Abstract
This review spans a wide arc from the first observations of the early anatomists to the present day. William Harvey was the first to describe the heart as the centre of the large and small circulatory system. He thus replaced the previously valid system of Galenos, It was Marcello Malpighi who first described that the capillary system connects the arteries with the veins. In 1688 Antoni van Leeuwenhoek (1632–1686) confirmed these results with a paper on capillary perfusion in the caudal fin of the glass eel.
It was then Hermann Boerhave (1668–1738, Leiden) who was the first to carry out microcirculation tests on patients. He studied the microcirculation in the human bulbar conjunctiva. Even today, microcirculation studies in the conjunctiva bulbi of patients are carried out today. Until 1831, it was never quite clear whether the observations reported belonged mainly to the field of microcirculation, which had not yet been defined. This was done in Great Britain by Marshall Hall (1790–1857). Technical Improvements allowed increasingly sophisticated studies of the morphological structure of the terminal vasculature. According to Gustav Ricker (1870–1948, Vienna), the terminal vasculature comprises the functional unit of the smallest arteries, arterioles, capillaries and venules. In 1921 it was still thought that the blood circulation was the sole response to the pumping action of the heart. Even the classic work by Bayliss on the myogenic hypothesis (later referred to as “blood flow autoregulation”) initially received little attention. More strikingly, even the findings of August Krogh, for which he received the Nobel Prize in Medicine in 1920 (for his discovery of the mechanisms of capillary motor regulation), were ignored. During an outstanding autoregulation symposium held in 1963 a broad consensus was reached on active and passive mechanisms, which is more or less valid till today. The mechanisms of regulation of capillary blood flow are now largely understood, although not completely resolved.
The development of video systems with recording capability and automated off-line recording of capillary erythrocyte velocities allowed the application of morphological and dynamic studies of cutaneous capillaries in humans. These reopened the field of physiological or pathophysiological questions again for many groups worldwide. Since 1955, many publications on “microcirculation (5423)” and “capillary microscopy (2195)” have been listed in pubmed.
Introduction
The Babylonians and Egyptians already had a rough idea of blood circulation. The papyrus of Ebers contains an anatomical description of the vessels from the heart to all parts of the body, including bleeding after injury to the vessels [1]. Erasistratos of Kos (320–250 B.C.), who worked in Alexandria [2], gave the first description of the blood vessels. Unfortunately, his works were lost in the fire of the Alexandrian library, so that his work is only known from quotations of others who still knew of it or had copies [2]. He has already described the vena cava and the pulmonary artery (“which resembles a vein”), the valves of the heart and he suspected something like “connections” that established communication between the arteries and the venous system. However, according to Rullière, this cannot be accepted as the first description of capillaries [3]. Erasistratos thus missed out on the first description of the circulation [2], particularly because he could not dispense with Hippocrates’ dogma that the arteries were filled with air.
Only W. Harvey (William Harvey, 1578–1657, Royal Physician and Professor of Anatomy and Surgery at the Royal College, London), in his paper “Exercitatio anatomica de motu cordis et sanguinis in animalibus” of 1628, presented the heart as the centre of two blood vessel systems, thus correcting Galenos’ previously valid circulation model [4].
Circulation model of Galenos of Pergamon (quoted according to [5]):
At that time, however, the capillaries had not yet been discovered. Harvey still believed that the blood entered the veins via arterio-venous anastomoses and porosities in the tissue. He himself never saw the blood pass from the arteries into the veins. Throughout his life he remained a fierce opponent; only towards the end of his life did the “circulationists” in Europe gradually gain the majority in Europe [2].
Four years after Harvey’s death in 1661, Malpighi (Marcello Malpighi, 1628–1694, professor of anatomy in Pisa) showed that arteries are connected to veins by a fine network of capillaries (on a preparation of the frog lung) [6].
The term capillary gets back to Andrea Cesalpino (1519–1603), who first used the term “capillamento” for the smallest vessels and also “circulatio” in 1569 to describe the small circulation. Cesalpino was a student of Realdo Colombo (1516–1559, anatomist) in Padua, a discoverer of the pulmonary circulation [7], where Harvey also studied (he first obtained his doctorate in Padua, later again in Cambridge). It can therefore be assumed that Harvey adopted both terms from Andrea Cesalpino. It should be pointed out here, that, in addition to Colombo, both Vesal (Andreas Vesal, German anatomist, 1514–1564 [8]) and Servetus (Michael Servet, Spanish anatomist, 1509–1553 [9]) described the pulmonary circulation before Harvey [2]. The same applies to Ibn an Nafis (1210–1296), whose much earlier writings were lost and rediscovered in 1924 [10].
However, his contemporaries were not convinced until he was able to show the movement of the blood in the capillaries (in the lungs, mesentery and bladder) in a live frogrmbox1\footnote []rmbox1Malpighi still considered the moving particles in the blood vessels to be fat clots. It was van Leeuwenhoek to be the first who described blood cells in the capillaries..
In 1688 (the letter appeared 2 years after his death) van Leeuwenhoek (Antoni van Leeuwenhoek, 1632–1686) confirmed these results with a paper on capillary perfusion in the caudal fin of the glass eel [11]. Thus, Galenos’s idea [5], which had been defended for so long by the “anti-circulationists”, was finally extinguished. The last proof for the correctness of Harvey’s creole model was provided by Stenon (Niels Stenon, 1638–1686, Danish anatomist and theologian): he showed that the heart is a hollow muscle that contracts to pump the blood through the entire vascular system [2].
First observations of the microcirculation
While Malpighi and van Leeuwenhoek were probably the first to publish studies in the field of microcirculation [6, 11], Boerhave (Hermann Boerhave, 1668–1738, professor of theoretical and later practical medicine in Leiden) was the first to study microcirculation in patients [12]. He studied the microcirculation in the human bulbar conjunctiva. His main work “De usu rationicii” was published in Leiden in 1703 [12]. Even today, microcirculation studies in the conjunctiva bulbi of patients are carried out using a slit lamp, a microscope specially adapted to the eye [13–17]. Boerhave described very precisely (see the translation by Levy [18], paragraphs 115, 117) that when the blood flow slows down the erythrocytes form aggregates:
In addition to this clear description of a phenomenon known in modern rheology as a non-Newtonian property of whole blood - namely the dependence of viscosity on the degree of shear due to the occurrence of cell aggregates when the flow is slowed down - Boerhave already described the phenomenon of inhomogeneous blood flow (see [18], paragraph 120), which was much later described as maldistribution by Albrecht Ehrly [19].
It is not widely known that Boerhave has also already described platelets quite clearly (see [18], paragraph 93):
It was not until 1841 and 1846 that descriptions of blood components smaller than red blood cells reappeared in the literature. Gulliver [20] in the UK and Simon [21] in Germany independently published that there were narrow discs in the blood that formed granular clumps with fibrin. Osler then observed in 1874 that these small particles formed aggregates [22]. The name “platelets” was coined by Bizzozero in 1882 [23]. He also described them as circulating in the blood and adhering to vascular lesions. He coined the term “
There are hardly any publications on microcirculation by Boerhaves students, although they learned the technique of investigation from him. Only Albrecht von Haller has his own studies on the blood flow in large and small vessels (but only in animals, not in patients), e.g. on the effects of bloodletting on the blood flow in small vessels 25. He described the blood circulation completely in the sense of his teacher H. Boerhave, i.e. purely mechanically (according to Boerhave, “mechanics are those who explain the functions of the body from its mass, configuration and speed by mathematical calculations based on rational premises or observations” [12]). This can be seen in a work from 1776, in which the term “iatromechanics”, is used for the first time:
Marie Francois Xavier Bichat (1771–1802) stands in complete contrast to this. He wrote in 1818:
“
Bichat was the first to clearly postulate that the blood circulation could not be explained by the laws of mechanics alone. He broadened the scope of vision of early circulatory research to include processes that are now part of biochemistry [27]. Based on Lavoisier’s (1743–1794) experiments on the oxidation of blood, he found that in the capillaries there was a transformation of light red blood into dark red blood, whereas in the lungs the opposite was observed (no explanation was given at the time).
Successive description of the anatomy of the capillaries
Until 1831, it was never quite clear whether the observations reported belonged mainly to the field of microcirculation, which had not yet been defined. This was done in Great Britain by Marshall Hall [28]. His definition - for the field of intravital microscopy - is still widely used today in highly anastomosed vascular areas such as the bulbar conjunctiva, tongue and inner lip:
As long as there are branches downstream, they are precapillary; as soon as there are connections to larger vessels, they are post-capillary vessels.
Improvements in microscopes and dissection techniques - in particular the irrigation technique (i.e. irrigation of freely dissected tissue with physiological fluid at body temperature) published by Thomas in 1878 [29] - allowed increasingly sophisticated studies of the morphological structure (and later the function) of the terminal vasculature. In 1879, it was the German surgeon C. Hueter who carried out capillary microscopic examinations of the inner lower lip of humans [30, 31]. He confirmed some observations already described by Bichat in animal models, e.g. that capillaries can “empty” (they are no longer visible under the capillary microscope once they are only filled with plasma [32]) and the colour change from red to purple in ischemia [30]. The intravascular aggregation of erythrocytes during flow arrest by Boerhave is also mentioned. Overall, his work is little known and not cited. Later, the Tübingen ophthalmologist von Schleich carried out investigations with the Zeiß corneal microscope [33]. However, no further work appears. Since Spalteholz, i.e. since 1888 [34], it has been assumed that the capillary network develops from ever more extensive dichotomous branches of the arterial system and then also flows dichotomously into the venous system (quoted from Illig [35]).
Subsequently, other model ideas were developed that deviated from this classical basic scheme. The first model was the bridge type by Chambers and Zweifach [36], followed by the “micromesh” model by Saunders et al. [37] (see Figs. 1, 2).
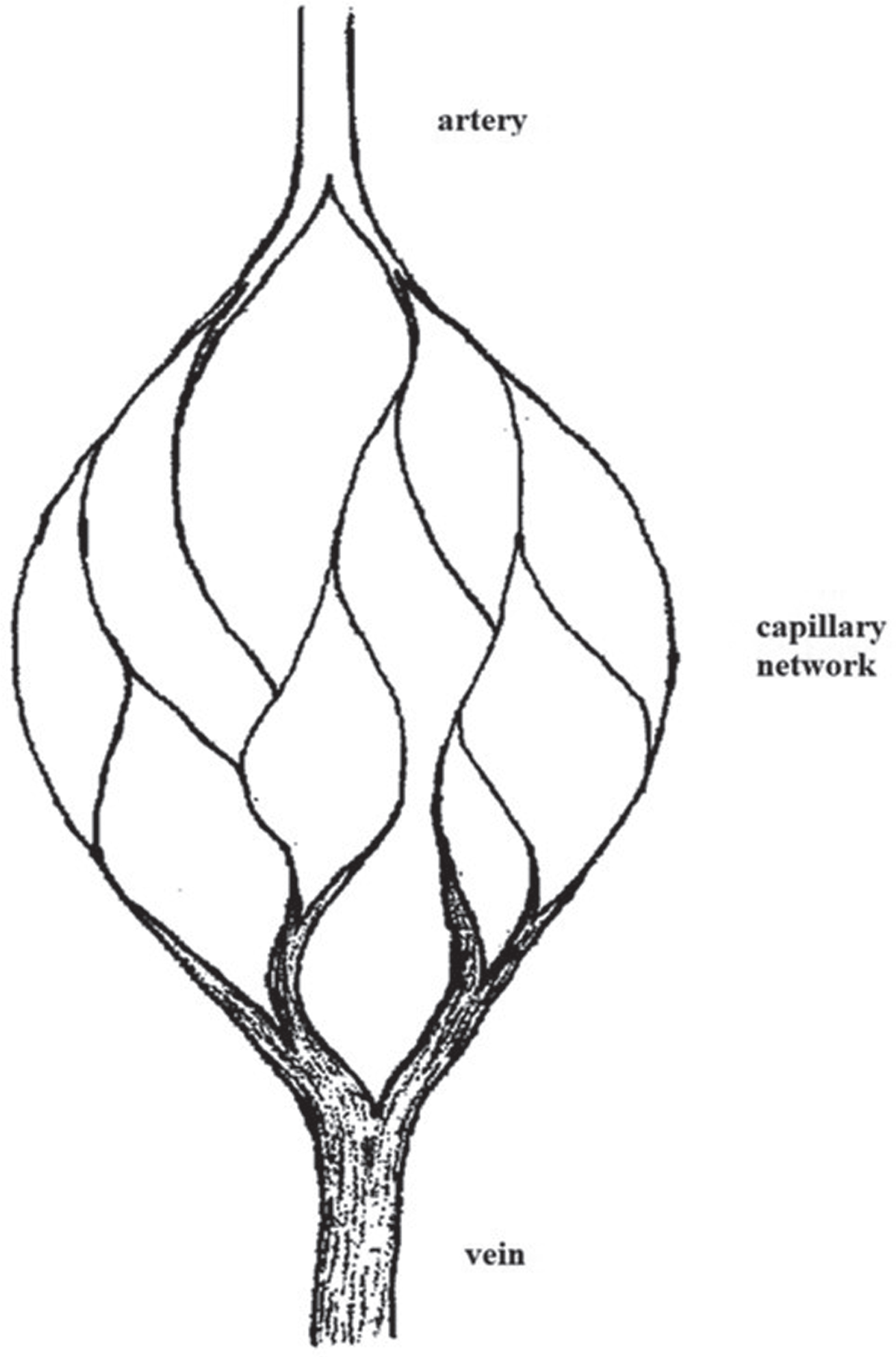
Classical basic scheme of microvascular architecture (modified according to [35]).
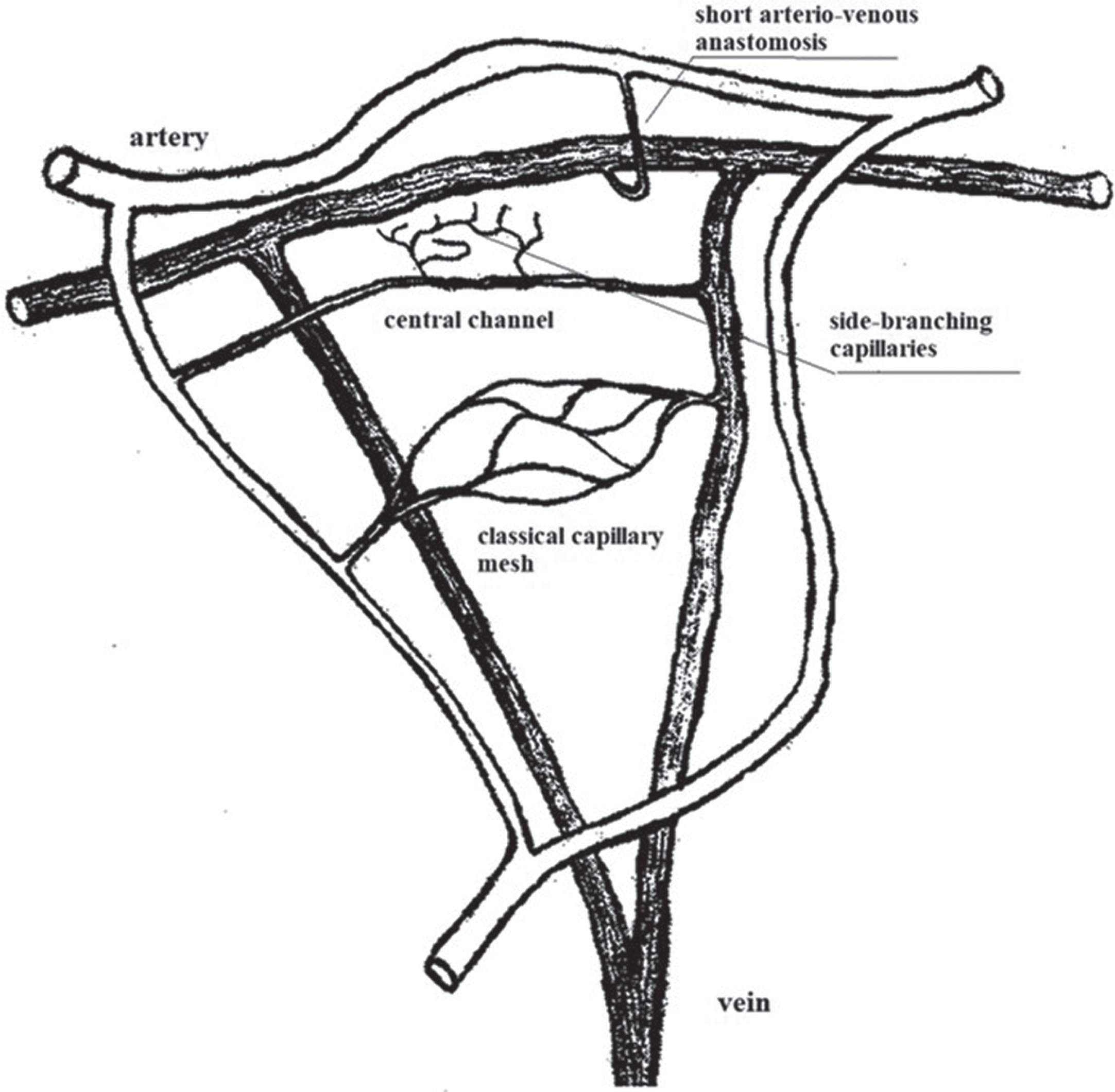
Central or Preference channel type according to Chambers and Zweifach (modified according to [36]).
The bridge-type model is a generalization of the observations made in animal models (rat mesentery, cat mesentery, dog mesentery) killed twice by Chambers & twice by Baez (see Fig. 3). A vascular structure as described in this model has been confirmed by Illig [38] and Baez [39], but not for other animal models or for humans. Table 1 summarizes the work on this subject.
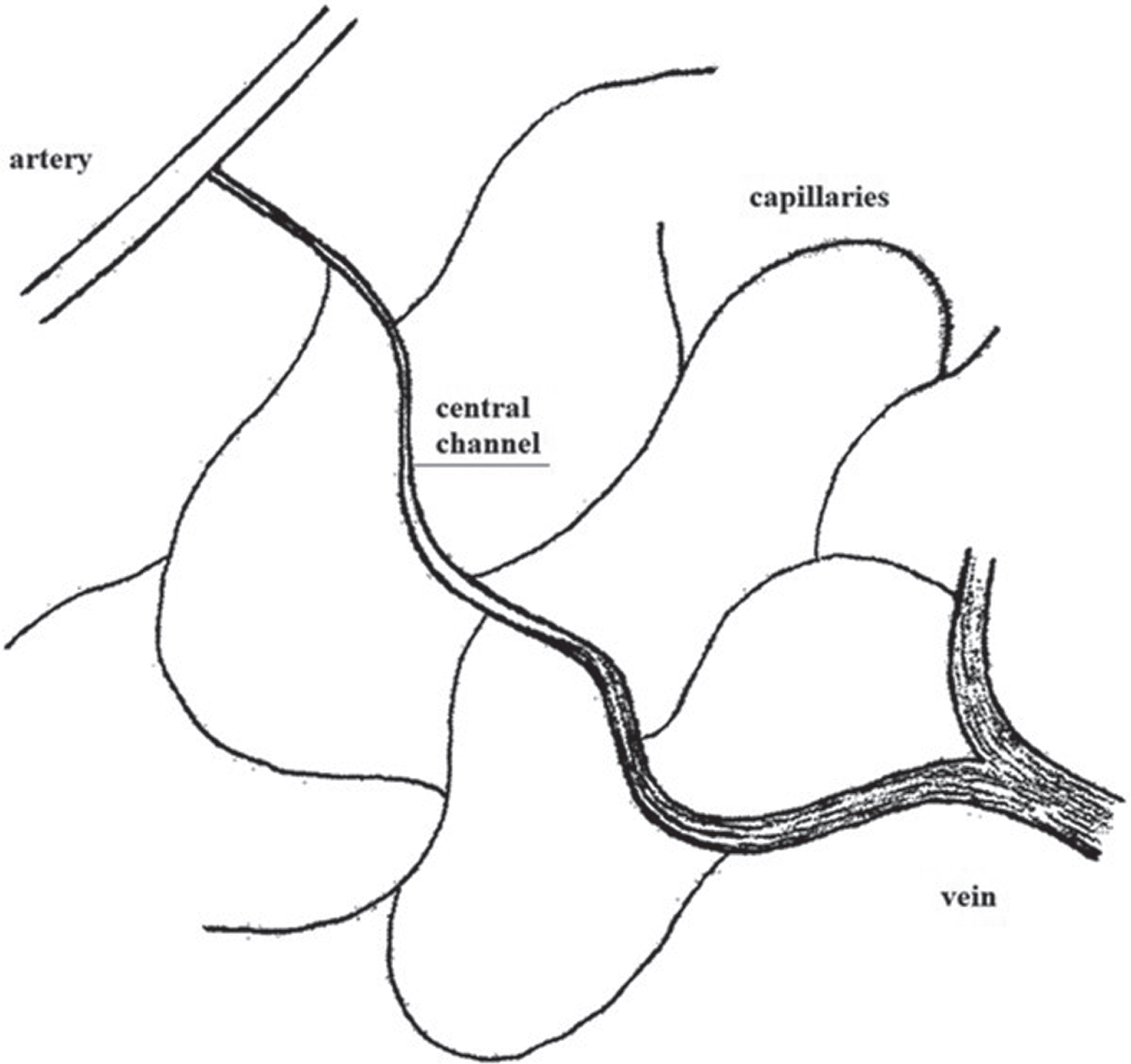
Macro- and micromesh type according to Saunders [37].
Vascularization in different organs of the animal or human with regard to the bridge type described by Chambers & Zweifach
The same applies to the Saunders network model. Again, generalization is not allowed.
Illig therefore concluded that, at least in humans, there is no rigid scheme for the capillary network, but that different characteristic design principles compete with each other [35]. In addition, there are apparently completely non-planar vascular regions, such as the mucous membranes [38].
When comparing work from intravital microscopy and anatomy, it is important to note that histologically, the transition from arterioles to capillaries is caused by the absence of muscle cells - generally, anatomical classification of vessels is based on wall structure. As this is not possible in intravital microscopy, differences in classification or characterization of vessels may occur [40, 46–48]. This definition is not applicable to intravital microscopy. Reliable microscopic detection and differentiation of the terminal arteriole is often very difficult. Therefore, vessels are usually classified intravital microscopically according to their diameter, dichotomous branching or union (i.e. position in the vascular network) and contractility. Despite these characteristics, it is difficult to clearly define capillaries, especially in the mucosal area. Von Hayek described “arterioles” which no longer had a muscular layer, but whose arrangement and circulatory function were to be regarded as capillary end vessels. He called these muscle-free terminal segments “precapillaries” [49]. In addition, there are arterio-venous shunts with very small diameters (in the range of capillary diameters), which are very difficult to distinguish from true capillaries [35, 50]. On the other hand, they can be clearly identified in the skin (e.g. in the nail fold of the fingers or toes) or in the retina [51, 52].
According to Ricker (Gustav Ricker, 1870–1948), the terminal vasculature (now synonymous with microcirculation) comprises the functional unit of the smallest arteries, arterioles, capillaries and venules [53]. In the American literature, the term “capillary bed” is not exactly the same as Ricker’s term “terminal vasculature”. However, the capillary bed includes more than just the capillaries: It includes not only the capillaries but also the entire submacroscopic part of the vasculature, and this is found in each of the definitions used today. According to Schmid-Schönbein vessels with a diameter of less than 30μm [54], according to Illig vessels smaller than 60μm [35] and according to Stromberg & Wiederhelm [55] or Gaehtgen vessels smaller than 100μm [56] are part of the microcirculation.
Illig’s work probably represents the last summary of the state of literature and his own extensive research in the field of microcirculation. It can only be warmly recommended to anyone working in the field.
In addition to the first qualitative descriptions of capillaries, information on their exact characterization, including their function, appeared very early. Bichat [27] was probably the first to observe that some capillaries were only temporarily filled with plasma. This phenomenological observation anticipates the later description of capillary recruitment by August Krogh [57]. The existence of a cell-free plasma layer near the vessel wall was also described in 1840 by a French researcher, Jean-Leonard Marie Poiseuille (1799–1869) [58, 59]. This was clearly confirmed by Heimberger in 1926 by vital staining of the perivascular tissue [50]. He also showed that the cell-free plasma layer becomes progressively smaller during prestatic flow deceleration.
Kiesewetter et al. later showed that during stasis, the diameter of the erythrocyte column and the capillary diameter are the same [60]. Nowadays, the cell-free plasma layer can be measured intravital-microscopically using different stains, which was first described by Alfred Bollinger’s group [61].
In 1850, Volkmann (Alfred Wilhelm Volkmann, German physiologist, 1800–1877) published a scathing critique of the theory of tissue attraction to blood and the ideas of Bichat and Claude Bernard [62]. Although Volkmann’s polemic was a dispute over theories rather than observations, it was so effective that, according to him, the theory of purely mechanical circulation was fully accepted. This was despite the fact that observations at the time already showed that there was more to the attraction theory. Still in 1921, R. Tigerstedt taught the following in the context of the physiology lecture (quoted after R. Müller [63]):
...
In 1921 it was still thought that the circulation of blood in the body was the sole response to the pumping action of the heart. This ignored findings published as early as 1813 and 1821 by A.W. Philipp [64], Thomson [65] and Hastings [66], which clearly indicated that there must be regulation of the microcirculation:
These observations were obviously incomprehensible at the time (nothing was known about local vascular regulation). However, the authors pointed out that the theory of attraction may still be relevant, as these findings clearly argue against a purely mechanically controlled blood flow. In 1868, Ostroumoff described quite clearly how a sudden increase in blood pressure, independent of vascular innervation, causes an immediate vascular contraction and vice versa [67]. However, this work did not find any resonance in the literature. The same applies to a subsequent paper by Ewald, who discussed these findings [68]. Even the classic work by Bayliss on the myogenic hypothesis (later referred to as “blood flow autoregulation”) initially received little attention [69] because of some weaknesses in the experimental part, which were presented in a critique by Anrep [70]. More strikingly, even the findings of August Krogh, for which he received the Nobel Prize in Medicine in 1920 (for his discovery of the mechanisms of capillary motor regulation), were ignored. These included: the transcapillary diffusion, the filtration and reabsorption and the regulation of capillary perfusion depending on the functional state of the tissue surrounding the capillary.
These findings, which originated in the animal model, had therefore not yet been included in physiology courses in Germany, and especially not in the field of clinical microcirculation research, which was gradually taking shape at that time (see chapter Clinical microcirculation research). The first effect of autoregulation on the microcirculation due to changes in blood pressure was described by the Dane Mogens Fog in 1934 [71]. He also associated these changes with the “myogenic theory” reformulated by Bayliss in 1932 [72]. Finally, an autoregulation symposium was held in 1963 where a broad consensus was reached on active and passive mechanisms [73]. In the following years, it was mainly the Scandinavian group around Björn Folkow that further investigated the mechanisms of “functional autoregulation” [73, 74]. The mechanisms of regulation of capillary blood flow are now largely understood, although not completely resolved (see e.g. [75]). Under physiological conditions, blood flow is controlled by autoregulatory and systemic regulatory mechanisms [76]. Blood flow is controlled by several mechanisms: mechanogenic and metabolic autoregulation, humoral and hormonal regulation, nervous regulation, extravascular pressure, and rheological factors.
These regulatory mechanisms are not to be taken as isolated entities but intertwined, interdependent and interrelated factors. In addition, each factor is focused on different parts of the vascular domain.
Mechanogenic autoregulation is the modification of the myogenic action of vascular smooth muscle cells by changes in transmural pressure. The smooth muscle cell is a functional syncytium of spindle-shaped muscle cells spirally attached to the endothelial tube. Their action is most effective in the vascular area of the arterioles. Under normal circulatory conditions, blood flow is controlled almost exclusively by vasomotor activity. The arterioles are thus a centre of blood flow control [76]. In contrast, metabolic autoregulation is characterized by a change in vascular smooth muscle cell activity in response to the concentration of various metabolites in the blood and/or interstitium. Thus, a decrease in the partial pressure of oxygen, an increase in the concentration of pyruvate and lactate, as well as ATP, ADP, AMP and adenosine lead to vasodilation. The accumulation of potassium or phosphate ions also causes smooth muscle cell relaxation and vasodilation [76].
Superimposed to these autoregulatory mechanisms are systemic control loops, on the one hand the nervous regulation. A resting tone of the vessels is maintained by a constant nerve activity of 1–3 impulses/s. An increase in this pulse rate leads to vasoconstriction.
On the other hand, humoral or hormonal regulatory mechanisms intervene in the circulation. Through α-receptors, noradrenaline and epinephrine are vasoconstrictive at high doses, whereas epinephrine is vasodilatory at most β-receptors and at low doses. Angiotensin is a very potent vasoconstrictor, as are kinins such as callidine and bradykinin.
The importance of another regulatory mechanism, which can be attributed to both metabolic autoregulation and humoral-hormoidal systemic regulatory mechanisms, has been recognised in recent decades. The pioneering work of Furchgott & Zawadski, which demonstrated the importance of the endothelium in the regulation of local blood flow [77], triggered an intensive period of research into endothelial cell biology that continues to this day [78]. L. Ignarro then demonstrated that the “endothelial derived relaxing factor, EDRF” was nitric oxide (NO) and showed its role in the human circulation [79]. Together with Ferid Murad, they were awarded the Nobel Prize in Medicine in 1998 for their discoveries about NO and its effects.
Contractility of capillaries
A controversial area since the beginning of microcirculation research has been a functional state formerly known as “capillary contractility”, now known as “segmental constriction of capillaries”. A separate section is devoted to the topic of contractility, as early animal experiments in this area still have to be used.
The theory of the iatromechanics was confirmed for a long time, for example by Wedemayer 1828 [80] but also by J. Müller in 1830 [81]. Even Cohnheim (discoverer of the diapedesis of leucocytes) was skeptical about the intrinsic mobility of the capillary wall in 1867 [82]. He attributed the change in the diameter of the erythrocyte column to a stronger or weaker filling of the capillaries.
In 1871, after observing the frog’s web, Riegel [83] also doubted that contractions occurred in the capillary region, but expressly pointed out that rhythmic contractions of small arteries and veins occur. In 1876 a paper was published by Stricker from Vienna [84] describing spontaneous contractions and dilations of capillaries in the cut nictitating membrane of the frog (Stricker was also the first to describe erythrocyte extravasation from capillaries). Lapinsky, in1899, interpreted Stricker’s contractions as purely passive adaptative processes following changes in arteries or venous lumen [85].
An increasing number of papers have been published describing active lumen changes in capillaries. These include a work by Rouget in 1874 [86] and by the Austrian S. Mayer [87], both attributed the lumen change to the contraction of certain cells, which are stored spindle-like from the outside around the capillary wall (these were first called Rouget cells, today pericytes). This was noted by Tannenberg in 1925 [88], who associated a throttling device at the capillary entrance (which he called a capillary gatekeeper) and as a mechanism with Rouget cells. In 1937, Zweifach described a special form of capillary vessels, which he called “precapillary sphincter” [89]. In subsequent work (see Illig [35] for a detailed description) it was found that this sphincter has the same spontaneous motor activity as the small arteries. According to Nicoll and Webb [47] this sphincter consists of a single cell, according to Lutz et al. [40] of several cells.
In 1903, Steinach and Kahn showed that capillaries were innervated [90], which reignited the debate. It was again discussed that an active contractile capacity does not necessarily require muscle cells. In 1921, August Krogh [91] described that frog capillaries are contractile and can contract independently of arterial blood pressure. They often show spontaneous but generally localized variations in diameter and respond to various local stimuli. The contractile capacity of capillaries was subsequently confirmed in 1922 [92], 1922 [93], 1934 [94], 1940 [95], 1957 [37], 1967 [96], 1969 [97, 98].
Animal studies in recent decades have shown that inflammatory stimuli (application of histamine, serotonin or bradykinin) cause dehiscence between endothelial cells in the post-capillary vascular bed [99–103]. The formation of dehiscence is the result of endothelial cell contraction with protrusion into the capillary lumen [99, 101]. This protrusion can be large enough to slow perfusion to the point of flow arrest [104–109].
The most recent work on this issue comes from Dietrich Lübbers’ group. They showed that electrical stimulation of frog mesenteric capillaries can induce capillary constriction [105, 111], confirming previous studies [35, 48]. Weigelt et al. described the function of these specialized endothelial cells as an additional regulatory mechanism for fine-tuning the microcirculation distal to the precapillary sphincter [108], i.e. in the area of the capillaries themselves. However, the density of these effector cells is rather low at 2% of capillary endothelial cells [108, 112]. They are characterized by a higher content of microfilaments, intermediate filaments and Weibel-Pallade bodies.
Comparable specialized capillary endothelial cells have not yet been demonstrated in warm-blooded organisms. However, there are a few descriptions of endothelial cell protrusions in mammalian capillaries that occur spontaneously [98, 113] or in response to electrical stimulation [105]. Overall, these findings remain controversial [114].
There is, however, an early paper from 1922 which identified a vascular region in the nailfold of patients with eclampsia where capillaries were constricted [115]:
“
This work by Nevermann [115] seems to have been ignored. It is not cited in the subsequent literature on capillary contraction. It seems to be the only work describing this phenomenon in humans over a long period of time.
In 2009, segmental constrictions on human capillaries could then be documented for the first time [116]. As part of a long-term study conducted over 5 years on a group of healthy subjects using intravital capillary microscopy, a segmental reduction of capillary lumina was documented in the nailfold of one participant. Serial images of the same capillaries were taken 1, 3, 4, 5, and 20 hours after the baseline measurement –until complete normalization of the capillary lumen (see Fig 4).
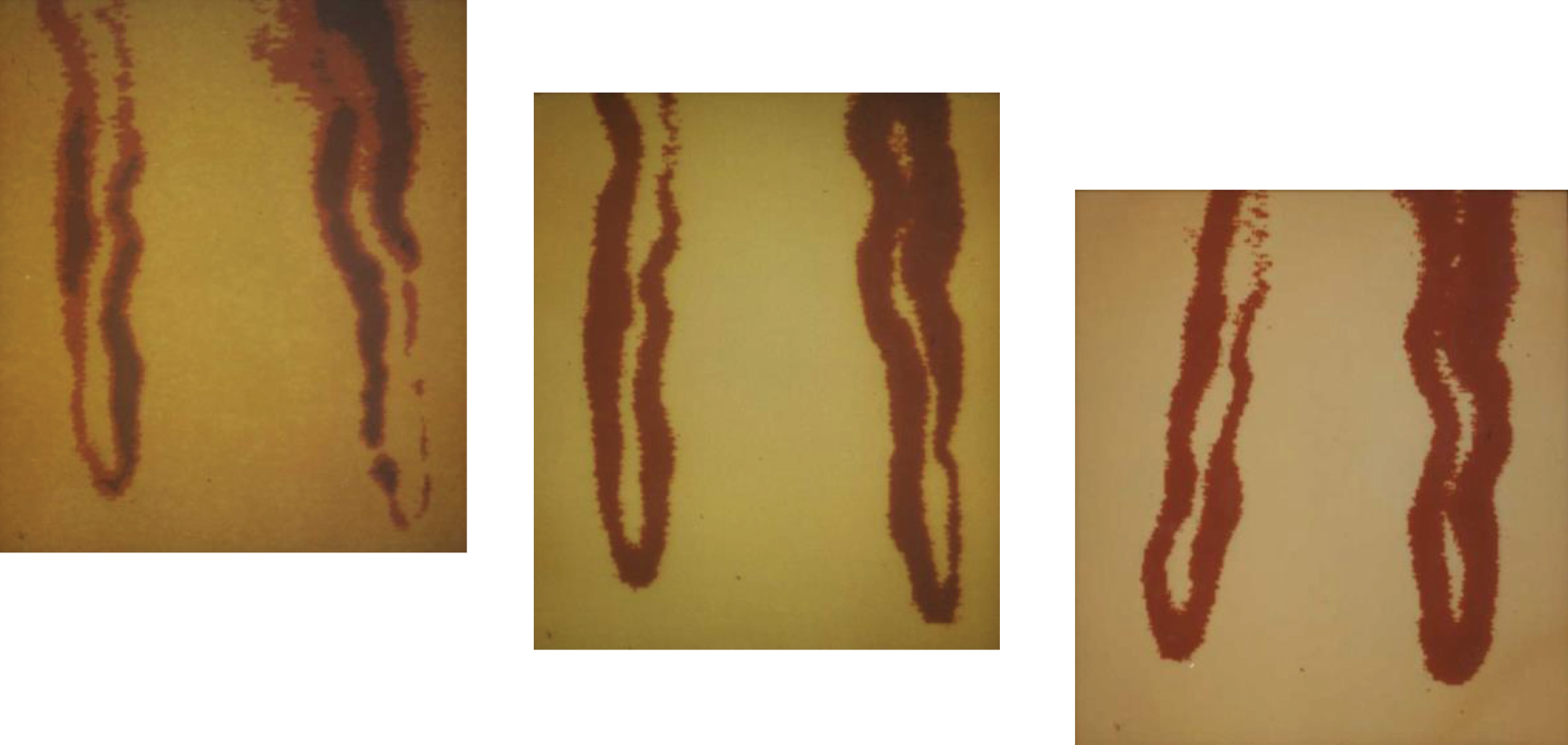
Nailfold capillaries documented at 3 time points (final magnification 1 : 570; modified according to [116]).
It is important to note that the demonstrated findings did not occur in all capillaries at the nailfold and it remained unclear whether such findings were restricted to skin capillaries or could occur in other regions of the body or even systemically. However, it was clearly demonstrated that segmental narrowing of capillary lumina can occur in humans, possibly leading to temporary cessation of perfusion.
Clinical microcirculation research was initiated around 1922, in particular by O. Müller (Otfried Müller, 1873–1945, internist in Tübingen, Germany). He, and his students worked exclusively on the human microcirculation, always studying the capillary form and -reaction in healthy people. The main focus of Müller’s work is the microcirculation research “in sick days” [63]. Results from the animal model - which he calls the rabbit clinic - are only used when human studies are not available [63]. The study of human capillaries at the nail fold had been made possible by two observations: Unna reported in 1881 that a drop of oil could make the skin transparent (quoted after Callandar [117]). The American Lombard was the first to see that the capillaries at the edge of the nail fold run parallel to the surface of the skin and can therefore be seen over a long distance [118] (which makes it possible to measure the velocity of erythrocyte).
At that time the observations made under the microscope still had to be drawn. Müller’s colleagues produced almost artistically coloured drawings of capillaries, which correspond to today’s microscopic pseudocolour representations of the capillary system (but without the same objectivity).
In 1926 Müller commented on a dispute that had flared up in German academia:
“
The background was as follows: the surgeon August Bier had been severely attacked by physiologists when he tried to explain experiments on reactive hyperemia after stasis by the fact that the tissue could obviously control its blood demand (a certain renaissance of Bichat’s theory of attraction [27]). A question that is still discussed in detail today: the transition from the metabolic to the mechanogenic to the NO-mediated hyperemia phase [74, 119].
After O. Müller, Thomas Lewis (1881–1945, British cardiologist) was the first clinician in Great Britain who - in contrast to Müller - dealt with human microcirculation with regard to physiological issues [120].
The year 1928 represents a certain summit in the field of clinical microcirculation research. The public health department of the city of Berlin planned to set up teaching courses to introduce capillary microscopy on a large scale for municipal health care physicians. This was supported by Hoepfner with the demand for capillary microscopic mass examinations of the entire population. There was rightly also public criticism of this project, e.g. by Pototzky:
“
Since 1973, the use of video technology has made it possible to measure capillary blood flow dynamically and with the advent of computer technology, quantitatively. In this context, working groups in Switzerland [121–125], Scandinavia [126–129], Great Britain [130, 131], and Germany are particular important [132–137].
In particular, Bollinger’s group developed methods to study the permeability of the capillary wall, which will not be discussed further in this manuscript [138–140].
Critique and decline of clinical capillary microscopy
In the years between 1922 and 1930, O. Müller repeatedly warned against uncritically conducted and interpreted investigations:
“
Many authors have tried to prove certain diseases by means of morphological abnormalities. However, this was only possible in the rarest of cases, as many of the changes described as pathological also occur in healthy people [35, 141]. By 1950 (quoted after Illig [35]) there were more than 500 often uncritical publications on the subject. In particular, the interpretation of individual findings (e.g. single or double crossing of capillary legs) was considered to be sensitive to certain clinical pictures without the specificity or sensitivity of these features having been sufficiently investigated. This led many users to abandon capillary microscopy as an invalid technique.
New recording and evaluation systems and the revival of clinical microcirculatory research
With the introduction of film and later video technology in the 1950 s and 1960 s, it became possible to perform dynamic investigations. In 1963 Zimmer et al. described a technique suitable for studying the blood flow in human capillaries [142]. A television camera was used to record microscopic capillary images transmitted through a prism system.
Wayland et al. [143] then presented a method for measuring the erythrocyte velocity in the cat mesentery. The groups led by Zweifach [144], Johnson [145] and Intaglietta [146] succeeded in establishing physiological and technical basis for documentation and systematic evaluation of capillaries in the animal model. Today the development has progressed to the point where the velocity of erythrocytes in capillaries is now automatically recorded by means of image computer analysis [131, 147]. Further development and improvement of the analyses is still ongoing [148].
The development of simple video systems with recording capability and automated off-line recording of capillary erythrocyte velocities allowed the application of morphological and dynamic studies of cutaneous capillaries in humans. Clinical studies were first performed by Bollinger [121] and Fagrell [149] (see Fig. 5). These reopened the field of physiological or pathophysiological questions in the field of microcirculation research again for many groups worldwide. Since 1973, many publications on “microcirculation (5256)” and “capillary microscopy (2179)” have been listed in pubmed.
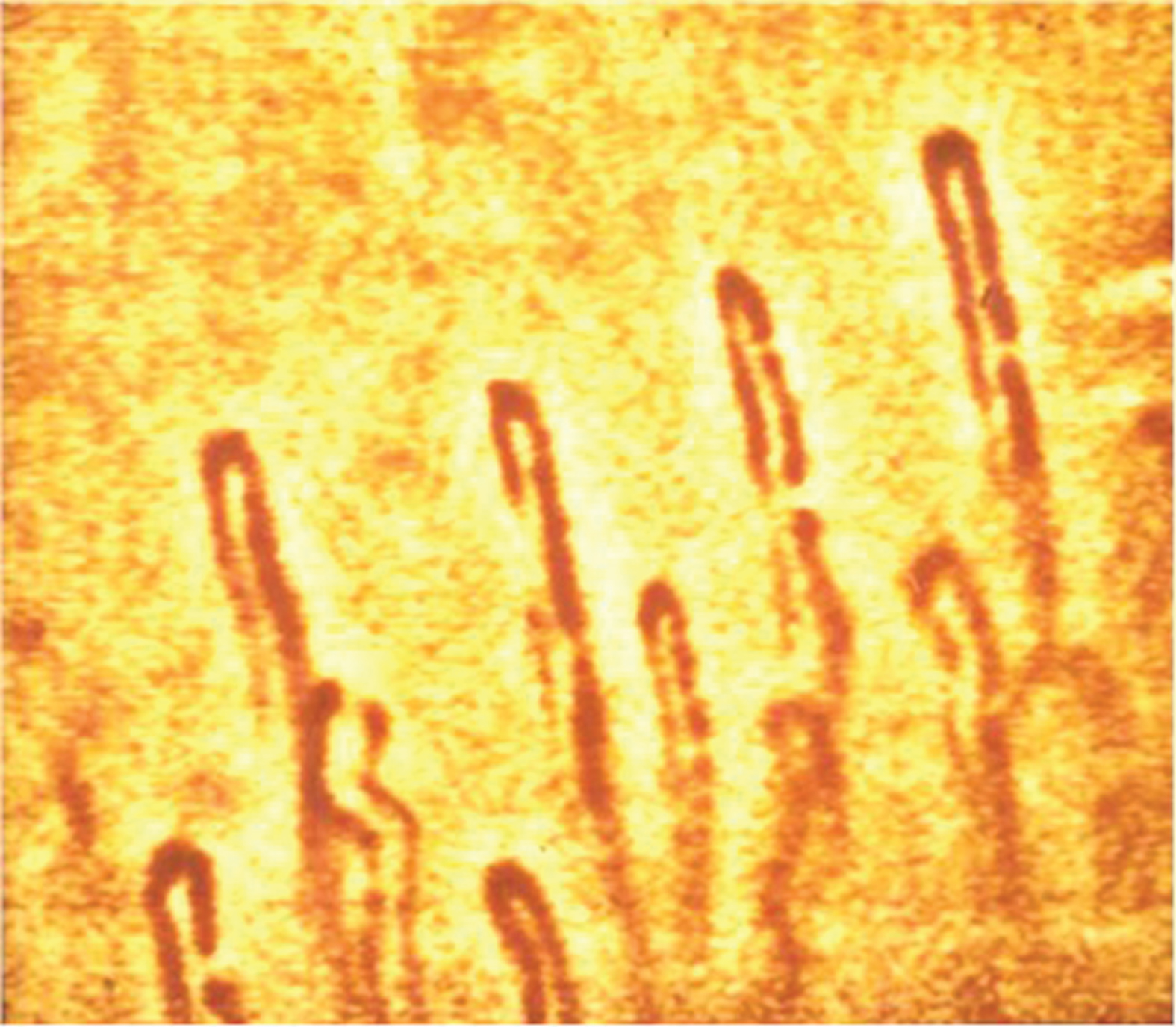
First row of capillaries at the rim of the finger nailfold of a healthy subject (total magnification 1 : 580).
In principle, images of cutaneous capillaries can be taken with any reflected-light microscope, although some adjustments to the specific conditions should be made. The system used by my working group consists of a very stable light microscope on an ACM stand (F. Zeiss, Oberkochen, Germany) with a Neofluar objective (6.3/0.20), an Optovar 1.0–2.0 (for rapid change of the total magnification), a cold light source (to avoid a temperature increase of the observation area) with green filter (for better contrast between red blood cells and tissue), a video camera and a monitor (details of the system are described in [32]). With such systems the capillary system in the nail fold can be visualised very well and also with sufficient magnification. Figure 5 shows the capillaries of the first row of loops at the nailfold edge of a healthy subject (pseudocolor image from a black-and-white camera), as they appear in the anatomy textbook: all arranged at equal distances with the same diameters, although the diameters of the arterial capillary legs appear slightly smaller than those of the venous ones.
With our system we were able to display fine details such as extravasated erythrocytes in subjects with increased capillary permeability and a capillary aneurysm (black arrow in Fig. 6).
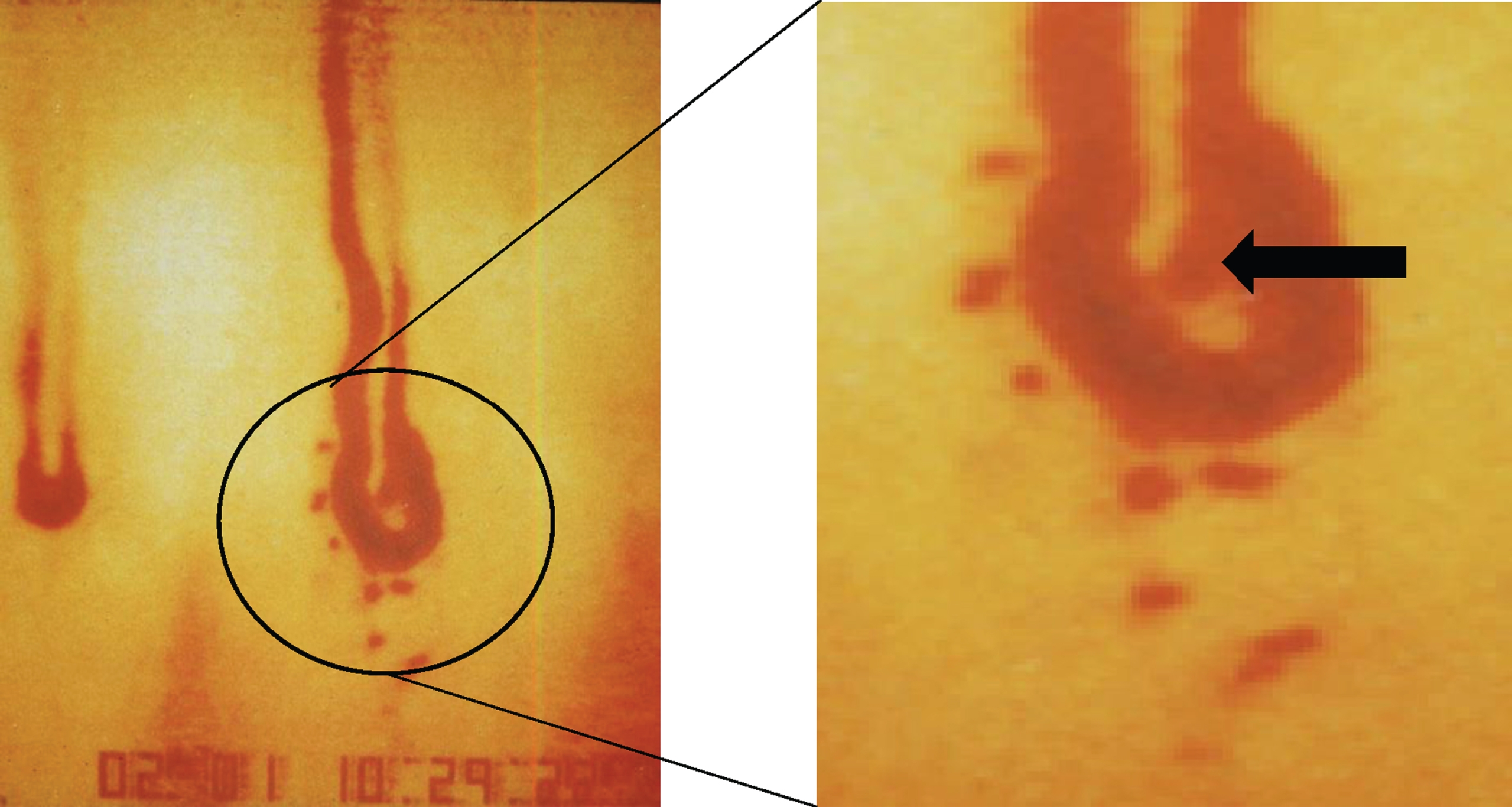
Capillary with extravasated erythrocytes and a small capillary aneurysm, see arrow in Fig. B (total magnification 1 : 580).
In various diseases, very tortuous capillaries are often visible, sometimes with fine ramifications (Fig. 7).
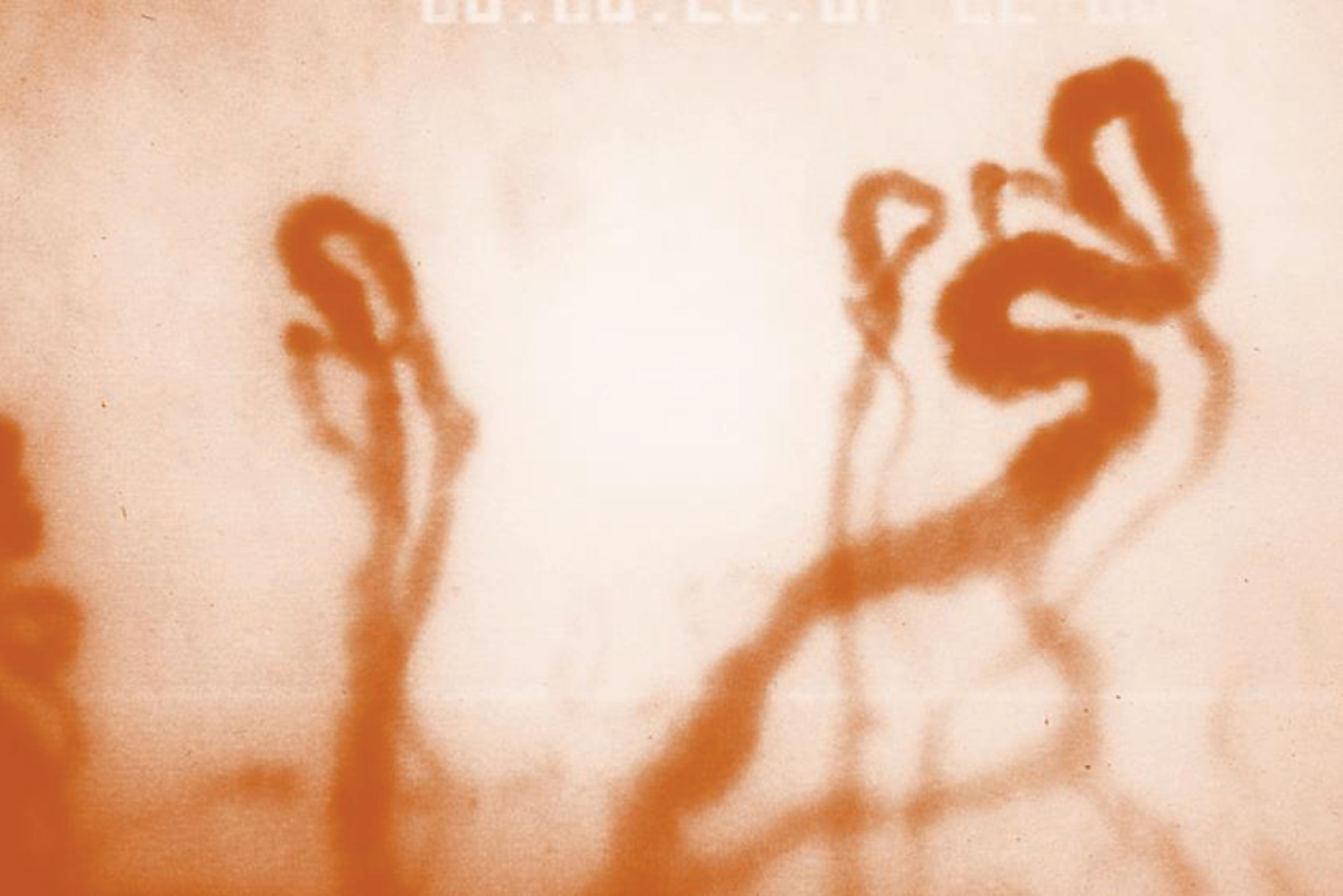
Both qualitative and quantitative parameters of the capillary geometry can be determined. Qualitative parameters include the arrangement of the loop pattern (regular, disordered, incomplete) or the description of conspicuous abnormalities (megacapillaries, capillary-free zones, capillary branches, erythrocyte leakage or capillary aneurysms).
Capillary density (as the number of capillaries per mm of epidermal margin), tortuosity index (for details on recording see [155]), capillary length (length of the visible capillaries) and the erythrocyte column diameter (the capillary diameter cannot be recorded with this method because the cell-free layer near the capillary wall and the capillary wall itself cannot be visualised). Capillary erythrocyte velocities used to be recorded very laboriously by frame-to-frame analysis, but today this is now automated. It is important to note the careful standardisation of the measurement process [156]; see Table 2.
Standardization of cutaneous video capillary microscopy
Under these standardized conditions, the reference range of mean erythrocyte velocity was determined in apparently healthy subjects in three age groups [156]; see Table 3.
Reference range of the mean erythrocyte velocity (
These values are broadly consistent with studies by other working groups: Duprez et al. (0,7 –0,8 mm/s,
In addition to measuring capillary erythrocyte velocity under resting conditions, it is also possible to assess the regulation of capillary blood flow. A possible provocative test of a physiological regulation is, for example, the ischaemia test, which measures the reactive hyperemia to compensate for the oxygen debt that has occurred (sketch in Fig. 8).
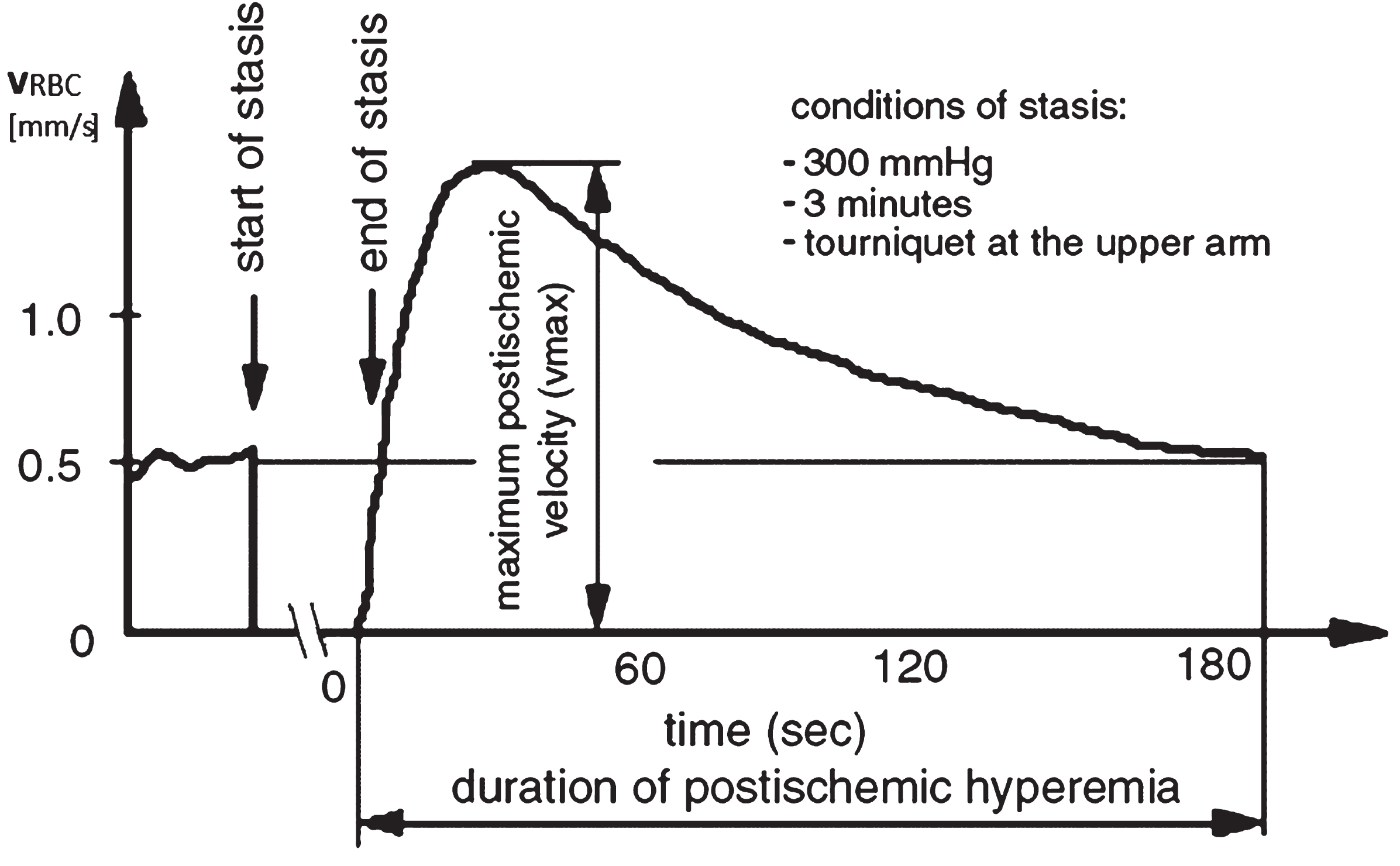
Typical course of a reactive hyperaemia after 3 min of stasis with characteristic parameters in a healthy subject.
To induce reactive hyperemia, ischemia is induced in the vasculature of either the finger or the whole arm for approximately 3 minutes. After the congestion is relieved, erythrocyte velocity is measured repeatedly to quantify characteristic parameters of the process. These include the duration and amplitude of the reactive hyperemia.
Blood circulation was described early on, and the existence of small vessels (still described as pores in the tissue) was suspected as early as 300 BC. It was left to William Harvey [161] to describe the small circulation (which had been described much earlier by Ibn An Nafis [10]), but without knowing about capillaries (Harvey still postulated arterio-venous anastomoses). Capillaries were first demonstrated histologically by Malpighi [6], who also showed the blood flow in the capillaries. Bichat was the first to clearly postulate that blood circulation could not be explained by the laws of mechanics alone. He broadened the scope of early circulatory research to include processes that are now part of biochemistry [27]. It was van Leeuwenhoek who first described blood cells and their movement in capillaries in 1688. Boerhave then introduced microscopy into the clinic [11], marking the beginning of clinical capillary microscopy. The vasculature, which includes the microcirculation itself, was finally defined by Marshall Hall in 1831 [27]. Much later, it was Otfried Müller who carried out systematic studies of the microcirculation in various diseases, always in comparison with healthy people, and summarized them in his famous textbook [63]. Since the 70 s of the last century, clinical examinations of capillaries have been methodologically advanced by the use of video and image computer technology, allowing quantitative analyses to be carried out in the following years.
Morphological changes of the cutaneous capillary system, disorders of capillary perfusion in microangiopathies or therapeutic influences on the nutritive skin circulation can now be reliably detected under standardized conditions. As a result, capillary microscopy is once again being used in universities, clinical centres and (rarely) even in private practices around the world.
However, the results of a capillary microscopic examination of the nail fold should not be overestimated or generalized. It only provides information about the capillary shape and blood flow in the microvasculature of the skin (fingers or toes) or mucous membrane (bulbar conjunctiva, lips, tongue [162–164]). Extrapolation of the results to other areas of the skin (e.g. the ankle area in chronic venous insufficiency) or even internal organs is uncertain. For the diagnosis of generalized microangiopathy, more and more procedures have to be used, as already discussed by O. Müller [63]. For him, the diagnosis at the nailfold alone was too narrow to draw conclusions about systemic diseases from the nailfold alone.
This is still true today. In hypertensive patients, for example, in addition to microangiopathic changes in the skin [165–168], comparable changes in the microcirculation of the skeletal muscle [163, 169] or the retina [170–173] have been demonstrated, so that in this case a systemic microcirculatory disorder can be assumed. In this context, the methods developed by Bollinger and colleagues for the diagnosis of capillary aneurysms and capillary permeability in particular should be mentioned, which were not considered as fluorescence microscopic methods in this review [138].
Footnotes
Acknowledgments
For fruitful discussions on the topic over a long period of time as well as valuable hints for literature research I would like to thank Prof. Dr. Ernst Wenzel (†), Prof. Dr. R.P. Franke, Prof. Dr. B. Fagrell, Prof. Dr. M. Rampling, Prof. Dr. D.W. Lübbers (†), Prof. Dr. Specht (†), Prof. Dr. A. Bollinger (†), Prof. Dr. BW Zweifach (†), Prof. Dr. RJ Xiu and Prof. Dr. A.M. Ehrly.
