Abstract
BACKGROUND:
Biocompatibility describes the influence of materials on their biological environment. Implant material in the human body can cause a foreign body reaction and the formation of a capsule around the foreign material. Since capsular formation is the most frequent issue after breast-implant insertion, knowledge and awareness of biocompatibility is crucial, especially since worldwide, breast augmentation continues to be the most popular plastic surgery, with over 1.6 million procedures performed in 2020, according to surveys by the International Society of Aesthetic Plastic Surgery (ISAPS).
MATERIAL AND METHODS:
This study includes 80 capsular samples of female patients who underwent revision surgery after breast-implant insertion at the University Hospital Regensburg. Capsules of breast implants with different surface structures (smooth, textured and polyurethane-coated) and shapes (round-shaped, anatomically-shaped) were analyzed histologically after hematoxylin-eosin-staining in respect to capsular thickness and layer formation.
RESULTS:
Capsular thickness and layering showed a statistically significant difference between polyurethane-coated and smooth as well as polyurethane-coated and textured implants. Capsules around polyurethane-coated implants presented greater thickness. However, the difference between smooth and textured implants was not statistically significant. Furthermore, the shape of the implants also indicated a statistically significant difference in capsular thickness. Implants of anatomical shape resulted in a thinner capsule than round-shaped breast-implants.
CONCLUSION:
In conclusion, this study demonstrated a thicker capsule around polyurethane-coated breast implants and no difference in capsular thickness between smooth and textured breast implants. Anatomically shaped breast-implants presented a thinner capsule than round shaped breast-implants.
Introduction
Worldwide, breast augmentation continues to be the most popular procedure in plastic surgery, with over 1.6 million procedures performed in 2020, according to surveys by the International Society of Aesthetic Plastic Surgery (ISAPS). In principle, there are many different indications for the implantation of breast implants. Breast implants are used not only for aesthetic reasons such as mammary augmentation, but also for the correction of congenital malformations of the breast and for breast reconstruction after mastectomy due to cancer or benign masses. Since breast reconstruction is of great psychosocial importance for affected patients, indications and therapeutic options should be adapted according to the needs individually.
Knowledge of biocompatibility of implants in human bodies is essential in order to ensure safety and reduce complications [1]. Biocompatibility describes the influence of materials on their biological environment [2, 3]. Implants introduced into the human body can cause a foreign body reaction and the formation of a capsule around the foreign material [4, 5]. Among other parameters, certain surface properties influence the biocompatibility of an implant [6]. Overall, polyurethane is one of the most widely used material in the manufacture of medical devices, due to its generally recognized good biocompatibility [7]. The worldwide most commonly used breast implants have smooth, textured, or polyurethane-coated surfaces [8]. Since capsular formation is the most frequent issue after breast-implant placement, knowledge and awareness of biocompatibility is crucial. Capsular contraction can cause discomfort to the patient, deformation of the breast and/ or pain, potentially resulting in additional surgeries. Thus, causing further distress to the patients.
Histologically, the immigration of immune cells immediately after surgery leads to capsular formation [9]. The goal of this acute inflammatory reaction is phagocytosis of the foreign material. Failure to do so leads to the activation of end-inflammatory cells, such as macrophages, monocytes and lymphocytes. This is followed by the release of proteases, chemotactic factors, complement factors, coagulation factors, cytokines, and growth factors [10]. These factors lead to the proliferation of fibroblasts, which synthesize more collagen, resulting in the formation of a capsule [11–14].
The aim of this study was to examine the influence of implant surface and shape on capsular formation. Polyurethane-coated implants were compared with smooth and textured implants. Furthermore, capsular formation was analyzed for round and anatomically shaped breast-implants.
Materials and methods
This study includes data of 80 female patients who underwent revision surgery after breast-implant insertion at the University Hospital of Regensburg between 2006 and 2019. Only patients for whom information on the duration of implantation, surface structure and implant shape were available were included.
The average age of the women was 47.9±12.9 years at the point of revision surgery.
During the revision surgery there was capsular tissue collected and prepared for histological analysis.
Biocompatibility
The biocompatibility of breast implants with different surface structures and shapes was investigated through histopathological analysis. Thus, capsular samples from 31 smooth, 41 textured and 8 polyurethane-coated breast-implants were analyzed.
Regarding the implant shape some cases needed to be excluded due to absence, or lack in quality of the capsule samples. Thus 23 capsular samples of round-shaped breast-implants were compared with 19 anatomically shaped breast-implants.
Postoperative the capsule samples were fixed in buffered formalin solution (3,7%), dehydrated using a tissue processor (Shandon Hypercenter XP/TH.SCI) and then embedded in paraffin (HistoStartrademark ThermoScientifictrademark). Slices from 2–5 μm thickness were cut using a microtome (Microm HM 340E, Thermo scientific) and placed on a microscope slide (Microscope Slides 76×26 mm/3×1 Inch, Engelbrecht medicine and laboratory technology GmbH Germany).
Hematoxylin-eosin staining was performed according to standardized automated procedure. Then, cover glasses were applied and the slides were digitalized using a Slide-Scanner (Pannoramic 250).
Capsular thickness was measured using the 3DHISTECH CaseViewer. To avoid mismeasurements due to oblique cuts through the capsule (which would measure a greater capsular thickness) the minimal capsular thickness was measured in five times magnification. After overview of the entire sample, ten fields of measurement were defined. The fields were chosen systematically throughout the entire sample, to receive the minimal capsule thickness. Furthermore, homogeneity of the capsules and capsular layering were recorded depending on cell nuclei density (Figs. 1–3).
Overview of a capsule sample after hematoxylin-eosin staining.
Capsule sample in five times magnification.
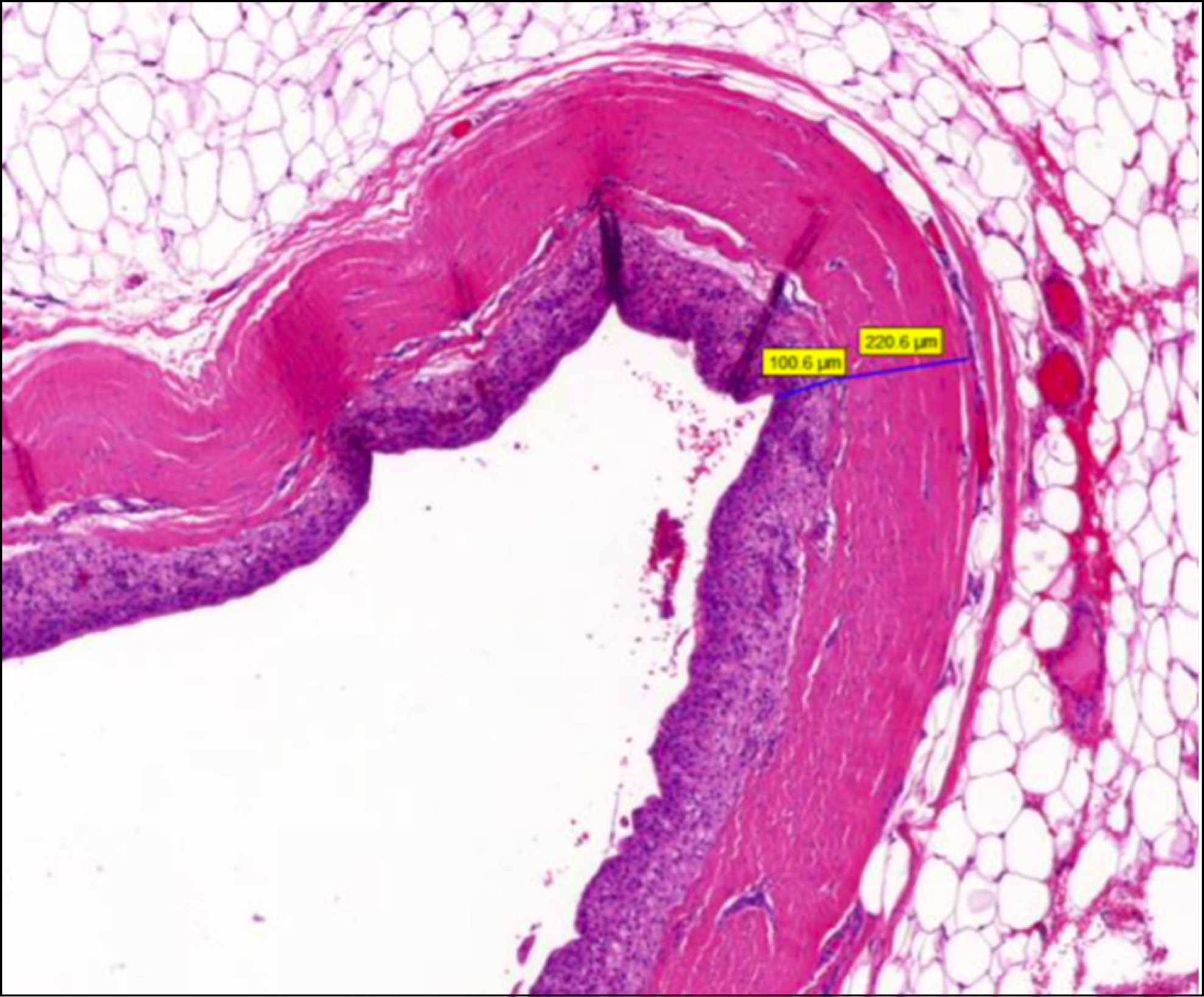
Measurement of a capsule sample. In this case, there is a capsule with two layers. The inner layer contains more cell nuclei. This can be recognized by the blue coloration.
Statistical analyses were performed using SPSS Statistics for Macintosh, Version 25.0 (IBM, Armonk, New York, USA).
Evaluation of the population were described using descriptive analysis.
Capsular layering was analyzed using a Pearson's chi-square test. Furthermore, pairwise comparison including a Bonferroni post-hoc correction was performed.
Capsular thickness was analyzed using a Kolomogrov-Smirnoff test. Statistical significance was evaluated by Kruskal-Wallis- or Mann-Whitney-U test. Paired comparison between two groups investigated the null-hypothesis that capsular thickness is not influenced through a different surface structure of the breast-implant.
Results
The time period from breast-implant insertion to revision surgery was 6.5±4.83 years for smooth implants, 4.27±4.82 for textured implants and 6.31±5.75 years for polyurethane-coated breast-implants.
The time period from breast-implant insertion to revision surgery averaged to be 10.87±5.09 for round and 9.89±4.19 for anatomically-shaped breast implants.
The mean volume of the breast-implants was 327.56±115.25 ml.
78,0 % of the breast-implants were placed subpectoral, 20,7 % were placed epipectoral, and 1,3 % were positioned underneath a flap.
Surface structure and capsular layering
The examination of 31 smooth, 41 textured and 8 polyurethane implants showed capsular layering in 2 smooth an 11 textured breast-implants (Fig. 3). However, no sign of layering was found in capsules around polyurethane implants. In 12 capsules formed around 3 smooth, 6 textures and 3 polyurethane covered implants layering was inhomogeneous.
There was a statistically significant difference of capsular layering depending on the implant surface structure using a Pearson's chi-square test (p = 0.027). A Post-Hoc-test was used for the Analysis of Variance. The value of the corrected type I error was 0.0056 calculated with a Bonferroni-Correction 0.0056.
Surface structure and capsular thickness
Histological measurements of capsular thickness from 31 smooth, 41 textured and 8 polyurethane implants showed no normal distribution, since the outcome of the Kolomogrov-Smirnoff test was lower than the defined p-value of 0.05. Further analysis, using the Kruskal-Wallis-test, indicates a statistical significant difference between the capsular thickness and the surface structure of implants (p = 0.015). Mean rank of capsular thickness in smooth implants was 35.77 μm, in textured implants 39.80 μm and in polyurethane covered implants 62.38 μm (Fig. 4).
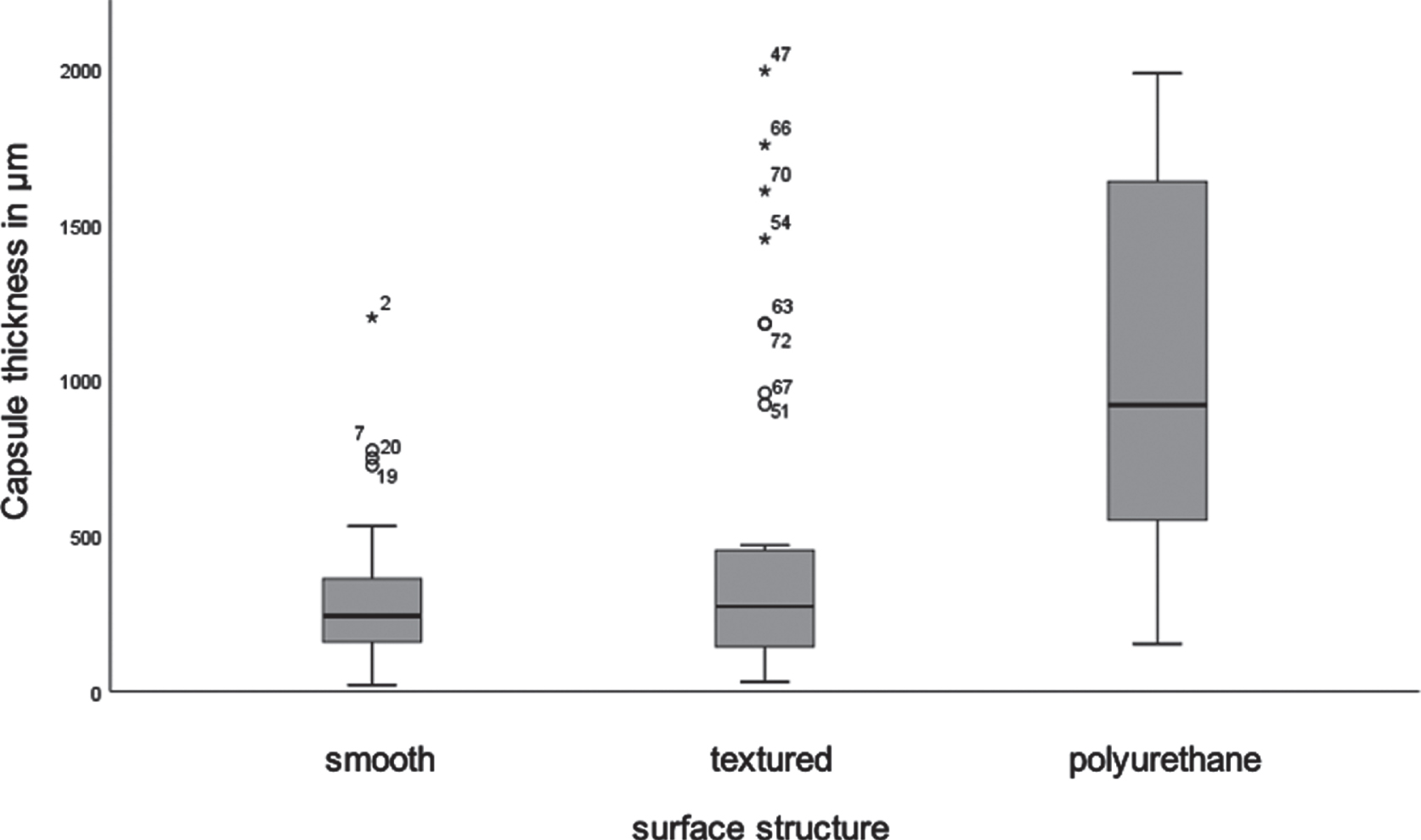
Box plot comparing the capsular thickness of implants with smooth, textured and polyurethane surface structure.
No significant difference was found between smooth and textured implants (p = 0.47). However, the difference between smooth and polyurethane surfaces was statistically significant (p = 0.004) with a medium effect size (r = 0.462). Furthermore, statistically significant difference was also found between textured and polyurethane surfaces of breast-implants (p = 0.012), as well with a medium effect size (r = 0.359).
Seven capsule samples depicted two different layers after hematoxylin-eosin staining. Three of them were capsules from round implants and four from anatomically shaped implants. A homogenous capsule, without layering, was visible in 32 capsules, 18 round and 14 anatomically shaped implants. Diffuse capsular composition was detected in 3 round and 1 anatomical implant. Chi-square test after Pearson indicated no statistical difference in capsular layering between the different shapes of breast-implants (p = 0.741).
Implant shape and capsular thickness
Capsular thickness of both, 23 round and 19 anatomically shaped implants, indicated no normal distribution according to the Kolomogorov-Smirnov test (p = 0.005, p = 0.000). The difference between the two shapes showed statistical significance (p = 0.045) in the Mann-Whitney-U-Test with a medium effect size (r = 0.31). Anatomically shaped breast-implants presented a lower average capsular thickness. The mean rank of capsular thickness in round implants was 24.96 μm and in anatomical implants 17.32 μm (Fig. 5).
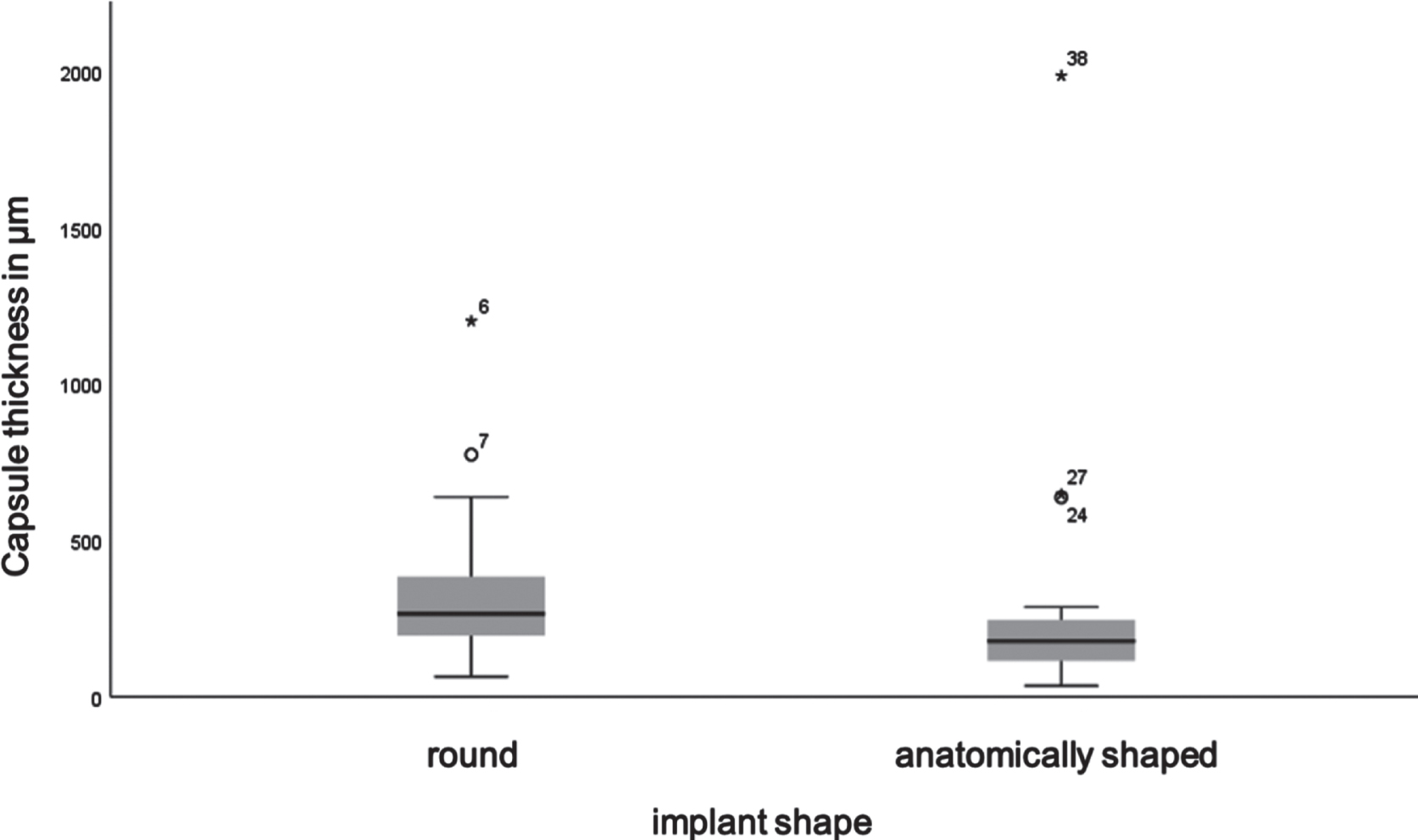
Box plot comparing the capsular thickness of round and anatomically shaped implants.
Thickness measurements of 72 capsular specimens were compared regarding biocompatibility of smooth and textured implants. No difference in capsule thickness was found between smooth and textured implants. Hwang et al. [15] also could not find a significant difference in capsule thickness, in 31 specimens, between smooth and textured implants. Handel et al. [16] examined data from 3495 implants in 1529 women and found no statistically significant difference in capsular fibrosis rates between textured and smooth implants. Rocco et al. [17] also failed to find a difference of statistical significance in his review. However, he only considered one study with 20 patients. Lista et al. [18] also found no difference between the two surfaces. However, only 12 cases of capsular fibrosis were included in their study. In contrast, Calobrace et al. [19] state that the implant surface structure is a risk factor for the development of capsular fibrosis. For textured implants, they determine a protective effect compared to smooth implants. Kuehlmann et al. found that aesthetic patients exhibit a significantly higher mean time period from implantation to revision surgery due to capsular fibrosis compared to breast cancer patients, suggesting that reconstructive-cancer patients in general develop capsular fibrosis earlier [20].
In this study, the described difference is investigated on the basis of 80 cases. In addition, it should be emphasized that the study design differs between the above-mentioned studies and the study conducted here. In this study, capsular thickness was measured after explantation, whereas the before mentioned studies investigated the clinical occurrence of capsular contracture. Another question is whether or to what extent one can correlate the clinical occurrence of capsular fibrosis with capsular thickness. Prantl et al. [21], Zahivi et al. [11], and Siggelkow et al. [22] investigated this and consistently concluded that a thicker capsule after explantation of smooth and textured implants is associated with a higher Baker score. To estimate the presence and severity of capsular contracture more objectively Prantl et al. also used ultrasound elastography. There was a high correlation between the Baker score and the objective scoring measured by ultrasound elastography [23].
However, polyurethane-coated implants were not part of their studies, so this relation requires further investigation, as the pathogenesis and structure of the capsules around polyurethane-coated implants differs from those of silicone-coated implants [24, 25]. Maybe the polyurethane surface leads to a thickening of the capsule with integration of the implant in the surrounding tissue. Our clinical experience is that capsular contracture is significantly less with polyurethane implants and that very rarely a higher Baker grade occurs.
The main focus of this study was the biocompatibility of polyurethane-coated implants. Measurements were based on 80 capsule samples. Compared to smooth and textured implants, a significantly thicker capsule was found in capsules of the polyurethane-coated implants.
In 1991, Handel et al. [26] already showed on the basis of 866 implants (of which 427 were polyurethane) that polyurethane implants led to statistically significantly less capsular fibrosis than smooth implants. Loreti et al. [27] reproduced this result using 358 implants. Vázquez and Pellón [28] studied this effect in long-term use and reached the same conclusion. In 2016, Duxbury and Harvey determined capsular fibrosis rates of 0.4–1% (polyurethane) and 2–15% (textured implants) in a systematic review [29]. Consequently, from a clinical perspective, polyurethane implants can be expected to decrease capsular fibrosis rates. In general, polyurethane is widely used in medical devices due to its good biocompatibility [13]. However, it should be noted that most applications of these products require only a short duration of use, unlike breast implants. There are few studies on the long-term use of polyurethane implants. Szycher et al. [30] and Sinclair et al. [31] demonstrated the biodegeneration of polyurethane by electron microscopy in a study of 10 respectively 75 capsular preparations. Polyurethane was detected in the capsules around the implant. Luke et al. [32] used infrared spectroscopy to detect polyurethane in all 9 capsules examined in their study. Silicone particles were also detected using this method. In contrast to textured and smooth implants, vacuolated macrophages were detected in all samples.
Regarding the histological appearance, there are some studies in rat models. Silva et al. [33] demonstrated a significantly thicker capsule formation around polyurethane-coated implants compared to textured implants. While in this study the difference between the two groups in terms of neoangiogenesis and myofibroblasts in HE-staining was not statistically significant, in 2019 Silva et al. was able to measure statistically significantly more myofibroblasts and mast cell expression using α-SMA and C-Kit immunomarkers, respectively, in the capsules of the polyurethane-coated implants than in the textured implants [34]. The studies for this were performed using 64 Wistar rats. Foley et al. [35] also showed in the rat model that mast cells promote fibroblast proliferation and thus collagen synthesis.
Hwang et al. [15] found a statistically significant relationship between the (tensile) strength of capsules and the rate of capsular fibrosis investigating 31 capsule samples. However, the relationship between capsule strength and capsule thickness was not statistically significant. Bucky et al. [24] examined this relationship in the hare model. The group correlated capsule strength with a modified Baker score and found a statistically significant relationship.
Conclusion
In conclusion, this study demonstrated a thicker capsule formation around polyurethane-coated breast-implants and no difference in capsular thickness between smooth and textured breast implants. Anatomically shaped breast-implants presented a thinner capsule than round breast-implants.
Further studies should be initiated to investigate the single components of the capsular structure around polyurethane-coated breast implants. Besides clinical grading of the capsular fibrosis should be compared with these histopathological findings.
Conflicts of interest
The authors declare no conflict of interest.
There are no other products to be declared.
Financial disclosure
This research received no external funding.
