Abstract
Background:
The progressive aging of the population will dramatically increase the burden of dementia related to Alzheimer’s disease (AD) and other neurodegenerative disorders in the future. Because of the absence of drugs that can modify the neuropathological substrate of AD, research is focusing on the application of preemptive and disease-modifying strategies in the pre-symptomatic period of the disease. In this perspective, the identification of people with cognitive frailty (CF), i.e., those individuals with higher risk of developing dementia, on solid pathophysiological bases and with clear operational clinical criteria is of paramount importance.
Objective/Methods:
This hypothesis paper reviews the current definitions of CF, presents and discusses some of their limitations, and proposes a framework for updating and improving the conceptual and operational definition of the CF construct.
Results:
The potential for reversibility of CF should be supported by the assessment of amyloid, tau, and neuronal damage biomarkers, especially in younger patients. Physical and cognitive components of frailty should be considered as separate entities, instead of part of a single macro-phenotype. CF should not be limited to the geriatric population, because trajectories of amyloid accumulation are supposed to start earlier than 65 years in AD. Operational criteria are needed to standardize assessment of CF.
Conclusion:
Based on the limitations of current CF definitions, we propose a revised one according to a multidimensional subtyping. This new definition might help stratifying CF patients for future trials to explore new lifestyle interventions or disease-modifying pharmacological strategies for AD and dementia.
Keywords
INTRODUCTION
Life expectancy has dramatically increased in the last decades thanks to improved nutrition, hygiene, living conditions, and widely available medical care. The proportion of elderly people, and in particular the oldest old, i.e., individuals older than 80 years, is growing fastest than any other age group and is expected to triple between 2015 and 2050 [1]. Alongside, the increased longevity carries several implications for the quality of life. Aging has been conceptualized as the progressive and overall physiological decline of an organism to produce adaptive responses to stressors due to accumulation of pathologies in different tissues [2, 3]. Older individuals therefore become more susceptible to adverse outcomes [4] and more vulnerable to mild endogenous and/or exogenous stressors, after which returning to baseline condition may be difficult [5]. As a result, aging represents the major risk factor for highly prevalent chronic conditions including cancer and cardiovascular disease [6]. Since aging is the most important and non-modifiable risk factor for cognitive deterioration, the burden of Alzheimer’s disease (AD) and other neurodegenerative disorders is expected to grow dramatically in the future. Reliable forecasts predict approximately 74.7 million dementia cases in the world by 2030, reaching 131.5 million by 2050 [7]. In this scenario, growing attention is being devoted to preventive and health promotion strategies to reverse, or at least improve, the age-related functional decline, reducing the duration of late-life morbidity and disability [8, 9], and increasing successful aging, which is characterized by life satisfaction, freedom from disability, mastery, active engagement with life, independence, social well-being, high cognitive and physical functional capacity [10].
Current pharmacological treatments for AD (i.e., cholinesterase inhibitors and memantine) are symptomatic ones with limited efficacy and considerable side effects, but do not increase life expectancy or slow progression to more severe stages of dementia [11]. Some potentially disease-modifying treatments have been tested to reduce amyloid deposition in AD, but no significant clinical effect has been achieved, and no disease-modifying drugs have been approved yet [12, 13].
According to the National Institute on Aging (NIA) and the Alzheimer’s Association (AA) criteria for preclinical AD diagnosis [14–16], research into future disease-modifying treatments for AD should target both amyloid deposition and tau pathology and recruit patients with early and preclinical disease stages [17]. Pharmacological and non-pharmacological interventions for preclinical stages of AD may target cognitively unimpaired individuals who carry high AD risk on the basis of biomarkers and/or genetic background to influence the disease trajectory [18, 19]. Clinical trials in cognitively unimpaired individuals at risk for AD may document a disease-modifying activity more effectively than those in clinically affected people, even at early disease stage [15, 21]. The window of therapeutic and preventive opportunity for AD may be represented by the very long pre-symptomatic period, and there is concern that promising drugs failed in trials because they were given too late [22].
From this perspective, identifying people with cognitive frailty (CF), i.e., those individuals who carry a higher risk of developing AD-related or other types of dementia on solid pathophysiological bases and with clear operational clinical criteria is of paramount importance. In the community setting, the prevalence of CF was reported to be 1.0–1.8% and to increase in the clinical setting [23]. In the present hypothesis paper, we will review the current definitions of CF, present and discuss some of their limitations, and propose a framework for updating and improving the conceptual and operational definition of the CF construct.
THE CONCEPT OF FRAILTY IN THE ELDERLY: DEFINITIONS AND MODELS
Preventive and health promotion strategies need specific indicators of early functional decline to be effective. In this context, the concept of frailty was developed and reached broad consensus in clinical and research settings [24–28]. First reported in the geriatric medicine literature in the 1950s and 1960s, the term frailty was further developed in the following decades, but its formal operational definition is more recent [29]. We will briefly review the main and most recent definitions of frailty.
The Fried’s phenotype model conceptualized frailty as a geriatric syndrome resulting from cumulative declines across multiple physiological systems and causing vulnerability to adverse outcomes such as falls, disability, institutionalization, and death [29]. The frailty phenotype model involved exclusively the physical domain upon five criteria, i.e., unintentional weight loss, self-reported exhaustion, slowness of gait speed, reduced hand grip strength, and physical activity, with no, 1–2, or ≥3 criteria defining robustness, pre-frailty, and frailty, respectively [29].
The more recent Rockwood’s deficit accumulation model considered multiple domains including medical, functional, and psychosocial aspects, but was also focused on measures of physical performance and the presence of functional deficits [30].
Aware of the limitations of models centered on physical domains only, the Gobbens’ model of frailty was based on a holistic view that included nutrition and cognition in addition to physical features with the elderly positioned on a dynamic continuum between a non-frail and a very frail status, which can be modified through preventive and therapeutic interventions [24, 31].
The biopsychosocial model approach to frailty combines the physical and psychosocial domains, expands the construct of frailty towards social sciences, and includes cognitive, emotional, motivational, and social features. According to this model, the risk of losing social and general resources, activities, or abilities that are important for fulfilling one or more basic psychosocial needs during the life span may increase the risk of developing AD or other neurodegenerative dementias and should be a target for the earliest intervention [32].
According to these models, frailty is a multidimensional construct that can be divided into three major phenotypes, i.e., physical frailty [29], CF [33], and psychosocial frailty [25, 34].
THE CONCEPT OF COGNITIVE FRAILTY: DEFINITIONS AND STATE OF THE ART
The concept of CF has undergone several changes over the years, and we will briefly review its definitions (Table 1).
Cognitive frailty: current definitions and operational criteria
AD, Alzheimer’s disease; CDR, Clinical Dementia Rating scale; CF, cognitive frailty; IAGG, International Association of Gerontology and Geriatrics; IANA, International Academy on Nutrition and Aging; MCI, mild cognitive impairment; SCD, subjective cognitive decline.
CF was first used in a 2006 review article to indicate a state of cognitive vulnerability in patients with mild cognitive impairment (MCI) exposed to vascular risk factors with an increased likelihood of progression to overt dementia [35].
The International Academy on Nutrition and Aging (IANA) and the International Association of Gerontology and Geriatrics (IAGG) reached a consensus in 2013 on the first definition of CF, which was conceptualized as a condition characterized by a heterogeneous clinical manifestation characterized by the simultaneous presence of physical frailty and MCI (i.e., Clinical Dementia Rating, CDR score = 0.5) without a concurrent diagnosis of AD-related or other types of dementia [36]. According to the IANA-IAGG definition, CF represents a state of reduced cognitive reserve, different from physiological brain aging and potentially reversible, occurring at an intermediate stage between age-related cognitive changes and neurodegenerative diseases [37–40]. A psychological component of CF may concur to increase the vulnerability of the individual to stressors [36]. Despite physical frailty and poor cognition appear to be associated in the IANA-IAGG definition, the causal link between these two entities is unclear.
Two subtypes of CF were later proposed by Ruan and coworkers, i.e., potentially reversible CF and reversible CF, with the former being represented by MCI (CDR score = 0.5) and the latter by subjective cognitive decline (SCD) and/or positive fluid and imaging biomarkers of amyloid accumulation and neurodegeneration, both resulting from physical or pre-physical frailty [33]. According to the SCD Initiative Working Group, SCD is characterized by subjective experience of worsening in cognition, typically in the memory domain, in the absence of objective cognitive deficits at formal neuropsychological evaluation (CDR score = 0) [41]. The potential reversibility of CF suggests it may be an important target for the prevention of dependency and other negative outcomes in older age.
There is increasing awareness that prevention of dementia is important, given the absence of disease-modifying treatments [12, 13]. CF is a topic of great interest in this context, but the current scenario is still far from being clear. Moreover, most studies consider CF as a geriatric condition, but the view of its potential reversibility [33] underscores the importance of neuropsychological rehabilitation. The aim of this hypothesis article is to address core issues in CF research and suggest a roadmap towards a CF redefinition that includes the geriatric, as well as the neurological and neuropsychological perspectives.
THE POTENTIAL FOR REVERSIBILITY OF COGNITIVE FRAILTY
After the proposal of Ruan and collaborators [33], research focused on the potential reversibility of CF [42]. MCI was originally defined as a transitional state between normal aging and dementia, mainly AD-related one, and characterized by mild cognitive disturbances without functional impairment [43]. According to this definition, trajectories of progression might include AD-related dementia for amnestic MCI [14, 44], and other neurodegenerative dementias (e.g., dementia with Lewy bodies, frontotemporal dementia) for non-amnestic MCI [45–47], with a higher yearly risk (5–10%) than the general population (1–2%) of conversion to dementia [48, 49].
Studies that focused on the neuropsychological predictors of MCI conversion to dementia showed that MCI can remain clinically stable for decades [50] or revert to normal cognition at follow-up, especially when a single cognitive domain is involved [51, 52]. However, MCI cases who revert to normal cognition have been demonstrated to carry a high risk of progressing to dementia later on [49]. Based on these reports, the data on MCI reversion rate to normal appear to be inconclusive, because of several bias factors, which include differences in MCI neuropsychological criteria, transient medical comorbidities, or neuropsychiatric symptoms that might lead to false positive results, length of follow-up [53, 54].
NEUROPATHOLOGICAL BIOMARKERS OF ALZHEIMER’S DISEASE AND RELATED CONDITIONS
Neuropathological changes associated with AD begin years or even decades before AD symptoms and signs appear and can be documented in the pre-symptomatic AD stage [55]. According to this view, amnestic MCI coupled with positive biomarkers of amyloid or amyloid and tau neuropathology represents the earliest symptomatic stages of AD, since progressive neuronal loss leading to irreversible cognitive impairment may have already occurred [15, 56]. There are, however, a number of open questions pertaining to the association between AD pathological changes and the onset of AD symptoms. A frailty accumulation index was reported to modulate the relationship between AD neuropathologic changes and dementia in a cohort of adults older than 59 years without known dementia at baseline, suggesting that individuals with even a low level of AD-related pathology might be at risk for dementia if they have high amounts of frailty [57].
The recent NIA-AA Research Framework introduced the A/T/N classification system, a biomarker classification scheme that groups different biomarkers (imaging and biofluids) by the pathologic process each measure [58, 59], and changed the view of AD pathology from a clinical-neuropathological to a clinical-biomarker entity. A/T/N classification divides AD biomarkers into three categories, according to the nature of the measured pathological process: A stands for amyloid biomarkers, T stands for tau biomarkers, and N stands for neurodegeneration or neuronal injury biomarkers [58]. Each type of biomarkers has different kind of representatives (Table 2) and is rated as positive or negative based on their presence in a single subject [59]. On the basis of the A/T/N classification scheme, the NIAA-AA Research Framework proposed AD staging across a continuum, and identified eight different biomarker profiles and three biomarker categories: individuals with normal AD biomarkers, individuals in the AD continuum (i.e., AD pathological change); non–AD pathological changes (i.e., with normal A biomarker but abnormal T and/or N; Table 3) [59]. The combination of cognitive status according to neuropsychological testing and the biomarker profiles according to the NIAA-AA Research Framework could lead to a better understanding of the complexity of AD and non-AD conditions, by focusing on the neuropathological features in parallel to the clinical/cognitive ones [59].
Biomarker classes according to the A/T/N classification system [59]
Aβ42, amyloid-β protein 42 peptide; CSF, cerebrospinal fluid; FDG, fluorodeoxyglucose.
The NIA-AA Research Framework proposed a six-level clinical staging that corresponds to the classical clinical syndromes (i.e., cognitively unimpaired, SCD or transitional cognitive decline, MCI, mild-to-severe dementia) and should be applicable only to individuals in the AD continuum (Table 4) [59]. Clinical stages do not run in parallel with the neuropathological changes, because AD biomarkers may be positive independently on the severity of clinical staging, e.g., cognitively unimpaired or SCD patients may have positive amyloid biomarkers [59].
Clinical staging and related clinical syndromes in the Alzheimer’s disease continuum [59]
Note that the clinical stages and syndromes do not run in parallel with the neuropathological changes. *The self-experienced persistent decline in cognitive functioning and/or behavioral changes must be unrelated to an acute event. †Reported by the patient or by an informant. CU, cognitively unimpaired; MCI, mild cognitive impairment; NIA, National Institute on Aging; SCD, subjective cognitive decline; TCD, transitional cognitive decline.
A greater number of abnormal AD biomarkers, indicating more severe AD pathology, have been reported to result in a higher risk of short-term cognitive decline [59]. We may hypothesize that a greater amount of AD biomarker changes at ages younger than 70 could characterize CF, although the longitudinal trajectories of AD biomarkers have not been investigated, to date [60]. Indeed, biomarker-based research should not be considered a template for all research into age-related cognitive impairment and dementia [59] and our hypothesis should be further explored in clinical trials.
Pre-frailty and frailty could represent a part of the same functional decline, and an early diagnosis and intervention could be possible considering the physical performances in addition to the cognitive framework of frailty.
COGNITIVE FRAILTY IN THE ERA OF ALZHEIMER’S DISEASE BIOMARKERS
The introduction of AD biomarkers changed the significance of MCI as a diagnostic category and the view that this condition may be reversible. According to the NIA-AA Research Framework [59], MCI patients with positive A biomarkers should be diagnosed as AD pathologic change with MCI, while those with positive A and T biomarkers should be diagnosed as prodromal AD, irrespective of N biomarkers status. Therefore, the term reversibility should be applied to MCI only if modifiable factors, such as polypharmacy, psychiatric conditions (e.g., depression), metabolic deficiencies, sleep disturbances, or sensory deficits could be identified [61]. These considerations imply that identifying MCI with CF and considering MCI as potentially reversible CF, as stipulated by the IANA-IAGG [36] and the Ruan et al. [33] definitions, without considering the underscoring neuropathology may be too simplistic and not reflect the multifaceted nature of this condition.
According to the definition by Ruan and collaborators [33], reversible CF corresponds to SCD, a non-specific condition characterized by self-report of persistent decline in cognitive capacity in comparison with a previously normal status and unrelated to an acute event, without objective impairment detected by standardized neuropsychological tests [41]. SCD may result from multiple causes, such as normal aging, preclinical AD, other psychiatric and neurologic disorders, side effect of drugs and substance use disorders (SUD) [41, 63]. Indeed, neurobehavioral changes should have a clearly defined recent onset, be persistent and not explained by life events [59]. Despite the absence of objective evidence of cognitive impairment, reversibility of SCD might be questionable in the presence of AD biomarkers [64], particularly in younger patients. Patients with SCD and positive A with or without T biomarkers corresponds to preclinical AD pathological changes or preclinical AD [59] that have a high likelihood of converting to overt AD [56, 65–67]. However, since neurodegeneration and neuronal damage may be negative in SCD cases and functional compensation is still possible [41], this population might be the most suitable for interventions aimed at preventing or at least postponing progression to dementia (Fig. 1). Most individuals with SCD appear not to follow the temporal biomarker order proposed by NIA-AA criteria, and a more parsimonious staging approach that does not presume all patients follow a singular invariant expression of the disease has been proposed [68].
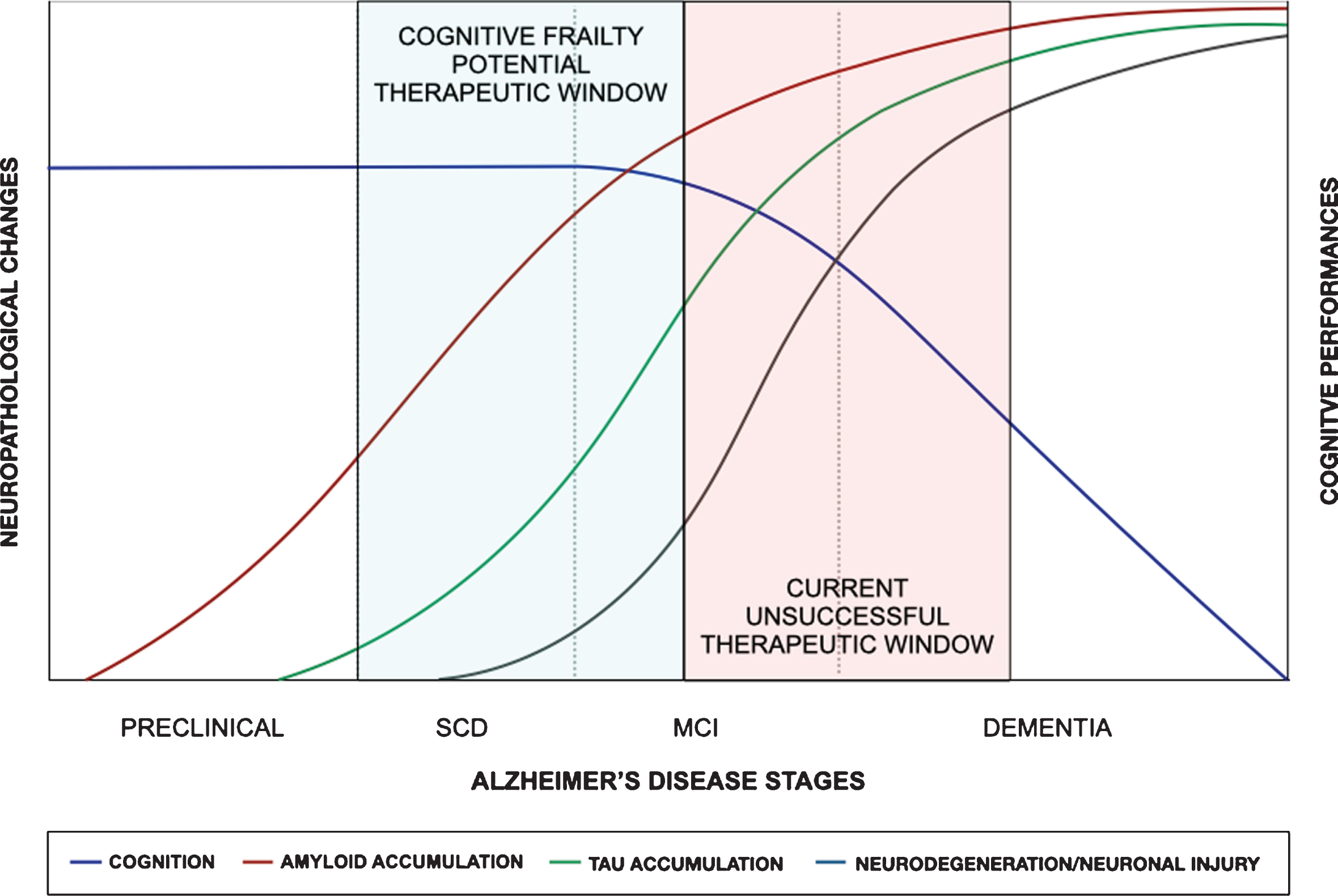
Cognitive frailty and the natural history of Alzheimer’s disease (AD). Here is schematically depicted the natural history of AD with the supposed timing of neuropathological changes in relation to the cognitive decline [59]. The definition of patients with subjective cognitive decline (SCD) and early mild cognitive impairment (MCI) at risk of dementia in the cognitive frailty stage could offer a potential therapeutic window to overcome the limitations of the current unsuccessful therapeutic window of late MCI and early dementia.
Cognitive impairment or decline, however, is not result of AD pathological changes alone, because it is often the result of mixed pathology, including AD and other concurrent degenerative pathology and/or vascular pathology and the scenario appears to be more complex and the topic for future research.
OPERATIONAL CRITERIA FOR COGNITIVE FRAILTY
The absence of robust and widely accepted operational criteria may limit the spread of the concept of CF [69]. At variance with the physical components of CF that were clearly operationalized by the Cardiovascular Health Study, the cognitive features of this construct were not well described, in that they include comorbid dementia, signs or symptoms of cognitive dysfunction, clouding, or delirium [42, 70]. Tools to detect CF were reported in approximately one third of studies and vary from self-reported cognitive-screening questionnaires to screening tests and neuropsychological batteries [71, 72]. Mini-Mental State Examination (MMSE) is the most widely used screening test, despite it has been shown to fail in detecting mild cognitive disturbances [73]; similarly, CDR, although widely adopted, may not be adequate, because it is too simple and not sensitive to mild cognitive deficits. Moreover, since they were developed as screening tools for AD-related dementia, MMSE and CDR are mainly focused on memory, being poorly sensitive to other cognitive domains, in particular to executive functions. To overcome this drawback, some researchers proposed the use of a wide range of neuropsychological tests [74], such as the frontal assessment battery [75], the five words test [76], the trail making test [77], the free and cued selective reminding tests [78], the digit symbol substitution subtest of the Wechsler adult intelligence scale revised [79], and the verbal fluency test [80], but they explore different cognitive domains with variable sensibility, specificity, reliability, and validity and some of them may be sensitive to the age of the patient [71, 81]. Since patients with CF may show mild levels of cognitive impairment, a more in-depth neuropsychological assessment including at least one test for cognitive domain would be more sensitive and could help understand if different CF subtypes can be documented.
An operational definition of SCD that captures both cognitive and functional decline has been recently proposed [68]. The analysis of qualitative and strategic aspects of cognitive performance such as process scores analysis, word-list intrusion errors, retroactive interference, and learning slope in SCD patients may increase the sensitivity and the earlier identification of cognitively normal older adults at risk for decline [82]. These findings suggest that widely accepted operational criteria and an in-depth neuropsychological evaluation may improve the prognostic evaluation of SCD patients.
Indeed, these figures indicate the absence of gold standard operational criteria for detecting CF, thus making the comparison of findings across studies difficult [71, 83]. Referring to common operational criteria for CF would be important to offer psychometrically adequate and widely shared clinical measures.
THE ASSOCIATION BETWEEN COGNITIVE AND PHYSICAL FRAILTY
The possible common etiopathogenesis between cognitive and physical frailty is a key feature on this topic that still remains unresolved [84]. Current definitions of CF stipulate physical pre-frailty or frailty in association with cognitive impairment [33, 36]. CF has been proposed as a potentially reversible clinical entity and represents an important target of secondary intervention in early or asymptomatic stage of dementia to promote healthy aging [85]. Common underlying mechanisms between physical and cognitive impairment include neuropathological changes, cardiovascular elements, nutrition, hormones, and chronic inflammation [36, 87]. A significant correlation between frailty and the global cortical atrophy was reported [88], and a CF model was demonstrated to have significant additional predictive effect on the risk of disability than the physical frailty model only [89]. Several cross-sectional studies demonstrated the interconnection between physical frailty and cognitive performance in people aged >65 years, but data about the possible causal or temporal relationship between these two components are scarce [70], and their dissimilar evolution suggest a more open view when considering their possible relationship [42]. Indeed, the prevalence of frailty according to the Fried’s criteria is on average less than 5% under the age of 70 years [90]. At variance, SCD and MCI frequently affect people younger than 70 years. A recent study reported the prevalence of SCD to be around 10% in adults aged 45–54 years with functional limitations in nearly 60% of those reporting SCD [91]. The different prevalence of physical frailty and SCD across the life span suggests that the concept of CF should be expanded to middle-aged adults with SCD, who could represent the most important population where therapeutic strategies to reduce the risk of conversion to dementia should be tested, as discussed above. This view might increase our understanding of how frailty and its risk factors develop during the earlier stages in life and contribute to the development of public health strategies aimed at preventing frailty and related adverse health outcomes [90]. Whether this approach is cost-effective should be the topic of future studies.
There is increasing evidence that poor gait performance is strongly associated with the incidence of cognitive decline and dementia, especially non-AD dementia that led to the definition of motoric cognitive risk syndrome, defined as presence of both slow gait and subjective cognitive complaints and absence of concurrent dementia or mobility disability [92]. A simple motor test of gait velocity combined with a reliable cognitive test was found to be superior than the CF construct (i.e., physical frailty plus CDR score = 0.5) to detect individuals at risk for dementia [93]. The correlation between lower limb motor function and the risk of dementia further casts doubt on the empirical basis of the CF syndrome [94]. These findings underscore that robust operational criteria for CF are needed, as discussed above. Longitudinal studies have indeed led to inconclusive results, underscoring both the parallelism and the dissonances between physical and CF, impeding firm conclusions on the correlation between the two components [42, 96].
Whether physical frailty and cognitive impairment represent a unique phenotype, as suggested by the CF construct, should be considered different phenotypes of a common underlying mechanism (i.e., vascular diseases, neurodegeneration), or are two separate conditions, co-occurring in older age, are still open questions with no definitive answer according to current knowledge.
Moreover, since subjects with SCD are often younger, they may complain of cognitive symptoms in the absence of physical disturbances. However, studies addressing the association between physical and CF in people aged less than 65 years are still lacking, probably because younger patients may be more unlikely to fulfill the criteria for physical frailty. Therefore, physical and CF should probably be kept separated in subjects <65 years.
COGNITIVE FRAILTY: TOWARDS A MULTIDIMENSIONAL REDEFINITION
Cognitive decline due to neurodegenerative disorders and coexisting vascular changes is expected to become one of the major health issues in the next few years [7, 97]. Because of the limited efficacy of currently available treatments for AD, the identification of potentially modifiable factors to be targeted by preventive and interventional strategies is gaining increasing attention. Among them, CF and associated physical frailty seem to represent a potential promising candidate. The current CF definitions present some conceptual and methodological issues that have been discussed above. Here we offer some proposals for updating and improving the CF construct.
First, the view that CF could be reversible or potentially reversible, either if the cognitive profile of the patient is MCI or SCD, should not rely upon clinical and neuropsychological profile only, but should take into account the presence or absence of AD biomarkers, according to the A/T/N classification system [58, 59] and other neurodegenerative or vascular changes, especially in younger patients, where these changes have a more robust pathological significance. At variance with Ruan’s definition of CF [33], some caution should be paid in considering SCD or MCI patients with positive biomarkers as reversible CF. In such cases, potentially modifiable factors, such as psychiatric comorbidities (e.g., depression or dysthymia), psychosocial (e.g., social isolation), biological (e.g., changes related to aging, metabolic deficits), or pharmacological factors (e.g., side effect of drugs, SUD) could represent the target for interventions aimed to reverse CF [41, 98]. In any case, the clinical construct of CF should be kept separated from the neuropathological changes and biomarkers, which should include the A/T/N framework, the other neurodegenerative and vascular changes. Indeed, the proposed CF construct differs from the SCD/MCI ones, in that operational criteria are available for the latter, but needs to be defined for the former. What’s more, CF may coexist with physical frailty, especially in elderly people.
Second, because of the absence of clear evidence that physical and cognitive components of frailty are correlated, they should better be considered as separate entities, instead of part of a single macro-phenotype [33].
Third, despite studies on physical frailty traditionally focused on patients older than 65 years, CF should probably not be considered a condition limited to geriatric population. Trajectories of amyloid accumulation are supposed to start earlier than 65 years and identifying patients with CF at higher risk of conversion to dementia may be important for future disease-modifying treatments [18]. It is worthy of note that CF does not simply reflect a condition of increased brain vulnerability due to aging. Brain aging indeed results in an unbalance between functional network integration and segregation in the medial temporal lobe memory and the frontostriatal executive systems, representing a physiological process without necessarily an overt cognitive impairment [99]. Conversely, CF refers to brain frailty that may be associated to neuropathological changes related to AD, cerebrovascular diseases, or other neurodegenerative conditions, making people more susceptible to cognitive, as well as motor, decline. Moreover, CF may affect people younger than 65 years.
Based on the lines of reasoning reported above, a proposal for a revised definition of CF according to a multidimensional subtyping is presented in Table 5 and Fig. 2.
Proposal for a revised definition of cognitive frailty according to a multidimensional model
A, amyloid; CF, cognitive frailty; MCI, mild cognitive impairment; N, neurodegeneration or neuronal injury; SCD, subjective cognitive decline; SUD, substance use disorders; T, tau. †Neurobehavioral changes should have a clearly defined recent onset, be persistent and not explained by life events [59]. *Not all A/T/N conditions could be considered as CF, since A+/T+ and A+/N+ conditions should more appropriately fit into Alzheimer’s disease continuum [59]; caution should be taken for the oldest-old people because these changes might be present despite normal cognition.
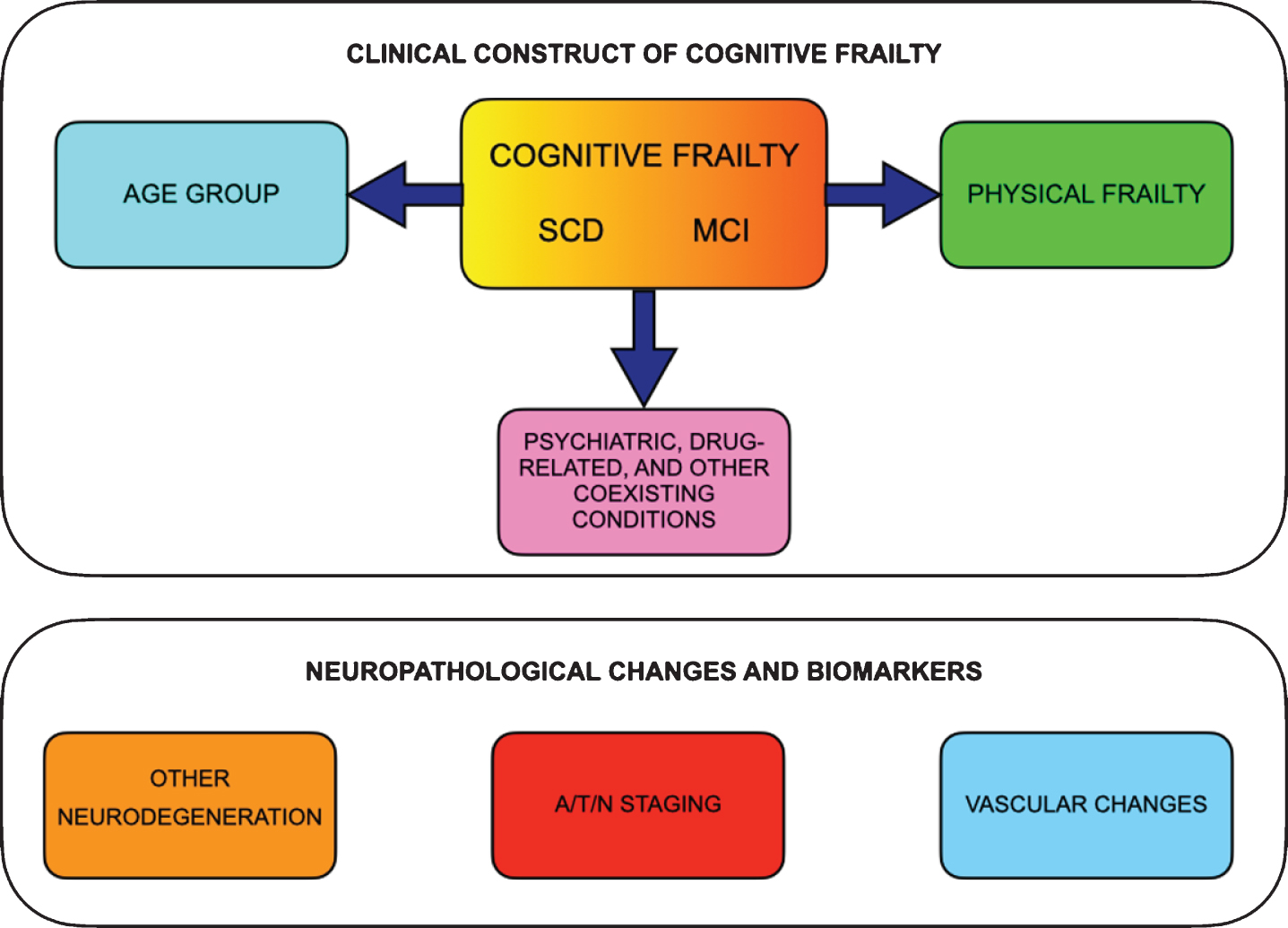
The proposed multidimensional clinical construct of cognitive frailty and the parallel neuropathological changes and biomarkers. Clinical construct of cognitive frailty should include subjective cognitive decline (SCD) or mild cognitive impairment (MCI) together with physical frailty and should consider age range and comorbidities (i.e., psychiatric, drug-related and other coexisting conditions). The neuropathological changes and biomarkers, if present, may offer additional prognostic information, e.g., stratifying the risk of conversion to dementia.
Finally, operational criteria for CF are important to offer a standardized assessment. In analogy to physical frailty [100], a simple, valid, reliable, and sensitive questionnaire, checklist, or abbreviated screening tool could identify patients that should undergo further testing throughout a comprehensive neuropsychological examination. In the absence of a gold standard neuropsychological measure of CF, future directions for clinical research should deal with the identification of the best neuropsychological battery with high sensitivity and specificity and short time for administration that could stratify CF patients according to the most involved cognitive domains and be tailored to patient’s age.
These changes to CF definition might help stratifying CF patients with a broader perspective to design future trials aimed at exploring lifestyle interventions for risk factors and mechanisms involved in conversion to dementia [101] and to test disease-modifying pharmacological strategies to target AD neuropathological changes at an earlier disease stages than they are currently performed [18]. Future studies should explore if the proposed redefinition of CF offers advantages in comparison to the simpler constructs of MCI and SCD, and if it can be easily applied in the clinical setting in diagnostic and therapeutic studies.
Footnotes
ACKNOWLEDGMENTS
The present study has been partially supported by the Italian Ministry of Research and University (MIUR) 5-year special funding to strengthen and enhance the excellence in research and teaching (![]() ). The funding source had no role in study design, in the collection, analysis, and interpretation of data, in writing of the report, and in the decision to submit the article for publication.
). The funding source had no role in study design, in the collection, analysis, and interpretation of data, in writing of the report, and in the decision to submit the article for publication.
