Abstract
Background:
Variable rate of cognitive decline among individuals with Alzheimer’s disease (AD) is an important consideration for disease management, but risk factors for rapid cognitive decline (RCD) are without consensus.
Objective:
To investigate demographic, clinical, and pathological differences between RCD and normal rates of cognitive decline (NCD) in AD.
Methods:
Neuropsychology test and autopsy data was pulled from the National Alzheimer’s Coordinating Center database from individuals with a clinical diagnosis of AD. Individuals with average decline of 3 or more points on the Mini-Mental Status Examination (MMSE) per year over 3 years were labeled RCD; all others were NCD.
Results:
Sixty individuals identified as RCD; 230 as NCD. These neuropsychology tests differed at baseline (RCD versus NCD): WMS-LM Immediate Recall (4.35[3.39] versus 6.31[3.97],
Conclusion:
Individuals with RCD have more severe pathology, more comorbidities, and lower baseline neuropsychology test scores of language and executive function.
INTRODUCTION
Alzheimer’s disease (AD) is the most common neurodegenerative disease of the central nervous system. Without the development of more effective treatments, the immense financial and emotional impact of this disease is predicted to dramatically worsen over the next several decades [1]. One of the most important problems impacting the management of AD is the variable rate of cognitive decline among patients [2].
Individuals with AD typically decline at a steady but slow rate, losing 1 point per year on the Mini-Mental Status Examination (MMSE) [3]. However, prior work has shown that 1/3 of individuals with AD decline at an accelerated rate, losing 3 or more points on the MMSE every year [4]. Individuals in this group are frequently labelled as having rapid cognitive decline (RCD). Identifying an individual as RCD is important because it is associated with worse functional outcomes [5] and higher mortality [6] than those with normal rates of cognitive decline (NCD). Additionally, the current inability to distinguish RCD in clinical trials increases the likelihood of a false negative outcome (type II error) [7].
Though risk factors are without consensus, prior research has shown onset before age 75 [6, 9], higher levels of education [3, 10], lower baseline cognition [8], and the presence of neuropsychiatric symptoms such as depression, anxiety, and psychosis [2, 11] to be associated with RCD. Other tools such as neuropsychology testing, apolipoprotein E (
Drawing firm conclusions from previous RCD research is problematic because most studies have relied solely on clinical diagnoses. Misdiagnosis rates of AD are high (20–40%), even in top dementia centers [13]. Thus, postmortem data are required to understand both the underlying pathology and clinical characteristics of RCD.
Autopsy confirmation of AD remains the “gold standard” for the diagnosis and is essential in the identification of comorbidities. The Neuropathology Data Set (NP) is derived from former and current participating Alzheimer's disease Centers (ADC) in accordance to standards set by the National Alzheimer's Coordinating Center (NACC) at the University of Washington, an important NIA-funded data repository of autopsy-confirmed AD and related dementia cases as well as cognitively normal participants [14, 15]. At the time that we obtained data, the NP included more than 4,000 individuals. To date, no large autopsy sample has been assessed for RCD using data on cognitive status and clinical characteristics.
In this study, data from NACC were used to investigate and compare the demographic, clinical, and neuropathological differences between RCD and NCD. We hypothesized differences between groups in demographic profile (particularly age at disease onset and education), baseline cognitive functioning, neuropsychiatric symptoms, AD pathology, and the number of medical comorbidities. We further aimed to identify unique characteristics present at baseline ADC visit that could be predictive of RCD.
METHODS
Participants
Data were requested from the NACC Database (visits conducted between September 2005 and March 2017). The NACC data sets used for this study were the Uniform Data Set (UDS), the NP Data Set, and the Genetic Data Set (RDD-Gen). Participants from NACC who had a total MMSE score between 12–30 at the initial visit, a presumptive etiological diagnosis of AD at the initial visit, and at least three visits with the UDS neuropsychology test battery were included. Participants unable to meet these criteria were excluded. The NACC dataset is fully de-identified, and all participants had provided written informed consent at their ADC, as approved by local institutional review boards [15].
Demographic information and clinical data
Clinical data from the NACC database were obtained from all participants meeting our above extraction criteria. This information included demographic information (age at initial visit, sex, race, level of education, age at cognitive onset, and age at death), baseline MMSE score, and clinical health history (variables of cardiovascular disease, autoimmune disease, psychiatric measures, and medications).
Neuropathological and genetic data
Pathologically determined AD was ascertained by creating a variable combining two NACC variables of amyloid plaques and tau proteins, ranked in severity by CERAD (C score) and Braak Stage (B score), respectively [16]. C Score ranks the density of neuritic plaques (neuron fragments surrounding amyloid-β protein) while B Score measures the cortical distribution of neurofibrillary tangles (aggregates of hyperphosphorylated tau proteins). Plaque and tangle scores in the NP database were derived according to the NIA–Reagan Institute criteria [17]. Pathologically verified AD was defined as a C score of C2 or higher combined with a B score of B2 (stage III) or higher [18]. Defining pathologically verified AD also allowed for the retrospective comparison of the pathological profile of a clinically diagnosed AD sample with a pathologically confirmed subset.
Variables of cerebral amyloid angiopathy, Lewy body distribution, atrophy (cerebral cortical atrophy measured 0–3 in severity and lobar atrophy measured as present/absent), multiple pathology and vascular pathology count variables, TDP-43, and
Pathologically verified AD was defined by creating a binary variable (1 = C Score of C2 and B Score of B2 (or stage III) or higher; 0 = not meeting this criteria). Count variables of cerebrovascular disease and “other” pathologies were created to assess for burden of cerebrovascular disease and multiple pathologies.
Neuropsychological tests
Tests selected from the UDS neuropsychology test battery are as follows: MMSE, Wechsler Memory Scale-Revised Logical Memory Ia Story Units Recalled and Logical Memory IIa-Delayed Story Units Recalled, the Boston Naming Test (total raw), Animal Naming Test and Vegetable Naming (total raw), Digit Span forwards and backwards (trials correct), Trail Making Test Part A and B (seconds to complete), and the Wechsler Adult Intelligence Scale-Revised Digit Symbol test (total raw). The test selection identified tests across cognitive domains.
Classification of groups
Participants were classified into two different groups based on a progressive loss of points on the MMSE. Consistent with prior research, individuals were categorized as experiencing RCD if they had an average decline of ≥3 points on the MMSE per year over their first three ADC clinic visits; individuals who did not experience such loss were categorized as experiencing NCD [12].
Statistical analyses
We compared participant demographic, clinical history, and pathological data between groups using Chi-square tests for categorical variables, and independent-samples
Subsequently, in order to identify potential independent baseline predictors of RCD, we constructed a multivariable logistic regression model using group membership as the outcome of interest. Independent predictors included in the model were determined in accordance with individual variable significance coupled with previous research on traits predictive of rapid decline in AD. Demographic factors, neuropsychology testing, and disease characteristics were added to the model in congruence with our stated hypothesis as described below:
Demographic factors of education, sex, and age were added to the model despite individual insignificance due to their correlation with neuropsychology testing.
We inspected statistically significant neuropsychological tests for multicollinearity as this could be problematic in the predictive model. Raw scores on the Wechsler Memory Scale-Revised Logical Memory Ia Story Units Recalled, the Animal Naming Test, and Trail Making Test Part B were included due to their significant baseline differences between groups and their relatively low collinearity.
Thyroid disease was included as a significant autoimmune disease measure that has also been cited in previous research as an indicator of worse cognitive trajectory [19, 20] along with neuropsychiatric measures of depression and nighttime behaviors (defined as presence versus absence from the Neuropsychiatric Inventory Questionnaire, NPI-Q [22]) added due to the frequent occurrence of these symptoms in Lewy body disease (LBD) and AD, respectively [21].
Statistical significance was established throughout at
RESULTS
Participant demographics and clinical data
This study included 290 participants clinically diagnosed with AD at their initial visit, 230 of which were classified as NCD, and 60 of which were classified as RCD. Those classified as NCD had a mean MMSE decline of 0.94 (SD:0.94) points per year, ranging from a loss of 2.67 points per year to a gain of 1.67 points per year, with mode loss of 0.66 points per year. Those classified as RCD had a mean MMSE decline of 4.28 (SD:1.23) points per year, ranging from a loss of 8 points per year to a loss of 3 points per year, with mode loss of 3.33 points per year. MMSE at initial ADC visit did not differ significantly between groups.
Time from initial diagnosis to death was significantly different between RCD and NCD, but other demographic variables did not differ significantly (Table 1.1).
Participant Demographicsa
aThe
Thyroid disease and AD medication use was more prevalent in the RCD group than the NCD group (30.0 versus 16.1%,
Participant Clinical Data at Baselinea
aThe
Neuropathological and genetic differences
There were differences in pathology between the RCD and NCD groups (see Table 2.1). However, differences in the Braak stage and plaque severity did not meet significance. Contrastingly, non-AD pathology did differ. Cerebral amyloid angiopathy was found to be significantly greater in patients with RCD than NCD (mean [SD]: 1.62[1.03] versus 1.13[1.02],
Neuropathological and Genetic Differences
In a subset of patients, atrophy at autopsy suggested that overall cerebral cortical atrophy (28 RCD/117 NCD: mean [SD] 1.54[0.92] versus 1.17[0.83],
No significant differences were observed in vascular disease count or multiple pathology disease count, TDP-43, or the presence/predominance of the
Among the original total study sample (
Neuropathological and Genetic Differences: AD Pathologically Confirmed Subset
aDue to missing data, sample sizes varied amongst characteristics:
Neuropsychological testing differences
Though baseline scores on the MMSE did not differ significantly, individuals with RCD had lower performance at baseline on the following neuropsychological measures: Wechsler Memory Scale-Revised Logical Memory Ia Story Units Recalled, the Boston Naming Test, the Animal Naming Test, and Trail Making Test part B (see Table 3 and Chart 1). Other neuropsychological measures did not vary at baseline.
Neuropsychological Testing Differencesa
aThe
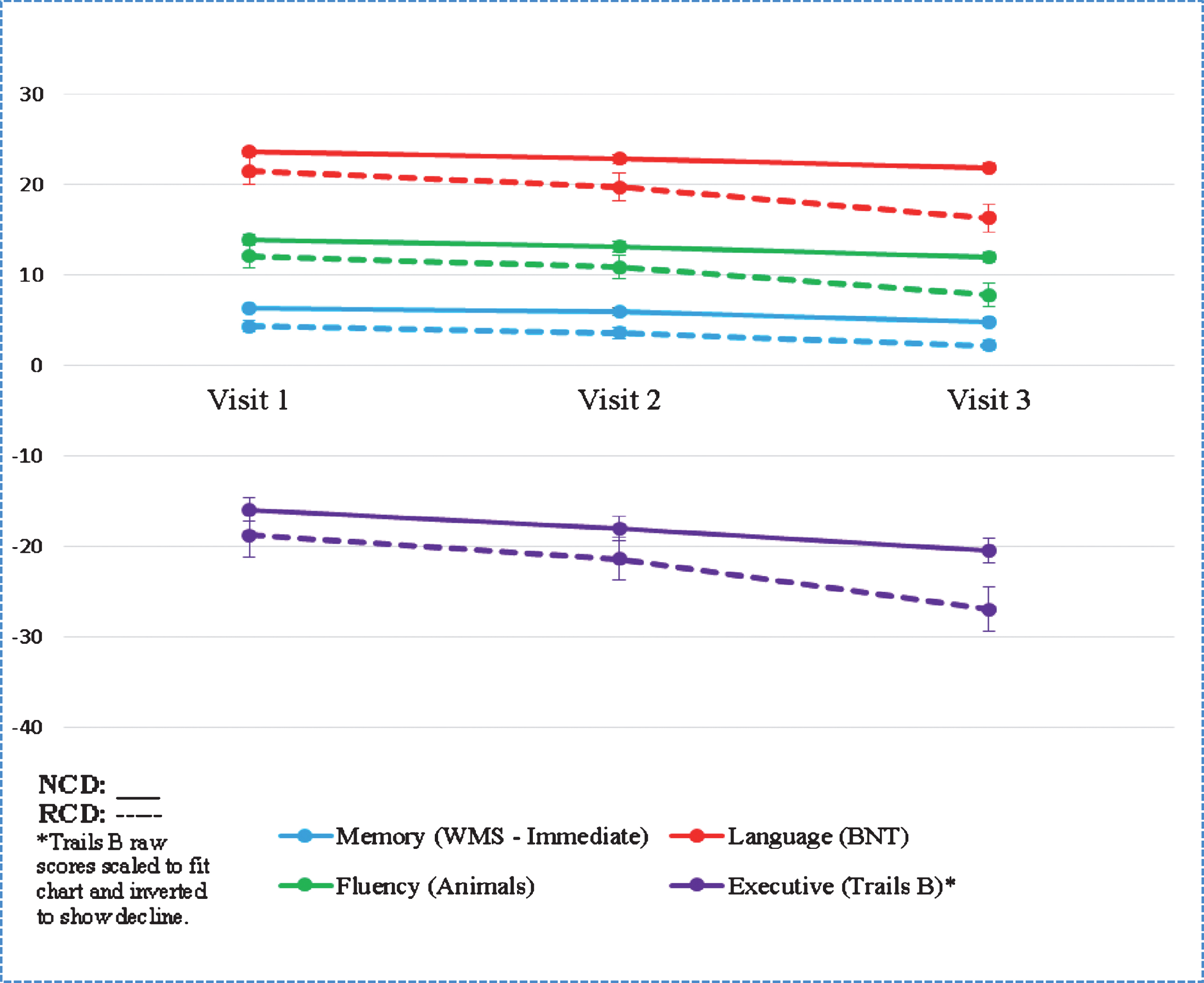
Neuropsychological Testing Differences
A model of clinical and neuropsychological predictors of RCD at baseline
A multivariable logistic regression model was fit to determine predictors of group membership, simultaneously entering related variables of disease characteristics, neuropsychological testing, and demographic information. Table 4 summarizes the results from the model, which was found overall to be statistically significant (
Logistic Regression Model for Predictors of RCD at Baseline
aDue to missing data, this model has the following sample size:
Presence of thyroid disease (odds ratio (OR): 2.87; 95% CI: 1.36–6.05;
Demographics of age, sex, and education were not associated with increased likelihood of an individual’s categorization as RCD within the model, nor were variables of nighttime behavior and depression from the NPI-Q, the Animal Naming Test, or Trail Making Test part B.
DISCUSSION
Central findings
This study looked at demographic, neuropsychological, and pathological correlates of RCD in AD in a large, well characterized sample with autopsy confirmation. The primary finding from our study is that patients with RCD had a more severe pathological signature than their NCD counterparts, specifically with more extensive comorbidity. This is evidenced in the autopsy data which demonstrated that RCD patients, despite having shorter time from diagnosis to death, had more cerebral amyloid angiopathy, greater atrophy, and more diffuse neocortical Lewy bodies. None of the observed demographic factors differed between the groups, which may indicate that RCD is driven primarily by biological factors (i.e., a more aggressive form of AD, with comorbidities) and less influenced by demographics (age, education, etc.) as previous research has suggested.
Secondary findings
Though RCD and NCD had similar baseline MMSE scores, individual neuropsychology test results suggest significant underlying baseline differences in language and memory (WMS Logical Memory Immediate Recall, Animal naming, BNT) and executive functioning (Trails B and WAIS-R Digit Symbol). Lower performance on these tests implies cortical dysfunction, which is consistent with the finding of more extensive extra-limbic atrophy, suggesting that disease has already progressed more extensively throughout the brain in the RCD group before their initial ADC visit. This demonstrates the importance of adequate neuropsychological characterization early on in the disease course, as well as the usefulness of and potential for the development of a predictive model of cognitive trajectory including similar measures.
Baseline cardiovascular risk factors and psychiatric symptoms also did not differentiate between groups, although a significantly higher portion of the RCD population was taking an AD medication at baseline visit. Observing that more RCD individuals are taking cognitive enhancing agents is indicative of other clinically distinguishing factors at baseline that were perceptible by a clinician, yet not captured in the database. This has been suggested in previous studies, which observe that cognitive symptoms may be indicated at baseline presentation without an obvious profile or causative effect [23].
Though a count of multiple pathologies did not appear significant, there was evidence of more comorbidity in the RCD group, with RCD presenting with more neocortical Lewy bodies and evidence of greater occurrence of autoimmune disease (thyroid disease). Other publications have also linked thyroid disease to cognitive decline [19, 20], though this is not consistent across studies [12].
While there is evidence of differences in clinically-identifiable factors at baseline, the regression model with a combination of factors of disease characteristics, neuropsychology testing, and demographic information was inadequately predictive of RCD. However, WMS logical memory immediate recall and thyroid disease were significant within the model, suggesting that further research is warranted, perhaps in different datasets.
Implications for current research
To our knowledge, the present sample is the largest with autopsy confirmation that has been examined for defining factors of RCD. Because of this, we sought to also examine many previous correlations with RCD found in previous publications. Our sample consists of 20% with RCD versus NCD, consistent with the proportions of previous research, varying between 11–40% [24]. We found that many of the distinguishing factors purported by past research did not significantly differentiate groups in our sample. Factors such as age, higher level of education, lower baseline cognition, neuropsychiatric status, and
Limitations
One limitation of the present study is the limited specificity of certain data points in NACC, namely variables of vascular pathology, multiple pathology, and thyroid disease. For example, it was difficult from the current dataset to accurately assess severity of vascular burden because the data available suggested that all participants were greatly, and similarly, burdened. More recent data being collected in NACC NP V10 allows for a more nuanced severity quantification of vascular pathology, but it will take time to accumulate an adequate number of participants. The same is true of the multiple pathologies count, though with the opposite limitation, with our sample having a mode of zero additional pathologies. Thyroid disease is also not parsed out into hypo/hyperthyroidism in NACC; prospective data collection would be required to obtain this data. It should also be taken into consideration that the NACC database subject education is likely above the population average, thus potentially limiting the generalizability of results [25].
In regards to the neuropsychology test data, it has been suggested in previous research that the MMSE may be too blunt of an instrument, thus being a questionable choice by which to define RCD. The MMSE has been criticized for its limited ability to assess global cognitive function, namely its lack of sensitivity to language and executive functioning [26]. This could attest to executive and language differences between RCD and NCD at baseline despite nearly identical MMSE scores, and begs the question of whether it should be the measure of choice in defining RCD. However, it can be observed that the neuropsychology database in NACC is also relatively limited in expansiveness, thus restricting the ability to probe deeply into cognition between groups [27].
It is also worth acknowledging the increased possibility of false-positives (Type I error) within our results due to multiple comparisons. The results of this study are hypothesis-generating so we did not adjust for multiplicity.
Implications for future research
Future research should look to biomarkers that might help predict the likelihood of one’s categorization as RCD
Overall, we demonstrated that individuals with RCD show more severe pathology, are more likely to have pathological comorbidity, and have lower baseline scores on neuropsychology tests of language and executive function. Despite evidence of baseline differences, a predictive model including these variables was not optimally sensitive to RCD categorization, suggesting that future research is needed to better capture the early profile of patients most likely to decline rapidly.
Footnotes
ACKNOWLEDGMENTS
The NACC database is funded by NIA/NIH Grant U01 AG016976. NACC data are contributed by the NIA-funded ADCs: P30 AG019610 (PI Eric Reiman, MD), P30 AG013846 (PI Neil Kowall, MD), P50 AG008702 (PI Scott Small, MD), P50 AG025688 (PI Allan Levey, MD, PhD), P50 AG047266 (PI Todd Golde, MD, PhD), P30 AG010133 (PI Andrew Saykin, PsyD), P50 AG005146 (PI Marilyn Albert, PhD), P50 AG005134 (PI Bradley Hyman, MD, PhD), P50 AG016574 (PI Ronald Petersen, MD, PhD), P50 AG005138 (PI Mary Sano, PhD), P30 AG008051 (PI Thomas Wisniewski, MD), P30 AG013854 (PI M. Marsel Mesulam, MD), P30 AG008017 (PI Jeffrey Kaye, MD), P30 AG010161 (PI David Bennett, MD), P50 AG047366 (PI Victor Henderson, MD, MS), P30 AG010129 (PI Charles DeCarli, MD), P50 AG016573 (PI Frank LaFerla, PhD), P50 AG005131 (PI James Brewer, MD, PhD), P50 AG023501 (PI Bruce Miller, MD), P30 AG035982 (PI Russell Swerdlow, MD), P30 AG028383 (PI Linda Van Eldik, PhD), P30 AG053760 (PI Henry Paulson, MD, PhD), P30 AG010124 (PI John Trojanowski, MD, PhD), P50 AG005133 (PI Oscar Lopez, MD), P50 AG005142 (PI Helena Chui, MD), P30 AG012300 (PI Roger Rosenberg, MD), P30 AG049638 (PI Suzanne Craft, PhD), P50 AG005136 (PI Thomas Grabowski, MD), P50 AG033514 (PI Sanjay Asthana, MD, FRCP), P50 AG005681 (PI John Morris, MD), P50 AG047270 (PI Stephen Strittmatter, MD, PhD).
Research reported in this publication was supported by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health under grant number 5P20GM109025.
