Abstract
Background/Objective:
The aim of this study was to determine if plasma concentrations of 5 surrogate markers of Alzheimer’s disease (AD) pathology and neuroinflammation are associated with disease status in African Americans.
Methods:
We evaluated 321 African Americans (159 AD, 162 controls) from the Florida Consortium for African-American Alzheimer’s Disease Studies (FCA3DS). Five plasma proteins reflecting AD neuropathology or inflammation (Aβ42, tau, IL6, IL10, TNF
Results:
Plasma tau levels were higher in AD when adjusted for biological and technical covariates.
Conclusion:
We identified effects of demographic and genetic variants on five potential plasma biomarkers in African Americans. Plasma inflammatory biomarkers and Aβ42 may reflect correlated pathologies and elevated plasma tau may be a biomarker of AD in this population.
INTRODUCTION
Alzheimer’s disease (AD) is a growing epidemic that disproportionately affects African Americans [1]. The prevalence of AD is estimated to be about twice as high in African Americans as in individuals of European ancestry. The United States (US) census projections estimate that by 2050, 42% of elderly adults will be members of minority groups [1]. Thus, the toll of AD is expected to rise considerably in the African-American population, underscoring the importance of discovering effective biomarkers and therapies that are relevant to this population.
Despite this rising prevalence, African Americans are underrepresented in AD research, including biomarker studies and clinical trials [2, 3]. A recent evaluation of major federally-funded biomarker studies in the US revealed that more than half of these studies did not achieve adequate recruitment of African Americans to reach the representation expected from the US census [3]. Due to this underrepresentation, the clinical utility and characteristics of biomarkers, which are primarily established in participants of European ancestry, are unclear for African Americans.
Emerging studies evaluating neuroimaging or cerebrospinal fluid (CSF) protein levels suggest that there may be population specific differences for these AD biomarkers between African Americans and individuals of European ancestry [4]. However, plasma biomarker studies in African-American AD participants are scarce, even though affordable and minimally invasive biomarkers are heavily investigated in individuals of European ancestry [5–7]. Furthermore, most plasma AD biomarker studies of African Americans are conducted in cognitively unimpaired or non-demented populations [8–10]. Although such studies are important to evaluate the effects of these biomarkers on cognition, investigations on the characteristics and diagnostic potential of plasma biomarkers in African-American AD study participants are sorely lacking.
In this study, we sought to fill this knowledge gap by investigating plasma protein levels in 321 African-Americans AD cases and controls from the Florida Consortium for African-American Alzheimer’s Disease Studies (FCA3DS) [11, 12]. We measured plasma levels of five proteins reflecting either core AD neuropathology (amyloid-β 42 [Aβ42] and tau) or inflammation (interleukin 6 [IL6], interleukin 10 [IL10], and tumor necrosis factor alpha [TNF
MATERIALS AND METHODS
Participants
A total of 321 self-reported African-American individuals (159 AD cases and 162 controls) were recruited at the Mayo Clinic, Jacksonville, Florida for this study (Table 1). All cognitively normal controls and AD cases included in this study were Mayo Clinic patients who consented to participate in AD research as part of the FCA3DS [11, 12], and were diagnosed by a neurologist as possible or probable AD according to the NINCDS-ADRDA criteria [15], or as controls if they were cognitively normal elderly participants with a Clinical Dementia Rating scale score of 0 at their last examination. Informed consent and approval by the Mayo Clinic Institutional Review Board was obtained.
Participant demographics and descriptive statistics of the plasma protein measurements
Data are shown for all samples from both batches combined, stratified by diagnosis. Age refers to age of participant at plasma draw. aUnpaired
Single molecule arrays by quanterix
Plasma was stored at –80°C until the protein quantifications. Plasma proteins were quantified using single molecule array (Simoa) assays on the Simoa HD1-Analyzer (Quanterix, Lexington, MA), with a single freeze-thaw cycle between the multiplex Simoa Human Cytokine 3-Plex A assay (IL6, IL10, and TNF
Genotyping
We extracted DNA from blood using the FLEX STAR system (AutoGen, MA, USA) and the FlexiGene DNA Kit (QIAGEN, Hilden, Germany) according to manufacturer’s instructions. We utilized TaqMan genotyping assays to obtain
Statistical analysis
Mean, standard deviation, and coefficient of variation (CoV) were calculated for every duplicate plasma protein measurement. Measurements with >20% CoV were excluded from the analysis. Statistical analyses for the five plasma proteins were performed on the log2-transformed mean of duplicate measurements, as these values were normally distributed (Supplementary Figure 1).
Separate multivariable linear regression models were used to examine independent predictors of our dependent variables, the plasma AD biomarkers. As aforementioned, the log2-transformed mean of duplicate plasma biomarkers measurements were used. All analyses included age of the study participant at blood draw (age), number of years of plasma storage (age of plasma), sex, plate, and batch variables. The primary analytic model (Model 1) included these variables, as well as diagnosis,
In the main analyses, batch 1 (AD only) and batch 2 (AD and controls) were analyzed jointly, while appropriately adjusting for potential batch effects (Table 2). Secondary analyses stratified by batch and by diagnosis were also conducted to determine whether the findings were driven by either of these strata (Supplementary Tables 2, 3).
Association of plasma protein levels with diagnosis, age, sex, and
Plasma protein levels (log 2) of all samples were analyzed using a multivariable linear regression analysis accounting for technical variables (plate, batch and years of plasma storage),
We also evaluated the relationship between each pair of biomarkers using multivariable linear regression where log2 transformed plasma levels of each biomarker were tested as the dependent variable in models which assessed all other biomarkers individually as independent variables. These analyses were adjusted for
RESULTS
A total of 23 plasma samples were excluded from the corresponding protein analyses due to quantification failure; 20 for IL6, 2 for IL10, and 1 for TNF
Controls were significantly older than AD cases both in the combined analysis (Table 1) and within measurement batch 2 (Supplementary Table 1). AD participants from batch 2 were also significantly older than AD cases in batch 1 (data not shown). There were no differences in the distribution of sex between the diagnostic groups in batch 2 or in the combined analysis (Table 1 and Supplementary Table 1). As expected [17],
Age was significant for three of the five tested plasma proteins in these African-American samples. In the combined analysis, higher plasma levels of Aβ42, tau, and TNF
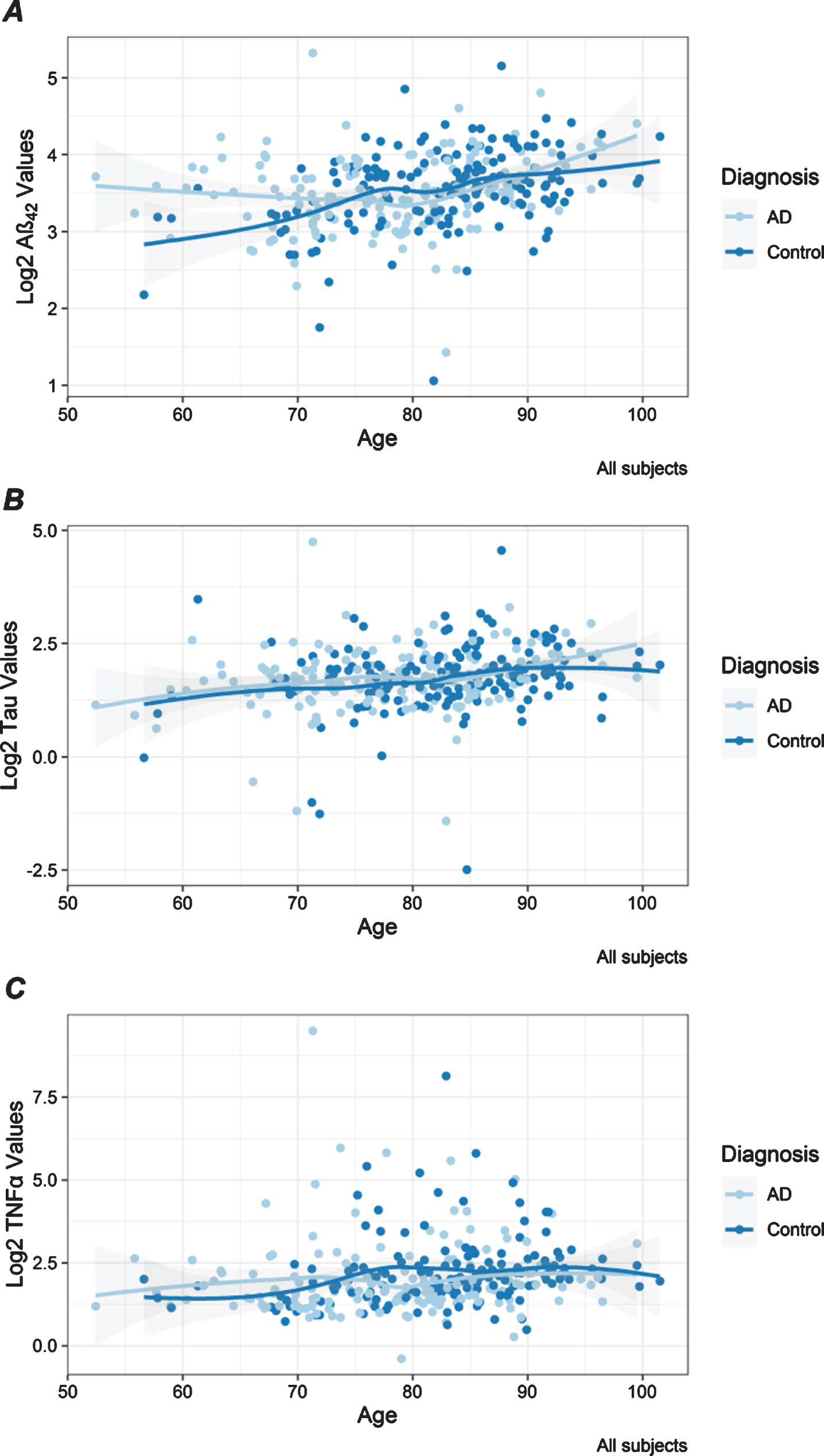
Distribution of plasma protein levels by age. Scatter plots are shown for Aβ42 (A), tau (B), and TNF
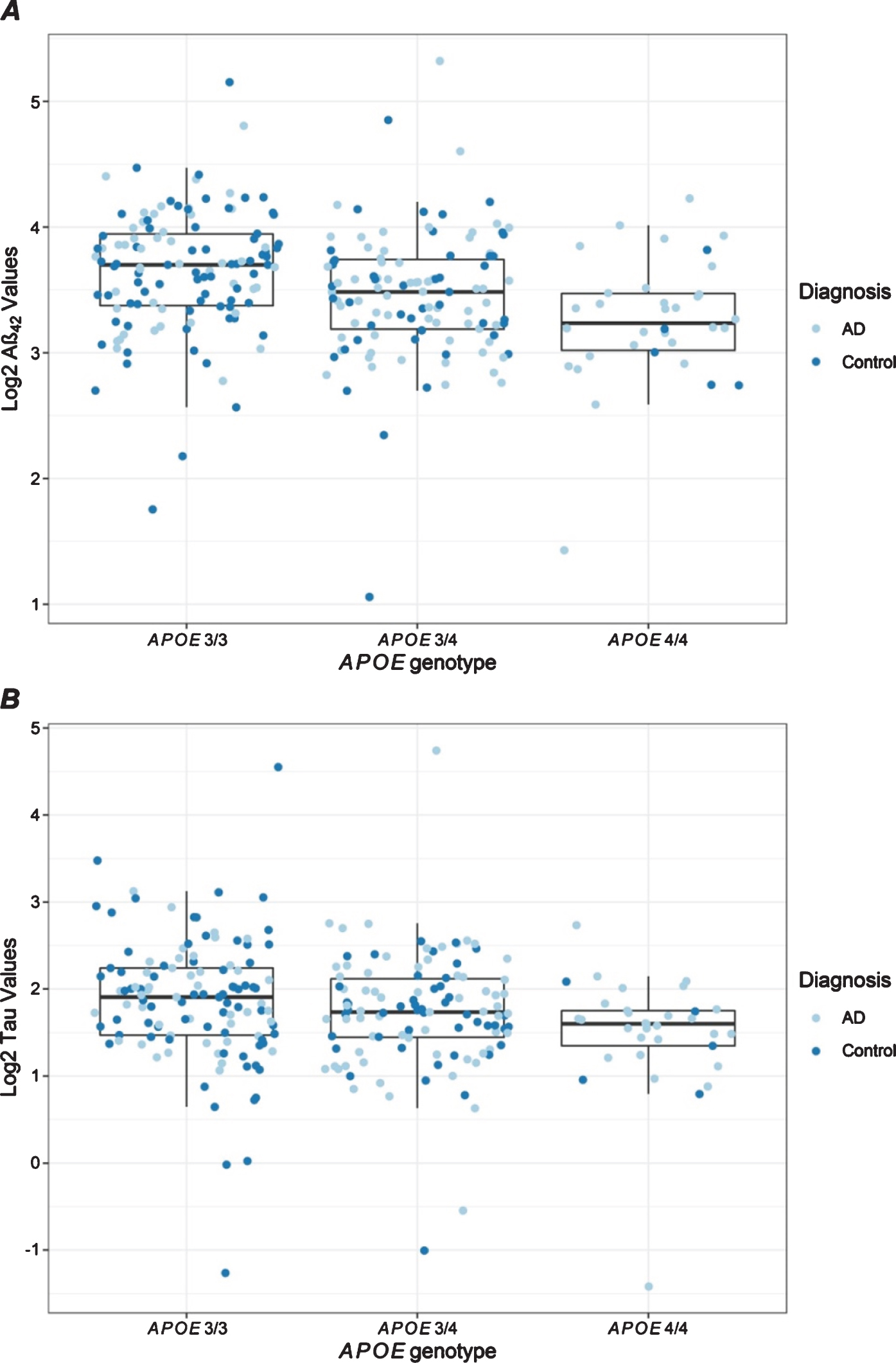
Distribution of plasma protein levels by
Female sex was associated with lower plasma IL10 levels in the combined analysis (Table 2, Supplementary Figure 4). Although the other two inflammatory biomarkers, IL6 (Supplementary Figure 5) and TNF
The only biomarker that showed significant association with diagnosis in the combined analysis was tau, which was higher in AD (Table 2, Supplementary Table 2). We note that AD cases had slightly lower, though insignificant, mean tau levels compared to controls (Table 1), when no covariate adjustments were made. However, in our study, controls were older and had lower
IL6 levels were significantly higher in AD cases than in controls in batch 2 (Supplementary Table 2), but only reached significance when
To evaluate the relationship of these plasma bio-markers, pairwise analyses were performed adjusting for biological and technical covariates. All observed protein-protein associations had positive regression coefficients (Table 3). Strong associations were observed between plasma Aβ42 and tau levels, whether Aβ42 or tau were analyzed as the dependent variable. Significant positive associations were also observed amongst the inflammatory biomarkers IL6, IL10, and TNF
Pairwise associations of plasma protein levels
Plasma protein levels (log 2) of all samples were analyzed for pairwise associations using a multivariable linear regression analysis. All analyses were adjusted for technical variables,
DISCUSSION
Our study sought to evaluate the diagnostic potential of five potential plasma biomarkers, namely Aβ42, total tau, IL6, IL10, and TNF
We found significantly higher levels of plasma total tau in African-American AD cases compared to elderly, cognitively unimpaired controls, after adjusting for biological and technical variables. AD cases had slightly lower, though insignificant, mean tau levels compared to controls, when no covariate adjustments are made. However, in our study, controls were older and had lower
Previous studies conducted in European [19], European-American [20], or Taiwanese [21] populations also detected significantly higher levels of plasma tau levels in participants with clinically diagnosed AD compared with elderly controls. More recently, a comprehensive meta-analysis of biomarker studies evaluated plasma or serum data from 271 AD participants and 394 controls [7]. This meta-analysis identified significantly higher total tau levels in AD participants, consistent with our findings. Interestingly, of the peripheral biomarkers meta-analyzed in that study (YKL-40, HFABP, Aβ40, Aβ42, MCP-1, NSE, and tau), total tau was the only one significantly associated with AD. In our study, we did not identify significant associations with AD for any of the other biomarkers assessed, namely Aβ42 and the inflammatory proteins, IL6, IL10, and TNF
To our knowledge, there are no studies in African Americans that evaluated cytokines as potential AD diagnostic biomarkers. Neuroinflammation is considered an integral part of AD pathophysiology [28], prompting investigators to look for inflammatory signatures in body fluids as potential disease biomarkers [13, 14]. A recent meta-analysis of peripheral cytokines and chemokines [14] evaluated 36 (
Among the biological variables tested as potential modifiers of plasma biomarker levels (age, sex,
We also observed a significant association of
An inverse relationship was also observed between
Furthermore, an association of female sex with lower plasma IL10 levels was observed in our study. IL10 is an anti-inflammatory cytokine, lower levels of which are thought to result in an enhanced inflammatory status [14]. IL10 was previously shown to be lower in African-American women who were disproportionately affected by metabolic conditions [39]. The implications of these findings, specifically for AD risk, and whether this is an indication of sex-specific differences in the general inflammatory status in this population remain to be established.
Underrepresentation of African Americans in biomarker studies poses a major barrier to the discovery of population-specific risk factors and translation of such findings to therapeutic targets, biomarkers and management approaches relevant to this population [2, 3] who is at greater risk of AD [1, 40]. Given the existing healthcare disparities for African Americans reflected in their delayed presentation for evaluation and lower likelihood to receive dementia treatment [41], the implications of this barrier are far-reaching. AD diagnostic and predictive potential of plasma biomarkers are unknown for African Americans, despite the fact that some plasma biomarkers have already been assessed in as many as >1,000 [42]–10,000 [43] participants that are primarily of European ancestry.
It is probable that utilizing multiple biomarkers jointly, rather than individually, will provide more robust outcomes and may also provide insight about disease pathophysiology [6], as exemplified by utilization of CSF Aβ42, total tau, and phosphorylated tau as combined biomarkers of AD pathophysiology both in the clinic and in research. In this context, it is also useful to characterize correlations or associations between different biomarkers. We identified positive associations between Aβ42 and tau with the inflammatory cytokines; as well as between TNF
In summary, our study provides detailed assessment of five plasma biomarkers in an African-American case-control series consisting of 159 AD participants and 162 controls. Our plasma biomarker study has a sizable representation of African-American AD cases, in comparison to prior studies of this population, which either lacked or had limited numbers of AD participants of less than 50 [26]. Our evaluation of both primary AD neuropathology biomarkers (Aβ and tau), and those of inflammation (IL6, IL10, TNF
Correlative plasma biomarker results generate hypotheses on concerted biological processes, which must be tested in future studies that assess complementary neuroimaging and CSF biomarkers in this and other populations.Despite these strengths, our study also has several weaknesses. Given that it is a cross-sectional study, the findings are prone to inter-individual variability of biomarkers that may not be related with AD, or to the other variables we adjusted for in this study. For this reason, longitudinal measurements of biomarkers and assessment of their prospective intra-individual variability is advocated as a more reliable biomarker [20, 43]. Another limitation of this study is that information regarding family history was not available for 80/159 (50%) of the controls. Among the 79 controls for whom family history was recorded, 40 had family history of dementia and 39 did not. This proportion of positive family history is higher than that estimated in a study by Tang et al. in which the frequency of family history of dementia in African Americans was found to be 15% [46]. This potential ascertainment bias in our study may have led to an underestimate of the effect size of the association of plasma tau protein in African American AD versus cognitively unimpaired controls. Although the association of plasma tau with diagnosis would not reach significance after correction for the five plasma proteins tested, the nominal associations we identified in the context of this understudied population are consistent with previously published work in European, European American, and Taiwanese cohorts and thus highly plausible. Despite having a sizable representation of AD participants and elderly controls, the sample size of our African-American case-control series is still a fraction of that collectively assessed in plasma biomarker meta-analyses of European ancestry cohorts [42, 43]. Although our study provides a thorough characterization of five plasma biomarkers in this understudied population, the field needs studies with even larger African-American series for replication of our results. Evaluation of other plasma protein biomarkers, such as ptau and Aβ40, is also needed; however, at the time when this study was conducted, the ptau assay had not been validated yet in plasma, and the amount of plasma available was insufficient for additional measurements. The findings from our study including effect sizes, which were previously lacking in the literature for this population, should help guide design of such future studies. Finally, future plasma biomarker studies of African Americans and other understudied minority populations should include markers of disease severity such as detailed neuropsychological and neuroimaging measures. Studies which can control for other factors that can influence plasma protein levels such as information on presence of infection or inflammation at the time of blood collection [6], blood liver enzyme and creatinine levels, are needed for all populations investigated for AD biomarkers.
This cross-sectional biomarker study on 321 FCA3DS AD cases and elderly, cognitively unimpaired controls provides a previously lacking detailed evaluation of five plasma proteins representing core AD pathology and inflammation in this understudied population. The findings of this study support further investigations of plasma tau as a potential AD biomarker and should help guide future studies of these biomarkers in African Americans.
AVAILABILITY OF DATA AND MATERIALS
Data may be made available after a reasonable and well-justified request to the principal investigator of FCA3DS, Dr. Nilufer Ertekin-Taner. Data cannot, however, be made freely available to the public, due to privacy regulations and informed consent.
Footnotes
ACKNOWLEDGMENTS
We thank the participants and their families for their participation in research. Without them this work would not have been possible. We are also thankful to the Mayo Clinic Memory Disorders Center Coordinators including Michelle Fudge, Rita Fletcher, Francine Parfitt, Kelly Smith, and Sylvia Grant. We thank Mayo Clinic Office of Health Disparities in Research for their support.
This work was supported by the National Institute on Aging [RF AG051504, U01 AG046139, R01 AG061796 to NET; P30 AG062677 to JL and NGR]; Florida Health Ed and Ethel Moore Alzheimer’s Disease grants [5AZ03 and 7AZ17 to NET; 7AZ07 to MC].
