verbal fluency).
The association between increased pathology in putamen and decreased performance in verbal learning and verbal retention (delayed recall) is in line with previous research [7, 11] and a review of functional magnetic resonance imaging (fMRI) studies regarding brain networks involved in episodic memory [36].
A second finding was the coupling between increased PiB PET in the right posterior cingulate gyrus and impaired performance in the TMTA task here interpreted as construct of attention. In a review of the role of posterior cingulate, it was reported that changes in posterior cingulate cortex are found not only in AD but many other diseases (e.g., traumatic brain injury, psychiatric syndromes, multiple sclerosis, etc.) in relation to regulating the arousal state, focused attention, and the breadth of attentional focus [40]. These changes have been shown using various methods like PET studies using glucose metabolism [41] and fMRI [36] and they have been shown to occur in the early stage of AD disease course [14]. Posterior cingulate has been pointed out as one part of a frontostriatal networks subserving orientation as well as goal-directed executive control and arousal/sustained attention [42].
A third finding was that performance in the TMTB test used to indicate executive function (demanding attention, mental control, and organization) was negatively associated with PET Aβ in nucleus accumbens, a part of the striatal region. This region has been suggested to be involved in three main phenomena: learning of associations between stimuli, actions and rewards; selection between competing response alternatives; and motivational modulation of motor behavior [39, 43], among which TMT performance is related to modulation of motor behavior. Similar observations have been reported by other researchers in various patient groups and based on clinical and experimental studies [44–48]. The TMTB test is regarded as a sensitive test for AD development and decline in the preclinical stage prior to the clinical diagnosis of AD [14–17].
Further, elevated PET Aβ abnormality in the right occipital lobe was negatively associated with copying of complex figural stimuli, for instance the copy task of the Rey-Osterrieth Complex Figure test and delayed recall of the same test (retrieval from visuospatial episodic memory). This finding may illustrate the brain activity in tasks requiring processing of visual stimuli [49, 50] occurring relatively late in the disease course [14].
Finally, there was a significant association between increased Aβ in the left parietal lobe and poor semantic fluency performance (Animal fluency) in agreement with previous research in memory clinic patient, when brain abnormality was measured by cortical thickness [51], brain atrophy [52, 53], and PET glucose metabolism [53]. The association between impaired parietal lobe and affected semantic processing seem to in the clinical stage of AD [14, 53].
In majority of relationships, PiB deposition was a negative factor for cognition. However, in two relationships, PiB Aβ was positively associated with cognition (attention versus right parietal lobe and executive versus left thalamus). In these two instances, there were both negative and positive ROI factors. The positive predictors are a paradoxical outcome, since PiB retention in all regions, except hippocampus, are negatively and significantly correlated with impaired performance in all seven cognitive tests. These positive effects are regarded as artefacts which is supported by statistical knowledge regarding the common reciprocal suppression phenomena in multiple regression analyses in which beta weights can become positive although they are expected to be negative under specific circumstances [54] and not a veridical neurocognitive phenomenon [55].
The effect of
The
In conclusion, five brain-behavior relationships were identified showing an association between amyloid pathology in specific subcortical and neocortical regions and impaired performance in specific cognitive tests. Aβ in subcortical compared to cortical regions seemed to occur early in the disease course. As already known, visuospatial processing is related to occipital pathology and retrieval from semantic memory is related to parietal pathology, which occur relatively late in close connection to the clinical diagnosis of AD. The abnormality of episodic memory versus Aβ in putamen, abnormality in attention versus Aβ in posterior gyrus cinguli as well as executive dysfunction versus nucleus accumbens are new findings.
Footnotes
ACKNOWLEDGMENTS
The original project and the multicentre analysis were funded by EC-FP6 Network of Excellence on Diagnostic Molecular Imaging (DiMI, LSHB-CT-2005-512146). The contribution of all the collaborators in the original European multicenter PET study, headed by professor Karl Herholz, is gratefully acknowledged. This specific study on PiB and cognition was funded by the Swedish Research Council (project 05817) and the Regional Agreement on Medical Training and Clinical Research by Stockholm County Council (ALF) as well as Swedish Research Council (VR/2017-02965 and VR/2017-06086).
