Abstract
Cell-based
Aim of the study was to identify effective DPI concentrations for CPR/CYP activity modulation and potentially associated dose and time dependent hepatotoxic effects. The cells were treated with DPI doses up to 5,000nM (versus vehicle control) for a maximum of 48 h and subsequently examined for CYP3A4 activity as well as various toxicological relevant parameters such as cell morphology, integrity and viability, intracellular ATP level, and proliferation. Concluding, the experiments revealed a time- and concentration-dependent DPI mediated partial and complete inhibition of CYP3A4 activity in CYP3A4 overexpressing HepG2-cells (HepG2-CYP3A4). Other cell functions, including ATP synthesis and consequently the proliferation were negatively affected in both
Keywords
Introduction
In humans, the liver is the main organ for the metabolization and elimination of pharmaceuticals and xenobiotics due to the high expression of phase-1 and -2 enzymes in hepatocytes [1]. For this reason, hepatocytes are the subject of intensive research efforts, and
In the context of phase-1 biotransformation, microsomal enzyme complexes in hepatocytes, consisting of cytochrome P450 oxidoreductase (CPR) and cytochrome P450 monooxygenases (CYPs), are essential components for a large number of oxidative metabolic conversions of pharmaceuticals or xenobiotics [4, 5]. Despite the large number of different CYPs expressed in the human organism (57 are known to date), only a few, mostly from CYP families 1, 2, and 3, are responsible for the oxidative metabolization of more than 75%of all clinically approved drugs [2, 7]. The microsomal flavoprotein CPR has a significantly lower diversity compared to CYPs with only one individually expressed polymorphic variant [8–10]. As the obligatory electron donor for CYPs, CPR is essential for the liver-mediated phase-1 metabolism. Further, CPR plays a vital role in both oxidative processes catalysed by several oxygenase enzymes as well as biosynthesis and metabolism of various endogenous substances of the hormone and fat metabolism [9, 11]. During phase-1 biotransformation several successive oxidative reactions take place in which electrons and activated oxygen are transferred to a substrate in an nicotinamide adenine dinucleotide phosphate (NADPH)-dependent process [12, 13]. In detail, two electrons are initially transferred from NADPH to the prosthetic group flavin adenine dinucleotide (FAD) contained in CPR before these are transferred to flavin mononucleotide (FMN), another co-factor of CPR, by means of interflavin electron transfer. Sequential electron transfer follows this via redox cycling to a heme-bearing microsomal CYP, which catalyses the oxidative conversion of a substrate [14–16]. For the prediction of the pharmacokinetics of new drug candidates, including relevant metabolites and hepatotoxicity, a clear understanding of the enzymatic phase-1 and -2 reactions interplay in the liver is crucial. In this context, preclinical drug screening with regard to biotransformation and toxicology is mostly based on physiologically relevant sensitive, reliable and in particular adaptable
DPI, a member of diaryliodonium salts, is an aromatic heterocyclic cation. Owing to their electron deficient properties at the iodine center, diaryliodonium salts are frequently used as aromatic electrophiles in aryl transfer processes [22]. Its chemical nature makes DPI a potent inhibitor of flavin bearing oxidoreductases, which are generally an integral element of electron transport chains. DPI have a wide spectrum of known cellular targets including CPR [13, 23], NADPH oxidase (NOX) [24–31], mitochondrial respiratory chain complex I (NADH ubiquinone oxidoreductase) [28, 32–34], and different types of nitric oxide synthase [13, 35]. It is assumed that DPI inhibition is achieved by covalent modification of flavin and/or heme prosthetic groups within enzymes based on radical formation. NADPH-dependent inhibition of CPR by DPI occurs via irreversible modification of reduced FMN, which effectively prevent electron transfer to their physiological targets [13, 36–38]. In these studies, DPI could be shown as an effective CPR inhibitor in recombinant expressed protein isolates, rat and human liver microsomes as well as in several
In the past, inhibitory effects of DPI were investigated with regard to a potential application in the therapeutic field, i.e. as an antibiotic [29, 41], anti-cancer [31, 43], anti-inflammatory [26, 30] and/or vasodilatory agent [23]. For the analysis of phase-1 biotransformation inhibition, studies were mostly performed in less complex model systems with recombinantly expressed and purified proteins or derived from microsomal fractions in order to clarify size and range of DPI effects and the mechanism of action.
Therefore, the aim of our study was to investigate DPI as inhibitor of phase-1 activity via CPR/CYP inhibition in an
Methods
Cell culture
Commercially available human hepatocellular carcinoma (HepG2) cells (HB-8065, ATCC, Manassas, VA, USA) as well as genetically modified HepG2 with stable recombinant overexpression of CYP3A4 (HepG2-CYP3A4), generated and kindly provided by the “Molecular Cell Biology” group from the BTU Cottbus-Senftenberg [44], were cultured under standard conditions (37 °C, 5 %CO2) in polystyrene-based tissue culture flasks (SARSTEDT AG & Co. KG, Nümbrecht, Germany) in Dulbecco’s minimal essential medium (D-MEM) supplemented with 10 %fetal bovine serum (FBS) superior, 6 mM L-alanyl-L-glutamine and 49.2 g/L NaHCO3, all purchased from Biochrom GmbH (Berlin, Germany). During standard cell culture the culture medium was replaced every second day. Prior to the inhibition studies with diphenyleneiodonium (DPI), the HepG2-CYP3A4 cell line was post-selected by adding 3μg/mL Blasticidin (AppliChem GmbH, Darmstadt, Germany) to the culture medium over a period of two weeks [45]. No Blasticidin was present in the culture medium during the experiments with DPI. For either cell passaging or experimental seeding, hepatocytes were harvested by trypsin/EDTA treatment (0.05%v/v Trypsin and 0.02%v/v EDTA in water, Biochrom GmbH, Berlin, Germany).
CPR/CYP inhibition studies with diphenyleneiodonium (study design)
The presented study was divided in three consecutive parts. For the assessment of DPI mediated influences on both CYP3A4 monooxygenase activity or toxicological relevant parameters in hepatocytes, HepG2 and HepG2-CYP3A4 cells were seeded in all study parts at a density of 62.500 cells/cm2 into either 96-well or 24-well plates (SARSTEDT AG & Co. KG, Nümbrecht, Germany) 24 h prior to DPI-treatment. The setup of the first study part initially aimed to determine the concentration range of an effective DPI-mediated inhibition of phase-1 biotransformation in the
Based on the findings from the first study part, regarding effective DPI concentrations and the DPI-related influence on the intracellular ATP level, as well as anticipating experimental planning for future metabolization studies of substrates/drugs (for which longer conversion times of up to 48 h often are required), the following study parts were performed with an extended setup to elucidate possible time dependent and toxic DPI effects on the HepG2 based
Determination of CYP3A4 enzyme activity and intracellular ATP level
For the assessment of DPI-induced inhibition of CYP3A4 monooxygenase activity in hepatocytes, HepG2 and HepG2-CYP3A4 cells were analyzed with the P450-Glotrademark CYP3A4 induction/ inhibition assay (Promega, Madison, WI, USA), used according to the manufacturer’s instructions. Briefly, after DPI treatment, cells were incubated with 50μl CYP3A4 substrate Luciferin-IPA diluted in culture medium at 37 °C, 5 vol-%CO2 for 60 min. Subsequently, 25μl of supernatants were transferred into a white-walled 96-well plate (SARSTEDT AG & Co. KG, Nümbrecht, Germany) and an equal volume of luciferin detection reagent was added followed by incubation for 20 min at room temperature in the dark. Luminescence was measured with a FLUOstar Omega microplate reader (Software version: 3.00 R2, BMG LABTECH GmbH, Ortenberg, German), followed by data analysis by MARS Data Analysis Software (Version: 2.41). In addition, the cells and the 25μl substrate solution remaining in the initial 96-well plate were mixed with 25μl ATP reagent solution of the CellTiter-Glo® 2.0 assay (Promega, Madison, WI, USA) and incubated for 10 min in the dark. ATP level was detected by measuring luminescence with the FLUOstar Omega microplate reader to allow normalization to the effective cell number or assessment of DPI mediated influences on the intracellular ATP level.
Determination of cell integrity by LDH assay
To determine a possible concentration and/or time dependent influence of DPI on cell integrity, the amount of lactate dehydrogenase (LDH) released from the cytoplasm into the cell culture supernatant was determined in the second and third study part. For this purpose, the LDH Cytotoxicity Colorimetric Assay Kit II (Biovision GmbH, Ilmenau, Germany) was used according to the manufacturer’s instructions. The experiments were performed in 96-well format (SARSTEDT AG & Co. KG, Nümbrecht, Germany) with both cell lines using triplicates in two independent experiments (
Viability and cell density determination by FDA/PI fluorescent staining
DPI-induced changes in proliferation behaviour and cell viability were determined by live-dead staining of the cells with Fluorescein Diacetate (FDA) and Propidium Iodide (PI), both purchased from Sigma Aldrich (St. Louis, MO, US). FDA as a cell-permeant esterase substrate served as a vitality probe, whereby it is hydrolysed into its fluorescent form by intact and metabolically active cells. PI was used to detect dead cells, as it is a DNA-intercalating fluorescent dye that is not cell-permeant. Viability staining was performed in 24 well format (SARSTEDT AG & Co. KG, Nümbrecht, Germany) with both cell lines HepG2 and HepG2-CYP3A4 in two independent experiments with
Statistical analysis
For statistical analysis, one-way ANOVA with Turkey’s multiple comparison test was used to calculate differences between groups using Prism 8 software (GraphPad Software, San Diego, CA, USA). Probabilities lower than 0.05 were considered statistically significant.
Results
Short-term exposure with high-dose DPI completely inhibits CYP3A4 activity and is slightly affecting ATP level
For the experiments with DPI, parental HepG2 and HepG2-CYP3A4 with recombinant CYP3A4 overexpression (described previously [44]) were used as cell models. Initially, the main focus was to determine the DPI concentration range showing an inhibitory effect on phase-1 monooxygenase activity after a 30 min treatment. CYP3A4 activity in the HepG2-CYP3A4 cell line seemed to be slightly decreased already at 5 nM DPI (Fig. 1). Starting with a concentration of 50 nM, a significant reduction of CYP3A4 activity was caused by DPI (
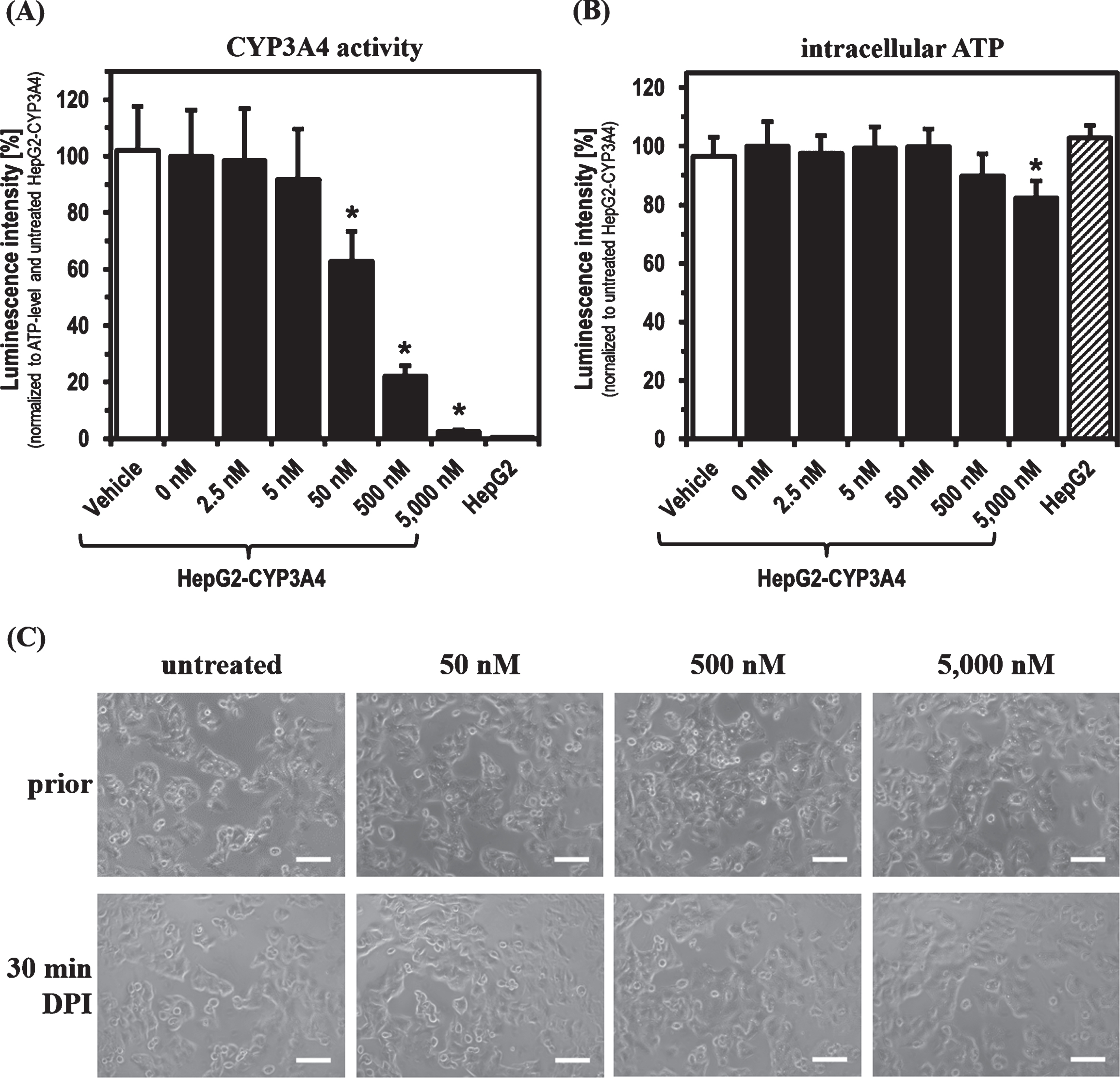
CYP3A4 activity and ATP level after 30 min DPI treatment. Determination of (A) CYP3A4 activity, (B) intracellular ATP level and (C) morphology of HepG2-CYP3A4 after 30 min DPI treatment (Mean±standard deviation; *
Next, we performed DPI treatments of HepG2 and HepG2-CYP3A4 for a longer period (48 h). In addition, we were interested to see if there could be a recovery of CYP3A4 activity as well as intracellular ATP level after short-term DPI treatment. For this, cells were treated with DPI concentrations between 1,000 and 5,000 nM for 30 min followed by 48 h of cultivation in DPI-free culture medium. As before, morphology of DPI-treated cells was analyzed and CYP3A4 activity as well as intracellular ATP level were measured. Moreover, a potential cytotoxic DPI effect on cell integrity was investigated by LDH assay, and the cellular viability status was analyzed with FDA/PI fluorescent staining.
As found with short-term treatments, DPI showed a concentration-dependent inhibitory effect on the CYP3A4 activity of HepG2-CYP3A4 also after 48 h of treatment (Fig. 2). A DPI concentration of 50 nM led to a significant reduction of CYP3A4 activity to about 60%(
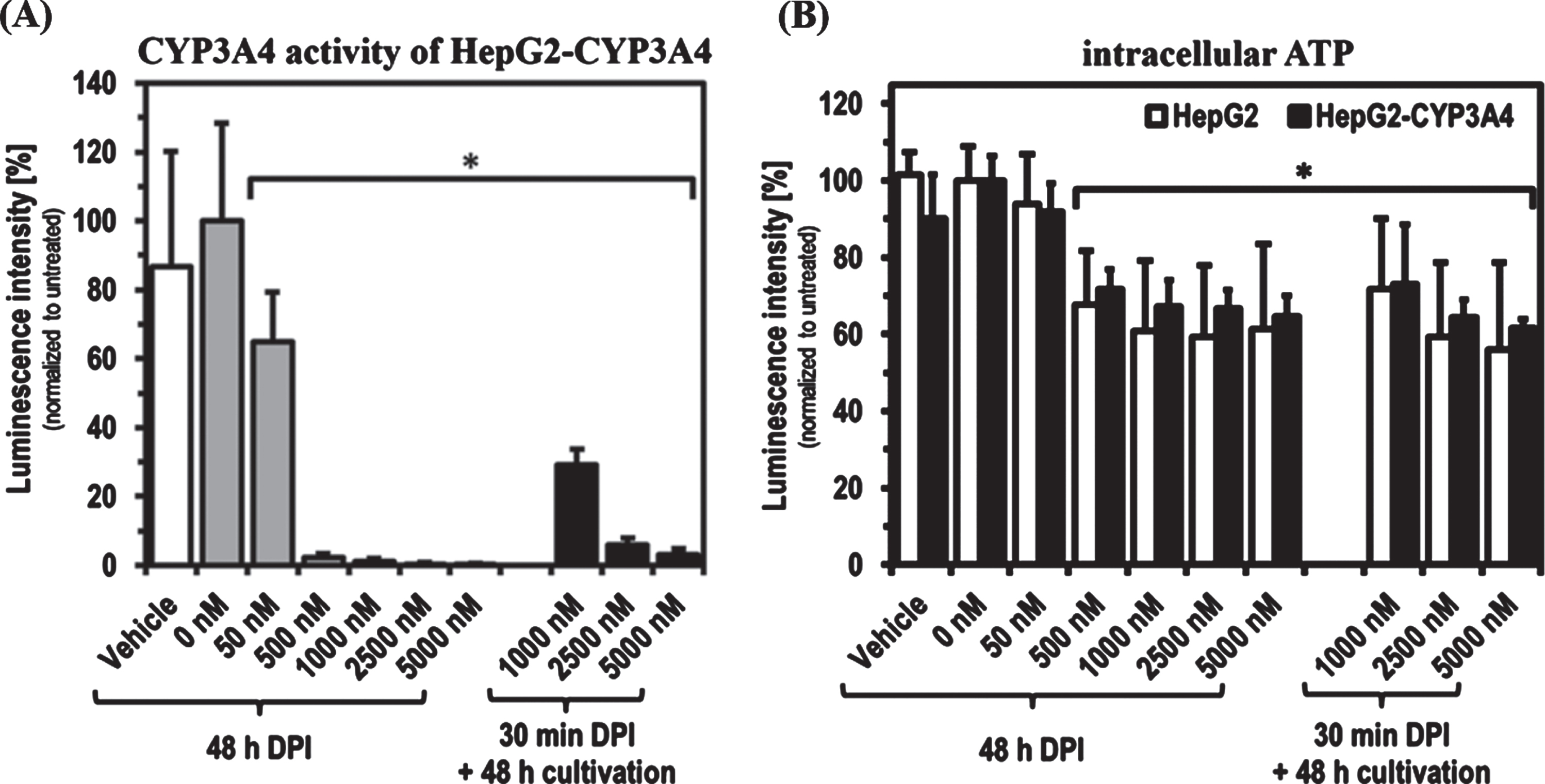
CYP3A4 activity and ATP level after 48 h DPI treatment as well as recovery after 30 min DPI treatment. Determination of CYP3A4 activity in HepG2-CYP3A4 (A) and intracellular ATP level in both cell lines (B) after DPI treatment for 48 h as well as for 30 min with following 48 h recovery in DPI-free medium (Mean±standard deviation; *
The experiments further revealed that, despite some DPI effects on ATP level, the cell integrity of both cell lines apparently was not negatively affected by DPI at any time (Fig. 3). The release of LDH was even slightly higher in the untreated cells and the vehicle controls (significant in HepG2 for all DPI concentrations). Direct comparison of the two cell lines showed only minor differences. Solely untreated HepG2 and its vehicle control tended to show an increased LDH release compared to HepG2-CYP3A4.
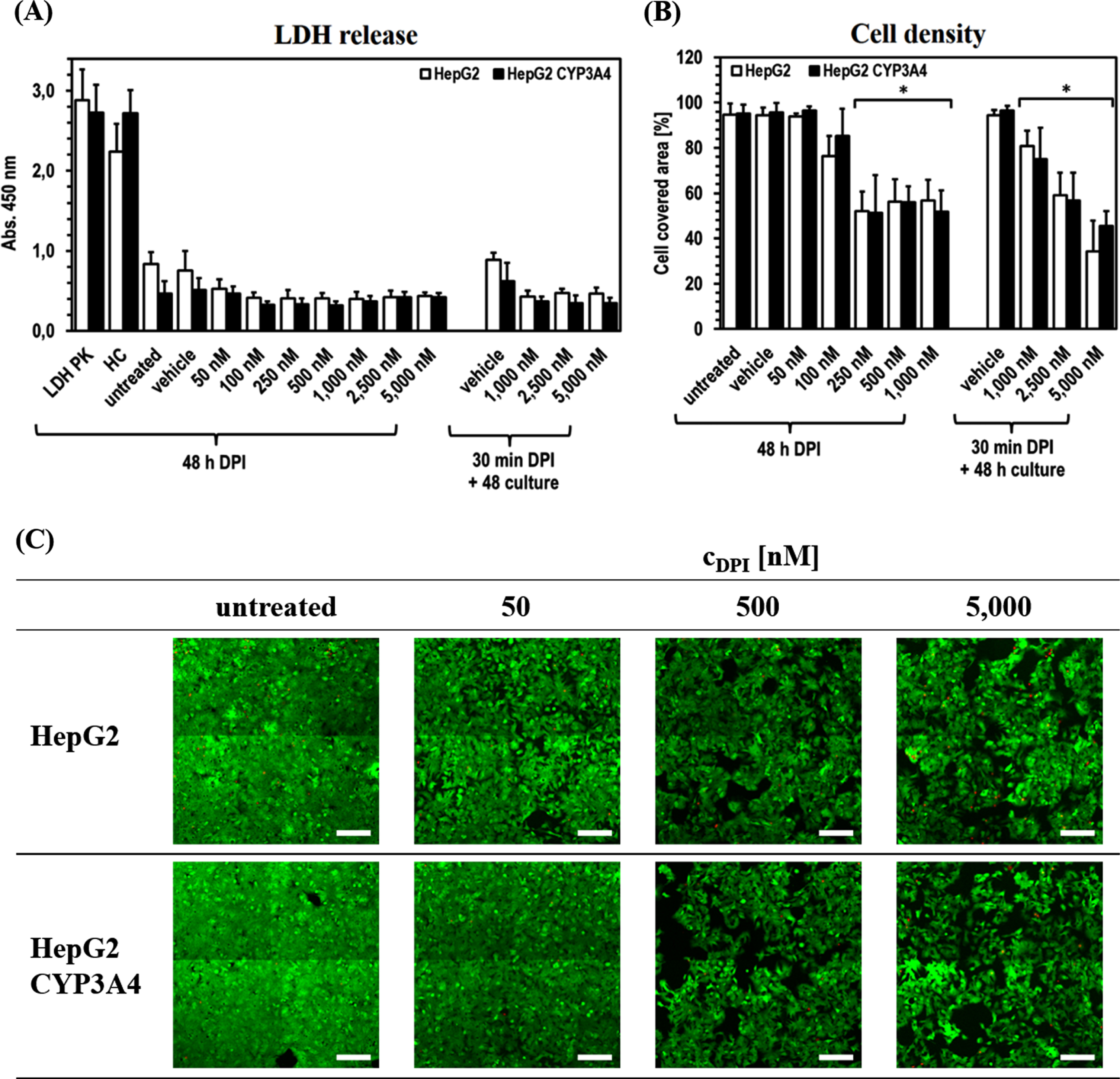
Cytostatic effect of DPI on HepG2 and HepG2-CYP3A4 cells. Analysis of the HepG2 and HepG2-CYP3A4 cell integrity via LDH release (A), metabolic activity via ATP level (B) and viability via FDA/PI staining (C) (Mean±standard deviation; *
The situation is different for the area covered by vital cells, which was used as a further evaluation parameter. In both cell lines, a comparable reduction of the covered area with increasing DPI concentration was observed. There was a significant difference for the area covered by vital cells to decrease to about 80%after 48 h of treatment with 100 nM DPI (pHepG2100 nM DPI < 0.0001). In HepG2-CYP3A4 only a slight tendency could be observed (pHepG2 CYP3A4100 nM DPI = 0.2710). At higher DPI doses in the range of 250–1,000 nM, a more extensive and in all samples significant reduction of cell density to ∼50%was visible (all
We were interested to evaluate the potential of diphenyleneiodonium (DPI) for the targeted modification of phase-1 monooxygenase activity in cell-based
In the first part of the study, we did not find any DPI effects on the cell morphology as analyzed by phase contrast microscopy. However, the strong CYP3A4 enzyme activity in the HepG2-CYP3A4 model could be significantly inhibited by DPI, depending on the concentration. For a relevant inhibition to approximately 20%of the original CYP3A4 activity of the HepG2-CYP3A4 cells, DPI concentrations of at least 500 nM were required. However, there was a negative effect on the intracellular ATP level at higher DPI concentrations detectable, which could have a serious impact on the on the energy balance and metabolism of hepatocytes. The aim of our study was to investigate not only a concentration but also a possible temporal dependence of the DPI effect on phase-1 activity. In addition, toxicological parameters such as cell integrity, viability and proliferation were analyzed to determine to what extent HepG2-CYP3A4 has the ability to regenerate phase-1 activity after a short 30 min DPI treatment and the extent to which toxicologically relevant effects emanate from DPI under these conditions.
With regard to the inhibition of CYP activity, there was no time dependence in the DPI effect when 50 nM was used. After both 30 min and 48 h DPI treatment the residual CYP3A4 activity was ∼60%, when compared to untreated HepG2-CYP3A4. The situation was different at higher DPI concentrations from 500 nM on, where compared to the 30 min treatment (∼20%residual activity) an almost complete inhibition of CYP3A4 activity was achieved after 48 h DPI treatment. Precisely in this concentration range, DPI mediated significant effects on intracellular ATP levels. This means that a substantial inhibition of phase-1 activity by DPI might have a negative impact on ATP synthesis. Higher concentrations of DPI did not further reduce the intracellular ATP level after 48 h of treatment. This could indicate that under the chosen experimental conditions 500 nM DPI was sufficient for maximum inhibition of CYP3A4 activity and the respiratory chain of the
With regard to the detected reduction of the intracellular ATP level after DPI treatment, experimental limitations result in ambiguities in the interpretation of the data. The decreasing intracellular ATP level with increasing DPI concentrations is probably partly due to the lower cell number after DPI treatment. A direct comparison of ATP levels between untreated and treated cells requires a comparable cell number. According to our cell density data, this is no longer given after 48 h treatment at least from 100 nM DPI and in the case of short treatment followed by 48 h cultivation in the third study part at higher DPI concentrations, as the cell density is already substantially lower. Since only the ATP amount in a complete well could be detected after 48 h, it is conceivable that the influence of DPI on the energy metabolism of the individual cell is less than suggested by the detected ATP level per well. However, it was already shown that DPI has an inhibitory influence on complex I of the respiratory chain [42], where the FAD cofactor of the mitochondrial localised NADH-ubiquinone oxidoreductase is a target for DPI [23]. In view of these findings on mitochondrial function in different cell types, as well as the observations from our experiments, it is clear that the ATP synthesis is directly linked to the proliferation [52–54]. The resulting conclusion is, that DPI reduces the ATP level within a short period of time, which has a negative effect on proliferation and results in a reduced cell density after 48 hours.
In our studies, a partial recovery of CYP3A4 activity of up to 30%could also be observed after 48 h of cultivation under DPI-free conditions, following an almost complete inhibition by 30 min treatment with 1,000 nM DPI. These observations do not necessarily contradict findings by others concerning irreversible inhibition of DPI targets [13, 15]. These measurements were made with protein isolates or microsomes. A cell-based system may has the possibility of reproducing enzymes and thus restore enzyme activity over time. At 2,500 and 5,000 nM DPI no recovery could be observed, as both phase-1 residual activity was still reduced below 10%after 30 min treatment followed by DPI-free cultivation as well as ATP levels and cell density were comparable to cells treated for 48 h.
Conclusion
The objective of the study was to investigate the potential of DPI as an inhibitor of phase-1 monooxygenase activity for
Footnotes
Acknowledgments
This work was funded by grants of the Ministerium für Wirtschaft, Forschung und Kultur (MWFK, state of Brandenburg, Germany) for the Fraunhofer Project Group “Pilzbasierte zellfreie Synthese-Plattformen –PZ-Syn” (project number 22-F241-03-FhG/005/001).
