Abstract
The noradrenergic (NE) locus coeruleus (LC) is vulnerable to hyperphosphorylated tau, and dysregulated NE-metabolism is linked to greater tau and disease progression. We investigated whether elevated NE-metabolism alone predicts memory decline or whether concomitant presence of tau and amyloid-β is required. Among 114 memory clinic participants, time trends (max. six years) showed dose-response declines in learning across groups with elevated NE-metabolite 3-methoxy-4-hydroxyphenylethyleneglycol (MHPG) with no, one, or two Alzheimer’s disease biomarkers; and no decline in the low MHPG group. Elevated MHPG is required and sufficient to detect learning declines, supporting a pathophysiologic model including the LC-NE system contributing to initial Alzheimer’s disease-related processes.
INTRODUCTION
Autopsy studies reported that the locus coeruleus (LC), the major site for norepinephrine (NE) synthesis, is one of the first brain regions to accumulate hyperphosphorylated tau pathology [1, 2]. The role of the LC in the late-life phenotypes of Alzheimer’s disease (AD) remains unclear, but recent work com-bining
We aimed to examine the relationship between hyperphosphorylated tau, Aβ, and MHPG, measured in CSF. In addition, we investigated whether memory decline shows a dose-response relationship depending on whether MHPG is elevated and whether MHPG is accompanied by AD biomarkers. Ultimately, these results will facilitate our understanding of the LC-NE system in the pathophysiological cascade of AD and whether NE-related markers can serve as indicator for disease progression.
METHODS
Participants
Participants (n=114) were included from a convenience cohort from the memory clinic of the Maastricht University Medical Center (MUMC), diagnosed with subjective cognitive decline (SCD, n=60), mild cognitive impairment (MCI, n=35) or probable AD dementia (n=19). Diagnoses were made by experienced physicians based on the core clinical criteria for MCI [8] and AD dementia [9]. Criteria for SCD included self-reported presence of subjective cognitive complaints and no impairments on cognitive tests (impairment is defined as a score below –1.5SD of the age-, sex-, and education-adjusted mean). Participants receiving a SCD diagnosis could have a CDR-rating >0 if cognitive tests were considered normal. Exclusion criteria were: major neurological disease, clinical diagnosis of other neurodegenerative disorders, recent transient ischemic attack or cerebrovascular accident, history of psychiatric disorders, and alcohol or drug abuse [7]. All participants provided consent, study protocols were approved by the Medical Ethics Committee of the MUMC and conform the World Medical Association Declaration of Helsinki.
CSF analyses and genetics
CSF was collected via a lumbar puncture and biochemical analysis of CSF Aβ1–42, p-tau181p, and MHPG was done blinded to diagnostic information and following standardized protocol [7].
Neuropsychological assessment
The cognitive assessment included the Clinical Dementia Rating (CDR) scale, Mini-Mental State Examination (MMSE), categorical verbal fluency task, letter-digit substitution test (LDST), and the verbal 15-word learning task (WLT, learning and delayed recall). Participants were invited for annual follow-up visits over a six-year period. Following our previous analyses, we focused on the WLT components in this study [7].
Statistical analyses
Statistical analyses were conducted in R version 3.5.1. We assessed the relationship between MHPG, CSF-p-tau or Aβ with linear regression (adjusting for age, sex, education, CDR-score, and
These groups were fed as a predictor into a linear mixed effects model with random intercepts for each subject and random slopes for time, with either WLT learning or recall as outcome and adjusting for age, sex, education,
RESULTS
Mean age of the sample was 62.97 year, with almost half of the individuals carrying at least one
Demographics of the MHPG/Biomarker groups
Mean and standard deviations of demographics of the entire cohort and the four subgroups. Group differences were tested with ANOVA (followed by pairwise
MHPG was not associated with Aβ (β=–0.07, t(108)=–0.93,
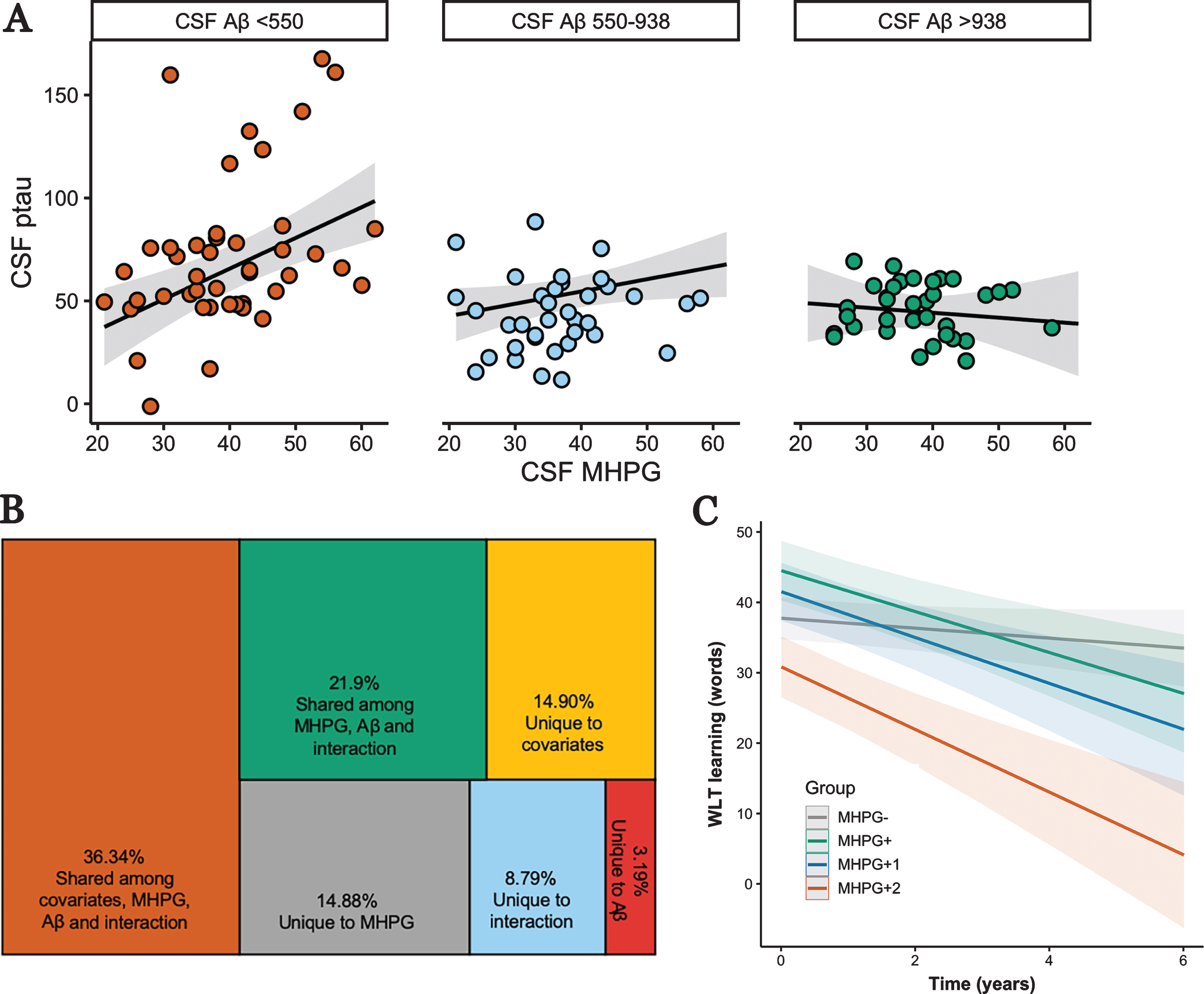
Associations between MHPG, Aβ, and p-tau and their relevance to memory decline. A) Interaction between CSF MHPG and CSF Aβ on CSF p-tau showing that a significant positive relationship between MHPG and p-tau occurs at Aβ-values <938pg/ml (derived from floodlight analyses; see Supplementary Figure 1). Analyses were done continuously but are grouped for visualization purposes; B) Variance decomposition of unique and shared contributions in explaining CSF p-tau of the model in A; C) Linear mixed effects model demonstrating that participants in the MHPG- group decline significantly less than the other three groups on the WLT learning over max. 6 years.
We then examined whether there is a dose-response relationship between elevated MHPG and having none, one, or two elevated AD-biomarkers in predicting memory decline (see Supplemental Material for relationships between each CSF marker and learning over time). To that end we created groups based on the median of the respective biomarker (Table 1). We assume missing at random given that the number of observations was not different among the groups (F (3,277) =2.01,
DISCUSSION
In this study, higher NE-metabolism was associated with higher CSF p-tau at Aβ values above the clinical cut-off. In addition, elevated MHPG was associated with declines in learning over a 6-year period, and memory decline was steeper in the presence of higher p-tau or Aβ pathology. Interestingly, AD pathology in the context of lower MHPG was not associated with significant memory decline. Based on these findings, we speculate that NE hypermetabolism by itself is a sufficient and required mechanisms related to decline in learning. Mounting postmortem and animal work suggests that the LC contributes to progression of pathology and symptomatology of AD [2, 11]. Our observations are consistent with a pathophysiology of AD in which changes in the LC-NE system may precede initial accumulation of tau pathology and subtle memory decline [12, 13], prior to significant deposition of Aβ. However, serial observations and experiments manipulating NE metabolism will be required to verify this pathophysiological sequence. Elevated NE metabolism may hold promise as early indicator of disease progression, at a stage where brain changes may be reversible and treatments be more efficacious.
Elevated MHPG with or without AD biomarkers was related to decline in learning, but the groups did not differ with respect to decline on delayed recall. Animal and human studies indicated that activation of the LC-NE system modulates all stages of memory processing, encoding, consolidation, and retrieval [14, 15]. In our data, all groups showed decline in recall performance. Recall scores are known to reflect short-term decline, whereas learning scores are more sensitive to long-term decline and have more clinical meaningfulness [16]. Examining relationships among NE-metabolism, AD-biomarkers and cognitive performance along the entire disease trajectory can shed light on the temporal dynamics of cognitive processes affected by LC alterations.
This study has strengths, including the combination of multiple CSF markers and longitudinal cognitive data, but, unfortunately, the modest sample size did not allow us to further split up the groups based on other thresholds, search for more complex interactions or consider nonlinear learning functions in a latent growth curve framework [17]. Replication in larger cohorts, ideally with longitudinal datapoints, will be necessary to investigate effect modifications, influence of aminergic mediation use, generalizability to other populations including those with lower proportions of
