Abstract
Background:
Right temporal variant frontotemporal dementia (rtvFTD) has been generally considered as a right sided variant of semantic variant primary progressive aphasia (svPPA), which is a genetically sporadic disorder. Recently, we have shown that rtvFTD has a unique clinical syndrome compared to svPPA and behavioral variant frontotemporal dementia.
Objective:
We challenge the assumption that rtvFTD is a sporadic, non-familial variant of FTD by identifying potential autosomal dominant inheritance and related genes in rtvFTD.
Methods:
We collected all subjects with a diagnosis of FTD or primary progressive aphasia who had undergone genetic screening (
Results:
Genetic variants in FTD related genes were found in 33% of genetically screened rtvFTD cases; including
Conclusion:
Our results demonstrate that rtvFTD, unlike svPPA, is not a pure sporadic, but a heterogeneous potential genetic variant of FTD, and screening for genetic causes for FTD should be performed in patients with rtvFTD.
Keywords
INTRODUCTION
Frontotemporal dementia (FTD) is a syndrome caused by degeneration of the frontal and/or temporal lobes [1]. Patients with predominant behavioral disturbances and frontotemporal atrophy on neuro-imaging are classified as behavioral variant FTD (bvFTD) [2], whereas the language predominant subtypes of FTD are classified under the umbrella of primary progressive aphasia (PPA) and have been associated with left hemisphere atrophy [3].
Over the years, the genetics of FTD have been broadly explored. The autosomal dominant inherita-nce pattern has been found higher in bvFTD, whereas semantic variant PPA (svPPA) is typically a non-familial sporadic disease [4
–7]. Pathogenic variants are most common in the microtubule associated protein tau gene (
Currently, diagnostic criteria for a variant of FTD presenting with behavioral changes, memory deficit, and prosopagnosia in the presence of right temporal atrophy (rtvFTD) are lacking [8]. Because of the atrophy pattern, theoretically, rtvFTD is considered a right variant of svPPA [3 , 10] and the general assumption is that it is also a sporadic disease.
Only one study focusing on the underlying genetic and pathological features in rtvFTD, showed a positive family history in 45% of the patients with pos-tmortem diagnostic confirmation [11]. Thus, we set out to investigate whether rtvFTD could be potentially a genetic disorder.
METHODS
In this report, out of 636 patients from the Amsterdam dementia cohort (ADC) with a clinical diagnosis of bvFTD (
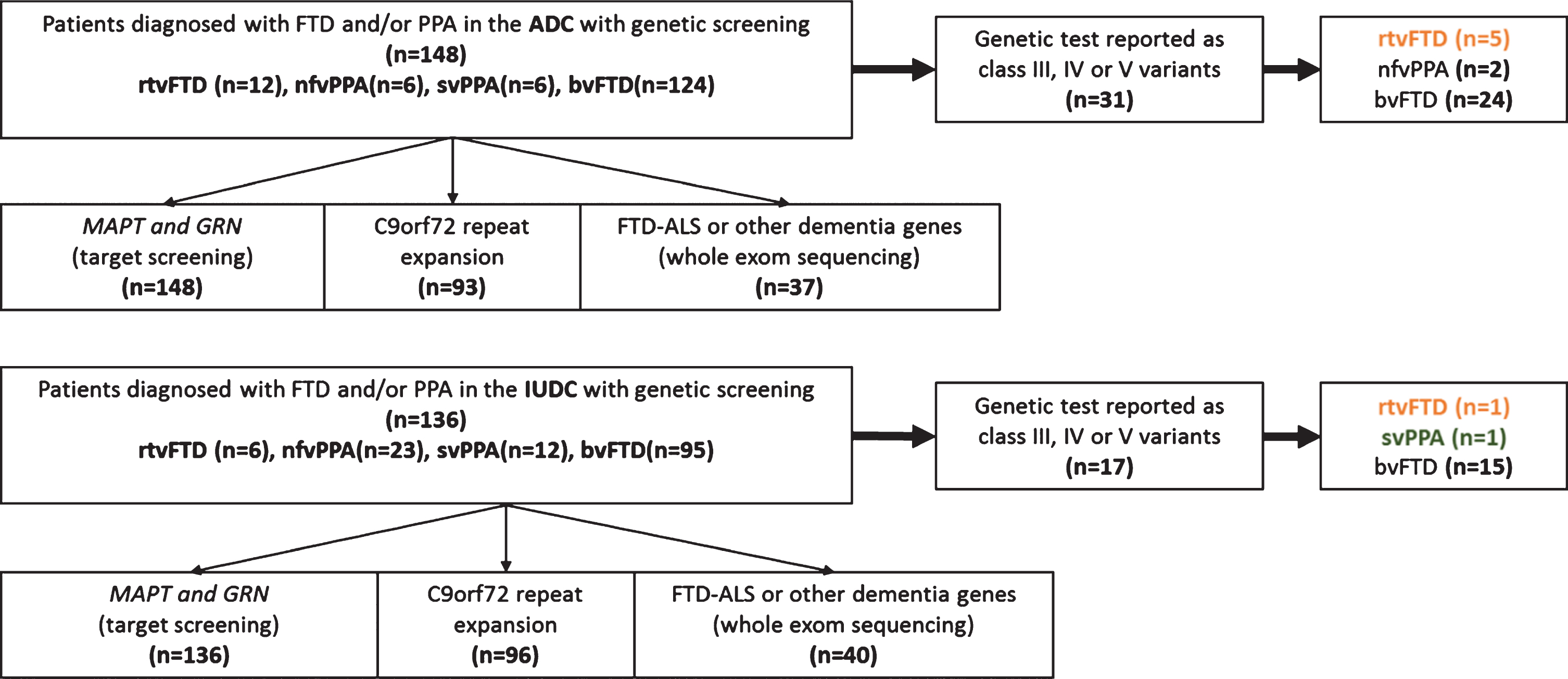
Patient selection.
RESULTS
Demographic, clinical features are displayed in Table 1, and detailed case histories are reported in Supplementary Material 2.
Demographic and clinical data
ADC, Amsterdam Dementia Cohort; IUDC, Istanbul University Dementia Cohort; MTA, mesial temporal atrophy; PET, positron emission tomography;
In our combined cohorts, genetic variants in FTD related genes were found in 33% of genetically sc-reened rtvFTD subjects (6 out of 18 genetically scr-eened rtvFTD), whereas only one svPPA (1 out of 18 genetically screened svPPA) subject had a genetic variant.
Summary of the cases
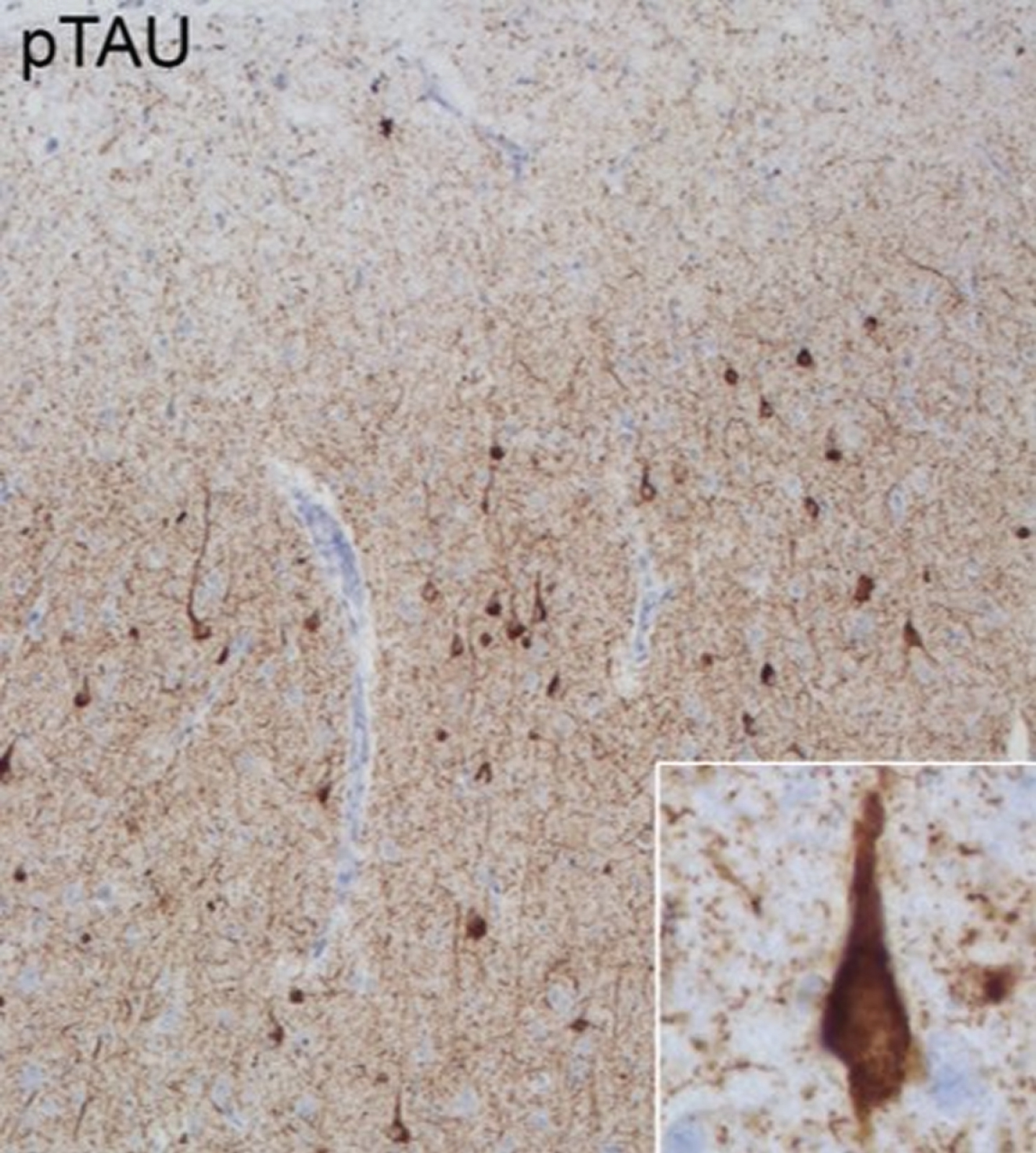
Pathological features of Case 3. Anterior cingulate cortex stained with phospho-tau (p-tau) monoclonal antibody (AT8: Pierce Biotechnology, Rockford, IL, USA). Extensive 3R and 4R tauopathy which is characteristic for
DISCUSSION
RtvFTD and svPPA are generally considered sporadic, non familial variants of FTD. In our combined cohorts, we can confirm that in svPPA rarely (∼5%) class III-V genetic variants in FTD related genes are found. However, 33% of rtvFTD patients that were screened for genetic mutations in FTD genes had a genetic variant. Moreover, these variants were in three different genes (
The genetic diagnosis of four out of six rtvFTD cases was FTLD-
Besides the
Although
In our study, four out of six patients had a strong family history for dementia. In the literature, a positive family history was reported in 37.5% (15 out of 40) of patients with rtvFTD [combined Chan et al. [34] and Josephs et al. [11]]. This percentage is quite high compared to svPPA in which a suggestive family history is identified in less than 5% of patients [6, 35]
Nonetheless, it is still unknown whether rtvFTD and svPPA share the same pathophysiology. A recent GWAS metadata analysis [36] has revealed that the svPPA gene network is uniquely associated with TAR DNA binding protein 43 metabolism. From this perspective, accompanying tauopathy in rtv-FTD resembles the heterogeneous pathophysiology of bvFTD, rather than svPPA. On the other hand, although C9orf72 is the most common worldwide cause of genetic FTD [5], it should be noted that this variant was not found either in our study or other rtvFTD cohorts [11, 34]. Therefore, further research into the pathophysiological background of rtvFTD and how this relates to the other FTD subtypes is warranted.
In conclusion, currently, there is no consensus on whether rtvFTD is a mirror variant of svPPA or should be lumped with svPPA. Although reminiscent of svPPA, our findings show that rtvFTD, unlike svPPA, often has a genetic basis and the genetic variants are found in multiple genes. Therefore, genetic screening is essential in patients with rtvFTD.
Footnotes
ACKNOWLEDGMENTS
We are very grateful for the generous contribution of the patients and their relatives. Research of the Alzheimer Center Amsterdam is part of the neu-rodegeneration research program of Amsterdam Neuroscience. The Alzheimer Center Amsterdam is supported by Stichting Alzheimer Nederland and Stichting VUmc fonds. WF holds the Pasman chair. Dr. HUE has received research support from the Turkish Neurological Society. FB is supported by the NIHR biomedical research center at UCLH.
