Abstract
Anosmia, stroke, paralysis, cranial nerve deficits, encephalopathy, delirium, meningitis, and seizures are some of the neurological complications in patients with coronavirus disease-19 (COVID-19) which is caused by acute respiratory syndrome coronavirus 2 (SARS-Cov2). There remains a challenge to determine the extent to which neurological abnormalities in COVID-19 are caused by SARS-Cov2 itself, the exaggerated cytokine response it triggers, and/or the resulting hypercoagulapathy and formation of blood clots in blood vessels throughout the body and the brain. In this article, we review the reports that address neurological manifestations in patients with COVID-19 who may present with acute neurological symptoms (e.g., stroke), even without typical respiratory symptoms such as fever, cough, or shortness of breath. Next, we discuss the different neurobiological processes and mechanisms that may underlie the link between SARS-Cov2 and COVID-19 in the brain, cranial nerves, peripheral nerves, and muscles. Finally, we propose a basic “NeuroCovid” classification scheme that integrates these concepts and highlights some of the short-term challenges for the practice of neurology today and the long-term sequalae of COVID-19 such as depression, OCD, insomnia, cognitive decline, accelerated aging, Parkinson’s disease, or Alzheimer’s disease in the future. In doing so, we intend to provide a basis from which to build on future hypotheses and investigations regarding SARS-Cov2 and the nervous system.
Keywords
INTRODUCTION
Severe acute respiratory syndrome coronavirus 2 (SARS-Cov2) is a novel betacoronavirus that causes a variety of symptoms in patients known as coronavirus disease (COVID-19) [1]. There is a growing set of observations that suggest these symptoms include a wide range of neurological manifestations [2 –5]. At the beginning of the current pandemic, the treatment of patients with COVID-19 focused on the management of fever, cough, shortness of breath, and respiratory failure. However, it is increasingly evident that SARS-Cov2 can contribute to a number of neurological issues including anosmia, seizures, stroke, confusion, encephalopathy, and total paralysis [6, 7]. Up to 20% of COVID-19 patients who require intensive care unit (ICU) admission due to their neurological issues, and COVID-19 patients in ICU who have neurological deficits, are at a higher risk of mortality [8, 9]. Patients who do leave ICU and recover from their respiratory symptoms are potentially at higher risk for long-term residual neuropsychiatric and neurocognitive conditions including depression, obsessive compulsive disorder, psychosis, Parkinson’s disease, and Alzheimer’s disease [10, 11].
In 2002, there was an epidemic of a coronavirus with severe acute respiratory syndrome (called SARS-Cov, here referred to as SARS-Cov1) which also caused a range of neurological conditions including encephalopathy, seizures, stroke, cranial nerve palsies, peripheral neuropathy, and myopathy [12]. It had a mortality rate of approximately 10% [13] that in part limited the spread of the disease [12]. In 2012, another coronavirus spread in the Middle East, called Middle East Respiratory Virus (MERS) [13]. This too resulted in clinical syndromes that involved multiple organs, including the brain, nerves, and muscles [14]. SARS-Cov2, possessing a high homology to SARS-Cov1 and MERS, appears to have the capacity to injure the central and peripheral nervous systems through direct and indirect ways [4 , 15]. Given that COVID-19 has created a worldwide health and economic crisis, its neurological implications have become the focus of intense clinical research.
In this review, we will summarize the published reports regarding COVID-19 patients with various neurological symptoms with a focus on our current understanding for pathophysiology of how SARS-Cov2 impacts the central and peripheral nervous systems. We will also address issues on how patients with Alzheimer’s disease or other neurological conditions are impacted by the current pandemic. We also discuss how non-infected individuals can make themselves more resilient for the short-term and long-term impact of SARS-Cov2, in case they become infected with this virus in the future. Finally, we will highlight the importance of identifying patients who have neurological issues without typical COVID-19 symptoms, in order to reduce the risk of virus spreading in in-patient and/or out-patient neurology units.
NEUROLOGICAL SYMPTOMS IN PATIENTS WITH COVID-19
Studies about anosmia or ageusia
An MRI case study from a patient with COVID-19 with acute onset of anosmia in Iran reported normal nasal mucosa (no congestion) and normal volume of olfactory bulbs bilaterally [16]. Dysfunction of smell and taste have been widely reported in patients with COVID-19 in European communities [17 –19] (Table 1). Lechien and colleagues found that among 417 patients with mild to moderate COVID-19, 85.6% reported having olfactory dysfunction and 88.0% had gustatory dysfunction [19]. Among 59 patients hospitalized with COVID-19 in a hospital in Italy, 33.9% reported having smell or taste impairment and 18.6% reported having both [20]. Among 202 patients with mild COVID-19 symptoms in another hospital in Italy, 64.4% had impaired sense of smell or taste [18]. However, among 214 patients hospitalized in China (Wuhan), with severe or non-severe COVID-19 symptoms, only 5.1% and 5.6% had smell or taste impairment; these impairments were more prevalent among patients with mild COVID-19 than those with severe COVID-19 (6-7% versus 3.0%) [6].
Studies on anosmia or ageusia
sQOD-NS, Short version of Questionnaires of Olfactory Disorders Negative Statements; NHANES, National Health and Nutrition Examination Survey; ARTIQ, Acute Respiratory Tract Infection Questionnaire; SNOT-22, Sino-nasal Outcome 22.
Studies about cerebrovascular disease
The formation of small or large blood clots, both in the brain and in multiple other organs, have been reported in a significant number of patients with COVID-19 (Table 2). In the study by Li et al., 13 of the 221 COVID-19 patients had imaging-confirmed evidence of cerebrovascular disease [21]. The majority of patients had ischemic infarcts in both small and large arterial vessels. They also noted that one patient had cerebral venous thrombosis, as confirmed by a CT venogram, and one patient had intracranial hemorrhage, confirmed by a head CT. In the study of 214 COVID-19 patients by Mao et al., five patients had ischemic strokes and one patient had intracranial hemorrhage [6]. Most of the patients with cerebrovascular disease (and other neurological deficits) in this study had severe COVID-19 symptoms. However, two of the six patients presented to hospital with signs of stroke, but did not initially have respiratory symptoms suggestive of COVID-19 [6].
Studies on cerebrovascular disease
CTA, computed angiography; CTP, computed tomographic perfusion; ICA, internal carotid artery; MCA, middle cerebral artery; PCA, posterior cerebral artery; ACA, anterior cerebral artery
Another study by Oxley et al. also highlighted that young healthy individuals can present with large strokes, with or without apparent COVID-19 symptoms such as cough or fever [22]. They report a 39-year-old man with no COVID-19 symptoms who developed hemiplegia, ataxia, and reduced level of consciousness. His brain MRI showed evidence of right posterior cerebral artery occlusion. He underwent a cerebral angiography and clot retrieval procedures followed by antiplatelet therapy with aspirin. He was transferred to ICU and remained in critical condition. Another patient, a 37-year-old man presented with right hemiplegia and reduced level of consciousness, also without any COVID-19 symptoms. His MRI showed evidence of a left middle cerebral artery occlusion. He received a clot retrieval procedure, was then started on apixaban, improved rapidly, and was discharged to home.
Seizure or encephalopathy
There are several case reports of COVID-19 patients presenting to hospitals with fever, stiff neck, confusion, changes in mental status, and/or seizures (Table 3). For example, Filtov et al. reported the case of a 74-year-old woman who presented to hospital with fever, cough, and confusion [23]. Her head CT showed evidence of an old large stroke in her left temporal lobes, but no new strokes. Moriguchi et al. reported the case of a 24-year-old man who presented to ER with headache, stiff neck, seizure, and loss of consciousness [24]. The patient’s brain MRI showed hyperintense signal in the right mesial temporal lobe and hippocampus as well as the wall of the inferior horn of the right ventricle. This patient’s nasal swab and cerebrospinal fluid (CSF) were positive for SARS-Cov2, as measured by PCR. He received supportive treatment in the ICU and later had significant improvements. It was noted that he had developed retrograde amnesia and did not remember events related to the onset of COVID-19 pandemic (personal communication with Dr. Moriguchi). Poyiadji et al. reported the case of a 58-year-old woman who presented to hospital in Detroit with cough, fever, and confusion. Her MRI was consistent with a diagnosis of acute necrotizing (hemorrhagic) encephalitis [25]. Duong and colleagues reported the case of a 41-year-old woman with COVID-19 who presented to hospital with headache, fever, disorientation, seizure, and hallucinations [26]. Her head CT and CSF were negative. Yin et al. described yet another patient, a 64-year-old man, with COVID-19 who presented to hospital with acute onset of lethargy, irritability, dissociated speech, and confusion. His head CT and CSF were negative too [27]. Mao et al. reported that among their 214 patients hospitalized with severe COVID-19, 14.8% had impaired consciousness and one patient experienced seizures[6].
Studies on seizures and/or encephalopathy
AMS, altered mental status; LOC, loss of consciousness; PD, Parkinson’s disease; COPD, chronic obstructive pulmonary disease; CSF, cerebrospinal fluid; PCR, polymerase chain reaction (for SARS-Cov2).
In a study of ICU patients with severe COVID-19 and Acute Respiratory Distress Syndrome (ARDS) (N = 58), 65% had confusion and 69% had agitation [28]. Among the 13 patients in this study who underwent MRI because of unexplained encephalopathic features, 62% (8/13) had leptomeningeal enhancement, 23% (3/13) had ischemic stroke, and all 11 patients who underwent perfusion imaging had bilateral frontotemporal hypoperfusion. Interestingly, two of their patients who had diffusion weighted MRIs were found to have experienced acute small strokes which were not expected based on their neurological exams. One patient had an asymptomatic acute ischemic stroke in his cerebellum [28].
Studies about peripheral nervous system
SARS-Cov2, similar to SARS-Cov1, can cause serious injury to cranial nerves, peripheral nerves, and muscles (Table 4). Facial weakness, difficulty breathing, being unable to stand or walk, or having difficulty weaning off respiratory ventilators may be in part due to Guillain-Barre syndrome (GBS) brought on by COVID-19. Gutierrez-Ortiz et al. report treating two patients who presented with eye movement abnormalities consistent with a diagnosis of Miller-Fisher Syndrome and polyneuritis cranialis. Their symptoms included anosmia, ageusia, areflexia, ataxia, internuclear ophthalmoplegia, and fascicular oculomotor palsy [29]. The blood tests for one of these patients was positive for GD1b. These patients promptly received IVIg and had a rapid recovery. Another patient with COVID-19, reported by Toscano et al., presented with severe facial weakness as well as sensory ataxia [30]. His brain MRI showed evidence of enhancement in the facial nerve. He too responded well to treatment with IVIg and improved within a week [30]. The four other patients in this report all had more typical GBS and variable degrees of typical COVID-19 symptoms. Overall, it appears that COVID-19 patients who presented with various degrees of cranial nerve and limb weakness and who were promptly diagnosed with GBS had a favorable prognosis.
Studies on peripheral nervous system
IVIg, intravenous immunoglobulin; EMG/NCS Study, Electromyogram and Nerve Conduction Studies.
In the observational study by Mao et al., COVID-19 patients in the ICU had multiple etiologies for lethargy, muscle atrophy, and weakness [6]. However, their muscular symptoms were above and beyond what would be expected in a critical care setting. They reported that 19.3% of their patients with severe COVID-19 had evidence of marked muscle injury [6]. Similar findings have been reported for COVID-19 patients in other ICU settings [31].
PATHOPHYSIOLOGY OF NEUROLOGICAL ABNORMALITIES IN COVID-19
The binding of SARS-Cov2 to angiotensin converting enzyme 2 (ACE2) is a critical step in the pathophysiology of clinical manifestations in patients with COVID-19 [32] (Fig. 1). The function of ACE2 in normal human physiology is to regulate blood pressure via inhibition of the angiotensin-renin-aldosterone pathways [33]. ACE2 facilitates conversion of angiotensin II to angiotensin(1-7) [33]. Higher levels of angiotensin II are associated with vasoconstriction, kidney failure, heart disease, apoptosis, and oxidative processes that accelerate aging and promote brain degeneration [33]. Several cardiovascular medications including statins and angiotensin receptor blockers exert their effects in part through ACE2 [34]. This enzyme protein in the cell membranes of multiple organs also happens to serve as the receptor for SARS-Cov2 [32]. ACE2 deficiency lessens the impact of SARS-Cov2 infection [32]. As such, ACE2 can actually serve as a target for therapeutic agents against SARS-Cov2[35].

SARS-Cov2: Cellular mechanism of action. SARS-Cov2 binds ACE2 to enter epithelial cells of blood vessels and cells in multiple other organs. 1) Once internalized, it can cause damage to mitochondria and lysosomes which in turn may result in increased reactive oxygen species (ROS), protein misfolding, protein aggregation, and cell death. 2) By binding to ACE2, SARS-Cov2 also downregulates and inhibits the metabolic conversion of Angiotensin 2 (AT2) to AT(1-7). The resulting higher levels of AT2 is associated with pro-inflammatory markers, vasoconstriction, vascular permeability and edema, vascular injury to cells in the lungs, brain, heart, and kidneys as well as processes involved in pro-apoptosis and aging.
ACE2 has a wide distribution in multiple organs including the nose, lungs, kidneys, liver, blood vessels, immune system, and the brain [36, 37]. After binding ACE2 in respiratory epithelial cells and then epithelial cells in blood vessels, SARS-Cov2 triggers the formation of a cytokine storm, with marked elevation in levels of interleukin-1, interleukin-6, and tumor necrosis factor [38, 39]. High levels of these cytokines increase vascular permeability, edema, and widespread inflammation with consequent damage in multiple organs [40]. The cytokine storm also triggers hypercoagulation cascades to cause small and large blood clots [39]. Combined hyperactivation of inflammatory markers, vascular injury, and coagulation factors contributes to ARDS, kidney failure, liver injury, heart failure, myocardial infarction, as well as multiple neurological conditions, which will be discussed below. A direct entry of SARS-Cov2 into the brain has been described for other coronaviruses and may play a role in SARS-Cov2’s possible contribution to demyelination or neurodegeneration [2, 12].
Patients who suffer from COVID-19 complications, including hospitalization and being isolated from their family members, experience a great deal of psychological stress [41]. Being isolated is similar to the stress of physical immobilization, which is associated with a sharp increase in cortisol and steroid levels [41]. Extreme stress levels also heighten the level of cytokines and contribute to medical complications in COVID-19 patients who are already experiencing organ damage due to a cytokine storm [42]. Sustained exposure to stress and high level of cytokines in these patients may contribute to a variety of neuropsychiatric and neurocognitive symptoms in the long term [43] (discussed below).
Anosmia and ageusia
Some of the anosmia in patients with COVID-19 can potentially be due to nonspecific upper respiratory infection symptoms. However, the recent literature suggests that the traditional nasal symptoms seen in influenza or rhinovirus are often absent in patients with COVID-19 [44]. In fact, patients who have COVID-19 do not develop significant nasal congestion or rhinorrhea [44, 45]. Moreover, patients with anosmia sometimes report having the false perception of altered or loss of taste [46]. The close relationship between olfactory and gustatory functions may account for the reason they misinterpret having loss of taste despite having experienced only loss of smell, as it can happen with some upper respiratory infection [46]. However, several studies have demonstrated that taste dysfunction in patients with COVID-19 appears to be more common than olfactory dysfunction and that 10.2–22.5% of COVID-19 patients have taste dysfunction without olfactory dysfunction [17 , 48]. Thus, ageusia is a specific symptom in patients with COVID-19 that is different from flu-like upper respiratory congestion or misinterpretation of taste perception due to loss of olfactory function [47].
Impairment in olfactory and gustatory functions in patients with COVID-19 are likely due to the SARS-Cov2 infection of the epithelial cells of nasal and oral mucosa [17, 47] (Fig. 2). High densities of ACE2 are present in olfactory epithelial cells, nasopharynx, and oral mucosa [49, 50]. By binding to the ACE2 in nasal and oral mucosa, SARS-Cov2 can inhibit the function of sensory receptor cells that mediate olfaction and gustation.
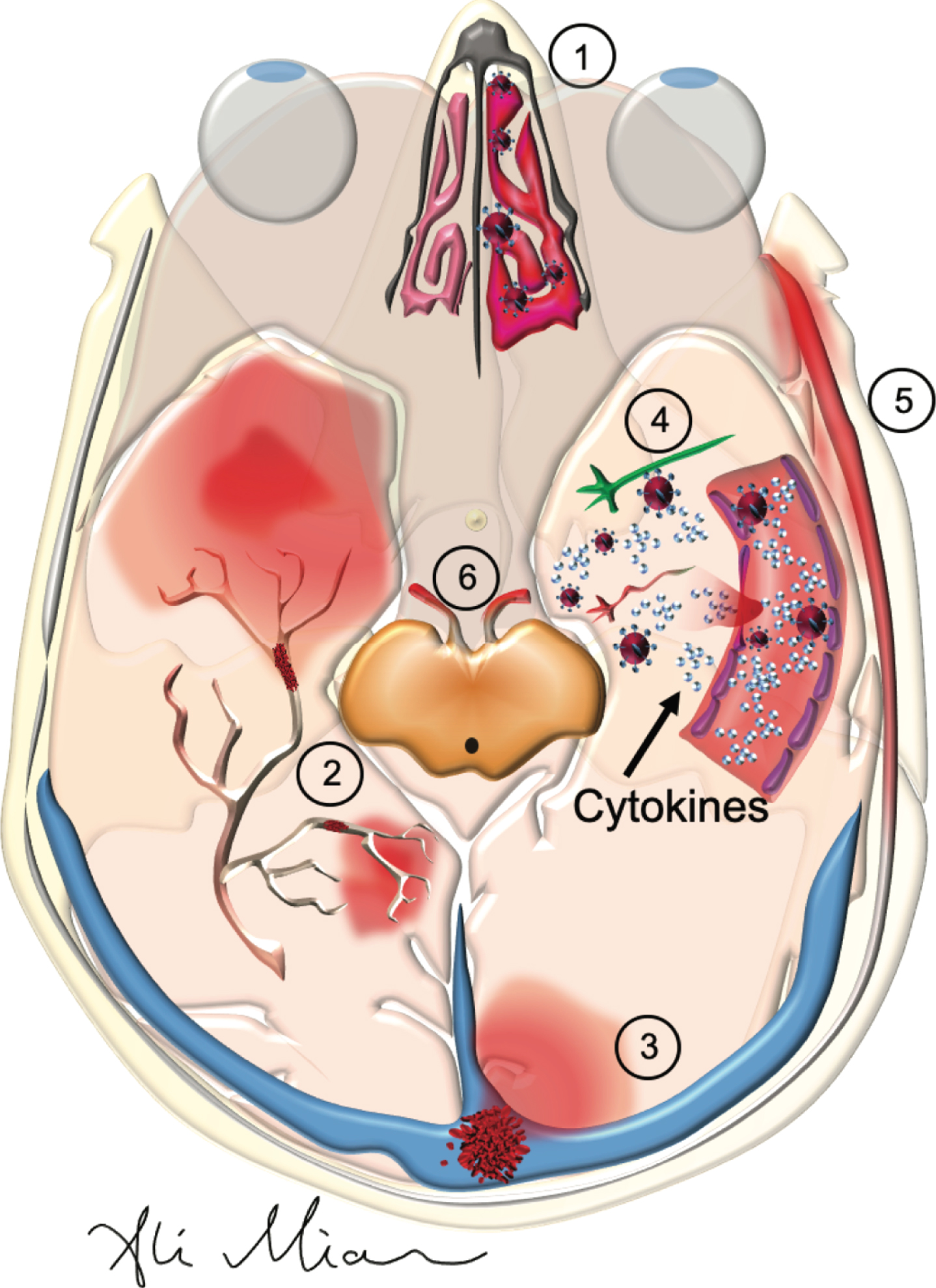
SARS-Cov2: Pathophysiology of action in the nose, cranial nerves and the brain. SARS-Cov2 can cause a variety of neurological symptoms in patients with COVID-19 such as anosmia, strokes, encephalopathy, meningitis, and cranial nerve injury. 1) By binding and inhibiting nasal (and gustatory - not shown) epithelial cells, it reduces the sense of smell and taste. 2) By activating the cytokines and hypercoagulation pathways in the blood, it results in the formation of small and large vessel occlusion in cerebral arteries. 3) Formation of blood clots in the cerebral veins can results in cerebral venous thrombosis. 4) High levels of cytokines in the cerebral vessels can damage the blood-brain barrier, and once infiltrate the brain, damage neurons and glia which results in seizures and/or encephalopathy. 5) Damage to arteries in meninges can result in meningitis. 6) Formation of auto-antibodies, known as molecular mimicry, may lead to damage to cranial nerves (see Fig. 3).
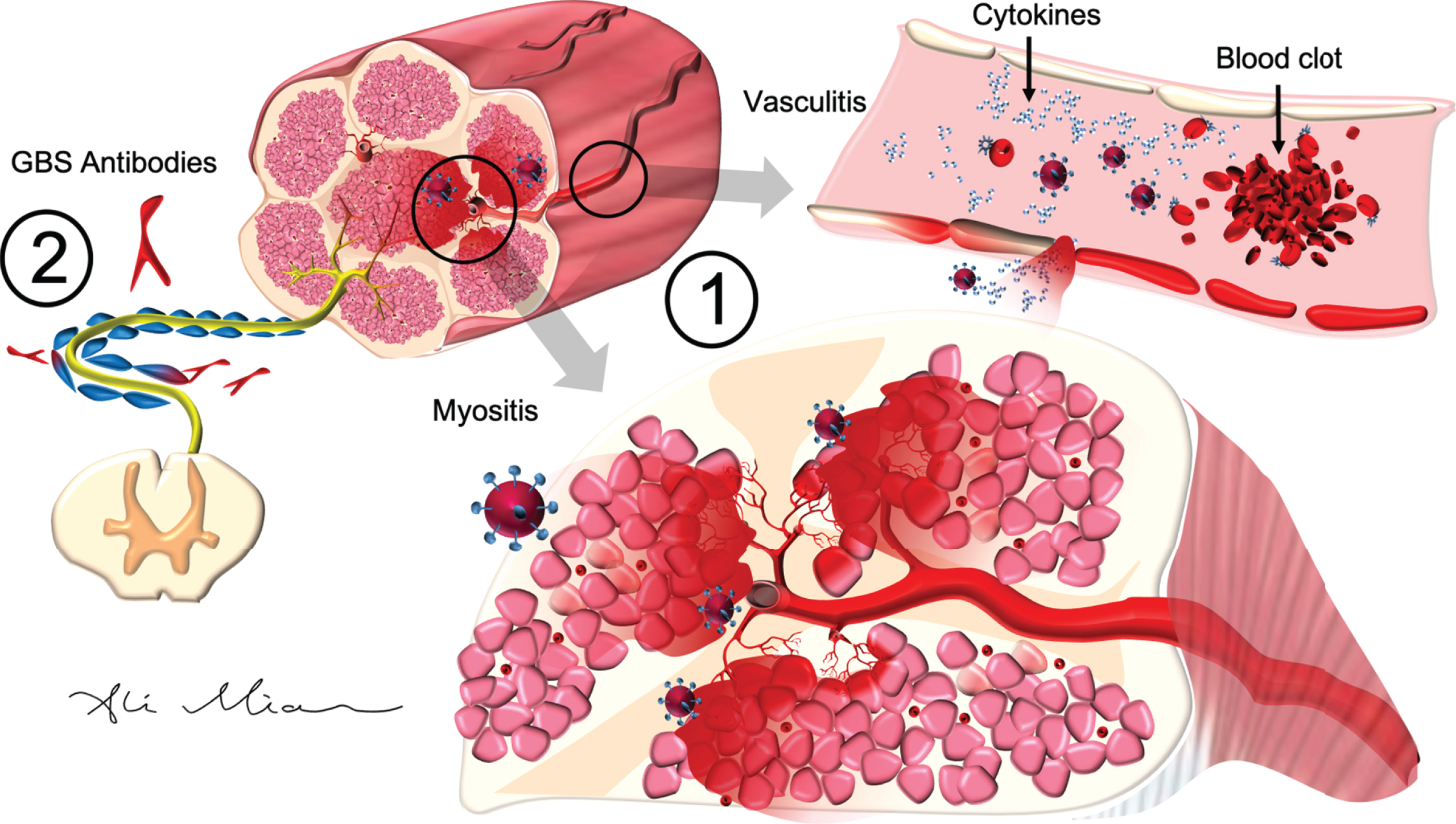
SARS-Cov2: Pathophysiology of action in peripheral nerves and muscle. 1) SARS-Cov2 activation of cytokines causes inflammatory injury to epithelial cells in the blood vessels (vasculitis) and muscles cells (myositis). In cardiac arteries and muscles (not shown), cytokine storm, triggered by SARS-Cov2, can result in hypercoagulopathy and formation of blood clots (myocardial infarction) or endocarditis. 2) SARS-Cov2 can trigger the formation of autoantibodies (such as GD1a) which react with antigens on axons and myelin cells to cause Guillain-Barre syndrome (GBS).
A retrograde transport from nasal mucosa to the brain has been described for SARS-Cov1 [51, 52]. As such, it is conceivable that SARS-Cov2 can also cross the cribriform plate and bind to neurons in the olfactory bulb, thereby reducing the perception of olfaction through a central mechanism [53]. Similarly, a retrograde transport of this virus from the gustatory receptor cells in the tongue to damage the neurons in the nucleus solitarius in medulla can potentially account for ageusia sensation in patients with COVID-19 [54]. A postmortem immunohistochemical study detected the presence of the SARS-Cov1 by electron microscopy in some neurons, but not in those in the olfactory bulb [55]. In one case report of anosmia in a patient with COVID-19, the brain MRI showed normal volume in olfactory bulb [16]. Further neuropathological and imaging studies with a with special focus on brain stem and olfactory bulb can help to determine if SARS-Cov2 is in fact capable of reaching the brain structure related to olfaction and gustatory function [52]. For now, given that smell and taste dysfunction are common among patients with COVID-19 and do often improve within weeks, the possibility for a central etiology remains unlikely.
The marked difference in the reported percentage of COVID-19 patients who suffer from anosmia or ageusia from Asia and Europe needs further evaluation [6, 17]. The one study from hospitalized patients with COVID-19 in China (Wuhan) found that only 5% of patients suffered from smell and taste impairments [6]. This is in contrast to the frequency of 33.9% to 88.0% for reported olfactory and/or gustatory dysfunction in three European studies of COVID-19 patients [9 , 18]. A marked variation between populations of patients with COVID-19 between Asia and Europe with regards to smell and taste, once confirmed with further studies and results from objective measures, can be explained by ACE2 polymorphism. Preliminary studies suggest that East Asian populations may have different allele frequencies of ACE2 [56] and that some ACE2 variants may have reduced capacity to bind SARS-Cov1 [57]. As such, symptoms and outcomes of patients with COVID-19 may vary greatly depending on which variant to ACE2 they have in different tissues. This possibility has strong clinical implications and needs to be further investigated.
Cerebrovascular disease
COVID-19 patients who experience cerebrovascular disease often have hypertension [58]. Strokes, as well as other neurological deficits, are also more common in patients with COVID-19 who suffer from diabetes [12 , 59]. Increasing evidence shows that higher body mass index is associated with more severe COVID-19, that mortality of COVID-19 is higher in obese patients, and that obesity by itself can be considered a risk factor for developing COVID-19 [60, 61]. As such, hypertension, pre-diabetes, and obesity are major risk factors for cardiovascular and cerebrovascular events in patients with COVID-19 [62]. Given these findings, we need to remind patients with these vascular conditions to be extra cautious not to get infected by SARS-Cov2.
There is now compelling evidence that COVID-19 patients are at increased risk for excessive level of hypercoagulopathy [40]. Cerebral angiography and venography findings suggest that blood clots for ischemic stroke in COVID-19 patients can happen in both cerebral arteries and cerebral veins [21, 63] (Fig. 2). Blood clots in these patients also lead to myocardial infarctions, pulmonary embolisms, and renal failure [40, 64]. Sometimes an ischemic stroke can happen to a patient at the same time as a deep venous thrombosis [59]. The hypercoagulable state in these patients has in turn been attributed to higher levels of inflammatory markers such as C-Reactive Protein, ferritin, interleukin-1, interleukin-6, TNF-alpha, and d-dimer [65]. More research is needed to determine how exactly binding of SARS-Cov2 to ACE2 triggers the cytokine storm and the secondary hypercoagulation—factors that contribute to a major morbidity and mortality in COVID-19 patients.
Though most of the reported strokes in patients with COVID-19 have been due to ischemic events, a handful of cases with intracranial hemorrhage have been reported as well [12 , 66]. The exact mechanism for how SARS-Cov2 causes intracranial hemorrhage remains poorly understood. One possibility is that by binding and downregulating ACE2, SARS-Cov2 slows the conversion of angiotensin II to angiotensin(I-7) [33]. Higher levels of angiotensin II are associated with vasoconstriction and peripheral vascular resistance (Fig. 1). The vasoconstriction associated with suppressing ACE2 can contribute to rupture of blood vessels in the brain. Another possibility relates to possible polymorphism of ACE and increased risk of intracranial hemorrhage, especially in Asian population [67].
Seizures and encephalopathy
Patients in ICU settings with multiple medical conditions, and on a long list of medications, can develop memory loss, slow processing speed, delirium, or even coma [68]. As such, a decline in mentation among patients with severe COVID-19 may not necessarily represent a direct brain injury brought on by the SARS-Cov2. However, it appears that these patients experience encephalopathy and delirium at a greater rate than would be otherwise expected in ICU settings [6, 69].
Analysis of the limited COVID-19 literature favors that SARS-Cov2 triggers an immune-mediated encephalopathy more than a direct viral encephalopathy [12]. SARS-Cov2 activation of cytokines such as interleukin-1, interleukin-16, and TNF-alpha causes injury to the blood-brain barrier (BBB) [12]. With increasing damage to BBB, cytokines penetrate the brain parenchyma, especially in temporal lobes where BBB is weaker [70, 71]. Strong inflammatory response and entry of blood material into the brain results in seizures and encephalopathy [12]. A direct viral infection of neurons to cause seizures is plausible as well. With the permeation of blood content into the brain, viral particles can enter and damage neurons directly (Fig. 2). Neurons do have ACE2, and postmortem pathological studies have detected SARS-Cov1 (by electron microscopy) in some neurons of patients with SARS-ARDS [72]. However, with the exception of two case reports with positive PCR in CSF for SARS-Cov2 in two patients with meningitis/encephalitis[24, 73], all other studies in which CSF was tested failed to find SARS-Cov2 by PCR [23 , 74] (Table 3). This failure to find traces of SARS-Cov2 in most reported studies of patients with COVID-19 encephalopathy could be due to a lack of optimal testing techniques in CSF for this virus, or more likely, a lessor role of large viral load in CSF/brain [75].
Patients with COVID-19 who present with acute headache, nuchal rigidity, seizure, and confusion may be experiencing meningitis [24]. The meninges are rich in blood vessels and have and also contain high levels of ACE2 [37]. Damage to these blood vessels and the inflammation in the meninges can in turn result in symptoms of meningitis [12].
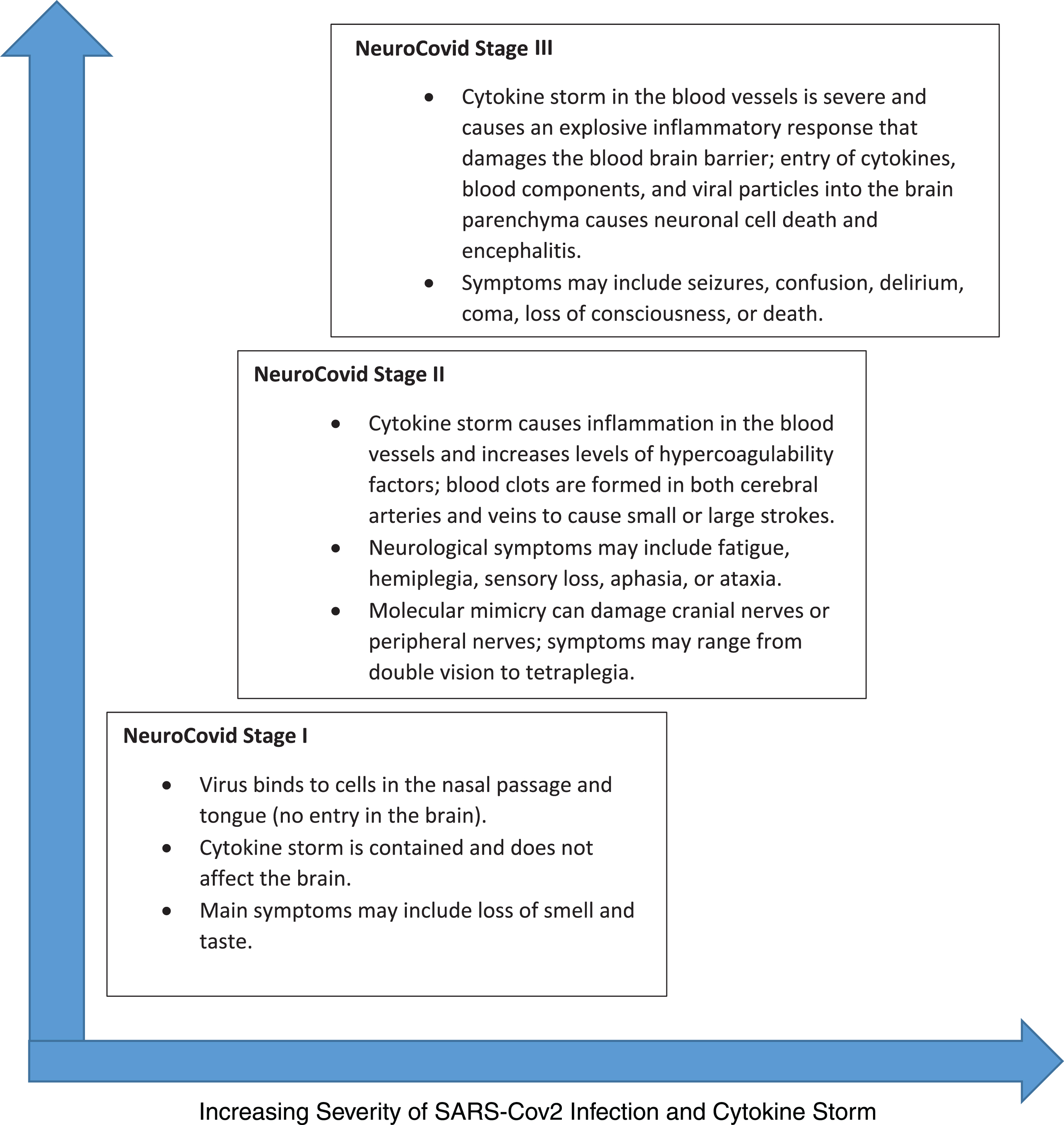
NeuroCovid Stage I, II, and III. SARS-Cov2’s neurological manifestation can be grouped into three stages. In NeuroCovid Stage I, the virus damage is limited to epithelial cells of nose and mouth. In NeuroCovid Stage II, patients may experience blood clots in their brain or have auto-antibodies that damage their peripheral nerves and muscles. In NeuroCovid Stage III, the cytokine storm damages the blood-brain barrier and patients may develop seizures, coma, or encephalopathy.
Cranial nerves, peripheral nerves, and muscle
Guillain-Barre syndrome (GBS), associated with ascending paralysis and some degree of sensory loss or cranial nerve injury, commonly occurs after certain bacterial or viral infections. GBS has been reported in patients who developed the SARS-Cov1 infection in 2002–2003 [12]. Now, several reports have outlined typical GBS axonal neuropathy, demyelination, or cranial nerve palsy in patients with COVID-19 [6 , 77] (Table 4). GBS is believed to occur as a result of “molecular mimicry,” which refers to cross-reactivity of natural immunoglobulins—formed in response to a bacterial or viral antigen—with specific proteins on the myelin, axon, or neuro-muscular junction [78]. The cytokines activated by SARS-Cov2 can also trigger vasculitis in and around nerves and muscles, with or without a molecular mimicry (Fig. 3) [78]. A direct invasion by the virus to the peripheral nerves can potentially occur, but the lack of any SARS-Cov2 finding in the CSF to date makes this unlikely [29, 30]. For now, the pattern of clinical presentations and rapid response to IVIg favors an immune-mediated etiology for peripheral and cranial neuropathy in patients with COVID-19.
Muscle injury and high levels of creatine kinase in COVID-19 patients in ICU can be attributed to critical care neuropathy and/or myopathy [79]. Sedating and paralyzing medications given to patients for ICU protocols can also make these patients weak and unable to stand or walk. However, the time course of severe muscle weakness in COVID-19 patients suggests that a vasculitis or myositis etiologies may be involved (Fig. 3) [79]. With regards to cardiac muscle, there is evidence that both myocarditis due to SARS-Cov2 as well as myocardial infarction due to cytokine storm, hypercoagulability, and ischemia can happen at the same time [64]. A neuroinvasion of brain stem neurons by SARS-Cov2 causing muscle weakness in ICU patients with ARDS is also under a great deal of investigation [52 , 80–82].
NEUROCOVID STAGING, FROM ANOSMIA TO ENCEPHALOPATHY
Based on the analysis of the potential pathophysiological mechanisms involved in neurological manifestations of SARS-Cov2, we propose a conceptual framework of “NeuroCovid Staging” that can serve as a basis for future discussions and investigations.
LONG-TERM COMPLICATION: NEUROCOGNITIVE AND PSYCHIATRIC CONDITIONS
Neurons contain significant levels of ACE2 and thus SARS-Cov2 can penetrate them and disrupt their cellular mechanism for energy production (mitochondria) and protein folding [83]. SARS-Cov2, as well as other corona viruses, can remain inside some neurons without being acutely toxic [11]. The abnormal misfolding and aggregation of proteins in patients who survive and recover from their acute SARS-Cov2 infection can thus theoretically lead to brain degeneration decades later[83]. Since some of the effects of SARS-Cov2 can manifest months or years after infection, it will be necessary to consistently follow-up with patients who have been affected by COVID-19. Keeping accurate registries of COVID-19 patients with neurological deficits may enable us to establish plausible connections with aging-associated and neurodegenerative disorders such as Parkinson’s disease in the future. This possibility has been raised as there has been a link between SARS-Cov1 and a higher risk of developing Parkinson’s disease[84] and multiple sclerosis [85].
We are still in early stages of the current pandemic and the focus of our medical interventions have been on the acute treatment of life-threatening consequences of COVID-19 such as pulmonary embolism, ARDS, myocardial infarctions, encephalitis, renal failure, paralysis, and coma. However, it is quite likely that the cytokine storm and the insults to the brain via small or large strokes, injury to BBB, and high levels of inflammation inside the brain would have long term neuropsychiatric consequences. Thus, the health care systems around the world may see in the coming years a wave of patients who present with depression, post-traumatic stress disorder, anxiety, insomnia, or psychosis as well as cognitive impairment or decline. As was found with SARS-Cov1 and MERS, not all patients with SARS-Cov2 infection who leave the hospital will return to 100% of their baseline emotional and neurocognitive function. A study of neuropsychiatric sequelae of SARS-Cov1 31-50 months after the acute infection found evidence for post-traumatic stress disorder (39%), depression (36.4%), obsessive convulsive disorder (15.6%), and panic disorders (15.6%) [10].
The cytokine storm in COVID-19 can cause a series of small punctate strokes without causing noticeable neurological deficits [28]. When these patients leave the hospital after an acute SARS-Cov2 infection, they may experience poor memory, attention, or slow processing speed. Thus, it would be helpful for these patients to see a neurologist or undergo neurocognitive testing 6-8 months after their hospital discharge if they feel they still have cognitive issues, slowness in processing information, or poor attention. Patients with low scores in certain cognitive domains can consider receiving brain rehabilitation in order to return to their baseline level of cognitive capacity. By doing so, they would reduce their risk for developing a worse case of age-related cognitive decline later in life [86, 87].
One of the most consistent findings in COVID-19 literature is that patients with vascular risk factors such as obesity, hypertension, and diabetes have a more dire outcome as compared to healthy and fit individuals who get infected with SARS-Cov2. As such, a strategy of regular exercise, eating a heart healthy diet, reducing stress, improving sleep, and following other recommendations for reducing risk of heart attacks and strokes now prove more critical than ever before [86, 87]. By becoming a host that is resilient to SARS-Cov2, COVID-19 patients can improve their odds of a faster and more favorablerecovery.
COVID-19 IN PATIENETS WITH ALZHEIMER’S DISEASE AND OTHER NEUROLOGICAL DISORDERS
Patients with Alzheimer’s disease may be at a higher risk of developing COVID-19 [88]. They may not be able to follow recommendations from public health authorities regarding prevention of SARS-Cov2 infection such as hand hygiene, covering mouth and nose when coughing, maintaining physical distance from others, or remaining at home. They may not understand, appreciate, or remember what they need to do. If they have depression, malaise, reduce mobility, and apathy, they may also be unwilling or unable to comply with any rules. Finally, some patients with severe Alzheimer’s disease who have agitation, wandering, or psychosis may refuse to be isolated. Their behavior may also put them at risk for further dementia related decline—especially if they are kept in a hospital environment and away from their family members or familiar surroundings. Prolonged hospitalization would have dire consequences for these patients. As such, caring for patients with Alzheimer’s disease, who are often older and have multiple risk factors for experiencing a poor outcome (or death) if they become infected with SARS-Cov2, poses a major public health challenge for caregivers, health care professionals, and nursing homes [88].
Patients with other neurological disorders are also at risk for multiple complications associated with COVID-19. Those with a previous history of cerebrovascular disease often have a poor outcome if they develop COVID-19 [89]. Patients with neuromuscular disorder such as myasthenia gravis may experience a relapse of their symptoms and there may be even an increase in incidence of this condition during the COVID-19 pandemic [79]. Patients with multiple sclerosis, amyotrophic lateral sclerosis, and respiratory dysfunction are likely to have difficulty surviving ICU hospitalization and those on immunosuppressive medications may decline faster [79, 90]. Neurologists need to be particularly mindful of COVID-19 issues that can directly impact the care of their patients with these disorders.
IMPLICATIONS FOR THE PRACTICE OF NEUROLOGY IN THE FUTURE
Given that cytokine-induced hypercoagulability and formation of blood clots in the lungs, heart, kidney, and brain pose significant morbidity and mortality in COVID-19 patients, treatment with antiplatelet or anticoagulant medications such as aspirin or heparin needs to be studied. The prevention of vascular events can lead to lower rates of pulmonary embolism, heart attacks, kidney failure, and embolic strokes. Clinical trials to test this hypothesis need to begin promptly. There is also a need for clinical trials which document and record the acute onset of neurological symptoms, detailed neurological test results, progression, and long-term recovery of symptoms in patients with COVID-19.
Given that some patients with COVID-19 can present to hospitals or outpatient clinics with neurological symptoms as their only symptom of SARS-Cov2 infections, neurologists therefore need to be mindful of the risk of infection spreading by such patients to staff or other patients in the clinical area [2]. In the future, we may need a pre-visit screening with questionnaires that check for anosmia, ageusia, fever, cough, shortness of breath, or living with family members who have been infected with SARS-Cov2. We may also make it mandatory that we measure the temperature, blood pressure, heart rate, and oxygen saturation in all patients who walk into a neurology practice.
Finally, it remains important to understand that while patients with COVID-19 can present with a wide range of neurological symptoms ranging from anosmia, cranial nerve palsy, weakness, strokes, to seizures or encephalopathy, they may still have other etiologies for their acute or chronic neurological issues. A patient with new onset of unilateral weakness, seizure, or diplopia may still have a non-COVID-19 etiology, even if they are found to have a recent SARS-Cov2 infection. We need to add COVID-19 to the list of differential diagnosis for our patients in a neurology unit and remain mindful that patients need to have a full standard work-up for their evaluation and treatment. Neurologists need to consider ordering blood tests for levels of cytokines, D-dimer, CRP, ferritin, and lymphocytes as well as SARS-Cov2 PCR and/or serology [7].
CONCLUSIONS
Patients with COVID-19 can present with a wide range of neurological manifestations that can be due to the injury to central and peripheral nervous system via a cytokine storm, blood clots, direct damage by SARS-Cov2, and/or molecular mimicry. This review, while presenting what is currently known about this virus and the related clinical neurology, represents only the base of what will eventually become a separate active field of research. Much work remains to determine a fuller understanding of the underlying neurobiology of COVID-19. These include better characterized COVID-19 cohorts with longitudinal follow ups. Standardized evaluations such as quantitative EEG, fluid biomarkers, cognitive evaluations, and multi-modal neuroimaging can also lend insight to possible long-term neurological sequalae in COVID-19 such as depression, memory loss, mild cognitive impairment, or Alzheimer’s disease.
Footnotes
ACKNOWLEDGMENTS
Dr. Mian receives education funding from the Washington University Carol B. and Jerome T. Loeb Teaching Fellows program. Dr. Meysami is supported by McLoughlin Cognitive Health Gift Fund and the Pituitary Injury Foundation. Dr. Raji is supported in his research by grants from the WUSTL NIH KL2 Grant (KL2 TR000450 – ICTS Multidisciplinary Clinical Research Career Development Program), the Radiological Society of North America Research Scholar Grant and the Foundation of the American Society of Neuroradiology Boerger Research Fund for Alzheimer’s Disease and Neurocognitive Disorders. We thank Ms. Melissa Hussey for her assistance in preparation of this manuscript.
