Abstract
BACKGROUND:
The majority of common agility tests include pre-planned change of direction (COD) maneuvers which require muscular coordination. However, muscular coordination in terms of organization of co-activated antagonistic muscle pairs in COD maneuvers have not been studied.
OBJECTIVE:
To evaluate muscle coordination by analyzing co-contraction at knee and ankle during three agility tests and to investigate the relationships between muscle coordination and test completion time.
METHOD:
Thirteen healthy young male subjects (22.1
RESULTS:
The findings indicated that concurrent activation costs a delay to complete the test faster. CCI was higher at right ankle and left knee and showed an increasing trend with advancing the phase when the test requires multiple CODs.
CONCLUSIONS:
Minimizing co-contraction would result in faster movement, however, there might be a trade-off between joint stiffness and test performance depending on changes in neuromuscular factors such as altered motor control strategies due to strength imbalances or occurrence of fatigue.
Introduction
Several studies have reported varying degrees of correlation between potentially dominant biomotor abilities for agility-linked skills (e.g. strength and speed) and performance in terms of test time in commonly used agility tests. Although not fully neglected while defining agility (e.g. [1]), coordination has been trivialized in the experimental studies focusing on its determinants. Such anticipated indicators of agility as leg muscle qualities such as strength, power, and speed have been widely studied [2, 3, 4, 5]. There are inconsistent results in the literature, however, regarding the relations of those aforementioned biomotor skills and performance in agility tests [6].
Differences in physical requirements of commonly used timed tests for the evaluation of agility have been proposed as a potential reason for the reported inconsistency [7]. Using test completion time (hereafter abbreviated to ‘test time’) as the sole criteria to assess one’s agility has also been criticized due to the fact that the test time is far from shedding light on the underlying biomechanical or neurophysiological factors that have potential to affect test performance [8]. Independent of whether dominated by running or hoping elements, quick change of direction (COD) maneuver is a common element in most of the agility tests. The execution of agility tests with multiple COD maneuvers requires muscular coordination [1]. However, dynamic lower extremity coordination has rarely been discussed from a neurophysiological point of view.
Recently, Lyle et al. [9] have pointed out lower extremity dexterity as an important attribute for deceleration and COD tasks, and proposed a tool to assess the ability to control the magnitude and direction of limb endpoint force. However, little is known regarding the lower limb muscular coordination in the regulation of these complex end point biomechanical outputs. Moreover, the relation between muscle coordination in lower limb muscles during COD maneuvers in common closed-skill agility tests (e.g. Illinois, 5-0-5, and hexagon tests) and performance, i.e. test times is not clear.
Among the definitions of muscle coordination, “a distribution of muscle activation or force among individual muscles to produce a given combination of joint moments” [10], and “purposeful activation of many muscles to produce a given motor task” [11] are the widely accepted ones. Both describe different levels of coordination from muscle space (from one to many dimensions) to joint space, while emphasizing different levels of analysis from muscle activation to forces and moments as well. For the purpose of this study, muscle activation level was deemed relevant level for COD maneuver and therefore muscle coordination is operationally defined as the organization of co-activated antagonistic muscle pairs that act at a joint. Specifically, an inverse relation between coordination and muscle co-activation was adopted, such that higher co-activation is related to poor coordination between antagonistic pairs [12].
Apart from the fact that muscle co-activation signals inefficient movement patterns as it counteracts the intended agonist action [13], it also plays role in injury prevention mechanism [14]. Indeed, joint stability that is interrupted by sudden external loads is known to be compensated by co-activation of antagonistic muscle pairs to increase joint stiffness and to maintain joint stability [15]. As a result, co-activation may decrease force production or cause a delay in the initiation of an action. Accordingly, if COD maneuvers dominate an agility test, it could be hypothesized that higher co-activation of antagonistic muscle pairs would be prominent-especially in slower trials, yet it would not be the case for agility tests that comprise mostly straight running elements. Based on this hypothesis, we aimed to investigate muscle coordination in antagonistic lower limb muscle pairs during COD maneuvers in closed-skill agility tests. Briefly, this study has two purposes: i) to evaluate muscle coordination in lower limb muscles by investigating muscle co-contraction in antagonistic pairs at the knee and ankle joints in three common closed-skill agility tests, namely Ilinois, 5-0-5 and Hexagon tests, and ii) to investigate the relationship between muscle coordination and agility test time.
Methods
Subjects
Thirteen undergraduate student athletes (mean
Instrumentation
Surface electromyography (sEMG) electrodes (Trigno, Delsys, Inc) having built-in linear tri-axial accelerometers were used to acquire simultaneous muscle activity and acceleration (ACC) data in EMGworks software (Delsys, Inc). The electrodes had the following specifications, outer dimensions: 37 mm
Agility test procedures and data collection
Each of the three commonly used agility tests, namely, Illinois, 5-0-5, and Hexagon tests were performed by the subjects twice. All three agility tests have pre-planned COD maneuvers without temporal uncertainty in common. In each trial, the test was initiated by the subject at a self-determined time. Figure 1 shows the setup details and procedures of each test which are explained briefly below in the same order.
Setup for agility tests a) IAT, b) 505T, c) HxT.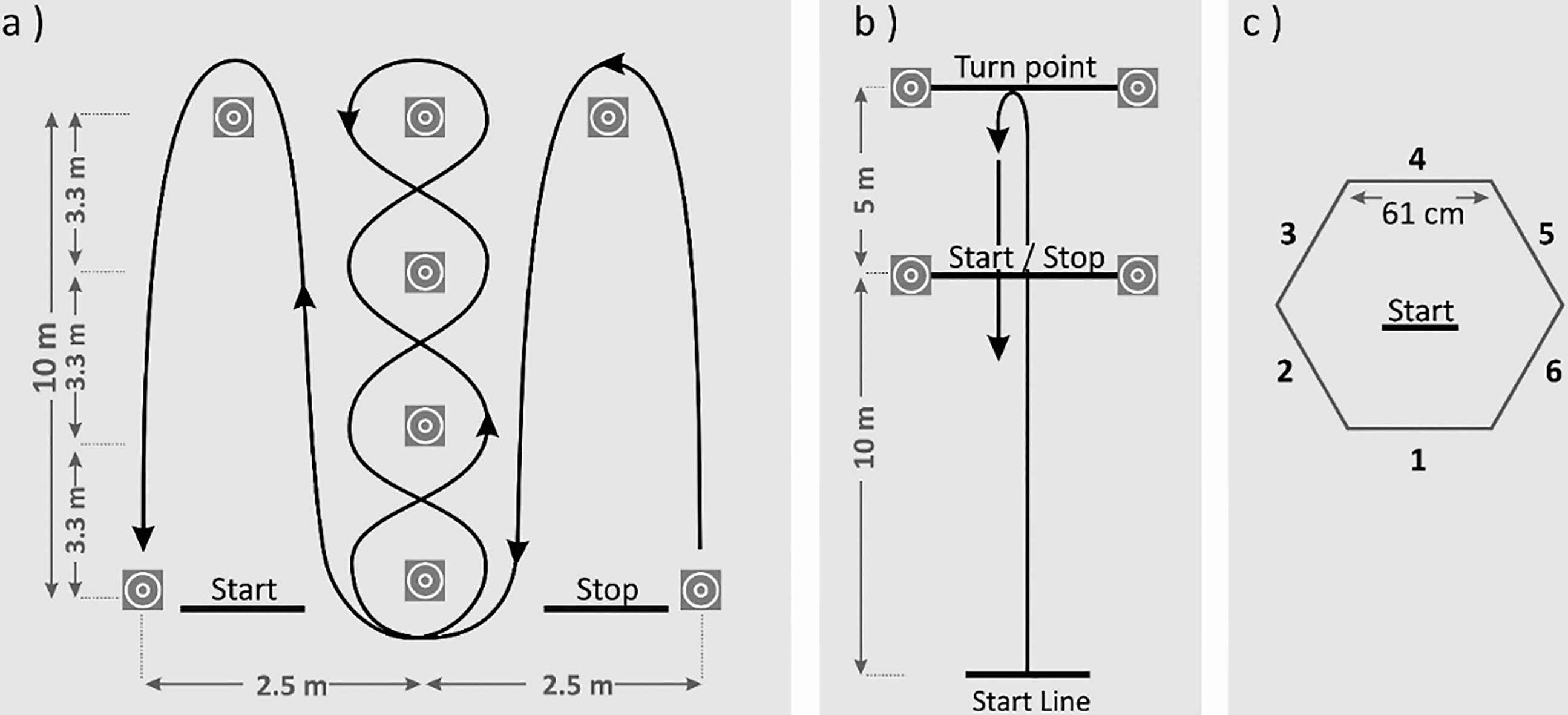
Illinois Agility Test (IAT): It requires negotiating the cones while running the course with maximum effort in the set path. It starts on prone position behind the start line, with elbows flexed, hands placed palm down on both sides and legs straight. The test also includes multiple rapid COD as well as accelerations and decelerations maneuvers. 5-0-5 Test (505T): This test includes single rapid COD as well as acceleration and deceleration maneuvers. It is executed by running as fast as possible on a 10-m go and back course, with a flying start of the performer from an upright position with the front foot on the start line. Hexagon Test (HxT): A double-leg test involves multiple rapid COD maneuvers. Subject starts in the middle of the hexagon, jump across six sides in a clockwise direction, and back to start position after visiting each side in order.
The IAT and 505T were electronically timed by using timing gates (Fusion Smart Speed Timing Gates System, QLD) located at start and stop points shown in the Fig. 1. The test times for the HxT were determined through the analysis of acceleration in the frontal axis and calculated as the time between the initiation and termination of the test (see data analysis for further explanation). Subjects’ best test times for each of the above-mentioned tests were presented in Table 1.
Subjects’ test times in seconds for their best trial
Muscle electrical activity was collected bilaterally by sEMG. Electrodes were affixed to the skin with a double-sided adhesive tape and also secured with surgical tape to minimize movement artifacts. The preparation of skin, sensor locations, and electrode placement was followed the recommendations of the SENIAM project. The electrodes were placed on the following agonist-antagonist muscle pairs at ankle (dorsal and plantar flexion) and at knee joints (flexion and extension), respectively: Tibialis Anterior (TA) and medial Gastrocnemius (GC), Vastus Lateralis (VL) and Biceps Femoris (BF).
Prior to execution of the agility tests, maximal Squat Jump (SJ) and Nordic Hamstring Exercise (NHE) were performed three times to determine a normalization factor (NF) for each muscle. SJ was started in a squat position with arms crossed across the chest and without counter-movement of the lower limbs. NHE was performed in an upright kneeling position with the assistance of the experimenter who applied pressure at ankle level to keep the lower legs stable and in contact with the ground. The subject was then instructed to fall forward as slow as possible. The peak EMG activity of GC, VL, and TA during SJ (including the ground contact phase) were used to calculate NF for each muscle, whereas the peak activity of BF during NHE was used to calculate NF thereof.
The raw EMG signals were rectified and then band-pass filtered (25–350 Hz) with a fourth order zero-lag Butterworth filter. As it is known that movement artifact influences the shape of the sEMG signal spectra up to frequencies of 20 Hz [16], while the movement artifact was attenuated, low- and high-frequency components of sEMG signal were preserved. The NFs were calculated from root-mean-square (RMS) of a 100-ms data window centered at the EMG peak amplitude. The EMG signal for each muscle’s activity during the agility tests were then normalized with the NF of the corresponding muscle.
Determination of change of direction (COD) time points
The ACC data was collected by using the tri-axial linear accelerometers which were embedded into hybrid sEMG sensors. In contrast to trunk-mounted acceleration based COD detection method [17], we used two accelerometers affixed to the TA muscles of both legs to collect EMG and ACC data simultaneously. In order to detect when a COD maneuver occurs during IAT and 505T, the envelope of the time history of the ACC data in the frontal axis was used. To do so, corresponding ACC data were filtered with a fourth order, zero-lag, low pass (5 Hz) Butterworth filter to locate instances of local minima in the acceleration traces. As expected, in IAT trials, there were five distinct COD time points (
The illustrative example for determination of time points by smoothing acceleration data (a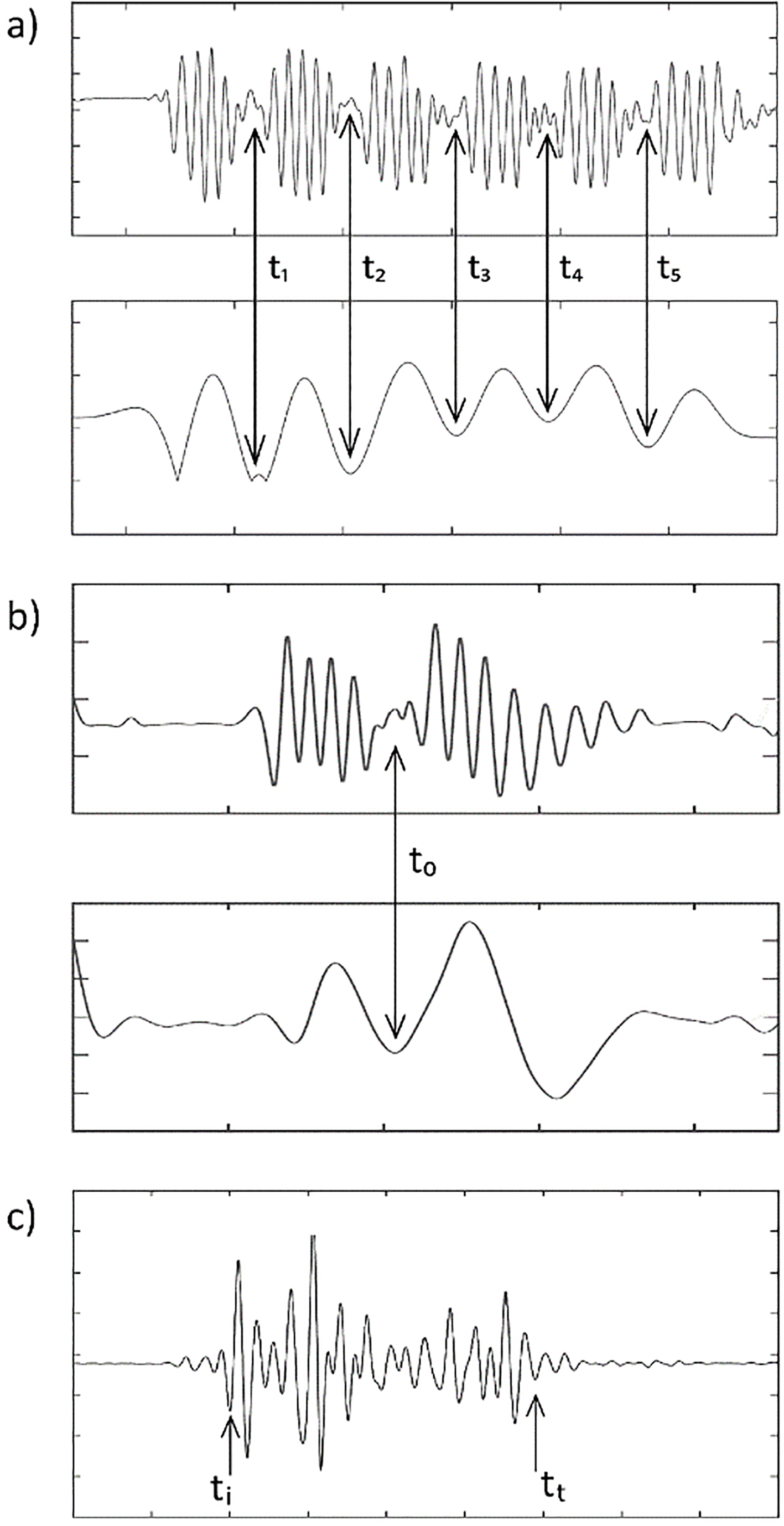
Co-contraction index (CCI) for antagonistic muscle pairs of ankle (between TA and GC) and knee (between VL and BF) was calculated in the above-defined time windows of corresponding agility test using Eq. (2.6) [18]. The normalized RMS EMG values were used to quantify CCI. The output of Eq. (2.6), CCI, defines co-activation of a pair of antagonistic muscles for each element in the EMG time series thereof rather than identifying which muscle has more activity.
“Muscle x Side x Phase” interaction for CCI calculated for different phases of IAT trials (estimated mean 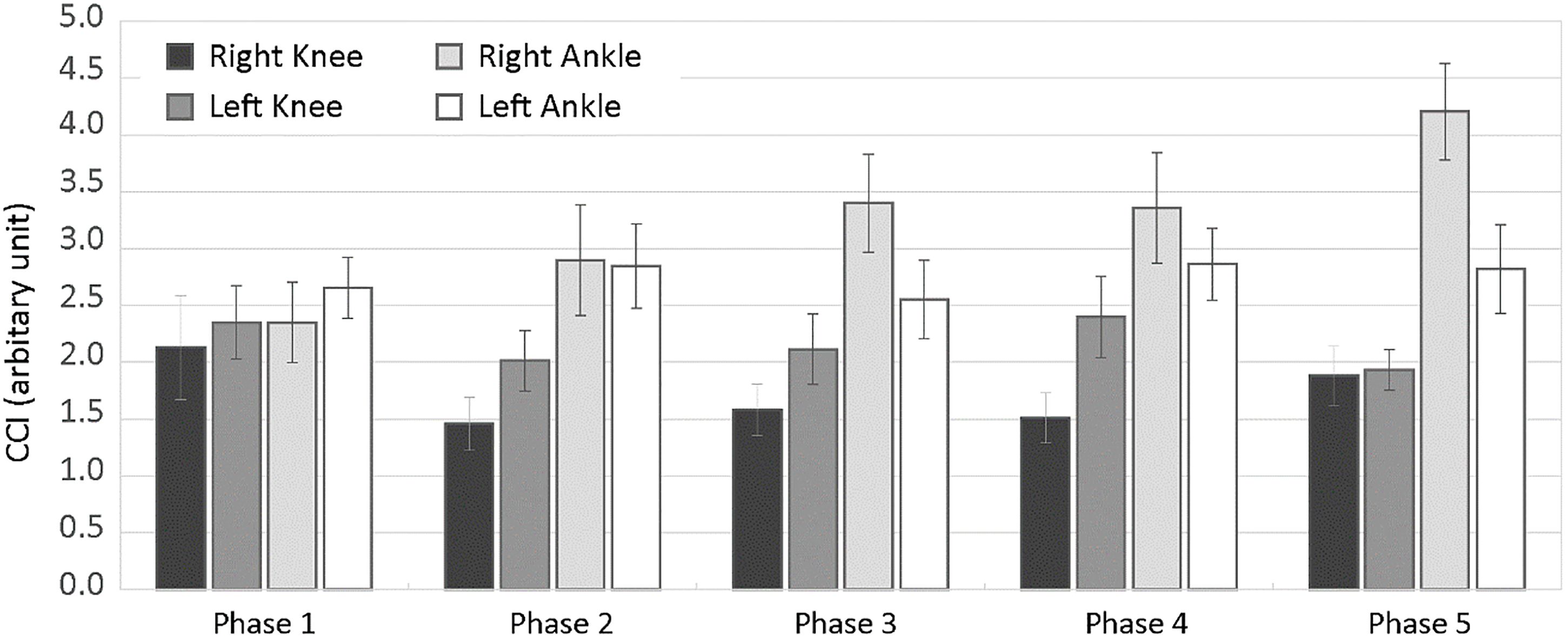
where ‘lowerEMG’ and ‘higherEMG’ stand for RMS EMG values corresponding to the less active and more active muscles, respectively. This method provides a sample-by-sample estimate of the relative activation of the pair of muscles as well as the magnitude of the co-contraction over the entire time window [18].
For IAT and HxT trials, the CCI was calculated on both sides. For the analysis of CCI for HxT trials, the time window between
For the analyses of 505T trials, CCI was first calculated for both sides to determine the trials which were led by the dominant foot. Such that, if a local minima of ACC trace was smaller at the dominant side and if CCI revealed a higher value on the same side, then corresponding trial was considered to be led by the dominant foot and selected for further analysis. In this way, we were able to determine the trials where the last foot stance before the turn and the first step after that turn were led by the dominant foot. At least one trial of 11 of 13 subjects met the above-mentioned selection criteria. If more than one trials met the criteria, only the fastest trail of the subject was further considered in the analysis. CCI was therefore analyzed for the dominant side of 11 subjects. Figure 5 illustrates CCI time history calculated for the knee and ankle, as well as the analysis window of 2 s which was centered at a local minimum. According to the modulation of CCI time-trace across subjects, that 2 s window was divided into three relative phases of 505T for further comparisons as follows: phase 1 (deceleration): between
Three-way [2 (ankle and knee)
Results
In IAT trials, as shown in the Fig. 3, CCI averaged across trials were higher at ankle than at knee in most of the phases. The contribution of right ankle during the analysis period had an increasing trend with advancing the phase. The ANOVA confirmed significant effect of joint on CCI (F [1, 24]
Illustrative example of CCI time series for ankle antagonistic muscles during different phases of a COD maneuver during 505T. The data were averaged across subjects (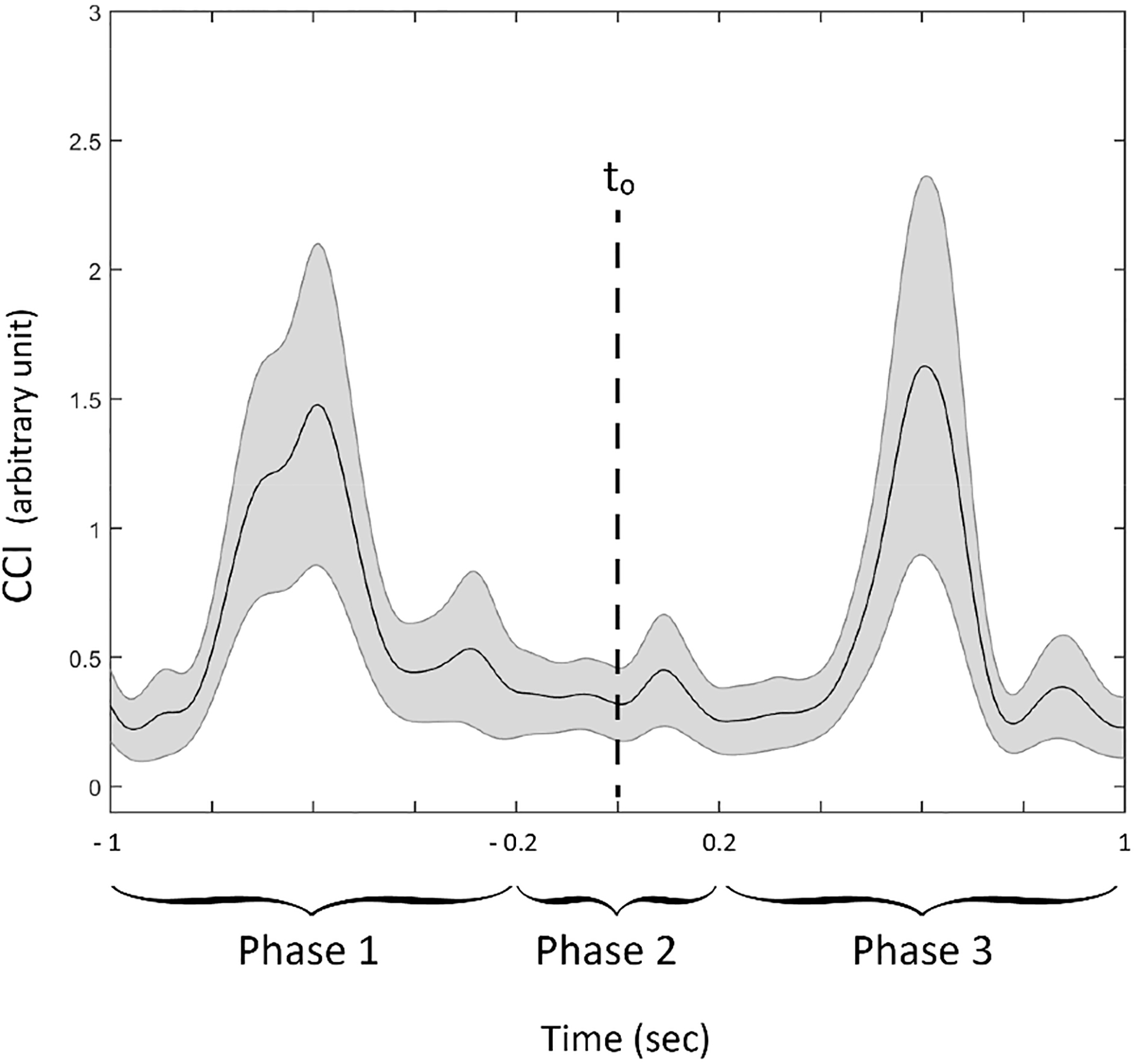
The CCI calculated for the HxT trials (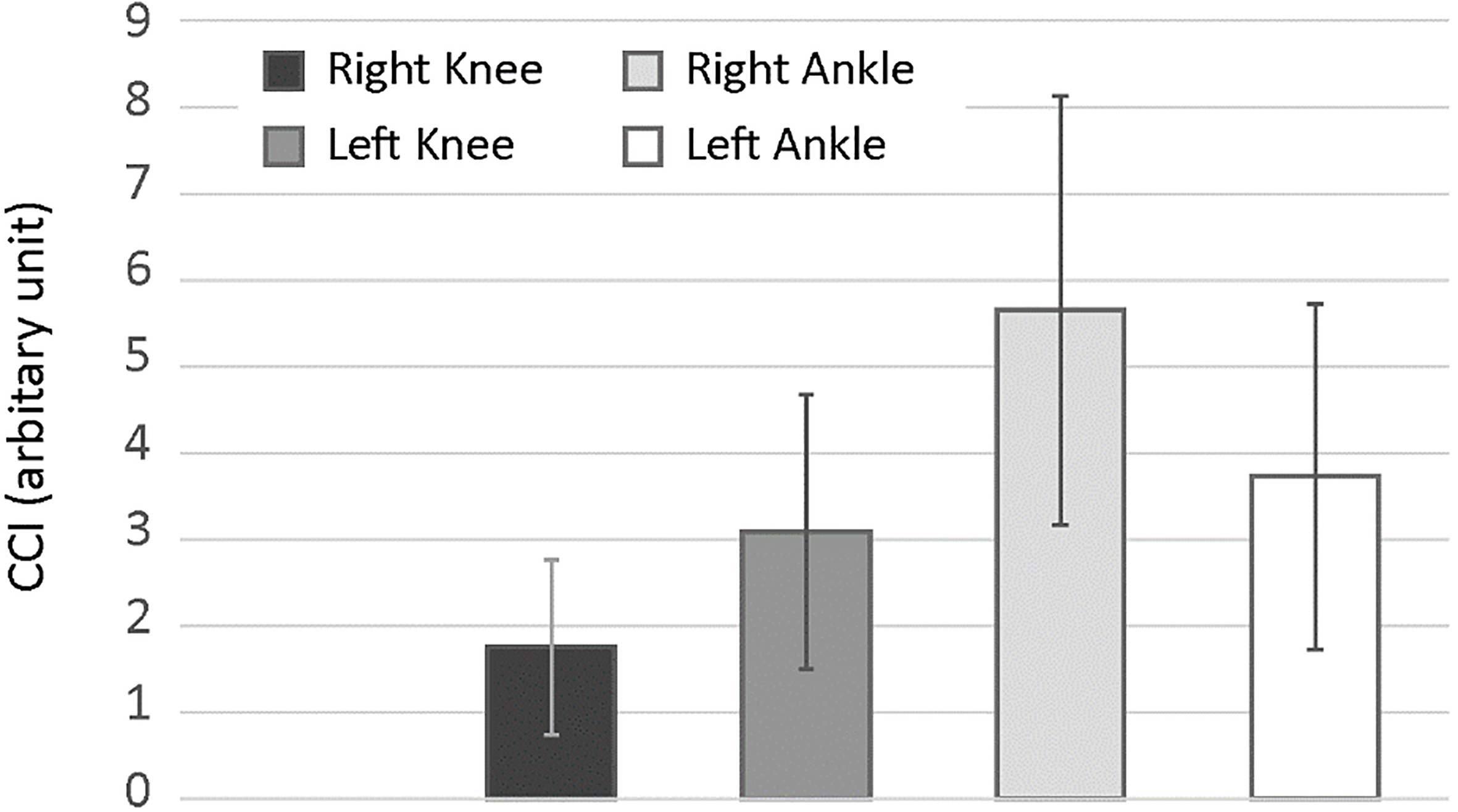
Figure 4 shows CCI averaged across subjects (
The CCI calculated for the HxT trials were followed the order of Ankle
Based on our adoption of an inverse relation between coordination and muscle co-activation, the results of this study demonstrate that lower co-activation of antagonistic muscles of leg might reflect an increased muscular coordination as lower CCI was related to how quickly multiple COD maneuvers were performed. The reduction in muscle co-activation can be interpreted as an achievement of a movement skill with progressive inhibition of muscular activity that is not related to the task [19]. Accordingly, we had expected to observe lower co-activation of antagonist muscle in the faster trials. In line with this expectation, we found that higher muscular coordination at both knee and ankle joints contributed to the agility test times to become faster in IAT and HxT. Despite the difference in number of movement elements, both tests consist of multiple quick COD maneuvers whether from single- or double-leg stance. Having in mind that small to moderate and mostly inconsistent correlations between mostly studied physical qualities of leg muscles (e.g. strength, power) and COD or between sprint time and COD [3, 7, 20, 21], we suggest that the agility test times might be influenced by contribution of some other factors such as muscular coordination in the execution of COD maneuvers.
To execute faster COD maneuvers, deceleration phase should be followed by transition of the body into a new direction as quickly as possible. While doing so, the combination of movement elements can be orchestrated in a number of ways through different joint kinematics as well as different contributions of the antagonistic muscle activity at both knee and ankle. On the other hand, such kinematic changes during COD maneuvers have also been related to joint instability [22]. Hence, whereas concurrent activation of agonist-antagonist muscle pairs costs delays in test times, that might be a consequence of a possible trade-off between joint stiffness and better performance in time times. During COD phase, knee extensor muscles act eccentrically to reduce hip and knee moment and the knee flexors contract concentrically to stabilize the joint. In this phase, both muscles display high activation levels and support joint stability by co-activating [23]. In a similar manner, at the ankle level, eccentric contraction of dorsal flexors are accompanied by concentric activation of ankle flexors to stiffen the joint for braking during COD maneuvers.
In regard to the kinematics of the COD maneuvers during IAT and 505T, both tests require to rapidly change velocity and direction of the whole-body center of mass by lowering and leaning the center of mass toward a new direction. Similar to 505T, the first COD phase of IAT also revealed slightly increased CCI among joints and sides. On the contrary, HxT are performed by rapidly shifting direction of gravitational moments while maintaining the center of gravity in a relatively constant position. Even though HxT consists of double-leg hopping, CCI was found to be higher at right ankle and left knee, similar to the values obtained for IAT. The difference for the dominant leg was at larger values for both IAT and HxT compared to the non-dominant side. Therefore, the imbalance between the activity of agonist and antagonist muscle pairs as well as imbalance between sides might affect joint stability and bilateral joint loading pattern during COD maneuvers.
The results of this study also revealed that co-activation of antagonistic muscle pairs around both knee and ankle joints had an increasing trend with advancing the phases of IAT. While the contribution of knee muscles increase, CCI was consistently larger at ankle for both sides. A possible strategy that is responsible from increased CCI at the ankle might be a trade-off on the level of joint or antagonistic muscle fatigue. Indeed, it has been known that muscle fatigue, or decrease in force generating capacity, may lead to reorganize the cooperation between knee and ankle joints [24]. To illustrate, Bonnard et al. [24] have shown that, during hoping tasks, the load sharing has been shifted to the knee extensors in order to compensate a decrease in muscle force produced by the ankle muscles. In our experiment, a progressive decrease in the ankle co-activation through the subsequent phases of IAT might also be the consequence of the heavier reliance on knee muscles with decrease in ankle muscle force. That was also supported by the overall normalized CCI on HxT where larger CCI values at the dominant ankle was compensated by increased co-activation at the ipsilateral knee. Fatigue has been shown to be among the responsible factors for the altered neuromuscular control of the lower limbs and changes in the ability of dynamic stabilization of the knee joint [25]. Kellis [26] demonstrated that the increased contribution of antagonist activity in fatiguing conditions concurred with the increased resultant joint moment possibly to stabilize the knee joint. Therefore, the effects of possible occurrence of fatigue on antagonist function is likely to confirm the trade-off between the agonist-antagonist muscles.
Agility is generally linked to coordination, yet there is still inefficient data available on this proposed relationship. Even though, this study showed some degree of correlation between test time and co-activation, as an adaptable physiological variable, classifying co-activation as a candidate measure for muscle coordination requires cumulative corroborative findings focusing on COD maneuvers. The major limitation of this study was that it relied on one set of antagonistic muscle pairs act at each joint. However, not only contribution of synergist muscles but also single and multi-joint muscles may also be considered for further understanding as well as more-detailed kinematic analysis should be incorporated to evaluate level of co-activation. Besides, since CCI used in this study is directionless (sEMG activity of a muscle with greater activity was used as denominator), it undermined the interpretation on the mechanics of COD maneuvers.
Further studies that focus upon the effect of muscle strength imbalances and the effect of occurrence of fatigue on muscle co-activation would allow greater understanding of the potential predictors of agility and coordination. Thus, empirical evidences on their interaction would provide a basis for practical applications and help to understand whether increased co-activation in antagonistic muscles of dominant ankle and ipsilateral knee is due to asymmetry in force production capacity or fatigability of these muscles, or whether the goal should be to minimize co-activation or reduce asymmetry between dominant and non-dominant side for a successful agility performance.
Conclusions
We suggest that the level of muscle co-activation can be viewed as an indicator of muscular coordination which is likely to be an important factor in closed agility tests that comprise multiple COD maneuvers. Since the ability to perform a COD maneuver quickly highlights sensorimotor capabilities associated with mobility and joint stability, muscle co-activation may be useful in evaluating competence in the coordination of lower limb muscles which is necessary for injury-free and quick execution of COD maneuvers.
Footnotes
Acknowledgments
The study was supported by TUBITAK (The Scientific and Technical Research Council of Turkey) (Project Number: 115S535). The authors would like to thank all subjects who volunteered to take part in this study, and our undergraduate student, Bedrettin Bulguru, for his assistance with subject recruitment and agility test setup.
Conflict of interest
The authors declare no conflict of interest.
