Abstract
BACKGROUND:
The accurate Tumor-Node-Metastasis (TNM) staging of colorectal cancer (CRC) is of great guiding significance for the judgment of tumor progression and prognosis, and the formulation of treatment strategies.
OBJECTIVE:
The aim of this study was to construct a recurrence risk scoring (RRS) system and prognostic prediction model to improve the accuracy of staging, prognosis prediction, and clinical decision making in resectable CRC.
METHODS:
CRC patients who underwent radical resection were retrospectively enrolled into study. Multivariable Cox regression model was applied to screen for independent prognostic factors. The RRS system is composed of independent prognostic factors which was awarded 1point each. A prognostic model composed of RRS and TNM staging system (RRS-TNM model) was applied to predict postoperative recurrence.
RESULTS:
TNM stage, tumor differentiation, preoperative elevated Carcinoembryonic Antigen, Carbohydrate Antigen 199, Prothrombin Time and Fibrinogen were the independent prognostic biomarkers. 173 of 540 patients had recurrence. The 5-year cumulative recurrence rate (5-y CRR) and disease-free survival (DFS) of postoperative p-TNM stage I, II, and III were 12.7% and 104.8 months, 26.5% and 89.3 months, and 55.5% and 57.3 months, respectively. The 5-y CRR and DFS of preoperative Low-risk (RRS 0-1score), Middle-risk (RRS 2-3scores), and High-risk (RRS 4-5scores) groups were 13.9% and 101.1 months, 40.9% and 75.5 months, and 70.2% and 41.1 months. The AUC (area under ROC curve) of RRS system was not inferior to that of TNM staging system (0.713 vs. 0.666;
CONCLUSIONS:
The RRS system accurately identifies CRC patients with high-risk recurrence preoperatively. Constructing a nomogram using the RRS system and TNM staging significantly improves the accuracy of staging and prognosis prediction, which is of great clinical significance for individualized clinical treatment and follow-up of CRC.
Introduction
Colorectal cancer (CRC) is one of the most common tumors, with the third highest morbidity and the second highest mortality worldwide [1], and ranks fifth in terms of morbidity and mortality in China [2], one of the main reasons for the poor prognosis of CRC is its high postoperative recurrence rate [3, 4]. As the gold standard of treatment decision and prognosis assessment, the Tumor-Node-Metastasis (TNM) staging system is widely used in clinical management of CRC [5]. However, the prognosis of CRC patients with the same pathological TNM (p-TNM) stage may be quite different in the clinic [6, 7, 8, 9], indicating that p-TNM staging alone cannot accurately reflect the true staging and predict the prognosis of CRC. Therefore, finding effective biomarkers to improve the accuracy of tumor staging system and accurately predict postoperative recurrence is of great significance for the individualized treatment and prognosis improvement of CRC.
Tumor markers are widely used in clinical tumor screening and monitoring of postoperative recurrence and metastasis. Carcinoembryonic antigen (CEA) and Carbohydrate antigen 199 (CA199) are abnormally elevated in several tumors, particularly those from the digestive tract [10, 11]. Studies have suggested preoperative CEA maybe an efficient predictor of recurrence of CRC after curative surgery [12, 13, 14, 15, 16]. Increased preoperative CA199 level was possibly associated with poor differentiation and prognosis in CRC [17, 18]. However, it is still controversial whether tumor markers can be used as biomarkers to predict postoperative recurrence of CRC. It is urgent to search for better biomarkers to predict recurrence and improve the accuracy of TNM staging. Studies have shown that coagulation parameters are valuable for predicting postoperative recurrence of CRC [13, 19, 20]. Furthermore, elevated preoperative prothrombin time (PT) level has been reported as a predictor of poor prognosis in CRC [19, 21] and increased level of fibrinogen (FIB) was associated with metastases and poor prognosis in patients with resectable CRC [13, 19, 22, 23]. The degree of pathological differentiation has been shown to be a reliable prognostic marker in CRC patients and significantly related with the overall stage of TNM system [24].
Due to the high heterogeneity of CRC, a single biomarker may not elevated in all patients, leading to limitations in its validity for prognostic prediction [25, 26]. The combined detection of multiple biomarkers may make up for the shortcomings of single biomarker detection to a certain extent [27, 28, 29], and has become an important tool for accurate staging and monitoring of recurrence of CRC [17].
The aims of the present study were: 1) identify and validate a recurrence risk scoring (RRS) system based on the most reliable prognostic markers in order to improve the accuracy of postoperative recurrence prediction. 2) combining the RRS system with TNM staging system (RRS-TNM model) accurately distinguishes patients with different recurrence risks and predicts the postoperative recurrence of CRC.
Materials and methods
Patients
This is a retrospective cohort study. The patients were selected from those stage I-III CRC patients who underwent curative resection in Zhongshan Hospital of Xiamen University from January 1, 2010 to December 31, 2018. The Ethics Review Committee of Zhongshan Hospital of Xiamen University approved this study (ZSYY20191102). These patients were included when: 1) with TNM stage I, II, or III CRC; 2) underwent radical resection. The exclusion criteria were: 1) without detailed preoperative data of coagulation indexes and tumor markers; 2) with preoperative chemotherapy or chemoradiotherapy; 3) with history of malignant tumor; 4) with coagulation disorders or history of anticoagulants or thrombolytic treatment. Ultimately, a total of 540 CRC patients were included (Fig. 1). All patients were given appropriate treatment according to the treatment guidelines and no additional clinical trials were performed on them.
Study design and research object selection. Abbreviations: CEA, carcinoembryonic antigen; CA199, carbohydrate antigen 199; PT, prothrombin time; FIB, fibrinogen; PLT, platelet.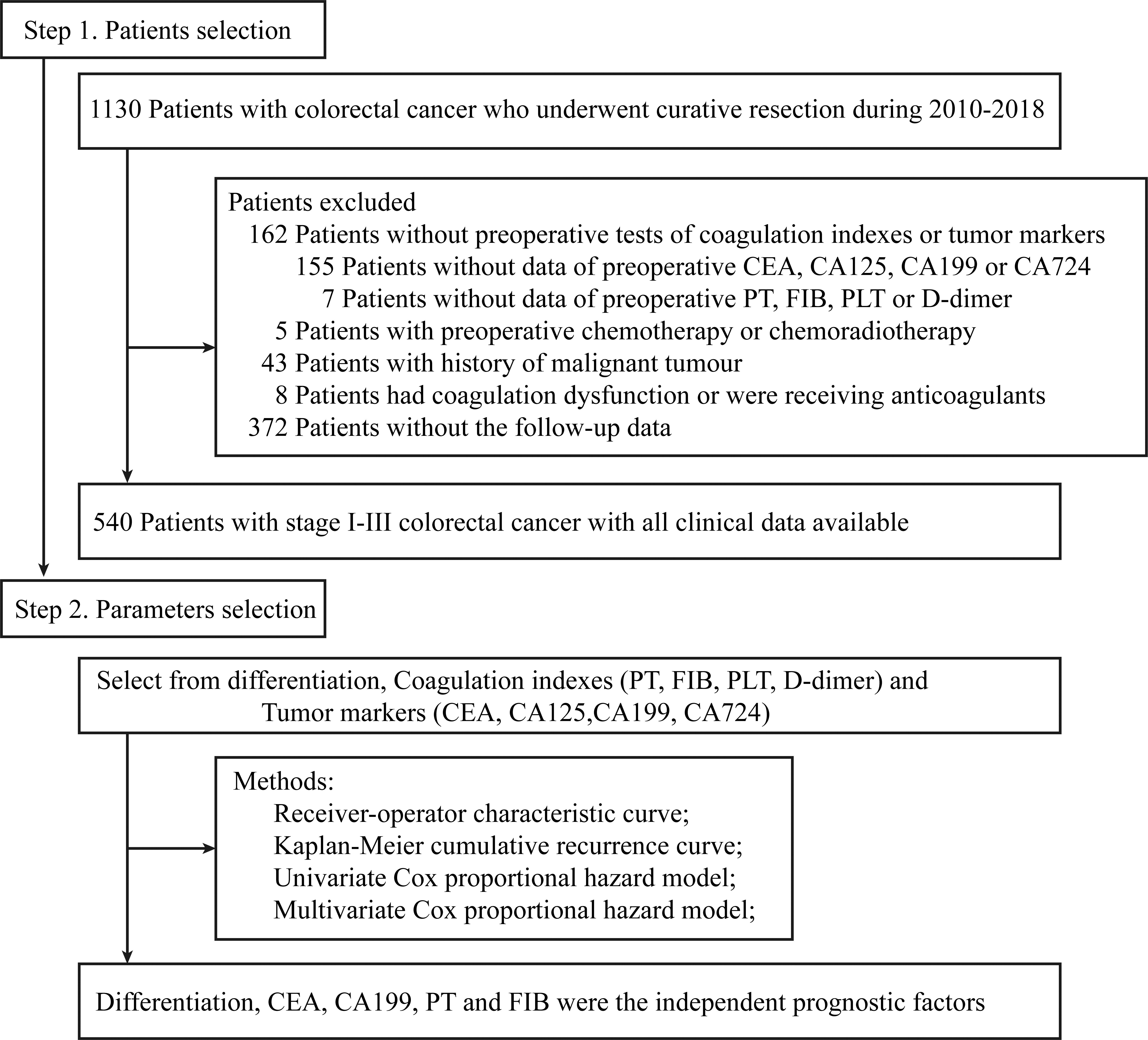
The clinical and pathological characteristics of the included patients were collected from the medical records, including gender, age, tumor location, tumor differentiation grade, T stage (the infiltration depth of primary tumor), N stage (lymph node metastasis), TNM stage, vascular and neural invasion. Tumor markers and coagulation indicators were analyzed, including CEA, CA125, CA199, CA724, platelet (PLT), D-dimer, PT, FIB. Through statistical analysis, five indicators (differentiation, CEA, CA199, PT, FIB) were selected as the components of the RRS system. Then the RRS system was integrated with TNM staging system into a prognostic prediction model (RRS-TNM model).
In our study, we used the 5-year cumulative recurrence rate (5-y CRR) and disease-free survival (DFS) to evaluate the outcomes of patients, DFS was defined as the time from operation to recurrence or metastasis. And this study was reported according to STROBE checklist [30].
Statistical analysis
All statistical analyses were based on IBM SPSS STATISTICS 26 software. Receiver-operator characteristic (ROC) curve was used to confirm the optimal sensitivity and specificity of these indexes, between the value corresponding to the maximum Youden Index and upper normal limits, the cut-off value of each included indicators is the one with higher sensitivity. The relationship of research parameters and clinicopathological indicators was depicted by the Pearson chi-square test. The effects of parameters on recurrence and metastasis were described by Kaplan-Meier cumulative recurrence curve, Log-Rank test depicted the difference between subgroups of parameters. Univariate and multivariate Cox proportional hazard model were used to analyze the prognostic factors of recurrence and metastasis. Nomogram and calibration curves were used to evaluate the prognostic model. Area under ROC curve (AUC) and Concordance index (C-index) were used to assess the discriminative ability. Risk score distribution was used to describe the accuracy of this prognostic model for recurrence, decision curve analysis was applied to compare the net benefit of patients using TNM stage and prognostic model.
Results
Clinicopathological characteristics of the included patients
A total of 540 stage I-III CRC patients who underwent curative resection were enrolled in this study. There were 228 females and 312 males. The median age of included patients was 66.5-year, ranging from 29 to 92, and 358 of them (66.3%) were older than 60 years. 454 participators (84.1%) had colon cancers in this study. The pathological report showed that well tumor differentiation was found in 442 patients (81.9%). A total of 205 patients (38.0%) had T stage1-3 primary tumors; lymph node metastasis was found in 230 patients (42.6%). There were 66 patients (12.2%) with TNM I staging, 230 patients (42.6%) with TNM III staging. Vascular invasion occurred in 299 patients (55.4%), and neural invasion was caught in 401 patients (74.3%). By the end of the follow-up, 173 patients (32.0%) had recurrence and metastasis (Table 1).
Clinicopathologic features and demographics of the included patients
Clinicopathologic features and demographics of the included patients
During variable screening, we compared the AUCs of 4 cancer markers and 4 coagulation markers, the AUCs of CEA, CA125, CA199 and CA724 were 0.646, 0.529, 0.613, and 0.537, respectively; and the AUCs of PLT, D-dimer, PT and FIB were 0.514, 0.551, 0.630, and 0.610, respectively (Fig. 2a). We selected the four metrics with the largest AUC and according to the Youden Index, the cut-off values of CEA, CA199, PT and FIB were 5 ng/mL, 20.6 U/mL, 13 s and 3.8 g/L, respectively. Then we analyzed the survival differences at different levels of these indicators, the results showed that they all were significant (all
The association of clinicopathologic characteristics and demographics with the research indicators
The association of clinicopathologic characteristics and demographics with the research indicators
CEA, carcinoembryonic antigen; CA199, carbohydrate antigen 199; PT, prothrombin time; FIB, fibrinogen.
Variable screening based on ROC curves and Kaplan-Meier curves. (a) ROC curves of CEA, CA199, PT and FIB. Kaplan-Meier curves of 5-y CRR according to preoperative levels of CEA (b), CA199 (c), PT (d), FIB (e) and tumor differentiation (f). Abbreviations: ROC, receiver-operator characteristic; CEA, carcinoembryonic antigen; CA199, carbohydrate antigen 199; PT, prothrombin time; FIB, fibrinogen; 5-y CRR, 5-year cumulative recurrence rate; DFS, disease-free survival; 95%CI, 95% confidence interval. 
The poorly tumor differentiation, elevation of CEA, CA199, PT and FIB was associated with recurrence and metastasis (all
Univariate and multivariate Cox proportional hazard analysis for DFS
CEA, carcinoembryonic antigen; CA199, carbohydrate antigen 199; PT, prothrombin time; FIB, fibrinogen; HR, Hazard Ratio; 95%CI, 95% confidence interval.
The RRS system was composed of the total scores of the five independent prognostic factors mentioned above, and each positive independent prognostic factor gets 1 point. Consequently, the enrolled patients were assigned a score from 0 to 5 (six groups) based on the number of positive indicators, we performed a survival analysis for these six groups, the results of log-rank test indicated non-significant difference in prognosis between scores 0 and 1 (
Kaplan-Meier curves of 5-y CRR according to different systems. (a) Kaplan-Meier curves of 5-y CRR according to the RRS system. (b) Kaplan-Meier curves of 5-y CRR according to the TNM staging system. (c) Kaplan-Meier curves of 5-y CRR according to the various risk levels in early-stage. (d) Kaplan-Meier curves of 5-y CRR according to the various risk levels in advanced-stage. (e, f) Kaplan-Meier curves of 5-y CRR according to the various risk levels in each TNM stage. Abbreviations: 5-y CRR, 5-year cumulative recurrence rate; DFS, disease-free survival; 95%CI, 95% confidence interval; RRS, recurrence risk scoring. 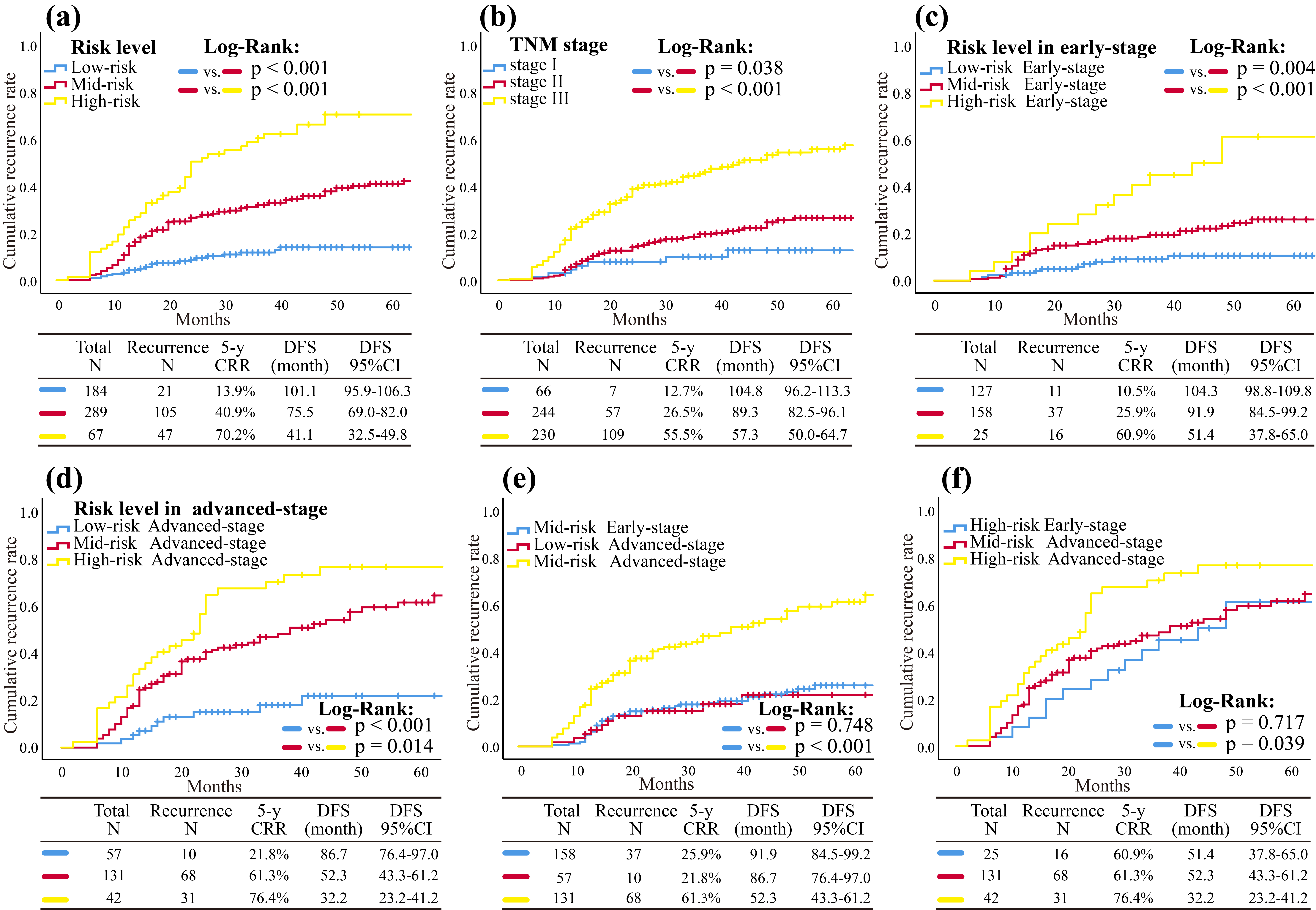
The prognosis of patients with different stages was significantly different (
Accurate screening of high-risk recurrent CRC patients at all stages of TNM using the RRS system as a biomarker
Prognostic differences at different risk levels for each TNM stage were also explored in depth. For early-stage CRC patients, the 5-y CRR and DFS in low-risk, medium-risk and high-risk groups were 10.5% and 104.3 months, 25.9% and 91.9 months, and 60.9% and 51.4 months, respectively (Fig. 3c). In advanced-stage CRC patients, the 5-y CRR and DFS in low-risk, medium-risk and high-risk groups were 21.8% and 86.7 months, 61.3% and 52.3 months, and 76.4% and 32.2 months, respectively (Fig. 3d). Interestingly, early-stage patients with higher risk had a worse prognosis than advanced-stage patients with lower risk. The differences in 5-y CRR and DFS between each risk level within early group were obvious (all
Construction and evaluation of a combined RRS-TNM model
Based on the results of the multivariate COX regression analysis, we formed the RRS-TNM model with all six independent prognostic factors, which is a combination of RRS system and TNM staging system. A nomogram was constructed for stage I-III CRC (Fig. 4a), calibration curves showed favorable constancy between the prediction of nomogram and actual conditions (Fig. 4b). The risk score distribution implied that this RRS-TNM model can well distinguish the prognosis of patients with different risk scores (Fig. 4c). The C-index value of the RRS-TNM model was 0.721 (95%CI, 0.701–0.742) and the AUC was the maximum in these three systems (0.770; 95%CI, 0.727–0.812), which was markedly higher than that of TNM staging system (0.666; 95%CI, 0.618–0.714;
Evaluation of the prognostic prediction model. (a) A nomogram for prognostic prediction of stage I to III CRC patients who underwent curative resection. (b) Calibration curves of 3-y and 5-y CRR for stage I to III CRC. (c) The risk score distribution and CRR status of this predictive model. (d) ROC curves of TNM staging system, RRS system and prognostic prediction model, and the comparison of AUCs of them. (e) Decision curve analysis for TNM staging system and predictive model (3-y CRR). (f) Decision curve analysis for TNM staging system and predictive model (5-y CRR). Abbreviations: ROC, receiver-operator characteristic; CEA, carcinoembryonic antigen; CA199, carbohydrate antigen 199; PT, prothrombin time; FIB, fibrinogen; 5-y CRR, 5-year cumulative recurrence rate; 95%CI, 95% confidence interval; AUC, Area under ROC curve; RRS, recurrence risk scoring.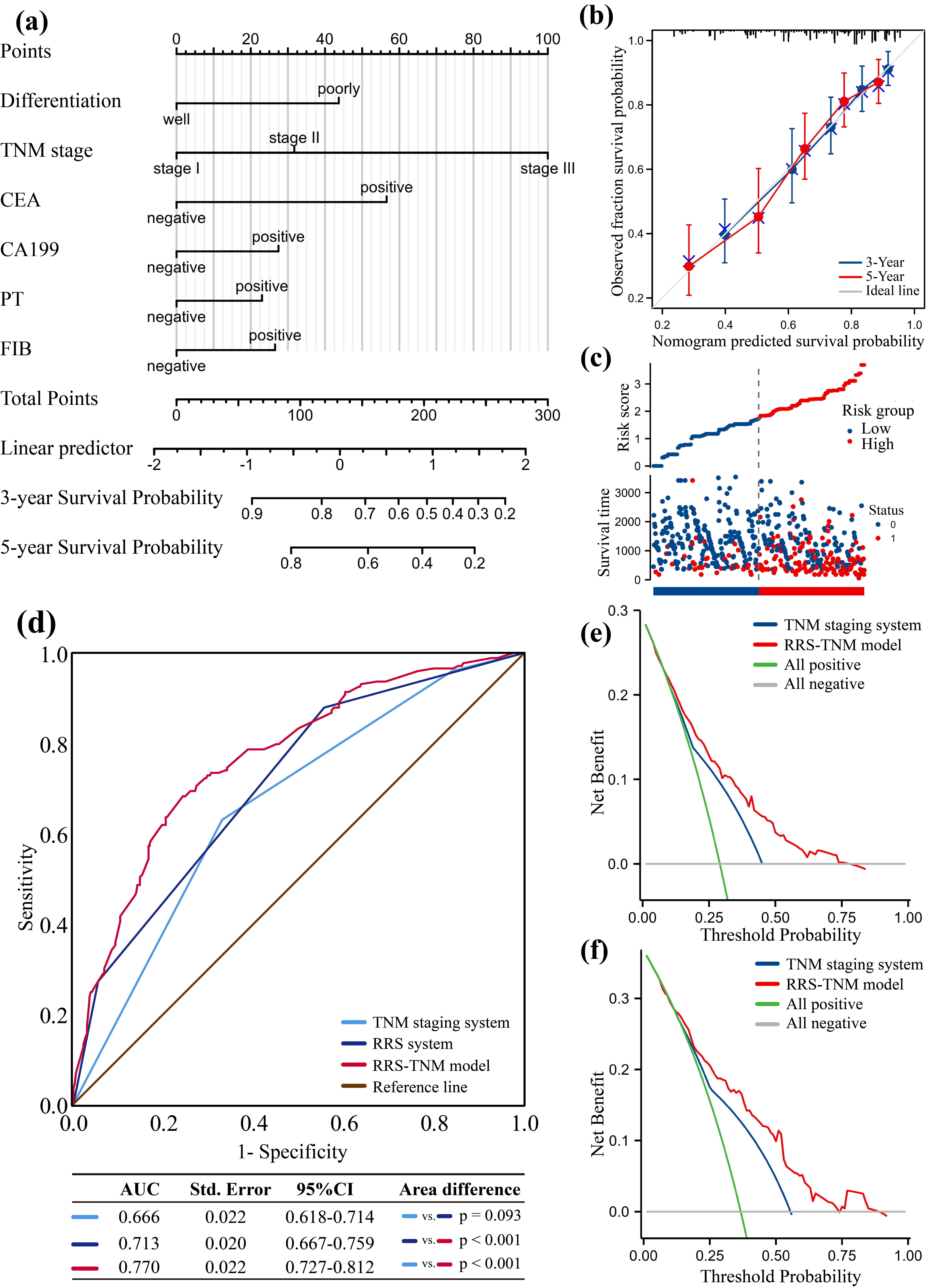
The accuracy of the TNM staging system, which consists of tumor infiltration depth, lymph node metastasis, and distant metastasis, is limited by the level of medical personnel and medical equipment. The differences in prognoses of CRC patients with the same TNM stage strongly appeal a novel staging system or an effective supplement to the TNM stage to achieve more accurate classification [31]. The progression of CRC may be accompanied by elevated levels of tumor markers and anomalous coagulation. Tumor differentiation, preoperative CEA, CA199, PT and FIB levels showed some effect in differentiating recurrent and metastases after radical resection in CRC patients. Many studies have suggested that pretreatment tumor markers including CEA and CA199 are prognostic indicators of CRC [12, 16, 17, 18, 32, 33, 34], these biomarkers can provide a certain predictive value for postoperative recurrence and metastasis of CRC; moreover, coagulation indexes are also associated with prognoses of patients [19, 21, 22, 23, 35, 36, 37]; but they don’t have satisfactory sensitivity and specificity.
In response to this clinical question, we selected several indicators significantly relating to prognosis to form a RRS system, then used this system to accurately classify patients with various prognoses, and combined with the TNM staging system to achieve higher accuracy for prognostic prediction.
In our study, we found that pathological grade of tumor differentiation, preoperative levels of CEA, CA199, PT and FIB were closely related to the lymph node metastasis, TNM stage and postoperative recurrence (all
Then we evaluated the prognostic differences between different risk levels. The results of statistical analysis indicated that patients with high-risk had a significantly worse prognosis than mid-risk patients (
We also used all six independent prognostic factors to compose an RRS-TNM model. The nomogram, as a predictive method, was widely used in recent years [38, 39, 40], we utilized nomogram to visualize survival analysis results. The C-index of RRS-TNM model was 0.721 and the AUC was 0.770, whereas, the AUCs of TNM staging system and RRS system was 0.666 and 0.713, respectively, which were both lower than that of RRS-TNM model. Moreover, the smooth calibration curves showed the consistency between predicted and actual probabilities, and decision curve analysis of the 3-y and 5-y CRR for both TNM staging system and RRS-TNM model also demonstrated a higher net benefit for the RRS-TNM model, indicating that using this model for prognosis prediction can achieve relatively realistic results. It also illustrates that the TNM staging system together with our constructed RRS system can significantly improve the accuracy of TNM staging system for prognosis prediction.
TNM stage is an indispensable parameter for describing the cancer. [8]; and the postoperative pathology reports include items on the tumor differentiation grade; CEA, CA199, PT, and FIB are common entries in preoperative examinations. Therefore, this model was constructed without the need to perform additional tests on the patients. Moreover, the levels of CEA, CA199, PT and FIB are objective data and are not influenced by the subjective judgement of medical staff, making this system more reproducible and actionable, and making the system a powerful complement to the TNM staging system. In addition, the RRS system, which consists of preoperative objective indicators, can be used to predict patient prognosis before surgery and serve as a good supplement to preoperative staging to guide the development of treatment plans. Although some literature has shown the value of these metrics, combining them into one model to predict prognosis is the first of its kind.
In a nutshell, the RRS system constructed in this study can be used clinically to supplement and modify the TNM staging system, and the prognostic model can be used after surgery to predict the prognosis of patients in order to develop corresponding treatment and follow-up plans.
Our study has certain limitations. First, the number of cases included in this study was small and the study was a single-center retrospective cohort study, and second, the recurrence rate of stage I CRC patients in this study was slightly higher than previously reported [41], which we attribute to the instability of the results due to the small sample. Therefore, this research work provides clinicians with a novel model, which has reference significance for the evaluation and prediction of stage I to III CRC prognosis, but its effectiveness and reliability need to be verified by further large sample studies.
Conclusion
In conclusion, pathological degree of tumor differentiation and the preoperative levels of tumor markers (CEA and CA199) and coagulation indexes (PT and FIB) are positively related to the prognosis of stage I to III CRC. The performance of RRS system is comparable to that of TNM staging system, demonstrating that the combination of these five indicators can provide an accurate classification of CRC patients’ prognoses. The prognostic model constructed from RRS and TNM staging system can accurately predict the prognosis of patients, which provides a better predictive value than TNM staging system.
Author contribution
Conception: JCC, YYZ, and KW.
Interpretation or analysis of data: KW, LLM, LYC, and YTJ.
Preparation of the manuscript: KW, YYZ, and LLM.
Revision for important intellectual content: KW and YYZ.
Supervision: JCC and YYZ.
Ethical approval
The Ethics Review Committee of Zhongshan Hospital of Xiamen University approved this study (ZSYY20191102).
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230116.
sj-pdf-1-cbm-10.3233_CBM-230116.pdf - Supplemental material
Supplemental material, sj-pdf-1-cbm-10.3233_CBM-230116.pdf
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81502039), the Natural Science Foundation of Fujian Province (2020J011214), the Health and Family Planning Commission of Fujian Province for Youth Research Project (2015-2-49), the Scientific Research Foundation for Returned Scholars from Ministry of Human Resources and Social Security (2015142), and the Digestive Center of Xiamen University Foundation. Thanks to Dr. Xujin Wei for his great support.
Conflict of interest
The authors declare that they have no competing interests.
