Abstract
BACKGROUND:
Necrotizing fasciitis (NF) is a rare but life-threatening condition in which extensive soft tissue destruction can occur very quickly if left untreated. Therefore, timely broad-spectrum antibiotic administration is of prognostic importance in addition to radical surgical debridement.
AIM:
This study evaluates the cases of NF in our hospital during the last ten years retrospectively with respect to the pathogens involved and their antimicrobial resistance. This approach aims to provide guidance regarding the most targeted initial antibiotic therapy.
METHODS:
We performed a retrospective microbiological study evaluating pathogen detection and resistance patterns including susceptibility testing of 42 patients with NF.
RESULTS:
Type 1 NF (polymicrobial infection) occurred in 45% of the patients; 31% presented type 2 NF (monomicrobial infection). The most common pathogens detected were E. coli, staphylococci such as Staphylococcus aureus and Staphylococcus epidermidis, Proteus mirabilis, enterococci, and streptococci such as Streptococcus pyogenes. Twelve percent presented an additional fungus infection (type 4). Ten percent showed no cultivation. Two percent (one patient) presented cocci without specification.
CONCLUSION:
Most pathogens were sensitive to antibiotics recommended by guidelines. This confirms the targeting accuracy of the guidelines. Further studies are necessary to identify risk factors associated with multidrug resistant infections requiring early vancomycin/meropenem administration.
Introduction
Necrotizing fasciitis (NF) is a fulminant inflammation of the subcutaneous tissue including the fascia system as well as deeper layers of tissue. Usually spreading from a small, inconspicuous lesion or injury, necrotizing fasciitis quickly spreads aggressively throughout the tissue and destroys adjacent structures. Nevertheless, a primary focus of inflammation or a portal of entry is not always detected in every case. Primarily, NF often presents as inconspicuous inflammation with overheating, redness, and swelling [1]. However, this is often accompanied by symptoms that are disproportionately pronounced, such as pain or a worsening of the general condition. Severe pain, caused by tissue ischemia, is a characteristic guiding symptom [1, 2]. Approximately two-thirds of cases involve the lower extremity [1]. The prevalence is low with 0.40 cases per 100,000, but the mortality rate is high at 20 or more than 30 percent, depending on the study [3, 4]. NF can be divided into several types with the most common being the polymicrobial form designated type 1 (with a prevalence of 80%). The most common bacteria are gram-positive streptococci, such as Streptococcus pyogenes, a group-A Streptococcus, or staphylococci, such as Staphylococcus aureus, and anaerobes such as Bacteroides fragilis or Enterobacteriaceae [1, 5]. Type 2 infections are monomicrobial in nature and occur in nearly 20% of the cases. The most frequently detected pathogens here are group-A-hemolyzing streptococci. Rarely occurring types are type 3, which occurs after contact with water polluted by vibrio species of bacteria, and type 4, which is caused by fungi such as Candida albicans, especially in patients under immunosuppression [2, 6]. Rapid diagnosis is essential to improve the prognosis of NF, although it is often under-diagnosed especially during the early stages of the disease [3]. In order to influence the course of the disease favorably and to minimize the mortality and lethality, early onset of therapy comprising four pillars is necessary. In addition to radical, and where necessary multiple, surgical debridements, intensive care therapy, early antibiosis, as well as supportive hyperbaric oxygen therapy (HBOT) is essential [1, 3]. Delayed diagnosis and thus delayed initiation of therapy can lead to organ damage, loss of limbs, and increased lethality rates [5]. Extensive debridement in particular should be performed as soon as NF is suspected [7, 8].
However, international guidelines differ in their recommendations on initial calculated broad-spectrum antibiosis [9]. The “Practice Guidelines for the Diagnosis and Management of Skin and Soft Tissue Infections: 2014 Update by the Infectious Diseases Society of America” [1] recommend vancomycin, or linezolid, or daptomycin, each in combination with piperacillin-tazobactam, or a carbapenem like meropenem, or ceftriaxone plus metronidazole, or a fluoroquinolone plus metronidazole. Penicillin G is administered with clindamycin when group-A streptococci are present [1]. The German Association of the Scientific Medical Societies in Germany (AWMF) guidelines “Calculated initial parenteral therapy of bacterial diseases in adults”, subchapter “Severe, (toxin-related) life-threatening necrotizing soft tissue infections” [2] recommends an acylaminopenicillin plus a betalactamase inhibitor like piperacillin / tazobactam or a carbapenem (group-one or two), each in combination with clindamycin or linezolid. Linezolid should be administered especially if MRSA is suspected. A third-generation cephalosporin may also be used in combination with metronidazole. If group-A streptococci are suspected, penicillin G should be used. According to both guidelines, the regimen should be adapted as soon as a pathogen diagnosis including susceptibility data is available [1, 2]. As stated by the German guidelines, vancomycin should not be administered as standard, as the substance is considered to be highly nephrotoxic and the therapeutic target concentration difficult to control. Since patients often already have impaired renal function due to NF or resulting sepsis, vancomycin may worsen this [2]. The in-house regimen of the level-I-trauma center investigated here at University Medical Center Regensburg, Germany, differs slightly from the two guidelines mentioned above, or can be seen as a combination of the two schemes. Until the pathogen has been identified, the Center recommends a combination of clindamycin, penicillin G, meropenem and vancomycin. The rationale for this study was to determine how well our current regimen fits the pathogens we detected and whether adjusting the initial antibiotic therapy would allow for a more targeted therapy that is also gentler on the patient. Figure 1 illustrates the different antibiotic regimens.
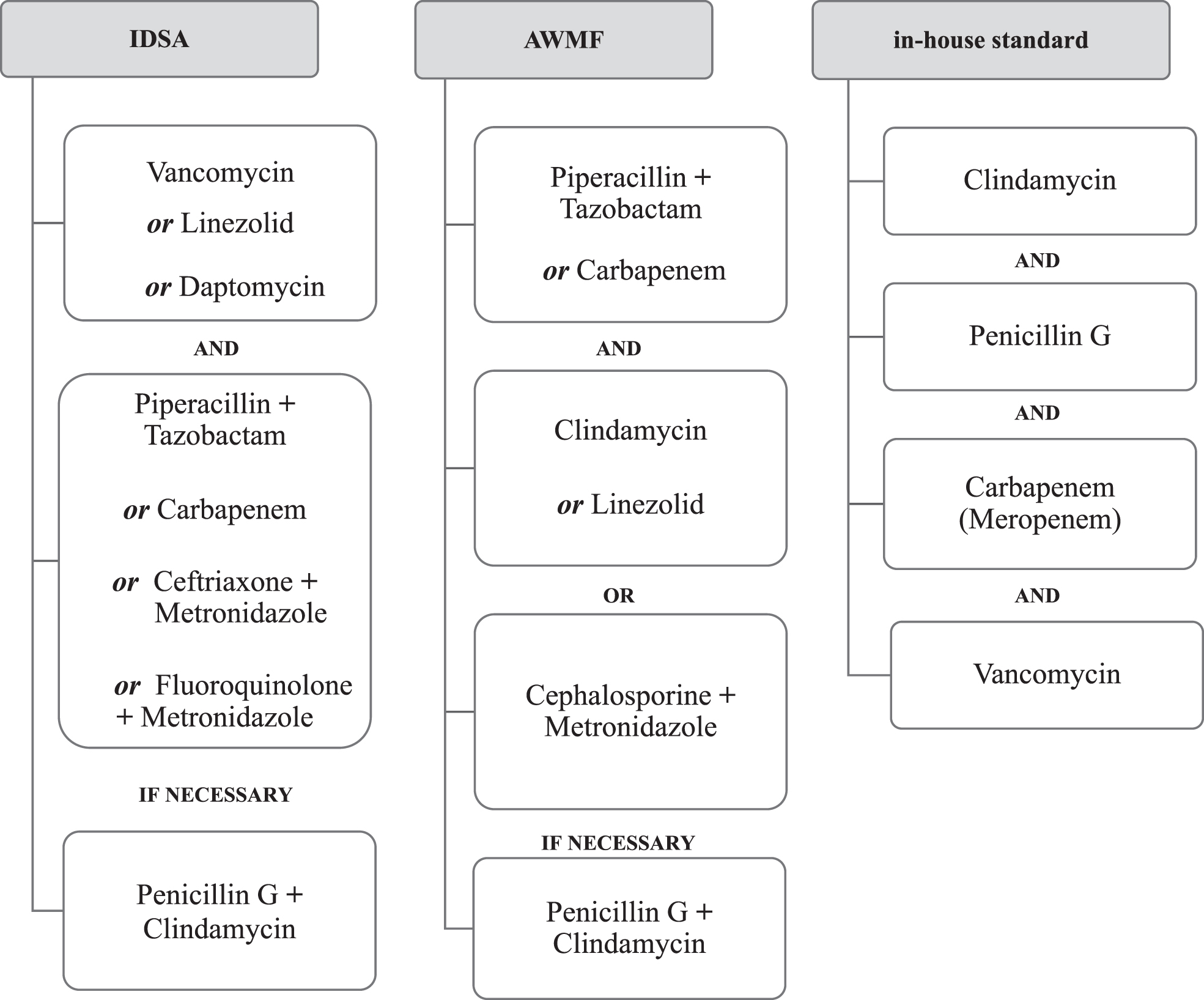
Overview of the antibiotic regimens.
The study was initiated following internal review of the in-house regimen employed in Regensburg. The aim of this study was to evaluate the cases of NF treated in our hospital during the last ten years with respect to the pathogens underlying the disease. To achieve this, we retrospectively evaluated microbiological tests performed on samples collected from infected areas as soon as possible following patient admission. Furthermore, we evaluated susceptibility and resistance tests performed for a range of antibiotics, in order to ascertain how well the recommendations of initial antibiosis correspond to the actual pathogens determined to be prevalent in our clinic.
Ethics
The institutional review board of the University of Regensburg approved this retrospective study (approval number: 23-3249-104). The patient data were processed and stored securely in house in pseudonymized form with access only through a unique assigned identification code. The study was carried out in accordance with the Declaration of Helsinki and in compliance with both local and EU data protection laws.
Patient selection
All patients diagnosed with necrotizing fasciitis in the last ten years at the Caritas St. Josef Hospital Regensburg, Department of Plastic and Aesthetic, Hand and Reconstructive Surgery, academic teaching hospital of the University of Regensburg, were included in the data collection. The survey period was set to ten years retrospectively until January 27, 2023. The period was limited to ten years, in order to determine which pathogens have been most prevalent in recent years and what the current situation is regarding antibiotic resistance. Necrotizing fasciitis is a clinical diagnosis. We only selected patients who were treated in the intensive care unit or underwent HBOT. Patients were selected using the ICD-10 code M72.6- and the procedure codes 8–93 or 8-98f for ICU treatment or 8–72 for HBOT. Patients with multiple differential diagnoses were excluded if, for example, no clear diagnosis could be made at initial treatment onset.
Characteristics of the study group
A total of 43 patients were initially included in the study. However, one patient was subsequently excluded because the date of the microbiology results was after the end of the inclusion period. Thus, 42 patients remained in the study. The mean age of enrolled subjects was 58.38 years (min. 35; max. 89, SD±12.34). Twenty-eight patients (67%) were transferred to our clinic from another clinic, one (2%) from a residential care home for the elderly, and 13 (31%) came from home to our emergency department. Fourteen of the 42 (33%) patients died while hospitalized, after spending a mean of 18 (17.79) days (range 1 to 45 days, SD±16) in hospital. Nineteen (45%) patients were treated with HBOT. HBOT in our clinic is mainly reserved for critically ill patients. Acute renal failure or the acute worsening of known chronic renal failure occurred in 29 patients (69%). The upper extremity was primarily affected by the soft tissue infection in three patients (7%), the lower extremity in 19 (45%), and the trunk including the genito-inguinal region in 18 patients (42%). In the remaining two patients, we could not determine any point of origin.
Microbiological testing
All patients had biopsy samples removed, which underwent microbiological testing to determine the pathogens involved in infection. Samples were collected from the infected areas as early as possible after admission, preferably as part of the workup in the emergency department, otherwise during the first surgical debridement. Where possible, the sample was obtained prior to initial antibiotic administration. In each case, we selected the microbiological examination that was performed closest in time to the date of admission to our hospital. Earlier microbiological examination results, for example from previous hospitalizations, were not considered, as these were not available from all patients and thus no uniform date could be determined. We also ignored whether patients had received antibiotic therapy at other hospitals, as this information was only available for a few of the admitted patients. The microbiology laboratory of University Hospital Regensburg carried out the microbiological evaluation, including testing each pathogen for susceptibility to common antibiotics.
Statistical methods
Statistical analysis was performed using Microsoft Excel for Mac, version 16.16.27, and IBM SPSS Statistics, version 29.0.0.0. The charts and illustrations were created using IBM SPSS Statistics, version 29.0.0.0 and Microsoft PowerPoint for Mac, version 16.16.27. To obtain an indication of the probability of survival in patients with NF and an indication of the effect of HBOT on disease progression, survival analysis was performed by calculating the Kaplan-Meier estimate. Day 179 was used as the observation endpoint, as this was the longest hospital stay for the enrolled patients. Further survival of patients was followed up to ensure that no patient died shortly after discharge as a direct result of NF. As far as we were able to determine, this was not the case. Contact information was not recorded for three patients and, accordingly, we could not follow them up. For the purposes of censoring in the Kaplan-Meier analysis, we assumed that these also belonged to the censored group (“death” event did not occur).
Spearman’s rank correlation coefficient was calculated for one ordinally (grade classification of “renal failure”) and one metrically scaled variable (“age”). Chi-squared was calculated for a nominally scaled variable (“HBOT” or “death”) and an ordinally scaled variable (“renal failure”). The point biserial correlation was calculated when one variable was scaled metrically (“age”) and the other was scaled dichotomously (“HBOT” or “death”). The phi coefficient was calculated for two dichotomous variables (“death” and “HBOT”). Cramér’s V is a correlation coefficient for a nominally scaled variable [10], but, in contrast to the phi coefficient, also applicable if the characteristics have more than two characteristic values (e.g. “type of infection”).
Results
Detected pathogens
Necrotizing fasciitis type 1, the polymicrobial variant, was present in 19 patients (45%). Thirteen patients (31%) presented with the monomicrobial type 2 variant. In five patients (12%), fungi were detected in addition to other pathogens, which is why these patients were designated as type 4. Cultivation resulted in no species detectable in the samples of four patients (10%). In one patient (2%), gram-positive cocci were detected without any further classification or species determination.
Table 1 portrays the pathogens detected and their respective detection frequencies. Physiological species as well as pathogens not normally associated with NF are also mentioned, because even such normally non-pathogenic species might become a problem in NF.
Detected pathogens
Detected pathogens
If the pathogens were determined as susceptible to amoxicillin (plus clavulanic acid if necessary) or ampicillin (plus sulbactam if necessary), susceptibility was also assumed for piperacillin and tazobactam, so this combination has not been explicitly mentioned in the Table 2.
Pathogens detected in type 2 NF (monomicrobial infection)
The significance level was set to α= 0.05. No correlation was found between the age of the patients and the occurrence of death (point-biserial correlation = 0.098). There was also no relevant correlation between age and the onset of renal failure (Spearman coefficient = 0.149). A low phi value of 0.271 was found indicating poor association of the onset of death with the performance of HBOT, although this was statistically insignificant (p = 0.079). A significant chi-squared value of 0.029 was found correlating the onset of death with the degree of renal failure. A Cramér’s V value of 0.236 was found for onset of death and type of infection (types 1 and 2). Table 3 portrays an overview of the correlations.
Correlation variables, coefficients, values, and their respective p-values, significance for a p value < 0.05
Correlation variables, coefficients, values, and their respective p-values, significance for a p value < 0.05
Mean survival was 121.93 days with a 95% confidence interval of 99.5 to 144.36 days.
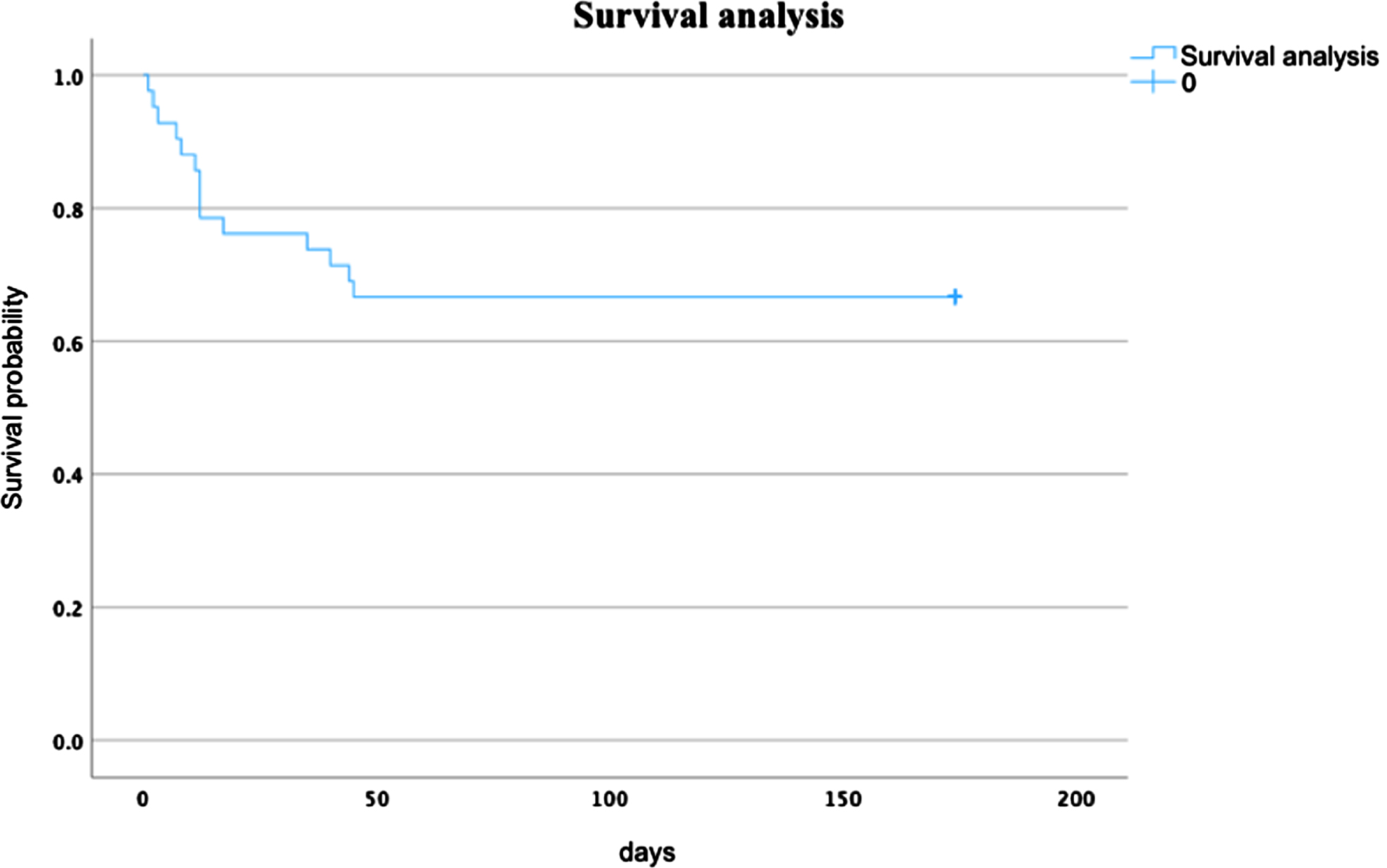
Survival analysis.
With HBOT, median survival was 99.11 days (95% confidence interval 63.31 to 134.5). Without HBOT, median survival was increased at 121.93 days (95% CI 99.5 to 144.36).
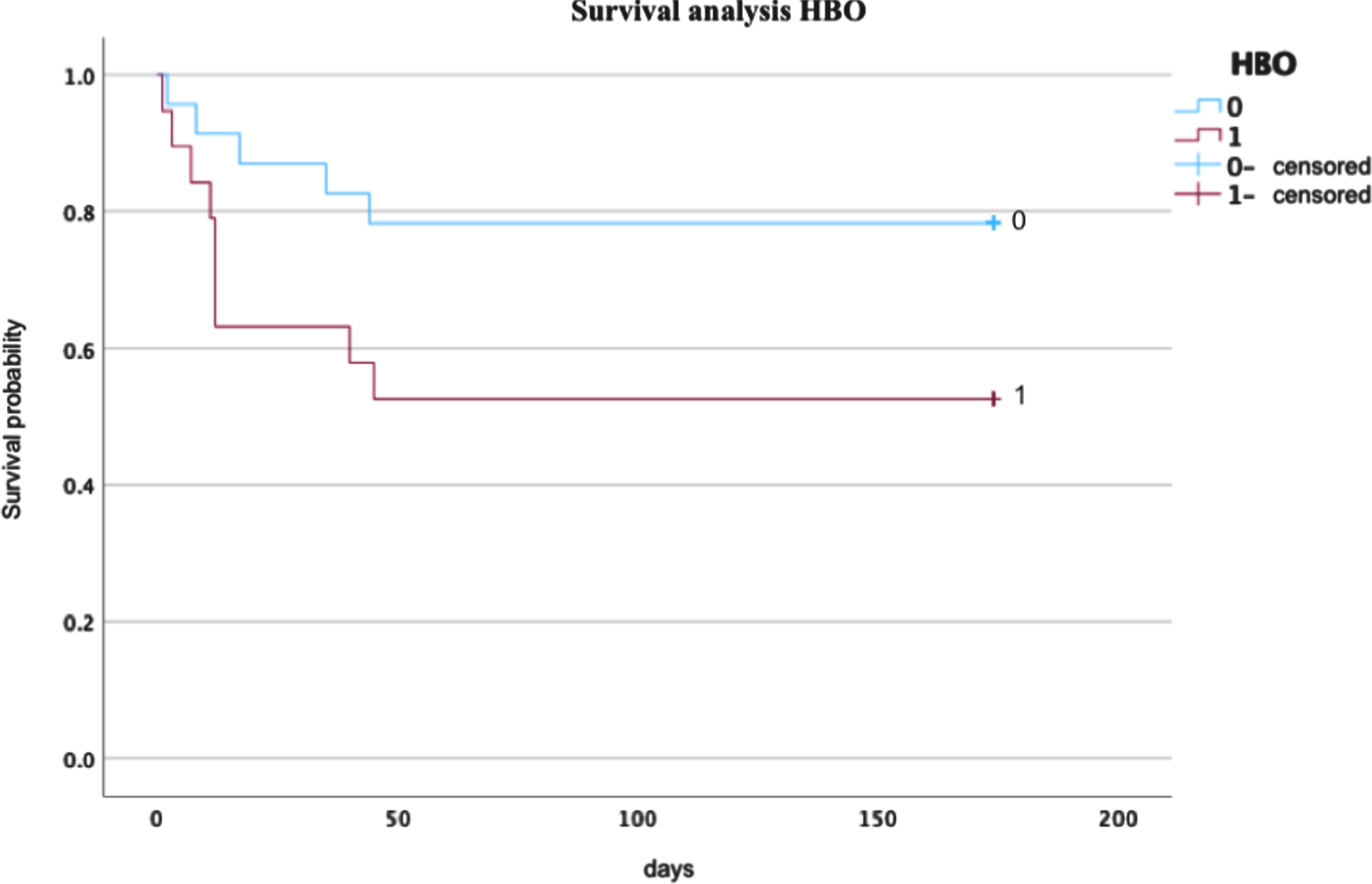
Survival analysis with respect to HBOT. “1” refers to patients undergoing HBOT, “0” without HBOT.
Necrotizing fasciitis is a rare but often fulminant inflammation of the soft tissues [8]. In addition to radical debridement, early calculated antibiotic therapy is of particular prognostic importance [1, 9]. The aim of this study was to evaluate the pathogen spectrum of necrotizing fasciitis seen in our hospital over the past ten years. For this purpose, the microbiology results and the corresponding susceptibility tests were considered, and the results were described accordingly.
The most common pathogens detected in our study were Staphylococcus species (13x), Streptococcus species (13x), E. coli (11x) and Enterococci (10x). This is consistent with findings from several other studies [11, 12]. In addition, Bacteroides were often found in other studies [12], but were only detected 5 times in our study. We also found rarer pathogens not known as typical pathogens in NF, such as Streptococcus anginosus. Rarer pathogens are often more to be expected in immunodeficient patients [13]. However, they have also been detected in immunocompetent NF-patients, especially in a subform of NF, Fournier’s gangrene [13]. Fournier’s gangrene is defined as necrotizing fasciitis of the genitalia, usually following an infection of the urogenital tract or the surrounding soft tissue, with rapid progression, extensive tissue damage, and high lethality [14]. In necrotizing fasciitis, even common, harmless species, such as Staphylococcus epidermidis, can be a trigger or aggravator of the disease. These otherwise harmless species can might have a pathogenic effect, especially in older or previously ill patients [15, 16]. Since our center is specialized in treating NF many patients of our cohort were transferred from other hospitals (67%), we must assume that previous antibiotic treatment might influence the sensitivity of detecting pathogens in microbiological analysis.
Most of the pathogens detected in our patients could be covered by the calculated antibiotic therapy of both the IDSA and the AWMF schemes. The majority of the pathogens could be covered specifically by piperacillin and tazobactam. In the case of streptococci, penicillin G is the antibiotic of choice. The staphylococci detected were susceptible to aminopenicillins and piperacillin, and in some cases required combination with a beta-lactamase inhibitor. An exception to this is Staphylococcus epidermidis, for which vancomycin should be administered. Caution is advised with Enterococcus faecium, which was determined four times, as this pathogen is typically resistant to (acylamino-)penicillins. Vancomycin or linezolid must be chosen here. However, in the case of vancomycin-resistant Enterococcus faecium, daptomycin or linezolid must be administered instead. It is nevertheless essential in clinical management to adjust antibiotics accordingly after receiving the results of susceptibility testing.
Of the six Staphylococcus aureus pathogens detected, one was methicillin-resistant Staphylococcus aureus (MRSA) and therefore only susceptible to, for example, vancomycin or linezolid. One of the reasons why the German guidelines do not recommend the standard administration of vancomycin, while the U.S. guidelines do, is the different susceptibility patterns of S. aureus in both countries: In contrast to Germany, where community-acquired infections account for 18–20% percent of MRSA cases [17], values of up to 78% are found for MRSA in the USA [18, 19].
Pathogens typical in nosocomial infections were detected three times (Pseudomonas aeruginosa twice and Acinetobacter baumanii once) [20, 21]. Both pathogens display increasing patterns of resistance to many common antibiotics and are thus often grouped as multidrug-resistant pathogens [22]. Acinetobacter was only susceptible to carbapenems like imipenem or meropenem but not to piperacillin and tazobactam. Piperacillin and tazobactam, as well as carbapenems, were effective against Pseudomonas aeruginosa. In total, 69% (n = 29) of the patients were admitted from external clinics or nursing homes and therefore had a higher risk of nosocomial infection. Unfortunately, we were unable to determine whether these were longer stays, which would increase the probability of nosocomial infection, or short initial admissions before immediate transfer to our hospital. It would be important to find out in further studies whether the pathogen spectrum in NF during long hospital stays differs from the standard expected spectrum and whether more multi-resistant pathogens occur. This would help to adjust the calculated antibiosis even more precisely.
Seven pathogens were found that could only be treated with vancomycin. Vancomycin is a glycopeptide antibiotic and is effective as a broad-spectrum antibiotic against gram-positive pathogens. However, resistance to vancomycin is also steadily increasing [23]. Especially in the intensive care unit, the increasing resistance to antibiotics represents a growing risk to patients [24, 25]. Vancomycin is also suspected of having a nephrotoxic effect [2]. Increased levels of reactive oxygen species and oxidative stress appear to be a major cause [26, 27]. This effect could be enhanced especially in combination with piperacillin-tazobactam, which is commonly used in soft tissue infections. Although the extent of nephrotoxicity of vancomycin remains unclear, discontinuation of vancomycin treatment should be considered in patients with a worsening of kidney function or the development of acute renal failure [26]. During the course of NF, many to most patients require intensive care [28]. In our study, 29 of the 42 patients (69%) suffered from worsening renal function. The NF patient population is at risk of potential organ damage, including renal damage, and therefore special attention should be paid to protecting organ systems, for example, through the use of medical treatments with the lowest possible rates of side effects. Thus, the German AWMF guidelines do not recommend vancomycin as first-line therapy [2], whereas the IDSA guidelines still do [1]. However, it is unclear to what extent renal and organ damage is aggravated by the administration of vancomycin. This needs to be carefully weighed. Overall, and especially if vancomycin is not used, it is important to carry out the microbiological and susceptibility testing as early as possible. Where necessary, appropriate antibiotic therapy can always be initiated quickly.
In all the national and international antibiotic guidelines considered here, piperacillin-tazobactam or a carbapenem such as meropenem are administered equally. In our analysis, there were three of 71 pathogens (4%) that were covered only by meropenem and not by piperacillin-tazobactam. Again, it would be controversial to prescribe piperacillin-tazobactam as standard and switch to a carbapenem only after appropriate susceptibility testing. Carbapenems are considered as reserve agents in the face of increasing resistance to many of the existing classes of antibiotics worldwide. They are often one of the last effective classes of antibiotics in multi-resistant bacteria [29]. To prevent further development of resistance, many studies are underway and more are needed to find suitable alternatives to the standard regimen of meropenem [30]. Therefore, the option of administering meropenem only after appropriate susceptibility testing should be considered to avoid overuse of the reserve drug. On the other hand, in the often very severe course of NF, it may be too late to wait for resistance testing with meropenem. Further studies are necessary to identify risk factors associated with multidrug-resistant infections requiring early administration of vancomycin and meropenem. Risk factors in other multidrug-resistant diseases, such as prolonged hospitalization, older age, or previous use of antibiotics [31], need to be considered and should be investigated. A limitation of this study is that underlying risk factors of the patients were not considered. It would be interesting to determine how the spectrum of pathogens varies with patient characteristics such as obesity, diabetes mellitus, or other pre-existing conditions. This may allow initial antibiotic therapy to be tailored more accurately to individual patients with unique risk factors. For this purpose, more detailed data on the existing potential risk factors and on the pathogens that occur should be investigated in further studies. Another limitation associated with the data collection for this study was the inability to determine retrospectively for all patients whether they had received antibiotics in previous clinics and, if so, which substance had been administered. This could be rather important, since the spectrum of pathogens presenting after initial antibiotic therapy may differ from the original spectrum.
Hyperbaric oxygen therapy (HBOT) has been considered as an adjunctive treatment in NF, although its effectiveness in treating this condition is still a subject of debate.
HBOT is defined as breathing 100% oxygen in a pressurized chamber, which increases the dissolved oxygen in the bloodstream by a factor of up to three, thus increasing tissue oxygenation. HBOT is considered to combat NF through several mechanisms. First, the increased oxygen levels delivered during HBOT enhance tissue oxygenation, potentially inhibiting the growth and survival of anaerobic bacteria commonly associated with NF. Second, HBOT may stimulate the formation of new blood vessels (neovascularization) in affected tissues, which may in turn promote wound healing and tissue repair [32].
Moreover, HBOT has immunomodulatory effects, such as enhanced white blood cell function and reduced inflammation, which could potentially aid in fighting the infection. Nevertheless, the use of HBOT in NF is controversial, and the evidence regarding its efficacy is limited and conflicting. Some studies have revealed improved outcomes, such as reduced mortality rates and tissue loss, while others have not found any significant benefits [33]. Mladenov et al. published results of a retrospective single-center study including 192 patients with NF or Fournier’s gangrene to assess in-hospital mortality and outcome dependent on patient, disease and treatment characteristics with or without HBOT. They reported that HBOT contributed to improved outcomes [34]. It is important to note that HBOT should not be used as a sole or primary treatment for NF. The mainstay of treatment for this condition is early and aggressive surgical intervention, including debridement, administration of appropriate antibiotics, and a high-end intensive care setting. HBOT may be considered in specific cases, particularly when extensive tissue involvement is present.
The survival time analysis of our study revealed that with HBOT, median survival was 99.11 days (95% confidence interval 63.31 to 134.5). Without HBOT, median survival was increased at 121.93 days (95% CI 99.5 to 144.36). We believe this to result from the fact that HBOT in our clinic is mainly reserved for critically ill patients with greater mortality rates.
Conclusion
In conclusion, this study aimed to evaluate the pathogens involved in NF cases and their antimicrobial susceptibility patterns. The results indicated that the most common pathogens detected in NF patients were Staphylococcus aureus, Streptococcus species, Escherichia coli, and Enterococci. The majority of these pathogens proved susceptible to antibiotics recommended by guidelines, such as piperacillin-tazobactam, penicillin G, and vancomycin. However, it is worth noting that vancomycin resistance is increasing, and caution should be exercised in its administration owing to the potential nephrotoxic effects. The findings of this study support the targeting accuracy of current guidelines on initial antibiotic therapy in NF cases.
Following the results of our study, we decided to change the regimen in our level-I trauma center investigated here. We decided to use following combination: Piperacillin/tazobactam, clindamycin, penicillin G. We will adjunct primary vancomycin only in cases of transfer between hospitals or transfer from inpatient facilities, as well as previous hospitalizations.
Further research is needed to investigate the prevalence of multidrug-resistant infections in NF patients, particularly those with prolonged hospital stays or other risk factors. This would aid in refining the antibiotic therapy guidelines to ensure appropriate coverage of emerging resistant pathogens. Additionally, future studies may explore alternative antibiotic regimens to reduce the reliance on reserve agents such as carbapenems.
Monitoring the prevalence and trends of multidrug-resistant strains in necrotizing fasciitis cases over time can shed light on the evolving resistance patterns. Understanding the emergence of multidrug-resistant pathogens can guide infection control measures and antibiotic stewardship efforts to prevent the spread of resistant strains.
Author contributions
VB: project development, data analysis and interpretation, manuscript writing, critical revision, final approval EE: data collection, data analysis and interpretation, manuscript writing, FH: critical revision of the article, final approval. SP critical revision of the article, final approval LP: project development, critical revision, final approval, MP: project development, statistical support, critical revision of the article, final approval.
Funding
This research received no external funding.
Institutional review board statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the institutional review board of the University Hospital of Regensburg (protocol code: 23-3249-104; 30. January 2023).
Informed consent statement
Considering the retrospective nature of the study and that the data were pseudonymized, the IRB did not deem patient consent as being necessary.
Data availability statement
Data are available on request from the corresponding author.
Conflicts of interest
The authors declare that there are no conflicts of interest.
