Abstract
BACKGROUND:
Lipedema is a chronic disease marked by symmetric enlargement of painful nodular and fibrotic adipose tissue, predominantly affecting the limbs. Since there is no specific test or biomarker for this condition, years often pass before the diagnosis of lipedema is established for the first time, thereby causing psychosocial distress, including depression, eating disorders, and social isolation. Over the last few years several advanced Doppler–based technologies have been developed to visualize slow flow blood vessels and superficial microvascular architecture undetectable by traditional color Doppler flow imaging (CDFI).
OBJECTIVE:
The aim of this study was to evaluate the superficial microvascular anatomy in lipedema patients compared to healthy controls and investigate the clinical significance of the Ultra Micro Angiography (UMA) technology in the diagnosis of lipedema. This new technique may contribute to reduce the diagnostic delay and, eventually, establish and guide treatment strategies toward a better therapeutic outcome in lipedema patients.
METHODS:
25 patients with lipedema and ten healthy controls with no history of lipedema were included in this study. All ultrasound examinations were performed on a novel high-performance ultrasound system (Resona R9/Mindray) using CDFI and the UMA technique.
RESULTS:
In all of the patients, Ultra Micro Angiography achieved the excellent visualization of microvascular structures, revealing that most lipedema patients showed grade 3 (n = 13) or grade 2 (n = 8) flow. UMA was superior to CDFI for depicting the microvascular structures.
CONCLUSIONS:
Here we show that UMA imaging characterizes the subcutaneous microvasculature with an unprecedented accuracy. The method has the advantage of being sensitive to small, slow-flowing vessels. This allows for the assessment of the course of vessels and vascular pathologies in great detail. Thus, UMA as a non-invasive diagnostic method can improve diagnostic accuracy in lipedema.
Introduction
Lipedema is a chronic condition characterized by a symmetrical enlargement of painful nodular and fibrous fatty tissue that predominantly affects the limbs but excludes the hands and feet. Lipedema often develops during hormonal changes such as puberty, pregnancy and menopause [1–3]. It is estimated that approximately 10% of the overall female population are affected by this disease [2]. The etiology of lipedema is still unknown, although some studies have suggested that polygenic susceptibility [4, 5] in combination with hormonal [6, 7], inflammatory, microvascular and lymphatic [8–11] disorders may be underlying etiologies. Affected women are often first confronted with the diagnosis of obesity and advised on the usual weight-reducing measures. Since there is no specific test or biomarker for this condition, years often pass before the diagnosis of lipedema is established for the first time, thereby causing psychosocial distress, including depression, eating disorders, and social isolation [3]. Usually, the condition is diagnosed based on history and physical examination results [12]. To date, ultrasound examination can determine the localization and extent of the fat proliferation and may help to distinguish lipedema from lymphedema at advanced stages of the disease [13–17]. In addition, the success of therapy can be monitored by ultrasound [18–20]. However, the potential to distinguish obesity from lipedema utilizing objective measurements obtained through ultrasound is yet to be demonstrated.
Current color- and power-Doppler techniques cannot accurately demonstrate vascularization of the dermis [21]. But in recent years, several advanced Doppler–based technologies [22, 23] including the novel Ultra Micro Angiography (UMA, Shenzhen Mindray Bio-Medical Electronics Co., Ltd., Shenzhen, China), have been developed to visualize slow flow blood vessels and superficial microvascular architecture undetectable by traditional color doppler flow imaging (CDFI).
The aim of this study was to evaluate the superficial microvascular anatomy in lipedema patients compared to healthy controls and to investigate the clinical value of UMA technology in the diagnosis of lipedema. This may contribute to reduce the diagnostic delay and establish treatment strategies toward a better therapeutic outcome in lipedema patients.
Materials and methods
Study design and participants
This prospective cross-sectional study was approved by the local Institutional Review Board and all participants provided informed written consent. Formal and documented ethical approval was obtained (reference number 22-3163-101). 25 patients with lipedema and ten healthy controls with no history of lipedema were included in this study. Following the german S1-guideline the inclusion criteria for lipedema patients are listed in Table 1 [24]. Clinical examination and conventional CDFI excluded other common causes of leg swelling such as chronic venous insufficiency and lymphedema. Healthy volunteers underwent clinical examinations to exclude the possibility of lipedema.
Inclusion criteria for lipedema patients (german S1-guidelines) [21]
Inclusion criteria for lipedema patients (german S1-guidelines) [21]
Patient characteristics
Values are mean ± standard deviation or absolute values.
All ultrasound examinations were performed by the same senior radiologist with more than 20 years of experience in dermatologic and musculoskeletal ultrasound, using Mindray Resona 9 ultrasound system with a linear L15-3WU probe (Mindray Bio-Medical Electronics Co. Ltd., China). The examination began with the participant in the supine position at the left saphenofemoral junction. The common femoral vein was evaluated for obstruction and reflux. To ensure reproducibility the following measurements were performed with the transducer placed longitudinally on the left anterior thigh on the middle third of the axis between the anterior superior iliac spine and the medial patella. UMA vascular blood flow images were obtained by applying the same imaging area in all examinations. During UMA evaluation, minimum pressure and a thick layer of ultrasound gel was applied on the transducer to prevent the vessels from collapsing. Color gain was increased until noise emerged and once maximum color flow signals were found, the transducer was held in the same scan position to observe and record UMA flow signals in the three sub-modes: color UMA (cUMA) demonstrates the velocity of microvascular blood flow, power UMA (pUMA) demonstrates the power intensity of microvascular blood flow and subtraction UMA (sUMA) demonstrates the power intensity of microvascular blood flow with higher sensitivity. The examinations were documented with single pictures or Cine-Loops and saved as digital DICOM files.
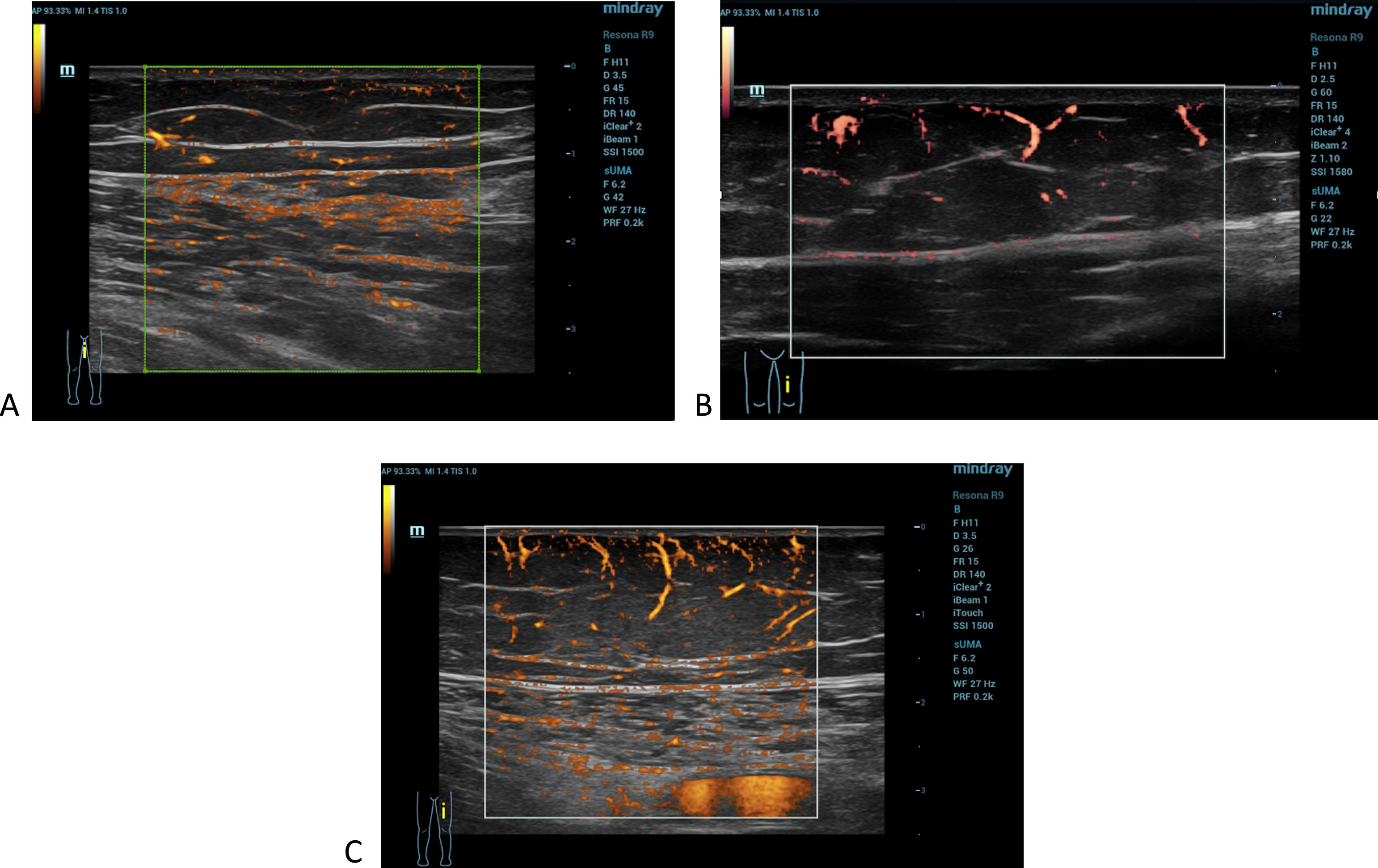
The UMA images according to each grade of flow: (A) grade 1, one or two defined vessels; (B) grade 2, three or four defined vessels; (C) grade 3, more than four linear vessels.
The images in sUMA mode were assessed for the presence of vascularity in the subcutaneous tissue layer. Any observed particularities were also noted. Vascularity was subjectively classified as follows: absent/ grade 0 (absence of defined vessels), minimal/ grade 1 (presence of one or two defined vessels, in which the course can be traced in its entirety over at least 5 mm), moderate/ grade 2 (presence of three or four vessels in which the course can be traced in its entirety over at least 5 mm) or marked/ grade 3 (presence of more than four vessels in which the course can be traced in its entirety over at least 5 mm).
Statistical analysis
Due to the explorative nature of this study, data analysis was mostly based on descriptive statistics. Continuous variables are presented as mean±standard deviation (SD) and minimum to maximum ranges. Categorical variables are presented as frequency (percentage).
Results
25 female patients with lipedema of the lower limbs (mean age 48.4 years, range 16-62 years) and 11 healthy controls (mean age of 47,1.6 years, range 21-82 years) with no signs of lipedema were studied. Among the 25 patients with lipedema, three patients presented with stage I, 17 patients with stage II and five patients presented with stage III lipedema. The majority of women with lipedema reported a family history of this condition (80 %). The onset of lipedema most often occurred during puberty (48%).
In all the participants, UMA achieved excellent visualization of subcutaneous microvascular anatomy, being superior to CDFI. The UMA image modes cUMA, pUMA and sUMA delivered clearer and more cohesive vessel branching detail in the subcutaneous tissue compared with CDFI (Fig. 2). In terms of presence and pattern of vascularity we observed a high variation among participants. UMA evaluation revealed that most lipedema patients showed grade 3 (n = 13) or grade 2 (n = 8) flow. In the healthy group five participants showed grade 1, three participants grade 2 and three participants grade 3 flow (Fig. 3).
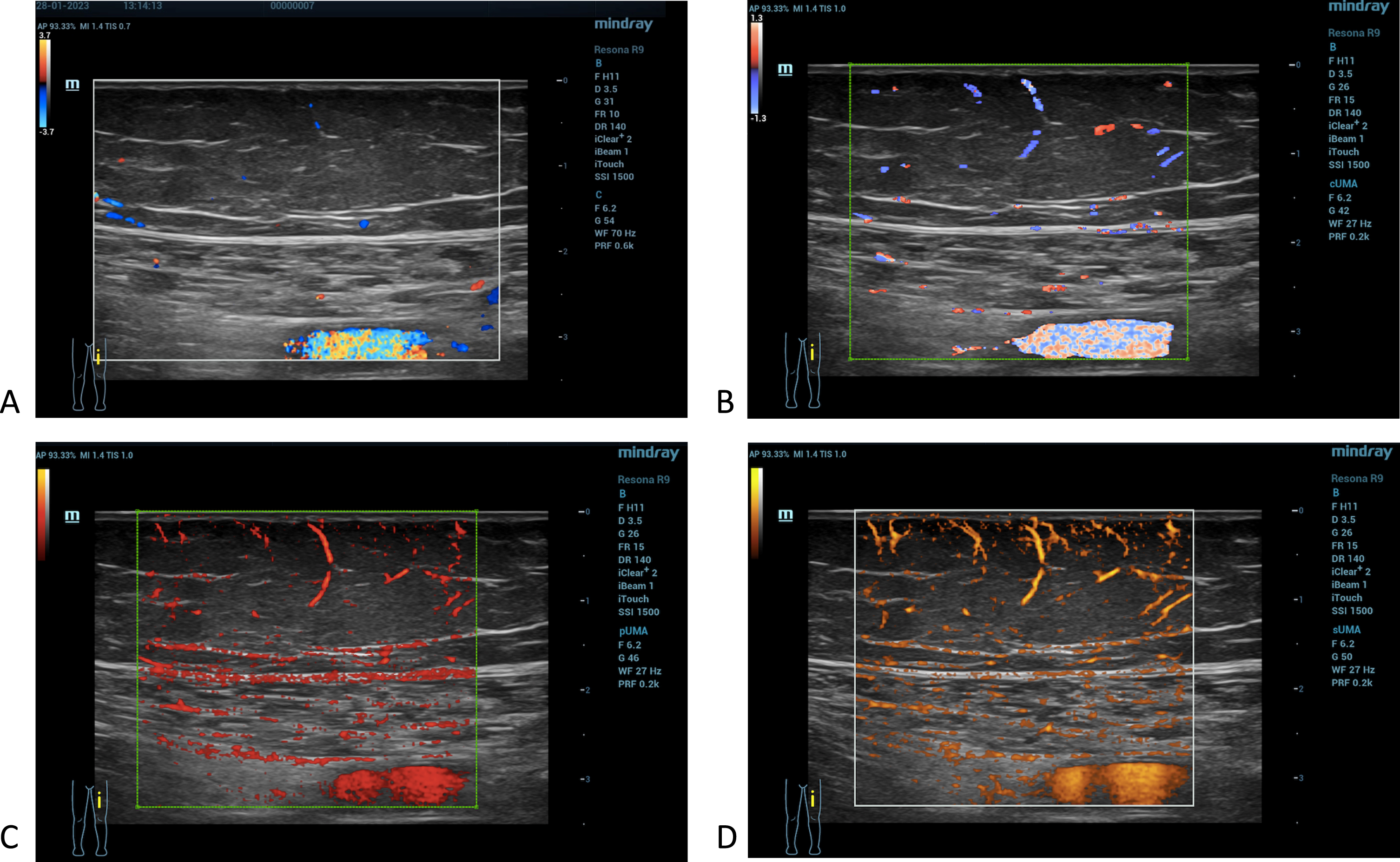
Ultrasound images of the left thigh of a 41-year-old stage III lipedema patient. The vascularity of the subcutaneous tissue was investigated using CDFI (A), cUMA image mode (B), pUMA image mode (C) and sUMA image mode (D).
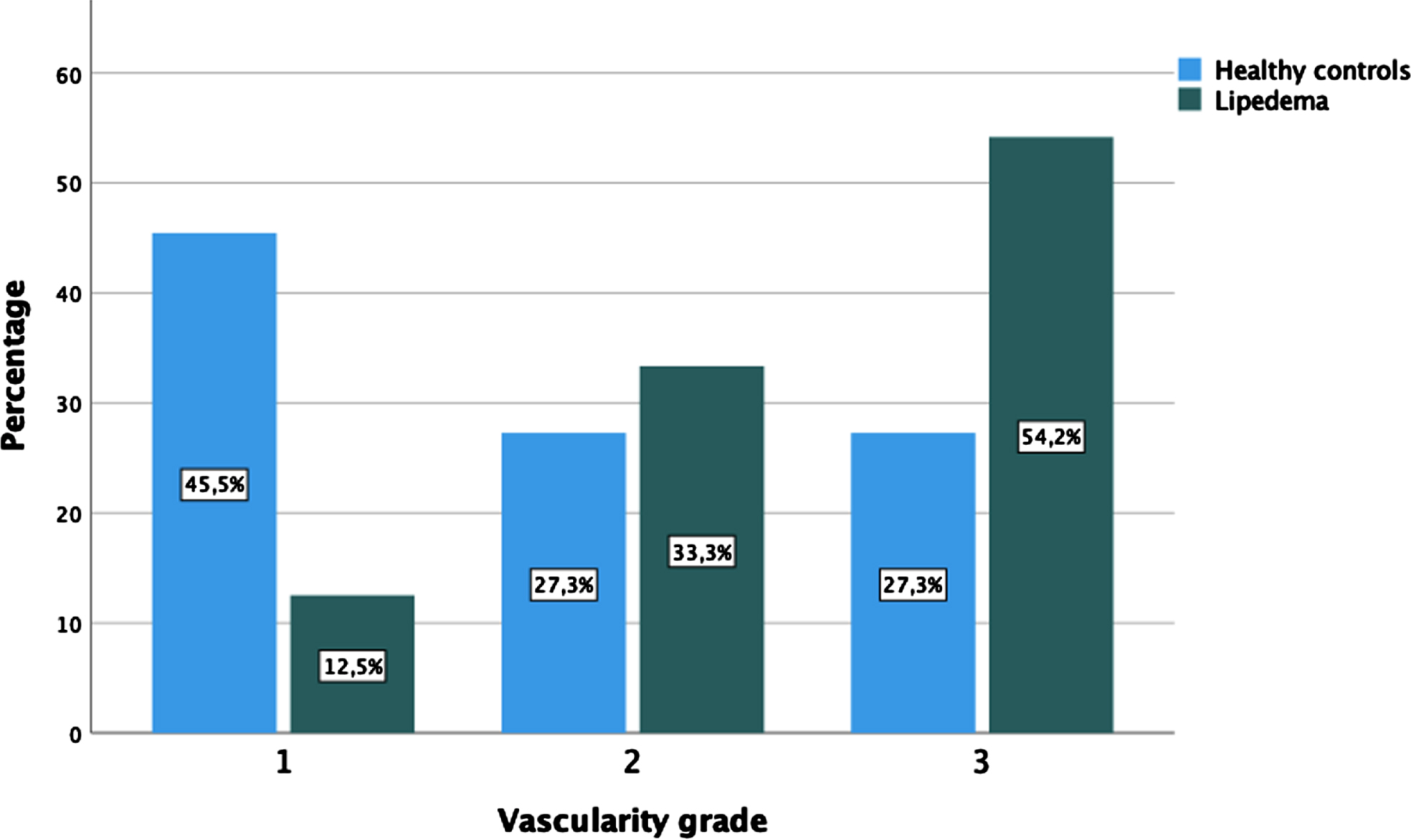
Bar graph displaying the frequency distribution of the UMA vascularity grades in the two participant groups.
In our study, we prospectively investigated the application of the Ultra Micro Angiography (UMA) technology to detect low velocity blood flow in subcutaneous tissue and microvascular (dys)function in lipedema patients compared to healthy controls. Our study is the first to investigate the utility of UMA for the assessment of subcutaneous vascularization in comparison to conventional color Doppler flow imaging (CDFI) in lipedema patients.
It is hypothesized that there is increased subcutaneous perfusion in the advanced stage of lipedema [11]. The hypothesis is consistent with our results (Fig. 3). Microaneurysms, calibre variations and fragility have been attributed to the subcutaneous vessels in lipedema in some studies [2, 11]. In our cohort, these pathologies were observed sporadically, but without clustering and without attribution to advanced stages. A striking feature in our cohort was the increased subcutaneous vascularization in grade 2 and 3 lipedema patients.
This increase in the number and size of blood vessels in lipedema, suggests increased inflammation burden with an accumulation of proinflammatory immune cells. Increased angiogenesis constitutes the fundament of the increased inflammatory burden [25–27] in lipedema. Angiogenesis, defined as the sprouting of new blood vessels from preexisting functional vessels and capillaries, is a multi-step process involving matrix degradation, vasodilatation, migration and activation of endothelia cells to form new lumens [28, 29]. The vascular endothelial growth factor (VEGF) is a crucial mediator and driver of angiogenesis in the physiological state and especially in tumor and inflammatory diseases [29, 30].
There are already data reporting increased neo-angiogenesis and an increased number of chronic inflammatory skin diseases e.g in psoriasis [32]. One study describes an increase in VEGF in lipedema patients compared to healthy people [33]. These data support the hypothesis of increased angiogenesis in lipedema patients and the increased number of blood vessels. We can confirm the increased number of subcutaneous blood vessels with our data. For vascular pathologies such as microaneurysms and calibre fluctuations, we could not define a pathognomonic pattern for lipedema.
To date diagnosis of lipedema is based on physical examination and searching for positive diagnostic criteria of lipedema. There is still no reliable test to make a certain diagnosis of this condition. The distinction between lipedema and obesity is very difficult in some cases, especially in women with greater BMI. Therefore, ultrasound analysis of skin thickness and hypoechogenicity of the subcutaneous fat can be valuable for the diagnosis of lipoedema (see Table 3) [13, 34]. Amato et al. have defined criteria that can be collected via ultrasound and consolidate the diagnosis of lipedema. The criteria allow the use of simple and reproducible ultrasound cut-off values for the diagnosis of lipedema of the lower extremities. Measurement of anterior tibial thickness, thigh thickness and lateral leg thickness have been recommended for the ultrasound diagnosis of lipedema in this study [14]. Iker et al. report that patients with lipedema have increased density and hypoechogenicity of subcutaneous adipose tissue. In particular the hypoechogenicity of the adipose tissue distinguished lipedema patients from lymphedema patients in this study. These differences could be detected via ultrasound [13]. Naouri et al. also reported that it is possible to differentiate between lymphedema and lipedema patients via ultrasound. In lymphedema patients, dermal hypoechogenicity was observed affecting the entire dermis. In lipedema patients, hypoechogenicity was localised at the ankle and predominated in the upper dermis [16].
Ultrasound features in lipedema and lymphedema
Ultrasound features in lipedema and lymphedema
To our knowledge subcutaneous blood flow in lipedema patients has not yet been investigated by ultrasound. There is profound evidence that subcutaneous microcirculation is increased and altered in lipedema [11]. Nowadays, different modes are available to evaluate the subcutaneous blood flow: CDFI, power Doppler imaging, B-Flow imaging and UMA.
CDFI is the most common method and is widely available. This technology, however, is dependent on the wall filter to decrease clutter and motion artifacts, and as a result, smaller, low-flow vessels are lost. To avoid a loss of signal related to slow flow the wall filter settings have to be kept at < 50 Hz, adapting the color gain setting until color noise is eliminated and decreasing pulse repetition frequency to < 1000 Hz. However, this would lead to an artifactual absence of blood flow (aliasing). Poor visualization in CDFI can also ocur when the angle of insonation to the vessel is at 90°.
Power Doppler Imaging (PDI) is an alternative to CDFI that shows elevated sensitivity to slow blood flow vessels, does not vary significantly with the direction of the blood flow and is less dependent upon the insonation angle.
B-flow technique is a digital subtraction imaging technique. Doppler artifacts such as aliasing are absent in B-Flow. Compared with the conventional color doppler ultrasound, the B-flow exhibits higher frame frequency and higher resolution but produces artifacts due to tissue motion and subtraction.
UMA acquires high-quality raw ultrasonic signals at high efficiency and utilizes an advanced tissue-rejection algorithm to remove tissue clutter from raw signals [35]. It has the advantage of detecting low-speed blood flow and displaying microvascular morphologies. The added value of this ultrasound technique has already been demonstrated in numerous diseases: rheumatoid arthritis [36], keloids [37], and thyroid nodules [38]. In our opinion, UMA could make a decisive contribution in diseases in which the microcirculation is altered. The successful visualization of superficial vessels in subcutaneous tissue using UMA technology may also be of great benefit for flap surgery [39, 40]. In a broader context, however, it must be emphasised that high-end ultrasound is only one piece of the puzzle in the diagnosis of the multifactorial lipedema disease. UMA may also be combined with contrast-enhanced ultrasound (CEUS) for the visualization of the microcirculation [41, 42]. However, CEUS requires intravenous injection of ultrasound contrast agents with its associated risks. UMA is a novel technique which can successfully depict microvascular blood flow signals without the use of contrast agents. The evidence for the widespread use of CEUS is given [42]; the UMA technique should be further investigated in prospective studies.
In conclusion, subcutaneous microcirculation in lipedema patients could be accurately assessed in our study using UMA. Increased subcutaneous perfusion was the leading pathology in the advanced stages.
Ultra micro angiography allows for the visualization of the subcutaneous microcirculation in lipedema, which is not visible in conventional color Flow imaging. The method has the advantage of being sensitive to small, slow-flowing vessels. Thus, the course of vessels and vascular pathologies can be studied in detail and characterized in relation to the clinical course and the response towards therapy. Of clinical relevance, UMA as a non-invasive diagnostic method can improve diagnostic accuracy in lipedema.
