Abstract
The growth of cyanobacteria can vary considerably depending on the ambient temperature. Since the optimal growth temperature for Arthrospira platensis (strain SAG21.99) is not yet known, this was investigated in the present study.
The study revealed that a process temperature of 30 °C seems to be optimal for the Arthrospira strain SAG21.99 cultivation in terms of a maximum biomass productivity. This was also true for the phycocyanin content which was at 30 °C significantly higher than at 20 or 40 °C.
Introduction
Various photosynthetic microorganisms have been extensively cultivated for commercial production, including Arthrospira platensis (AP, often called Spirulina), Chlorella sp., Haematococcus pluvialis and Dunaliella salina [1]. Specifically, AP, a filamentous cyanobacterium, is one of the most commercially successful microalgae as food for humans and animals as it contains large amounts of proteins, vitamins, lipids, pigments and further biologically active ingredients [2–4]. As AP inhabit soda lakes, they are cultivated under highly alkaline conditions at pH values between 8–11, a condition which prevents the culture from toxic contamination [5]. In contrast to many cyanobacteria, there is no report of toxicity of Arthrospira for humans, animals or environments [6].
In order to maximize the productivity, the process operating conditions like light, temperature or the AP strain should be optimized and strictly controlled [7]. The cultivation system for AP mass production is usually conducted outdoors, where cells frequently encounter fluctuating temperature and light intensity. Low temperatures at winter time as well high temperature during summertime can pose a critical problem for growth. Therefore, the optimal temperature has to be chosen for the different AP strains and controlled at which the highest yield can be achieved. The optimum temperature for AP growth is reported to vary in a very broad range depending on the strain used. For a lot of different strains, the optimum temperature was described to be at 30 °C [9–12] or at 35 °C [13, 14]. Richmond [15] reported an optimum growth temperature for Arthrospira of 35–37 °C or even of about 39 °C [16]. Thermotolerant strains of Arthrospira have been cultivated between 35 and 40 °C [17]. Torzillo [18] reported that the problem of temperature control in summer in Italy has been greatly alleviated with the selection of a strain of Arthrospira able to grow up to 42 °C and to withstand temperatures of 46 °C for a few hours without adverse effect. On the other hand, a new strain of AP –O9.13F –was detected in Solonoye lake in western Siberia [19], which differs extremely from the strains known so far according to their optimum growth temperature. This strain O9.13F showed the highest relative growth index upon cultivation at 20 °C. This demonstrates the broad adaptability of AP to environmental conditions.
As the optimum temperature for the strain used (SAG21.99 from the “Algenstammsammlung der Universität Göttingen”) is not yet analyzed according to their optimum growth temperature. Therefore, we measured the dependence of the growth and of various ingredients of AP from the temperature of the culture medium.
Material and methods
Arthrospira platensis used for cultivation was obtained from the Culture Collection of Algae at Göttingen University (strain: SAG21.99). The stock suspension of AP was cultured in Zarrouk medium consisting of (per liter): 16.8 g NaHCO3, 0.5 g K2HPO4, 2.5 g NaNO3, 1.0 g K2SO4, 1.0 g NaCl, 0.2 g MgSO4·7H2O, 0.04 g CaCl2, 0.01 g FeSO4·7H2O, 0.08 g Na2EDTA and 1.0 ml of trace metal solution. The trace metal solution consisted of (per liter): 2.86 g H3BO3, 1.81 g MnCl4·4H2O, 0.22 g ZnSO4·4H2O, 0.0177 g Na2MoO4, 0.079 g CuSO4·5H2O. All the reagents used were of analytical grade. The growth medium was initially sterilized at 121 °C in a HV-50 autoclave (SYSMEX VX-95, Sysmex, Norderstedt, Germany) for 15 min.
AP was cultured in bioreactors of 2 l each at temperatures of 20, 30, and 40 °C [8]. All preparations were gassed with 1% CO2/air mixture (20 l/h) to exclude possible carbon limitation.
The dry weight of biomass (DW) was determined after centrifugation with 17.000 g, washing the cell pellet and a second centrifugation (again at 17.000 g), drying and then weighing. The pH values were monitored continuously over the cultivation time (EGA 133, Sensortechnik Meinsberg, Meinsberg, Germany). The nitrate concentration was determined according to AOAC methods [20]. The phycocyanin concentration was photometrically determined according to Bennett & Bogorad [21]. Fatty acids present in the cells in lipid form were converted to fatty acid methyl esters (FAME) and determined according to Bligh & Dyer [22]. These were analyzed with a gas chromatograph (Agilent 7820A, GC column SP2560 from Supelco, use of biscyanopropyl polysiloxane).
For all samples mean value and standard deviation are given. Statistical significance was evaluated by one-way ANOVA coupled with a post hoc Bonferroni correction analysis using Graph Pad Prism 8 software. P values below 0.05 are considered significant.
Results and discussion
Figure 1 shows that the biomass productivity markedly depends on the respective cultivation temperature; the increases (value at the end of the culture period minus baseline value) of the three samples differed significantly (p < 0.001). At a temperature of 30 °C, biomass growth was significantly higher than in the comparison cultures grown at 40 °C and 20 °C, respectively (p < 0.001 each). A cultivation temperature of 20 °C resulted in a significantly reduced biomass growth rate while an increase in temperature to 40 °C led to a stagnation of biomass production after only 4 days both in comparison to the growth at 30 degree Celsius (see Fig. 1).
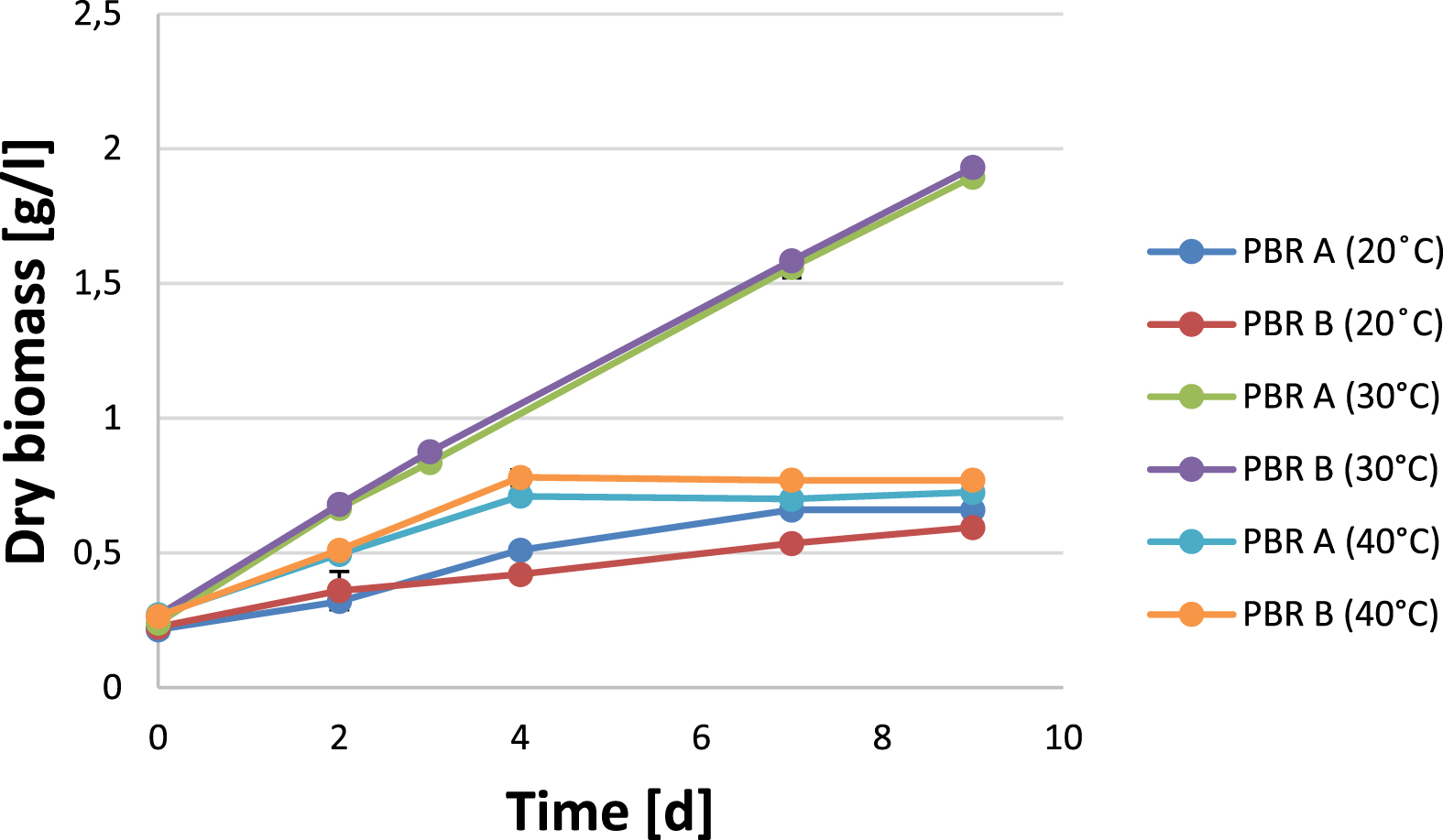
Dry biomass of Arthrospira platensis cultures at varying culture temperatures in Zarrouk medium up to nine days (two independent experiments, measurements in duplicate).
Parallel to the decrease in temperature, there is a decrease in the pH value of the culture medium. This is probably due to the temperature dependence of the solubility of carbon dioxide. Increasing the temperature decreases the solubility of CO2 and therefore it increasingly enters the gas phase. Thus, the equilibrium reaction is shifted from hydrogen carbonate (
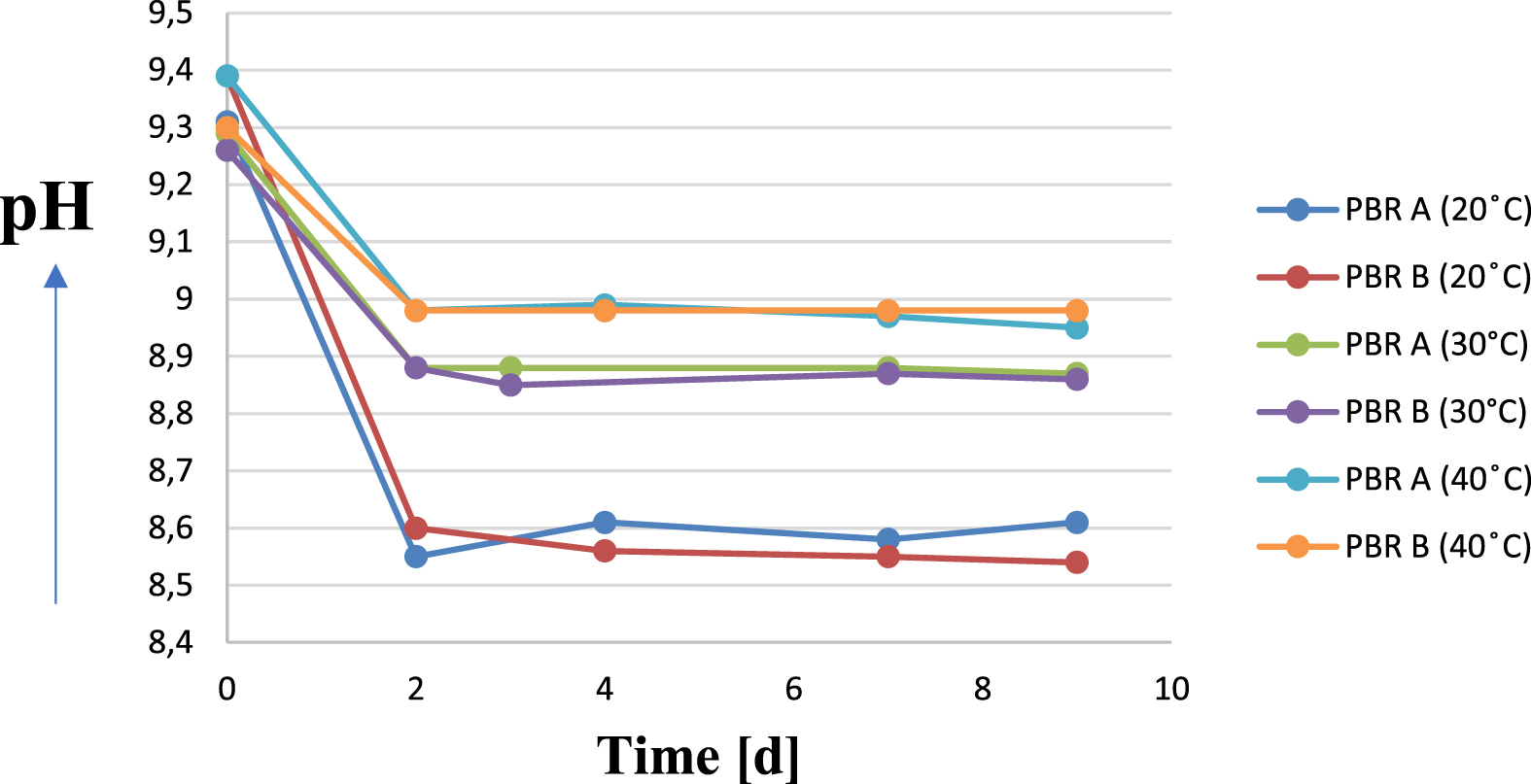
pH kinetics of Arthrospira platensis cultures at different temperatures in the culture medium up to nine days.
The nitrate uptake from the culture medium by the Arthrospira cells is shown in Fig. 3. Here, it can clearly be seen that a higher nitrate uptake by the cells occurred at a cultivation temperature of 30 °C, which led to a decrease in the nitrate ion concentration of approx. 950 mg/l within 9 days (see Fig. 3). In the trials at 20 °C and 40 °C growing temperature, respectively, the nitrate concentration decreased only by about 400 mg/l during the same experimental period, which is consistent with the reduced cell growth and also appears consistent with respect to the approximately equal final dry biomass concentrations under these temperature conditions.
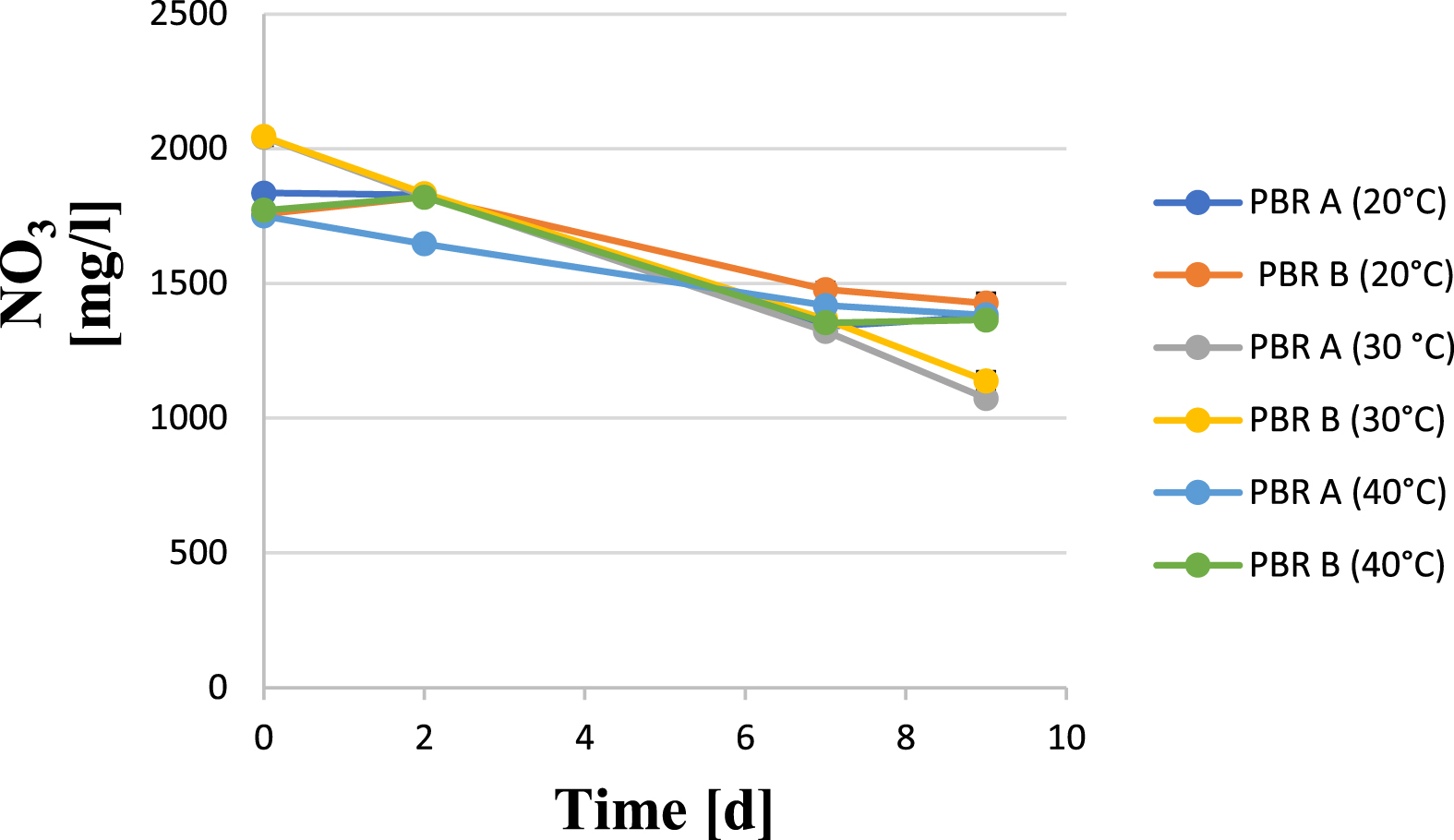
Nitrate uptake of Arthrospira platensis cultures at different temperatures in the culture medium up to nine days.
The material composition of the Arthrospira biomass was also significantly affected by the different cultivation temperatures. After extraction of the respective biomass samples, both phycobiliproteins and fatty acids were quantified by photometric and chromatographic analytical methods.
Starting from the cultures at baseline, in which the biomass-specific phycocyanin content was approximately the same, it was found at the end of the experimental period that it reached the highest value, of about 80 mg /g dry biomass, at 30 °C growing temperature. In the cultures grown at 20 °C and 40 °C, the phycocyanin content was 28 and 35 mg/g dry biomass, respectively, and was thus significantly lower. The generally apparent decrease in phycocyanin levels over time, is likely due to light-adaptive regulatory mechanisms induced by the dilution of starter cultures and the associated change in relative light levels (Fig. 4). These results are well in line with former studies by Oliveira et al. [23] as well as by Ogbonda et al. [16].
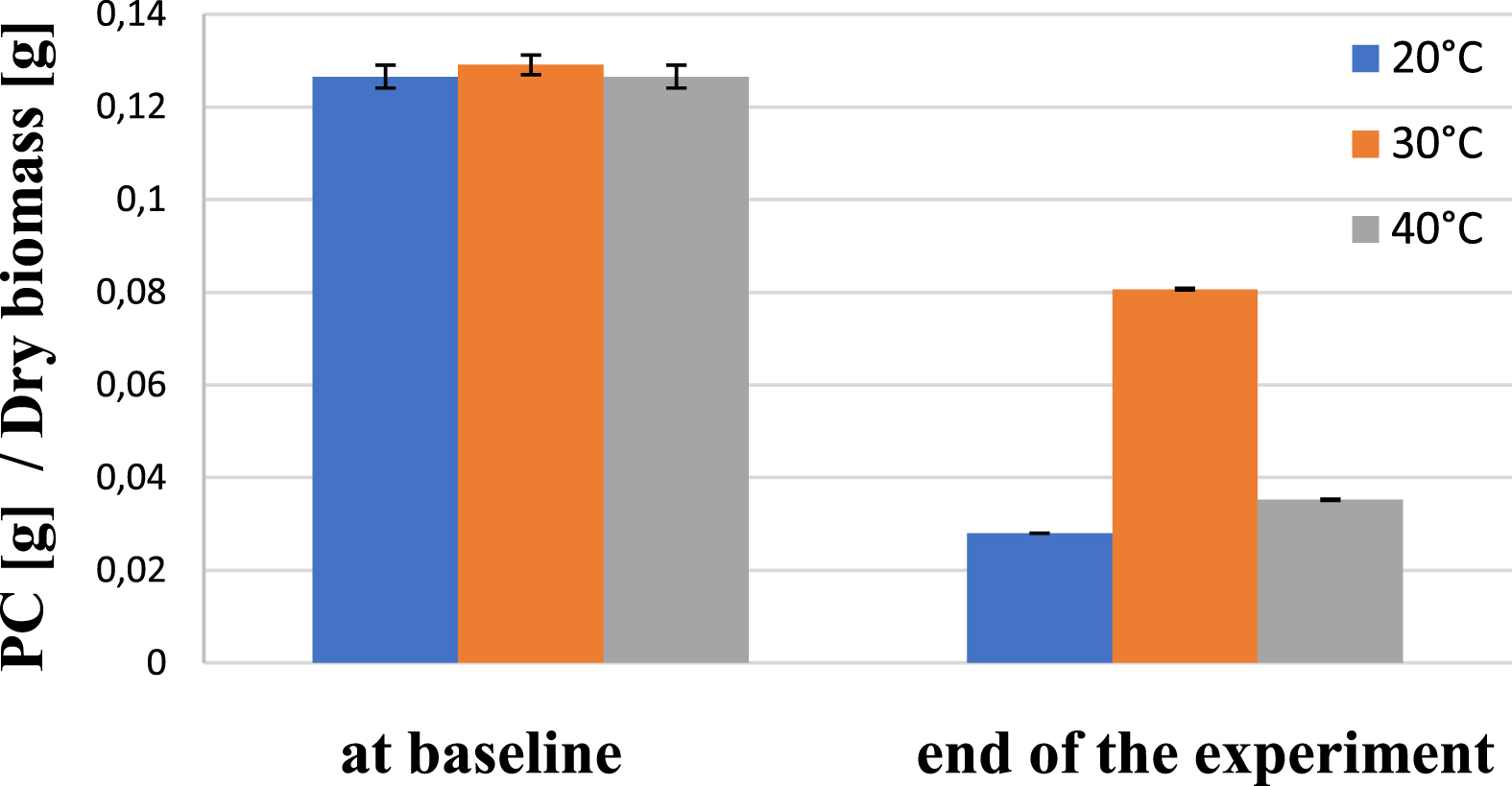
Phycocyanin content of AP at baseline and at the end of the experiment at different temperatures.
Gas chromatographic analysis of fatty acid methyl esters showed that with increase in temperature, the total fatty acid content of Arthrospira biomass also increased significantly, reaching the highest value of 56 mg fatty acid per g dry biomass (equivalent to 5.6%) on average at 40 °C cultivation temperature (Fig. 5).
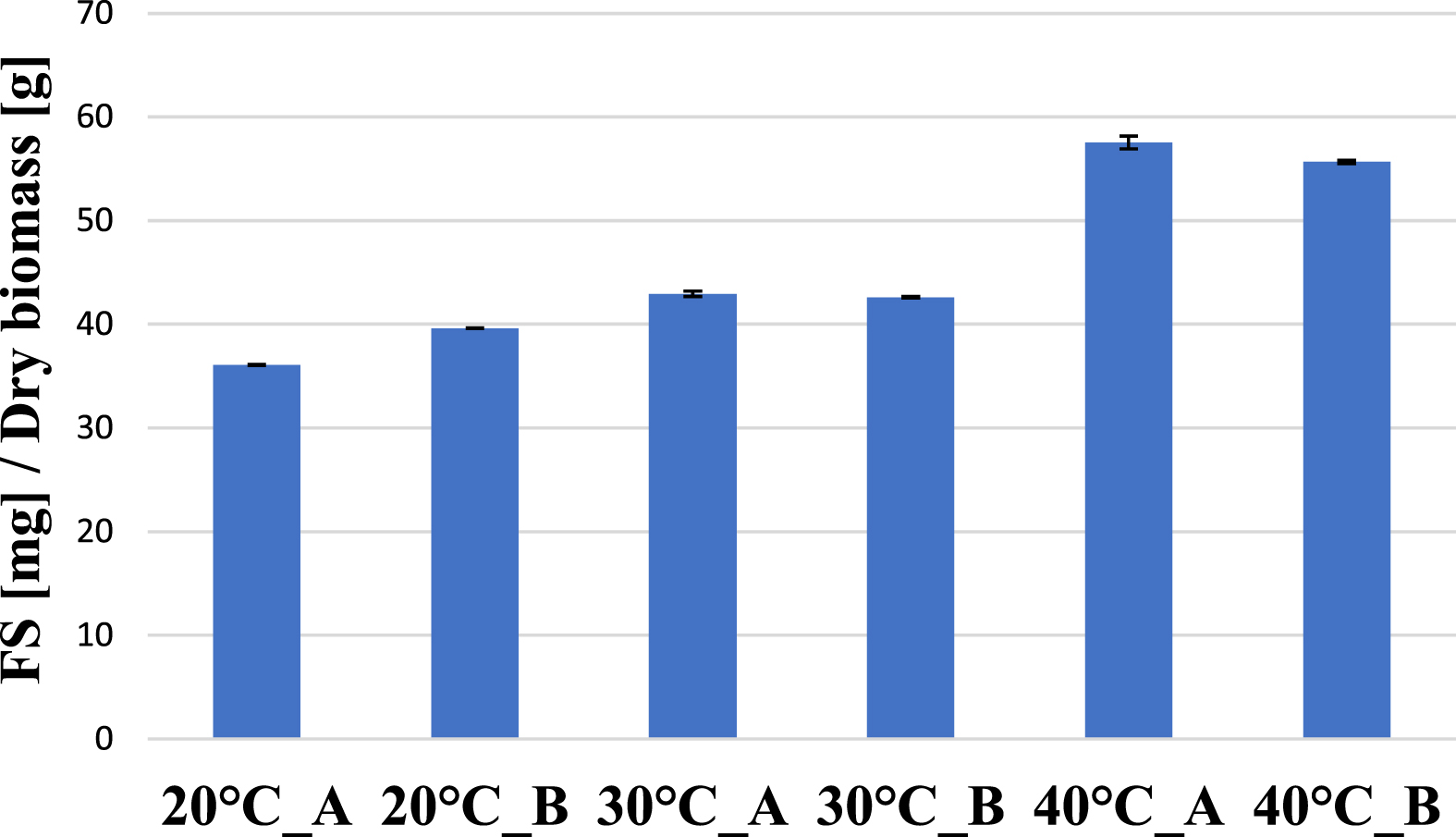
Total fatty acid content (FS) of Arthrospira platensis biomass at different growing temperatures at the end of the experiment.
The fatty acid composition of Arthrospira was also influenced by the cultivation temperature. The largest proportion of fatty acids in the biomass was palmitic acid (C16 : 0) whereby lowering the temperature to 20 °C resulted in a reduced content of this fatty acid. At 40 °C, the content of dihomogammalinolenic acid (C20 : 3n6) and gamma-linolenic acid (C18 : 3n6) was significantly reduced and was approximately 1.2% and 13%, respectively, relative to the total fatty acid content. At a growing temperature of 30 °C, the content of these two fatty acids was 2.6% and 22%, respectively, and was thus significantly increased compared to the other two temperature ranges tested. The synthesis precursor of these fatty acids, essential linoleic acid (C18 : 2n6), was present at a lower level of approximately 11.5% at 30 °C (Fig. 6).
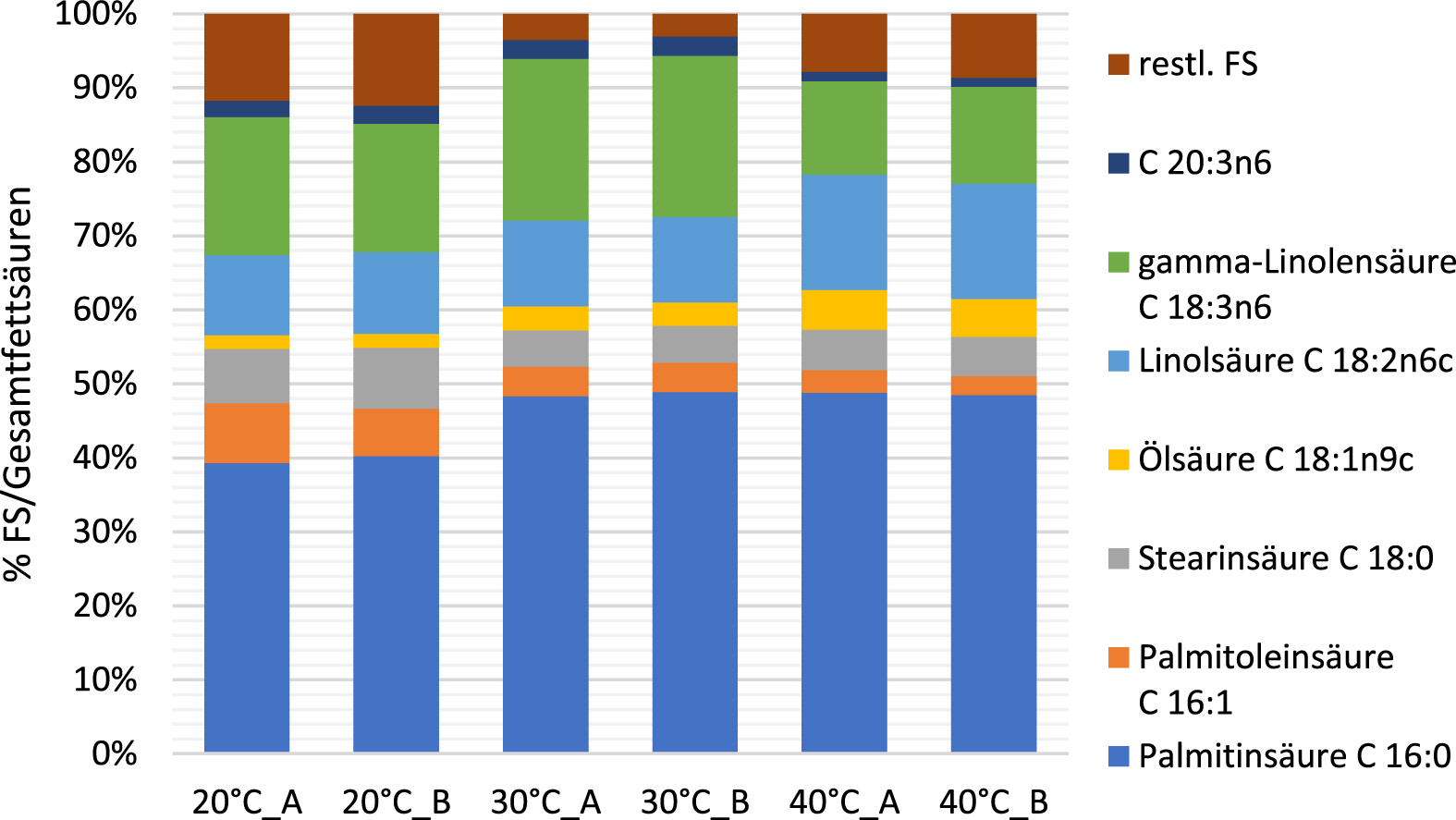
Fatty acid composition of Arthrospira platensis biomass at different growing temperatures at the end of the experimental period.
The change in cultivation temperature generally resulted in changes in fatty acid composition, possibly indicating that specific enzymatic desaturation and elongation steps in Arthrospira fatty acid synthesis are subject to temperature-dependent regulation.
In contrast to light, temperature is less specific, as it will affect all metabolic processes, including cell division. The wrong temperature limits algae growth and photon use even more than the suboptimal light intensities [24]. Generally, too high temperatures are harmful to algae. Too high temperatures –starting above 30 °C [25] reduce photosynthesis by inactivating the photosynthetic proteins thereby disturbing the energy balance of the cell [26]. Heat disintegrates functional molecules (e.g. enzymes), can cause reduction of the carboxylase activity of Rubisco resulting in a loss of biomass productivity and high-value compounds [27, 28]. Lastly algae can die. For example, most algae die within less than 30 minutes when exposed to 50 °C [29].
On the other hand, too low temperatures slow down carbon assimilation activity and metabolic reactions limiting photosynthesis [25]. In addition, the fatty acid composition of Arthrospira was also influenced by the cultivation temperature. Low temperatures led to an increase in the unsaturation of fatty acids of cyanobacteria, which affects the physical and biochemical properties of the lipid membrane [30] and has been discussed as a precautionary measure of microalgae to increase the membrane fluidity of the cell [31]. Careful temperature management is therefore crucial for the cultivation of microalgae.
In summary, it can be concluded that a process temperature of 30 °C seems to be optimal for the Arthrospira strain SAG21.99 cultivation in terms of a maximum biomass productivity. This was also true for the phycocyanin content which was at 30 °C significantly higher than at 20 or 40 °C.
