Abstract
BACKGROUND:
Local ablation is an important treatment for liver cancer in cases of cirrhosis. Liver failure may occur after ablation, especially in advanced liver diseases. Currently, there is no standardization for peri-interventional risk assessment. The liver maximum capacity test (LiMAx) is used to assess functional liver capacity, but there is a lack of exploration of its use in this context.
OBJECTIVE:
The aim of this study was to retrospectively evaluate the usefulness of peri-interventional LiMAx measurements in patients with primary or secondary liver cancer who underwent ablation treatment.
METHODS:
A LiMAx test was performed at 24 hours pre- and postablation in 49 patients. Blood parameters were collected to determine liver function using MELD and ALBI scores. The results of the LiMAx test were related with these scores and to critical postintervention LiMAx values.
RESULTS:
LiMAx values correlated strongly with MELD and ALBI scores before the intervention and reflected the change in liver function, as shown by an increase in scores after the intervention. Notably, LiMAx values decreased during the intervention. AUC analysis for patients at risk of reaching a critical liver level after the intervention showed a cutoff value of 186μg/kg/h.
CONCLUSIONS:
The LiMAx test may be a valuable tool in liver ablation for both peri-interventional monitoring of liver function and preintervention risk assessment.
Introduction
Globally, hepatocellular carcinoma (HCC) is the sixth most common cancer and the third leading cause of cancer-related death, and colorectal cancer is the second leading cause of cancer-related death in developed countries [1]. Approximately 20–55% of patients with colorectal cancer develop liver metastases [2, 3], and the incidence of primary and secondary liver malignancies is therefore high. Surgical resection is considered the standard of care for curative treatment of malignant liver tumors. However, the surgical procedure is significantly limited if the patient’s general condition is impaired, the tumor is progressing rapidly, the anatomical location of the tumor lesion is unfavorable, previous extensive abdominal surgery has been performed or the function of the postoperative residual liver is inadequate [2, 4]. Thus, local ablative procedures are becoming increasingly important in treatment of primary and secondary malignant liver tumors, especially in the case of preexisting liver damage (cirrhosis). Local ablative procedures for treatment of HCC are effective, with low rates of morbidity and mortality, and are tissue sparing with regard to the liver parenchyma, which can be critical for patients with inadequate liver reserve. Currently, local ablative procedures are an alternative for secondary liver tumors when surgery is contraindicated, as a bridging measure or for recurrence after hepatectomy, and an alternative in treatment of HCC when transplantation is not possible [5–8]. Microwave ablation (MWA) and irreversible electroporation (IRE) are well-established percutaneous ablative techniques in oncology used for treating various malignancies, particularly liver cancer. However, despite their widespread use, complications are associated with these techniques, including risk of posttreatment hepatic failure (PTHF), particularly in patients with advanced cirrhosis [9–11].
In patients undergoing ablation, assessment of liver function is critical for treatment planning and follow-up. Such assessment is typically performed by using various laboratory values and scoring systems, such as the Model for End-Stage Liver Disease (MELD) and albumin bilirubin (ALBI) scores [12–14]. Pre-operative ultrasound has also been shown to be useful [15–17]. However, Liver function tests, including indocyanine green (ICG) and LiMAx (Liver Maximum Capacity Test), can provide additional insight into liver function [18–21]. In particular, the LiMAx test, which is based on respiration of 13C-methacetin, provides assessment of functional liver capacity. Although it has been shown to be useful in various clinical scenarios, it has not been extensively studied in the specific context of patients undergoing MWA or IRE. In general, a reliable and comprehensive assessment of liver function prior to the procedure is needed to determine potential risk of liver function deterioration following ablation. Currently, this assessment relies primarily on global liver function parameters derived from laboratory values as well as MELD and ALBI scores [22]. Nevertheless, these methods have limitations, and certain nuances of liver function that could be captured by the LiMAx test might be missed. Therefore, wider implementation of the LiMAx test may provide a more accurate and nuanced understanding of liver function in patients undergoing procedures such as MWA and IRE.
The aim of this study was to investigate whether ablation-induced deterioration in liver function, as measured by blood-based scores such as MELD and ALBI, is associated with detectable changes in LiMAx values. To this end, we sought to evaluate whether the LiMAx test is suitable for monitoring liver function in the context of ablation in the peri-interventional setting. In addition, whether a preintervention LiMAx can help to identify patients who are likely to reach a critical liver value after the intervention was investigated.
Methods
Patients and study design
Approval for this retrospective study was obtained from the local ethics committee, and all cases were discussed by a local tumor board. All study participants provided written consent to undergo ablation and the LiMAx test. Only patients who had no contraindications to ablation or LiMAx testing and whose laboratory values and LiMAx data were available were included in the study. Of the 70 patients enrolled between 1 October 2017 and 31 December 2018, 49 met these criteria. The patients underwent a LIMAx test and blood sampling within 24 hours before and after ablation. Of the laboratory values collected, bilirubin, creatinine, international normalized ratio (INR), aspartate transaminase (AST), gamma-glutamyltransferase (γ-GT), alanine transaminase (ALT), alkaline phosphatase (AP) and albumin were examined in this study.
LiMAx testing
In accordance with previously published recommendations, the LiMAx test was performed within 24 h before and after ablation [18]. The patients fasted for 3 hours prior to testing. To calculate the delta over baseline, the respiratory ratio of 13CO2:12CO2 was determined at ten minutes before i.v. injection of 13C-methacetin. After reaching a constant value, 2 mg/kg body weight methacetin was injected via i.v. bolus and flushed with 20 ml 0.9% NaCl. Analysis of 13CO2 production was then initiated using the LiMAx device (Humedics GmbH, Berlin, Germany). The measurement was performed for a maximum of 60 min. A LiMAx value > 315μg/kg/h reflects healthy liver function; the lower the LiMAx value is, the more impaired the liver function is [19]. The LiMAx difference was calculated as follows: LiMAx postintervention minus LiMAx preintervention. The LiMAx drop was calculated as LiMAx preintervention minus LiMAx postintervention, and this difference was divided by LiMAx preintervention and is expressed as a proportion.
ALBI and MELD scores
Johnson et al. analyzed and validated prognostic factors for survival in advanced liver disease in a large international study [23], showing that albumin and bilirubin are the most important prognostic factors. Based on this finding, the ALBI score was developed with appropriate weighting; it is calculated as follows: ALBI=(log10 Bilirubin [μmol/l] x 0.66)+(Albumin [g/l] x -0.0852). The results can be divided into 3 groups: ALBI score< -2.6 corresponds to ALBI grade 1 (low mortality risk); ALBI score> -2.6 to -1.39 corresponds to ALBI grade 2 (intermediate mortality risk); and ALBI score> -1.39 corresponds to ALBI grade 3 (high mortality risk).
The MELD score is used to assess the severity of liver disease and the associated mortality rate without liver transplantation and was therefore calculated as a supplement [24, 25]. The MELD score can expressed as integer values between 6 and 40. The higher the MELD score is, the higher the degree of liver damage is or the more likely it is that the patient will die within the next three months without a liver transplant.
Ablation procedures
IRE procedures were performed with the NanoKnife® system (Angiodynamics®, Latham, NY, USA) using up to six 19-G electrodes. The IRE ablation parameters applied were as follows: voltage, 1650 to 3000 V; pulse duration, 90μs; pulses per cycle, 70. Microwave ablation was performed using the Acculis microwave tissue ablation system and a 14-G applicator (AngioDynamics, Latham, NY, USA) operating at 2.45 GHz with a maximum output power of 140 W. Radiofrequency ablation was performed using the StarBurst©, RITA©, 1500X ablation system (AngioDynamics, Latham, NY, USA) with monopolar perfused 14-G electrodes (StarBurst©, Talon Semi-Flex, AngioDynamics, Latham, NY, USA). The ECT procedure was performed with a Cliniporator (IGEA® Clinical Biophysics, Capri, Italy) and five electrodes under intravenous administration of a total of 30,000 IU of bleomycin.
Statistics
GraphPad Prism 9.5.0 (GraphPad Software, LLC, San Diego, CA, USA) was used for statistical calculations and presentation. All variables are described as proportions, means with standard deviations or medians with interquartile ranges. According to the distribution of the test variables, two-tailed dependent and independent samples t tests were used for analysis, and Pearson’s correlations were determined. The correlation coefficient was interpreted as follows: up to 0.1 as weak; 0.3 as moderate; and above 0.5 as strong. ROC/AUC analysis was also carried out, and the results were interpreted as follows: 0.9-1 as excellent; 0.8-0.9 as good; 0.7-0.8 as fair; 0.6-0.7 as poor; and 0.5-0.6 as fail. A value of
Results
Baseline data
A total of 49 patients underwent LiMAx testing before and after minimally invasive percutaneous liver therapy. The majority of the patients received MWA (92%), three patients (6%) received IRE, and one patient received ECT. The underlying tumor entities are compiled in Table 1. Most cases (71%) were HCC, 20% were colorectal carcinoma (CRC), 6% were cholangiocarcinoma (CCC), and 2% were hemangioendothelioma. The laboratory parameters used to calculate scores and to assess liver function are also shown in Table 1.
Patient demographic and clinical baseline characteristics (n = 49)
Data are presented as n (%) for categorical variables and the mean (±standard deviation) or median (interquartile range; Q1-Q3) for continuous variables. CCC = cholangiocarcinoma, CRC = colorectal cancer, HCC = hepatocellular carcinoma, MELD = model for end-stage liver disease, INR = international normalized ratio, AST = aspartate transaminase, γ-GT=gamma-glutamyltransferase, ALT = alanine transaminase, AP = alkaline phosphatase.
Patient demographic and clinical baseline characteristics (n = 49)
Data are presented as n (%) for categorical variables and the mean (±standard deviation) or median (interquartile range; Q1-Q3) for continuous variables. CCC = cholangiocarcinoma, CRC = colorectal cancer, HCC = hepatocellular carcinoma, MELD = model for end-stage liver disease, INR = international normalized ratio, AST = aspartate transaminase, γ-GT=gamma-glutamyltransferase, ALT = alanine transaminase, AP = alkaline phosphatase.
The mean LiMAx value was 221 (±112) μg/kg/h before the intervention and 198 (±99) μg/kg/h after the intervention. The median MELD score was 8 (7-10) before the intervention and 10 (8-13) after the intervention. The ALBI score could only be determined in 44 patients before the intervention and was on average -2 (±0,63); after the intervention, the ALBI score could only be determined in 41 patients and was on average -1.7 (±0,67). The values are given in Table 1. When divided by ALBI grade, most patients were in the ALBI grade 2 group (
Pairwise comparison of the means of all three scores before and after the intervention were significantly different: LiMAx t(48)=2.582
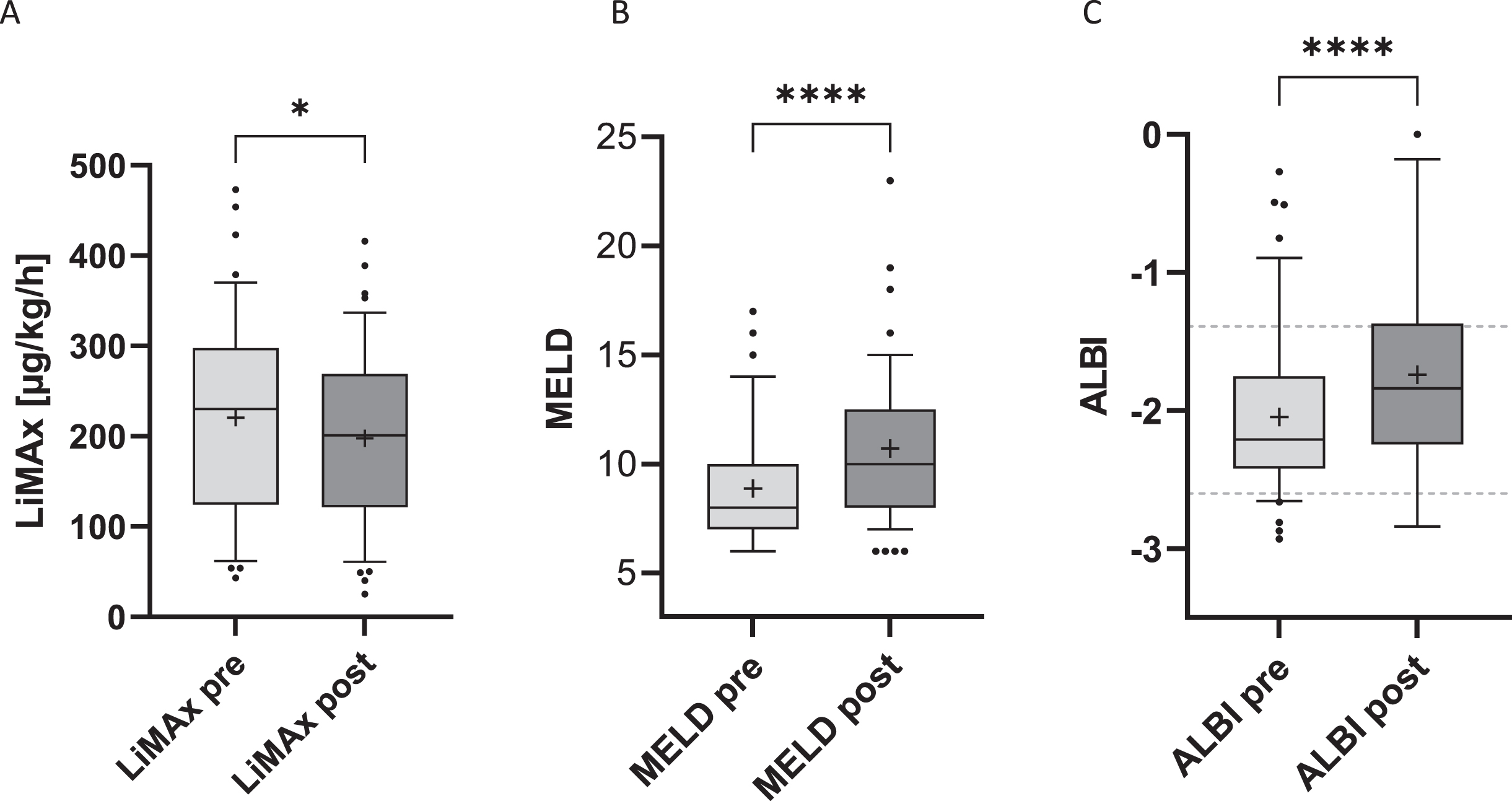
Pairwise comparison of LiMAx (A), MELD score (B) and ALBI score (C) before (pre) and after (post) the intervention. Except for the ALBI score, which is
Pearson correlations (r). (A) The correlation of LiMAx pre with MELD and ALBI preintervention values is shown. (B) The correlation of the LiMAx post to MELD and ALBI postintervention values. The correlation coefficient is shown with the 95% confidence interval indicated in square brackets, and the asterisks represent two-sided significance analysis
**** Two-tailed
The patient population was divided into two groups to further investigate the performance of the LiMAx test in the peri-interventional setting, particularly in relation to critical postintervention liver values. For this purpose, patients were divided according to their LiMAx value after the intervention depending on whether it was greater than/equal to or less than 150μg/kg/h. This value was chosen because previous publications have reported that patients with a postoperative LiMAx value of more than 150μg/kg/h on the day after surgery have a better outcome in terms of days in intensive care or hospital and in-hospital mortality [18, 19]. Therefore, patients with a level below 150μg/kg/h are considered to have critical liver values.
The baseline and liver function values for the two groups are shown in Table 3. Of the 49 patients, 63% had a postinterventional LiMAx greater than or equal to 150μg/kg/h; the other 37% had a lower value. The percentage decrease (LiMAx drop) was similar in both groups (t(47)=0.156
Characteristics of the study population grouped according to the postinterventional LiMAx value (greater or equal to 150μg/kg/h or less)
Data are presented as n (%) for categorical variables and the mean (±standard deviation) or median (interquartile range; Q1-Q3) for continuous variables.
Characteristics of the study population grouped according to the postinterventional LiMAx value (greater or equal to 150μg/kg/h or less)
Data are presented as n (%) for categorical variables and the mean (±standard deviation) or median (interquartile range; Q1-Q3) for continuous variables.
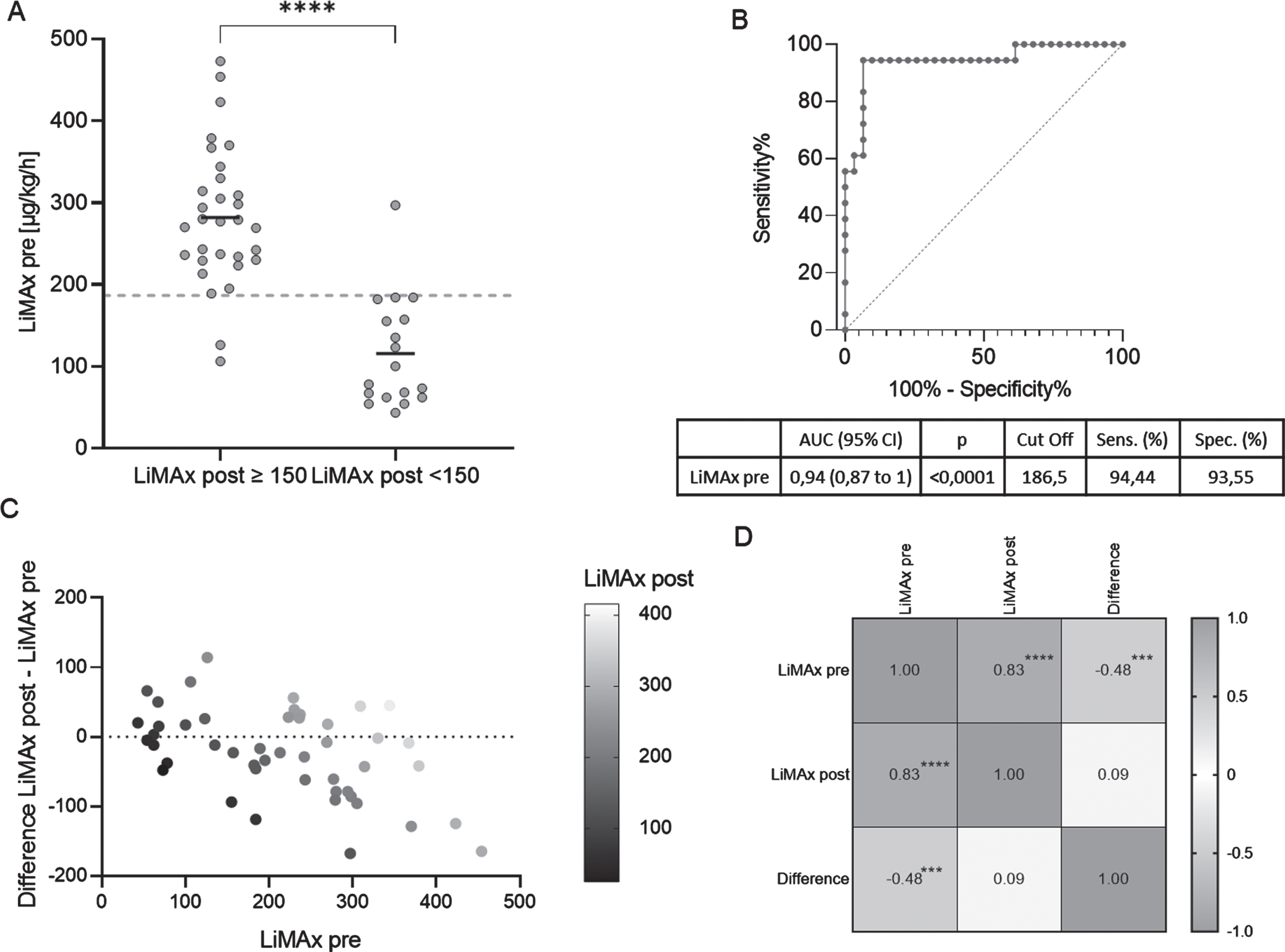
(A) Paired comparison of LiMAx preintervention values for groups subdivided by LiMAx postintervention values (greater or equal to 150μg/kg/h or less). Group means are shown by short horizontal lines, and the ROC/AUC analysis cutoff is shown by the dashed line. (B) ROC curve with AUC analysis of LiMAx preintervention and the calculated cutoff value to identify patients who may reach a critical LiMAx postintervention under 150μg/kg/h value after the intervention. The cutoff was calculated using the Youden index [38]. CI = confidence interval. (C) Illustration of differences between LiMAx preintervention and LiMAx postintervention in relation to the LiMAx preintervention values. (D) Pearson correlation matrix of LiMAx preintervention, LiMAx postintervention and the difference between the two. Two-sided p values obtained from the significance analysis are shown as ****<0,0001; ***<0,001.
Figure 2C shows the relationship between pre- and postinterventional LiMAx values in terms of the difference between them, whereby the smaller the LiMAx preintervention value was, the smaller the decrease after intervention was. It should be noted, however, that some variability was observed and that not all LiMAx pre values decreased. Again, a higher LiMAx preintervention value was associated with a greater decrease, with a significant negative correlation, as shown by the Pearson correlation (r(47)=-0.48 [-0.67 to -0.23],
ROC/AUC analysis was chosen to determine whether the LiMAx preintervention value might serve as a measure for identifying patients who might reach a critical level of less than 150μg/kg/h after the intervention. The results are illustrated in Fig. 2B. LiMAx preintervention displayed a very good discriminatory ability, with an AUC of 0.94 [0.87 to 1.00]. The cutoff value was determined using the Youden index and indicates the LiMAx preintervention value below which a critical liver value might be reached after the intervention. This value was 186.5μg/kg/h, with a sensitivity of 94.44% and a specificity of 93.55%. The MELD score had a lower discriminatory ability, with an AUC of 0.71 [0.55 to 0.87],
Following the introduction of the LiMAx test as a direct assessment of liver function in 2009, the test has become established for presurgical assessment of liver function, allowing for good risk assessment and prediction of postsurgical liver failure [18, 27]. Compared to its peri-interventional use in surgery, use of the LiMAx test in other liver treatments is less well studied. However, there are some recent publications showing that the LiMAx test can be used in transarterial chemotherapy (TACE) [28–32], but for other therapies, such as ablation, the evidence is sparse. One early study that explored use of a 13C-methacetin breath test for both TACE and RFA exist [33]. In this publication, no change in 13C-methacetin metabolism was observed before RFA or one day afterward, as was the case for TACE. In more recent TACE/LiMAx studies, deterioration in liver function was also measured at one day after the procedure [28, 31]. In the present study, LiMAx was measured in the setting of ablation, and deterioration in liver function was observed. This effect was also seen in MELD and ALBI scores, which were used as surrogates for liver function. Blood values for liver markers can deteriorate after an intervention, and this deterioration is reflected in scores based on blood values [28, 34]. In this study, the preintervention LiMAx value correlated strongly with MELD and ALBI scores, with a similar strength as previously published [29, 35]. However, these previous publications were mostly limited to the correlation before the intervention, whereas in this study, the LiMAx score at one day after the intervention also correlated strongly with MELD and ALBI scores after intervention. Thus, the LiMAx test was shown in the present study to provide valid values in the context of ablation and, for the first time to our knowledge, to correlate with MELD and ALBI. The increase in MELD and ALBI scores after the procedure is mainly due to destruction of tumor cells and an increase in bilirubin occurring postintervention [28, 34]. The numerous influencing factors and limitations of the LiMAx test summarized by K. Gorowska-Kowolik et al. seem to play a minor role in ablation [36]. Furthermore, this study shows that the LiMAx test can reflect the change in liver function associated with ablation as well as the change in liver function indicated by an increase in MELD and ALBI scores after the intervention. Importantly, this study provides initial insight into the use of the LiMAx test in the context of liver ablation.
LiMAx values below 150μg/kg/h are described in the literature as critical. For example, a correlation was found between lower pre-TACE LiMAx levels and bilirubin as a surrogate for liver failure after TACE [31]. In another study, patients with HCC classified as BCLC A (Barcelona Clinic Liver Cancer) with a LiMAx value of less than 150μg/kg/h had a lower 30-day survival rate than patients with higher LiMAx values [29]. To assess the diagnostic value of LiMAx for peri-interventional use in ablation, we analyzed which patients achieved a critical LiMAx level below 150μg/kg/h after the procedure and divided them into two groups based on this value. Our analysis showed that these patients already had a low LiMAx level prior to the procedure. Interestingly, the average decrease in LiMAx was comparable between the groups, regardless of the initial value (LiMAx drop 3,1 vs. 4,9). However, the LiMAx drop (3.8%) for all patients analyzed in our study was lower than that of Reichert et al. (7.1%) and lower than that of Barzakova et al. (10%). It is crucial to note that the patients with a low preintervention LiMAx score also tended to have a low postinterventional LiMAx score. Although the absolute decrease was less than that in patients with a high preintervention LiMAx score, the low preintervention value in these patients suggests an already limited liver reserve; thus, further decrease might be critical for these patients. This indicates the importance of preintervention evaluation of liver function in patients undergoing liver ablation. Nevertheless, further studies are needed to confirm these findings and to fully explore the potential of the LiMAx test in this setting.
Furthermore, this study found that the preintervention LiMAx value can be used as a predictor of postintervention critical liver function. Our ROC/AUC analysis indicated that a pre-LiMAx value of 186μg/kg/h can be used as a cutoff to identify patients who may reach a critical liver function value after the intervention. The cutoff obtained in this study showed high sensitivity and specificity in predicting critical liver values after the intervention. Therefore, the LiMAx test may be a valuable tool in the preinterventional setting to identify patients who may reach critical liver values after the intervention, thereby improving risk assessment and potentially influencing treatment strategies. It would also be interesting to evaluate whether its predictive value can be improved by combining it with other markers of liver function or disease severity.
This study has numerous limitations that need to be considered when interpreting the results. Importantly, this study was retrospective and included a relatively small number of patients. Therefore, the findings need to be validated in prospective studies with larger patient cohorts. The LiMAx test is widely used in clinical practice because of its speed, lack of toxicity, ease of administration and cost-effectiveness. However, there is considerable variability in the results of the LiMAx test, which adds another layer of complexity to interpretation of the results. This variability might be due to many factors, including age, physical activity, diet, medical conditions, anemia, oxygen supplementation, hepatic extraction of methacetin, liver blood flow and gastric emptying rate, among others [36]. Furthermore, as xenobiotics are known to affect cytochrome P450 activity, and therefore, LiMAx results are affected by numerous factors, including cigarette smoking and alcohol consumption. Additionally, certain drugs can affect cytochrome P450 activity [37]. Another limitation is the small number of patients and the heterogeneity of the diseases in the patient population. This heterogeneity may have introduced numerous confounding effects that may influence the results, thus limiting the generalizability of our findings to more specific patient populations. Another limitation is that we did not quantify the size of ablation defects, such as their volume. Overall, the size of the ablation area may influence outcomes [31], and without this information, it is difficult to fully understand the impact of the procedure on each patient. Although the study was designed with a relatively short observation period, importantly, recovery and normalization of certain values after ablation may require longer times. Therefore, the short observation period might also limit the accuracy and completeness of the results. Nevertheless, despite the small sample size, the MELD and ALBI values obtained are comparable to those reported in the literature, demonstrating the validity of the data collected. The limitations identified in this study are the subject of ongoing research. Larger and more controlled studies are needed to address these limitations, improve understanding of ablation outcomes, and refine predictive models in this area. Despite these limitations, our study provides valuable insights and adds to the growing body of evidence on the effects of ablation procedures.
Conclusion
This study demonstrates that the LiMAx test can provide valuable information in the setting of liver ablation. The test has the potential to reflect peri-interventional changes in liver function, as indicated by MELD and ALBI scores. Importantly, our results suggest that the preintervention LiMAx score can be used to predict critical postinterventional liver function. These findings highlight the potential of the LiMAx test as a tool for risk stratification and management of patients undergoing liver ablation. In our study, the LiMAx value decreased during ablation. To avoid falling below a critical liver function value of less than 150μg/kg/h, ROC/AUC analysis of our data suggested a cutoff value of 186μg/kg/h before the intervention. However, further research is needed to confirm these findings and to optimize use of the LiMAx test in this clinical setting.
