Abstract
Background:
High sensitivity liquid chromatography mass spectrometry (LC-MS/MS) was recently introduced to measure amyloid-β (Aβ) species, allowing for a simultaneous assay that is superior to ELISA, which requires more assay steps with multiple antibodies.
Objective:
We validated the Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 assay by LC-MS/MS and compared it with ELISA using cerebrospinal fluid (CSF) samples to investigate its feasibility for clinical application.
Methods:
CSF samples from 120 subjects [8 Alzheimer’s disease (AD) with dementia (ADD), 2 mild cognitive dementia due to Alzheimer’s disease (ADMCI), 14 cognitively unimpaired (CU), and 96 neurological disease subjects] were analyzed. Aβ species were separated using the Shimadzu Nexera X2 system and quantitated using a Qtrap 5500 LC-MS/MS system. Aβ1-40 and Aβ1-42 levels were validated using ELISA.
Results:
CSF levels in CU were 666±249 pmol/L in Aβ1-38, 2199±725 pmol/L in Aβ1-40, 153.7±79.7 pmol/L in Aβ1-42, and 9.78±4.58 pmol/L in Aβ1-43. The ratio of the amounts of Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 was approximately 68:225:16:1. Linear regression analyses showed correlations among the respective Aβ species. Both Aβ1-40 and Aβ1-42 values were strongly correlated with ELISA measurements. No significant differences were observed in Aβ1-38 or Aβ1-40 levels between AD and CU. Aβ1-42 and Aβ1-43 levels were significantly lower, whereas the Aβ1-38/1-42, Aβ1-38/1-43, and Aβ1-40/Aβ1-43 ratios were significantly higher in AD than in CU. The basic assay profiles of the respective Aβ species were adequate for clinical usage.
Conclusion:
A quantitative LC-MS/MS assay of CSF Aβ species is as reliable as specific ELISA for clinical evaluation of CSF biomarkers for AD.
INTRODUCTION
Cerebrospinal fluid (CSF) amyloid-β (Aβ)1-42 is a definite biomarker of Alzheimer’s disease (AD). A decrease in the Aβ1-42 level, which begins 25 years earlier, allows the prediction of the onset and progression of Aβ amyloidosis in the AD brain. CSF Aβ1-42 is the earliest and most reliable biomarker for the prediction and diagnosis of AD [1–3]. Low CSF Aβ1-42 and cortical amyloid PET ligand binding have been adopted as biomarkers of Aβ plaques (labeled “A”) in the ATN classification system of the NIA-AA research framework and have been proposed as diagnostic criteria based on the biological definition of AD for observational and interventional research [4].
However, antibody-based immunoassays, such as ELISA, have several disadvantages [24, 25]. Currently available ELISA systems using antibodies against different epitopes or the conformation of Aβ species measure the levels of various forms of Aβ species. The specific characteristics of Aβ with self-aggregation, binding to tubes, post-translational modifications, and cross-reactions with endogenous components of CSF decrease assay sensitivity and lead to variable results depending on the ELISA systems. Therefore, discrepant findings have been reported across each assay procedure and laboratory. This variability represents a major issue for global clinical observational and interventional studies. Commercial ELISA kits and systems are expensive, time-consuming (often include overnight incubations), and indicate the variation of measured values due to the different lots of the ELISA kits.
An approach based on liquid chromatography mass spectrometry (LC-MS/MS) has been developed to overcome these methodological issues [26–29]. The direct confirmation of Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 is possible, and detailed comparisons can be made with advanced LC-MS/MS equipment [8, 17–19]. However, the lower sensitivity and intra-assay variability of this system compared to immunological methods have yet to be reliably improved [26–29]. Therefore, we herein developed and validated a novel LC-MS/MS method to assay CSF Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43, and further confirm their contribution as biomarkers for AD pathological changes by comparing the results obtained by the conventional ELISA system for Aβ1-40 and Aβ1-42.
MATERIALS AND METHODS
Subjects
One hundred and twenty CSF samples from 106 patients with neurological diseases and 14 cognitively unimpaired subjects (CU) were examined. The AD group consisted of 4 cases of autosomal inherited ADD with PSEN-1 mutations, 4 of ADD, and 2 of AD due to mild cognitive impairment (ADMCI) based on the criteria of NIA-AA [4, 31]. They were all defined by positive
Subject profiles. Subjects consisted of 10 cases of Alzheimer’s disease with dementia (ADD) and AD due to MCI (ADMCI), 17 of amyotrophic lateral sclerosis (ALS), and 9 of corticobasal degeneration (CBD). Six cases of encephalopathy (ENC) consisted of meningoencephalitis, Creutzfeldt-Jacob disease, leukoencephalopathy, superficial siderosis, or cerebral angiitis. Three cases of frontotemporal dementia (FTD), 14 cognitively unimpaired healthy controls (CU), 14 cases of multiple sclerosis (MS) and related disorders including neuromyelitis optica in the multiple sclerosis group, 6 of multiple system atrophy (MSA), 11 of Parkinson’s disease (PD), 4 of peripheral neuropathy (PN), 11 of progressive supranuclear palsy (PSP), and 15 of spinocerebellar degeneration (SCD) were analyzed. No significant differences were observed in age among the CU and other groups
LC-MS/MS chemicals and reagents
Synthetic human Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 were purchased from the Peptide Institute (Osaka, Japan). Uniformly stable isotope-labeled peptides manufactured by rPeptide (Watkinsville, GA, USA), corresponding to each peptide (Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43), were used as internal standards (ISs). Artificial CSF was purchased from Tocris Bioscience (Bristol, UK), and rat plasma was purchased from Charles River Laboratories Japan (Yokohama, Japan). Other materials and reagents were purchased from Kanto Chemical (Tokyo, Japan), Kokusan Chemical (Tokyo, Japan), and Sigma-Aldrich (St. Louis, MO, USA). Ultrapure water was prepared using Milli-Q Integral 10 (Merck, Darmstadt, Germany).
LC-MS/MS sample preparation by solid-phase extraction
Human CSF samples (300
LC-MS/MS analysis
Chromatographic separation was performed using the Shimadzu Nexera X2 system (Shimadzu Corporation, Kyoto, Japan) with a column of Acquity UPLC BEH C8 (1.7
MRM transitions. Mass spectra were obtained by the Qtrap 5500 system with the positive electrospray ionization (ESI) and multiple reaction monitoring (MRM) mode. MRM transitions are shown, respectively
ELISA
Sandwich ELISA was used to quantify Aβ1-40 and Aβ1-42 levels using Human β Amyloid (1-40) ELISA Kit Wako II and a Human/Rat β Amyloid (1-42) ELISA Kit Wako, High-Sensitive according to the manufacturer’s instructions (Wako Pure Chemical Industries, Ltd., Osaka, Japan). Microplates were pre-coated with monoclonal BAN-50 (IgG, anti-Aβ1-16) and sequentially incubated with 25
Data analysis
Simple linear regression models were used in statistical analyses. The linear regression line was drawn by the method of least squares. Comparisons among the groups of assay values were performed using the Kruskal-Wallis test with multiple comparison tests for non-parametric data. Two-tailed
RESULTS
The minimal to maximal LC-MS/MS assay ranges of Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 were 193.231∼1,224.771, 391.624∼3,560.087, 29.326∼328.450, and 0.714∼20.624 pmol/L, respectively (Table 3). CSF levels as mean±standard deviation values of CU were 666±249 pmol/L in Aβ1-38, 2199±725 pmol/L in Aβ1-40, 153.7±79.66±79.7 pmol/L in Aβ1-42, and 9.78±4.58 pmol/L in Aβ1-43. The ratios of Aβ1-38/1-42, Aβ1-38/1-43, Aβ1-40/1-42, Aβ1-40/1-43, and Aβ(1-40/1-43) /Aβ(1-38/1-42) were 4.66±0.94, 71.7±15.8, 15.7±3.7, 241±58, and 53±13, respectively. The ratio of the amounts of Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 was approximately 68:225:16:1 (Table 4).
Basic profile of the LC-MS/MS analysis. The minimal to maximal assay ranges, median, mean, and standard deviations of Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 values measured by LC-MS/MS assay and ELISA
Assay data obtained by LC-MS/MS. Mean±standard values (pmol/L) of Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43, and the ratios of Aβ1-38/1-42, Aβ1-38/1-43, Aβ1-40/1-42, Aβ1-40/1-43, and Aβ(1-40/1-43)/Aβ(1-38/1-42) in respective disease groups including with ADD and ADMCI, ALS, CBD, 6 ENC, 3 FTD, MS, MSA, PD, PN, PSP, and SCD
**a
First order regression analysis showed a correlation between Aβ1-38 and Aβ1-40 (
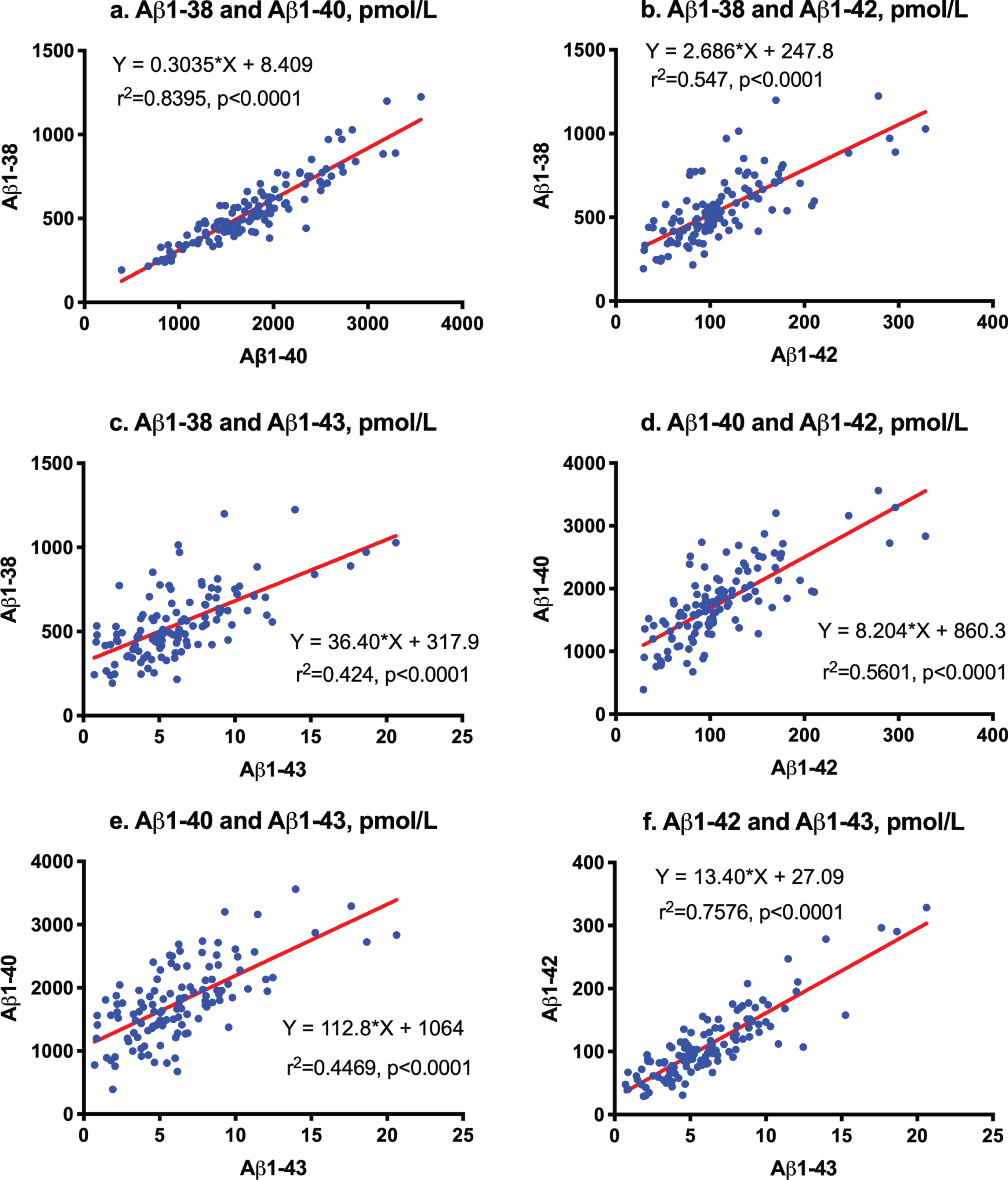
Correlation among Aβ species measured by LC-MS/MS. First order regression analysis between Aβ1-38 and Aβ1-40 (a), between Aβ1-38 and Aβ1-42 (b), between Aβ1-38 and Aβ1-43 (c), between Aβ1-40 and Aβ1-42 (d), between Aβ1-40 and Aβ1-43 (e), and between Aβ1-42 and Aβ1-43 (f).
Aβ1-40 and Aβ1-42 values measured by LC-MS/MS were strongly correlated with those obtained by ELISA. The first order regression analysis was

Correlation between LC-MS/MS and ELISA. Aβ1-40, Aβ1-42, and Aβ1-42 + Aβ1-43 values measured by LC-MS/MS were strongly correlated with those obtained by ELISA (a:  represent sporadic ADD, red triangles
represent sporadic ADD, red triangles  represent sporadic ADMCI, brown circles
represent sporadic ADMCI, brown circles  represent familial AD). Correlation analysis between measured levels by LC-MS/MS and ELISA in Aβ1-40/Aβ1-42 and Aβ1-40/Aβ1-42 + Aβ1-43 showed close relationships, but most values were present in the leftmost regression curve (e and f).
represent familial AD). Correlation analysis between measured levels by LC-MS/MS and ELISA in Aβ1-40/Aβ1-42 and Aβ1-40/Aβ1-42 + Aβ1-43 showed close relationships, but most values were present in the leftmost regression curve (e and f).
The regression curve between the Aβ1-38/1-42 ratio and Aβ1-40/Aβ1-43 ratio measured by LC-MS/MS was
We then examined the efficacy of LC-MS/MS assay values as biomarkers for AD by comparisons with assay values among ADD/ADMCI, CU, and other neurogenerative diseases. No significant differences were observed in Aβ1-38 and Aβ1-40 levels among ADD/ADMCI and other neurological diseases and CU (Fig. 3a, b). Aβ1-42 levels were significantly lower in ADD/ADMCI than in CU (

Values of CSF Aβ species and ratios among neurological diseases and controls. No significant differences were observed in Aβ1-38 and Aβ1-40 levels among ADD/ADMCI and other neurological diseases and CU (a, b). Aβ1-42 levels were significantly lower in ADD/ADMCI than in CU ( represent sporadic ADD and ADMCI, cyan circles
represent sporadic ADD and ADMCI, cyan circles  represent familial AD).
represent familial AD).
DISCUSSION
Mass spectrometry is a device that is capable of detecting the masses of various substances, as well as accurate peptide sequence lengths. Due to advances in the operability and analytical devices for extremely small amounts, this method has been clinically applied to the measurement of various substances in recent years. MS/MS is the combination of two mass analyzers in one mass spectrometric instrument. The combination with a highly selective separation method by LC allows for more accurate and reliable quantification for clinical application. Therefore, the LC-MS/MS technique has been used to measure Aβ peptides [7–9, 26–29]. This method eliminates time-consuming immune assays with a large number of steps and multiple antibodies or immunoprecipitation steps for pre-clinical studies. This approach also allows for performance of a single assay to measure several different Aβ species simultaneously in one sample with selectivity, specificity, and in a high-throughput format, while still achieving the high sensitivity required for low level endogenous Aβ peptides (such as CSF Aβ1-43), plasma Aβ species, and modified tau products.
Comparative assays using the same samples between the LC-MS/MS assay and commonly used ELISA systems for Aβ1-40, Aβ1-42, and Aβ1-42 + Aβ1-43 confirmed a strong correlation (
We previously proposed that Aβ1-38/1-42 and Aβ1-40/1-43 were distinct product/precursor pairs and reflected
Comparisons between 66 cases of early-onset AD and 25 of late-onset AD by ELISA showed that CSF Aβ1-43 significantly varied with age in AD. However, Aβ1-43 levels were not superior to Aβ1-42 for diagnostic accuracy [15]. CSF Aβ1-43, but not Aβ1-42, differentiated between patients with amnestic MCI who progressed to AD during a 2-year follow-up [16]. Goda and Kobayashi evaluated dog CSF Aβ1-38, Aβ1-40, Aβ1-42, and Aβ1-43 using peptide adsorption-controlled LC-MS/MS and showed that the Aβ assay was reproducible [17]. Aβ quantitation by LC-MS/MS showed satisfactory intra- and inter-day reproducibility as well as freeze-thaw and room temperature stability [17, 18]. The findings of a recent large study by Janelidze et al. using three different immunoassays revealed that the CSF Aβ42/Aβ40 and Aβ42/38 ratios were significantly better than CSF Aβ42 alone for the detection of brain amyloid deposition in prodromal AD and differentiating AD dementia from non-AD dementias [20]. Widely used ELISA for the quantitation of Aβ1-42/total tau, Aβ1-42/Aβ1-40, Aβ1-42/Aβ1-38, and Aβ1-42 showed the greatest accuracy for identifying amyloid-positive subjects. Aβ1-40 and Aβ1-38 showed significantly lower discriminative powers [21]. The first report of ELISA assays for Aβ1-38, Aβ1-40, and Aβ1-42 showed only decreased levels of Aβ1-42 compared with Aβ1-38 and Aβ1-40 in CSF from AD [22]. CSF Aβ1-42/Aβ1-40 and Aβ1-42/Aβ1-43 were positively correlated with in gait variability and ADL in mild ADD [23]. A UPLC-MS-MS assay of CSF Aβ1-42 showed equivalent diagnostic utility to those of commonly used AlzBio3 ELISA systems [26]. Oe et al. reported that the validated methods had limits of quantitation of 44 fmol/mL for Aβ1-42 and 92 fmol/mL for Aβ1-40 in CSF. A strong correlation was observed between LC/MS/MS and ELISA for Aβ1-42 (r2 = 0.915), and those correlations were weak for Aβ1-40 (r2 = 0.644) [27]. The accuracy and precision of LC-MS/MS will contribute to the clinical diagnosis and monitoring of therapeutic interventions [27].
The present results suggest that the quantitative LC-MS/MS assay for CSF Aβ species is as reliable as specific ELISA for the clinical evaluation of CSF biomarkers for AD. Since the present study was not a large scale or prospective control study, further large-scale studies are necessary. However, the reliable and convenient LC-MS/MS multiple assay of Aβ species is equivalent to specific multiple ELISA assays. Therefore, the LC-MS/MS multiple assay of Aβ species may be applicable and contribute to kinetic [7, 8] and drug monitoring [44] studies on Aβ species. The establishment of a multiple Aβ and tau species assay by LC-MS/MS is warranted.
Footnotes
ACKNOWLEDGMENTS
We thank Yasuhito Wakasaya for his research assistance. This study was supported by the Amyloidosis Research Committee Surveys and Research on Special Diseases, the Longevity Science Committee of the Ministry of Health and Welfare of Japan; Scientific Research (C) (18K07385 MS, 19K07989 T.K.) from the Ministry of Education, Science, and Culture of Japan, the Hirosaki University Institutional Research Grant, and the Center of Innovation Science and Technology-based Radical Innovation and Entrepreneurship Program from the Japan Science and Technology Agency. This study was approved by the Ethics Committee of Hirosaki University (2017-112) and Geriatrics Research Institute and Hospital (2019-78). All participants provided written informed consent.
