Abstract
Cancer has become a leading cause of morbidity and mortality in recent years. Its high prevalence has had a severe impact on society. Researchers have achieved fruitful results in the causative factors, pathogenesis, treatment strategies, and cancer prevention. Semaphorin 3F (SEMA3F), a member of the signaling family, was initially reported in the literature to inhibit the growth, invasion, and metastasis of cancer cells in lung cancer. Later studies showed it has cancer-inhibiting effects in malignant tumors such as breast, colorectal, ovarian, oral squamous cell carcinoma, melanoma, and head and neck squamous carcinoma. In contrast, recent studies have reported that SEMA3F is expressed more in hepatocellular carcinoma than in normal tissue and promotes metastasis of hepatocellular carcinoma. We chose lung, breast, colorectal, and hepatocellular carcinomas with high clinical prevalence to review the roles and molecular mechanisms of SEMA3F in these four carcinomas. We concluded with an outlook on clinical interventions for patients targeting SEMA3F.
Keywords
Introduction
Malignant tumors have a major impact on human health, with a relatively high mortality rate [1, 2]. Therefore, research, prevention, and treatment of malignant tumors have become a global concern. According to the data reported in 2020, China accounts for 24% of new cancer diagnoses and 30% of cancer-related deaths worldwide [3]. Cancer is expected to become the world’s leading cause of death around 2060 [4]. Incidence and mortality rates of lung, breast, liver, and colorectal cancers are among the highest in all studies and reports [1]. Current research on the molecular aspects of these four clinically prevalent cancers is focused on oncogenes and anti-oncogenes [5, 6, 7]. Semaphorin 3F (SEMA3F) was first identified as an anti-oncogene in lung cancer and has since been linked to the growth, invasion, and metastasis of these four clinically prevalent cancers in several studies [8, 9, 10, 11, 12]. It has also been reported that the oncogenic effect of SEMA3F may be related to its receptors, Neuropilins protein and Plexins protein [13, 14]. The article discusses the regulatory role of SEMA3F and related receptors in cancer and their molecular mechanisms.
Search strategy
We obtained 1257 records by entering keywords through the electronic database. Duplicate records were removed using EndNote software. Next, we removed articles that were not relevant to the study content by browsing titles and abstracts, and finally included 76 articles by full-text evaluation (Fig. 1).
Workflow for literature search and study selection.
Semaphorins (SEMAs) were initially proposed as proteins secreted by axons of the central nervous system [15]. However, later studies revealed that these proteins play a role in tumor growth, angiogenesis, and cell metastasis and are involved in immunoreaction [16, 17, 18, 19]. SEMA3 is the only group of secreted proteins among vertebrate signaling proteins and is classified into seven subclasses (A–G). Studies have shown that SEMA3 functions in neuromodulation, tumor microenvironment, cardiovascular development, and bone homeostasis [16, 20, 21, 22, 23, 24]. In addition, SEMA3A inhibits tumor progression by suppressing tumor angiogenesis, such as breast, lung, and prostate cancers [25, 26, 27, 28, 29]. However, studies analyzing clinical samples from patients with rectal and pancreatic cancers have shown that high expression of SEMA3A is associated with poor patient prognosis [30, 31, 32]. SEMA3B has been more frequently studied in lung and breast cancers, where it inhibits tumor progression by inhibiting cancer cells [33, 34]. It has also been reported that SEMA3B has tumor-suppressive effects on gastric and liver cancers [35, 36]. SEMA3C mainly promotes tumor progression, such as promoting the growth of cervical cancer and accelerating the growth, invasion, and metastasis of prostate cancer [37, 38, 39]. In addition, it promotes the invasion of breast and stomach cancer [40, 41] SEMA3D has received less attention in cancer research. SEMA3D binds to membrane associate protein A2 (ANXA2) in pancreatic cancer to promote cancer cell invasion and metastasis [42, 43]. SEMA3E mainly binds to its receptor PlexinD1 to promote cancer progression [44, 45]. For example, SEMA3E-PlexinD1 accelerates the invasion and metastasis of colorectal cancer, endometrial cancer, and the proliferation and metastasis of gastric cancer [46, 47, 48]. SEMA3G has been little studied in cancer and has been reported to inhibit the metastasis and invasion of glioma cells [49]. SEMA3F, a member of class III signaling, has been widely recognized for its antitumor effects and was first reported to inhibit lung cancer progression as an antitumor factor that inhibits tumor cell growth and metastasis by competing with VEGF for the binding of NRP2 [50, 51, 52]. What’s more, in a later study of clinical samples, SEMA3F expression was reduced in patients with lung, breast, colorectal, prostate cancer, and osteosarcoma and often had a poor prognosis [9, 53, 54, 55, 56]. Its role in inhibiting tumor progression has also been found in recent studies of oral squamous cell carcinoma, esophageal squamous carcinoma, and head and neck squamous carcinoma [57, 58, 59]. In vitro studies have shown that SEMA3F inhibits lung, breast, and colorectal cancer growth, invasion, and metastasis by various mechanisms [12, 51, 60, 61, 62, 63]. After summarizing the in vivo and in vitro studies, we found that SEMA3F inhibited the progression of the lung, breast, and colorectal cancers and could be used as an anti-oncogene to focus our research. In contrast, a recent study showed that SEMA3F promotes metastasis of cancer cells in hepatocellular carcinoma [11]. Hence, further research into the specific regulatory mechanisms of SEMA3F in lung, breast, colorectal, and hepatocellular carcinomas is warranted.
SEMA3F-related receptors
Neuropilins include NRP1 and NRP2, a transmembrane protein originally reported as a mediator of axon guidance in the signaling family [64, 65]. NRP1 is located on chromosome 10 and NRP2 on chromosome 2 [66] closely related to angiogenesis, tumor growth, and viral transmission [67, 68, 69, 70]. NRP, as a receptor, can bind to the signaling family and the angiogenic factor VEGF family [71, 72, 73]. The SEMA3F-specific binding receptors are mainly NRP2 [74]. Recently, it was found that the binding of NRP as a receptor with VEGF can promote tumor angiogenesis, while NRP2 and SEMA3F can prevent tumor angiogenesis [18, 75]. In addition, SEMA3F is an inhibitory ligand for NRP2, which regulates NRP2 expression [76, 77]. The reduced expression of NRP2 reduces its ability to bind to VEGF, further preventing tumor angiogenesis.
Plexins are a large class of transmembrane proteins whose family consists of nine members (A1, A2, A3, A4, B1, B2, B3, C1, and D1) [78]. It is reported to be the only known transmembrane receptor that interacts directly with small intracellular GTPases [79]. Plexins are surface receptors of SEMA, and their binding is related to cell adhesion and migration. Except for SEMA3E, which binds directly to plexins, all other members of the SEMA3 family are known to bind first to Neuropilins, which then bind to PlexinA or PlexinD to form a complex [21]. The molecular mechanism of the biological function of SEMA3F as an anti-cancer gene is the SEMA3F/NRP2/PlexinA complex, in which NRP2 acts as a binding receptor and PlexinA as a signal transduction element [13, 14].
SEMA3F may suppress lung cancer growth, metastasis and invasion
Lung cancer is the leading cancer with a high risk and death rate [1, 80]. Initially, a high frequency of 3p21.3 deletion was found in the early stage of lung cancer patients. Further studies revealed that this deletion sequence had a high tumor suppressive effect, and a related fragment was isolated from lung cancer strains and named SEMA3F [81]. Next, we investigated the expression of SEMA3F in lung tissues and found that SEMA3F was expressed in normal human lung tissues in the cytoplasmic membrane and cytoplasm. However, it was only expressed in the cytoplasm of cells in lung cancer tissues, and its expression was reduced. This low expression in lung cancer tissues was often associated with the high aggressiveness of the tumor [82, 83]. Also, in clinical samples, elevated expression of NER and VEGF was found in tissues with low SEMA3F expression [83, 84]. At the same time, NRP is a co-receptor for SEMA3F and VEGF [85]. The combination of SEMA3F and NRP inhibits the expression of VEGF, thereby suppressing angiogenesis and inhibiting tumor proliferation. In addition, H157 cells (non-small cell lung cancer cells) stably expressing SEMA3F and H157 cells not expressing SEMA3F were injected into the lungs of mice, and its showed that the survival days of mice injected with cancer cells stably expressing SEMA3F were significantly greater than that of mice without SEMA3F expression [8]. These findings demonstrate that SEMA3F inhibits the progression of lung cancer and improves the prognosis of patients.
At the molecular level, the results show that SEMA3F can down-regulate integrin levels [8]. There is a consensus that integrins promote cancer development by multiple mechanisms, accelerating cell migration and invasion, leading to accelerated progression of many cancers [86, 87, 88]. In addition, integrins can also promote VEGF expression, accelerate angiogenesis, and lead to tumor cell proliferation [89, 90]. The decrease in integrin levels undoubtedly plays a key role in inhibiting lung cancer progression.
In lung cancer signal transduction, SEMA3F attenuates integrin ligase (ILK), leading to decreased phosphorylation of ERK and decreased phosphorylation of AKT, leading to decreased AKT/HIF-1
It was reported that ZEB-1 could suppress SEMA3F expression in lung cancer cells by directly binding to the conserved E-box site at the 5’ end of the gene [96]. Zinc finger E box binding homology box (ZEB)-1 is a major transcriptional repressor of E-cadherin in cancer [97, 98]. E-cadherin plays a crucial role in inhibiting cancer cell metastasis by enhancing intercellular adhesion and reducing the metastasis of cancer cells. In addition, loss of E-cadherin is a significant feature of tumor mesenchymal-epithelial transition (EMT), which plays a key role in tumor invasion and metastasis by enhancing cell motility and aggressiveness [99]. ZEB-1 inhibits the expression of E-cadherin, which accelerates the metastasis and invasion of cancer, and SEMA3F, which accelerates the progression of lung cancer. These findings suggest that ZEB-1 could be a promising therapeutic target in future lung cancer research and development.
In conclusion, previous study results demonstrate that SEMA3F has a strong tumor-suppressive effect on lung cancer from several aspects [8, 51]. Furthermore, it can inhibit lung cancer growth, metastasis, and invasion. This prompts us to consider SEMA3F a key breakthrough point in basic and clinical research and targeted drug development in lung cancer. SEMA3F can also be considered a diagnostic and monitoring indicator in diagnosing and managing lung cancer. Of course, the previous results are rigorous and plausible, but constructing a knockout mouse model of SEMA3F may round out the study.
SEMA3F may suppress breast cancer growth, metastasis and invasion
As one of the most dangerous diseases for women’s health [100, 101], studying the therapeutic targets of breast cancer is particularly important. The role of SEMA3F as an anti-oncogene was first reported in lung cancer [81]. Later studies reported a high frequency of deletion in the 3p21.3 region in breast cancer patients and demonstrated the presence of SEMA3F in the deletion region [102] suggesting that SEMA3F may also have an anti-oncogenic role in breast cancer. In in vitro cellular assays, the results showed that SEMA3F significantly inhibited the ability of breast cancer cells to move and spread, leading to a decrease in the migration ability of cancer cells [63, 103] and we also demonstrated that SEMA3F inhibited the invasion of breast cancer cells [12]. Furthermore, in a later study of SEMA3F and its receptors in mammary hyperplasia, mammary carcinoma in situ, and invasive mammary carcinoma, it was discovered that the expression of SEMA3F. Its receptors Plexin-A1 and Plexin-A3 in the epithelium decreased with the severity of the lesion, and NRP1 decreased in invasive carcinoma, while NRP2 did not change [54]. Based on the above in vivo and ex vivo findings, we confirmed that SEMA3F inhibited cancer progression in breast cancer. It could inhibit the growth, metastasis, and invasion of cancer cells. Therefore, increasing the expression of SEMA3F could improve the prognosis of patients.
A mechanistic study found that retinoid orphan nuclear receptor alpha (ROR
Previous studies have shown that SEMA3F binds specifically to the receptor NRP2 to inhibit cancer progression. While in breast cancer studies, SEMA3F was also found to bind to NRP1 to exert anti-cancer effects [103]. It has become a consensus that NRP1 activation in breast cancer promotes cancer growth, metastasis, and invasion. NPR1 is an independent marker of poor prognosis in breast cancer patients [108]. Its activation promotes MAPK pathway and NF-
SEMA3F may suppress colorectal cancer growth, metastasis and invasion
In recent years, the prevalence of colorectal cancer has been increasing year by year due to the great abundance of material resources and the complexity of dietary behavior of the population [112, 113, 114]. Similarly, the tumor suppressive effect of SEMA3F exists in colorectal cancer, where it specifically binds to NRP2 to inhibit tumor growth and metastasis [60]. Notably, in studies using lung and colorectal cancer cell lines, SEMA3F was a target gene of p53, which negatively regulates tumor angiogenesis through the SEMA3F-NRP2 pathway [61] P53, a key anti-oncogene, has been studied and reported since its discovery to induce cell cycle arrest, apoptosis, and DNA repair [115]. SEMA3F acts as a target gene of p53 against tumor angiogenesis which is valuable in inhibiting tumor progression and improving patient prognosis. Furthermore, it has been demonstrated that NRP2 induces lymphangiogenesis in the colorectum without SEMA3F by activating integrin
SEMA3F regulates information table in breast, lung, colorectal and hepatocellular carcinomas
SEMA3F regulates information table in breast, lung, colorectal and hepatocellular carcinomas
Mechanism diagram of SEMA3F regulating breast, lung, colorectal and hepatocellular carcinomas. (Created with BioRender.com).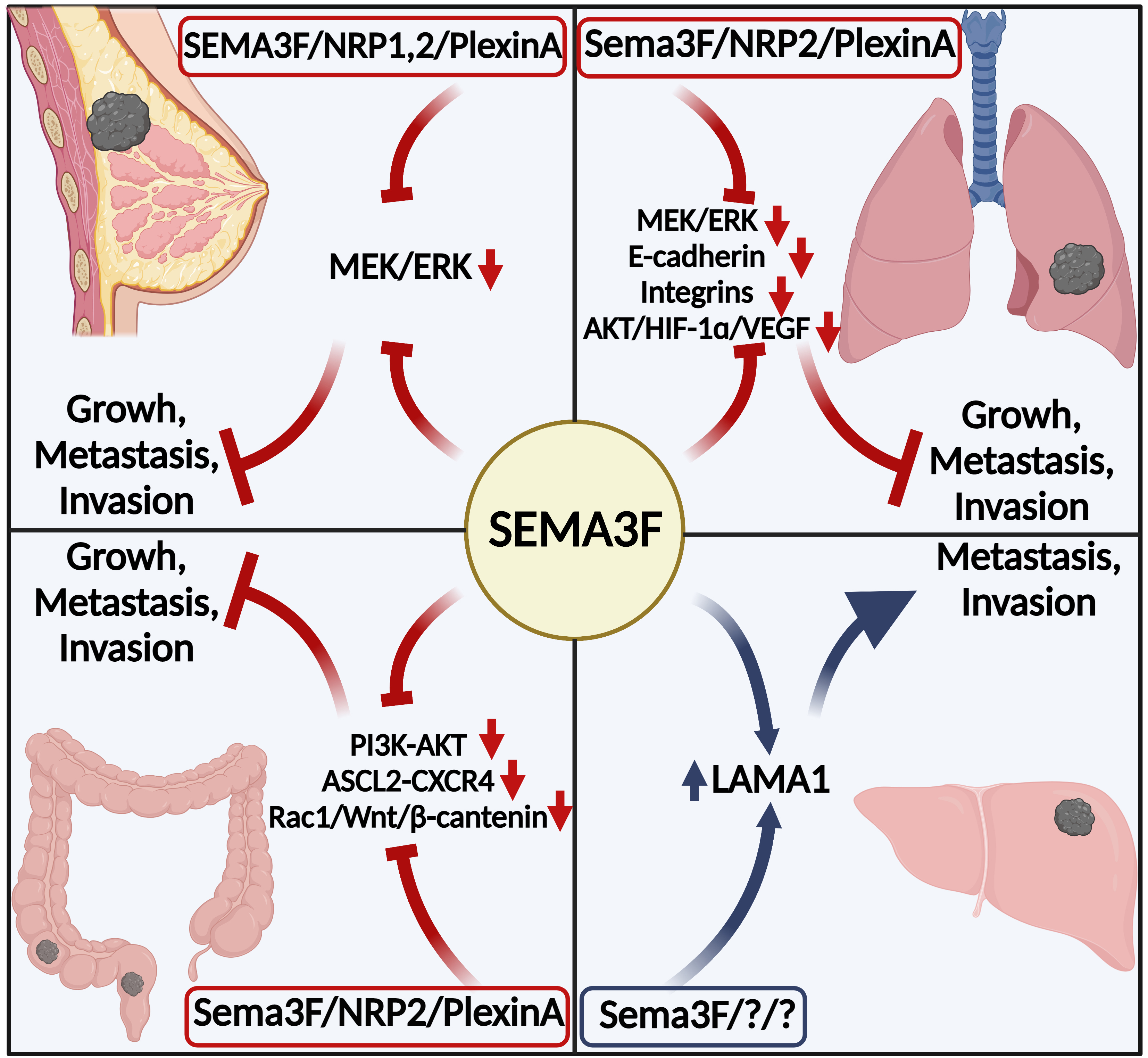
The mechanism of SEMA3F inhibition in colorectal cancer has been revealed. It has been reported that SEMA3F inhibits colorectal invasion and metastasis by downregulating the ASCL2-CXCR4 axis through the PI3K-AKT pathway [9] The transcription factor ASCL2, a regulator of colon cancer progenitor cells, down-regulates the inhibition of colorectal cancer progression, including cancer cell proliferation, metastasis, and invasion [116, 117]. In addition, the chemokine receptor CXCR4 has the property of inducing cancer cell chemotaxis, invasion, and proliferation [118, 119]. ASCL2 can upregulate the expression of CXCR4 in colorectal cancer [120]. Therefore, inhibiting the activity of ASCL2 further inhibits the progression of colorectal cancer. The PI3K-AKT pathway regulates cell proliferation, growth, and metabolism and is commonly and abnormally activated in cancer patients. PI3K or AKT phosphorylation inhibition can inhibit cancer progression [121, 122]. Inhibition of the PI3K/AKT pathway by SEMA3F inhibited cancer progression, while downregulation of PI3K/AKT led to a decrease in ASCL2/CXCR4 expression. This cascade of responses led to a severe inhibition of colorectal cancer progression, which was certainly attributed to SEMA3F. In addition, we have verified in clinical patient specimens that low expression of SEMA3F, high expression of ASCL2, and high expression of CXCR4 in colorectal cancer patients often represent a poor prognosis for the patients [9]. This affirms the presence of SEMA3F as an anti-oncogene in colorectal cancer, providing more options for treating colorectal cancer and a basis for developing targeted therapies.
Modulating the stemness of cancer cells has now become a breakthrough point to inhibit cancer progression. A study claims that SEMA3F can inhibit the stemness of rectal cancer cells by inactivating Rac1 [123]. Stemness of cancer cells means their potency to act as stem cells, having tumorigenic abilities, including independent self-renewal and multi-differentiation [124]. Increased stemness of cancer cells may promote carcinogenesis, metastasis, and invasion, leading to accelerated cancer progression and recurrence [125, 126]. Rac1, a member of the Rho family, is involved in physiological activities such as cell adhesion, proliferation, migration, and motility [127]. Furthermore, Rac1 promotes cancer angiogenesis, invasion, and metastasis [128]. Therefore, inhibition of Rac1 activity by SEMA3F necessarily stops the progression of colorectal cancer. In addition, overexpression of SEMA3F in colorectal cancer cells inhibits the Wnt/
Hepatocellular carcinoma, one of the most lethal cancers, is often associated with viral hepatitis, alcohol abuse, cirrhosis, and other factors [130, 131, 132]. Antitumorigenic properties of SEMA3F have been reported in various cancers, including lung, breast, and colorectal cancers The role of SEMA3F in hepatocellular carcinoma was rarely reported until recently, when bioinformatics analysis of hepatocellular carcinoma samples revealed that SEMA3F expression was upregulated in hepatocellular carcinoma and that SEMA3F expression was positively correlated with metastasis and pathological stage of patients, and that patients with high SEMA3F expression tended to have a poor prognosis [11, 133, 134, 135]. This is inconsistent with our review of the international literature on SEMA3F as an anti-oncogene for other types of cancer. However, most of these findings are based on bioinformatics analysis, and more basic research is required to prove them. NRP2 acts as an affinity receptor for SEMA3F. In a previous report, NRP2 was highly expressed in cancer tissues, and the results in hepatocellular carcinoma tissues were the same as before [11]. This differs from previous reports that elevated SEMA3F expression can downregulate NRP expression. However, existing research is insufficient to explain the mechanism.
Regarding molecular mechanism, SEMA3F upregulates LAMA1 expression in hepatocellular carcinoma [11]. Laminin alpha 1 (LAMA1) is a major component of the extracellular matrix (ECM), which is involved in metastasis and invasion of cancer cells and promotes cancer development in gastric, colorectal, esophageal, and ovarian cancers [136, 137, 138, 139]. Therefore, activation of LAMA1 expression by SEMA3F in hepatocellular carcinoma accelerates cancer metastasis. This reveals that patients with hepatocellular carcinoma with high SEMA3F expression in clinical practice often have a poor prognosis.
There are very few reports on SEMA3F in hepatocellular carcinoma. Hence, we cannot thoroughly analyze its role and regulatory mechanism, and we hope that more studies will follow. Nevertheless, these few reports are significant and break our original knowledge. Furthermore, SEMA3F does not inhibit tumorigenesis in all tumors, generating new insights for the subsequent research and treatment of hepatocellular carcinoma.
Conclusion and prospect
Cancer has affected human health at many levels, causing physical and mental pain to patients and severe economic burdens to families and society. Cancer has clearly become a problem that must be solved for medical progress to continue. We are looking forward to the emergence of more effective treatments. However, we are looking forward to researching targeted drugs at this stage. Targeted therapy is the latest trend in cancer treatment and is emerging as a new treatment strategy that significantly improves the efficiency of cancer treatment and the prognosis of patients. The role of SEMA3F in inhibiting tumor cell growth, invasion, and metastasis exhibited in several malignancies should be of strong interest to us, especially in the lung, breast, and colorectal cancers where the molecular mechanisms are quite well-defined. Although the existing studies cannot fully and concretely explain the mechanism of SEMA3F regulation of tumors, they can be used to develop and test targeted drugs that induce the expression of positive SEMA3F regulators and inhibit the expression of negative regulators, increasing SEMA3F expression. In addition, we can use SEMA3F as a biomarker for clinical diagnosis and patient prognosis assessment by measuring the expression of SEMA3F in tissues or in serum. Notably, most of the existing studies reported that SEMA3F binds to the receptor NRP2 to inhibit tumor angiogenesis, and only in colorectal cancer was SEMA3F mentioned to inhibit cancer lymphangiogenesis. As lymph node metastasis is one of the major pathways of cancer metastasis, we hope that more studies will explore the mechanism of SEMA3F in tumor lymphangiogenesis in depth. Further research and evidence are needed to verify the latest report of SEMA3F promoting tumor progression in hepatocellular carcinoma. Our report summarizes SEMA3F regulation of clinically high-incidence (breast, lung, colorectal, and liver with decreasing incidence order) cancers and their associated molecular mechanisms based on existing research (Fig. 2 and Table 1). Our report may be insufficient, but we look forward to reporting more findings.
Footnotes
Acknowledgments
First and foremost, the authors would like to express their gratitude to all Section colleagues who have investigated and reported on the SEMA3F gene. I appreciate Guangxi Medical University’s Regenerative Medicine Laboratory and my instructors Prof. Qian Liu and Prof. Dezhi Song for providing the research environment.
Conflict of interest
Authors have no conflicts of interest to report.
Author contributions
Conception: Qian Liu.
Interpretation or analysis of data: Chaofeng Wang. Preparation of the manuscript: Chaofeng Wang. Revision for important intellectual content: Qian Huan, Dezhi Song. Supervision: Dezhi Song.
