Abstract
BACKGROUND:
The incidence of thyroid cancer has risen rapidly over the last decades. Although mortality rates are relatively low compared to other cancers, the rate of new cases started to increase in the early 2000s. While tumor suppressors and oncogenes were recently identified in thyroid cancer, the potential roles of these genes in thyroid cancer remain unclear.
OBJECTIVE:
Analyze the roles and functions of tumor suppressors and oncogenes in thyroid cancer.
METHODS:
Thyroid cancer data were collected from public databases, such as the UCSC Xena database of TCGA thyroid cancer, TISIDB, and UALCAN. The genes frequently associated with unfavorable thyroid cancer were examined and validated. The association of these target genes with thyroid tumorigenesis, stages, subtypes, and survival rates were analyzed. Additionally, the genes aberrantly expressed in thyroid cancer and significantly involved in thyroid tumorigenesis, stages, subtypes, and survival rates were identified.
RESULTS:
Female sex was identified as a risk factor for thyroid cancer. The expression of PAPSS2, PDLIM3, COPZ2, ALDH1B1, ANTXR1, GUF1, and SENP6 negatively correlated with thyroid cancer prognosis.
CONCLUSION:
Female sex was a risk factor for thyroid cancer. In addition, our analysis suggested that PAPSS2, PDLIM3, COPZ2, ALDH1B1, ANTXR1, GUF1, and SENP6 are negatively correlated with the prognosis of thyroid cancer. The expression of ANTXR1, GUF1, and PDLIM3 was weakly associated with thyroid cancer’s immune and molecular subtypes.
Introduction
The incidence of thyroid cancer is rapidly increasing in the United States. According to the National Cancer Institute (NCI) report, there were around 915,664 individuals with thyroid cancer in the United States in 2019, and an
The 5- and 10-year trends of thyroid cancer during the last decade
The 5- and 10-year trends of thyroid cancer during the last decade
Data source: SEER 22 2015–2019. By Sex, Delay-adjusted SEER Incidence Rate, All Races, All Ages, All Stages.
estimated 43,000 new cases will occur in 2022. Thyroid cancer is an abnormal growth of thyroid cells, forming a lump within the thyroid. Most of the thyroid nodules are non-cancerous (benign), and only 5% are cancerous. Based on the histological examinations, four types of thyroid cancer were identified: papillary, follicular, medullary, and anaplastic. As the most common type of thyroid cancer, papillary thyroid cancer (PTC) accounts for 90% of thyroid cancers and has a better prognosis than the other types [1]. Genes associated with thyroid cancer have been identified, including BRAF, RET/PTC, and RAS [2, 3, 4]. However, no significant association between these genes and PTC subtypes and sex was observed [5].
Thyroid cancer is one of the few cancers that are more common in females, with a 2.9-fold higher risk of developing thyroid follicular cancer in women than in men (female: male ratio 16.3:5.7) [6]. Based on SEER’s 2019 cancer prevalence proportions in 12 Areas and the 2019 US population estimates from the US Bureau of the Census, 85714 females (60%) and 31,673 males (30%) were living with cancer. The estimates included white individuals, including Hispanics, 65–74 years old, and less than 27 years since the initial diagnosis. Gender was associated with thyroid cancer incidence (Table 1), as well as aggressiveness and prognosis [7, 8]. Recent studies have reported that both chromosomal and gonadal factors contribute to sex bias in disease traits such as obesity, nonalcoholic fatty liver disease, cardiovascular diseases, diabetes, and COVID-19 infection [9, 10, 11, 12, 13]. It seems that sex chromosomes play a relatively more influential role in these age-dependent diseases because older individuals undergo menopause and reduced hormone production [14, 15]. Most sex chromosome effects result from X-chromosome inactivation (XCI) escape, whereby some genes escape from XCI during aging. XCI is the inactivation of one of the X-chromosomes in females to balance X dosage in males [18]. Some genes on the X chromosome can escape XCI under specific conditions, resulting in imbalanced gene expression between males and females [16, 17]. It has been proposed that the sex hormones and the X chromosome could be involved in the sex-associated thyroid cancer risk [19, 20]. However, the mechanisms underlying the male preponderance are not entirely known yet.
Recently, bioinformatics analysis using pan-cancer next-generation sequencing (NGS) database has been used for cancer research, which could provide more information about molecular features than conventional histological classification. To better understand the gene functions within the thyroid tumor and improve survival, we screened and explored the function of tumor suppressors and oncogenes, then used these genes to identify potential thyroid cancer predictor genes. We aimed to identify gene expression aberrations and interpret their significance on the growth and development of the thyroid tumor.
In this study, we performed a comprehensive bioinformatics analysis of thyroid cancer-associated genes to identify potential tumor suppressors and oncogenes associated with serous thyroid cancer. In addition, the expression data analysis provided specific information on the molecular basis and subtypes of thyroid cancer and elucidated the genes driving thyroid tumors.
Ethical statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (Seventh revision (2013)). The study was approved by the Ethics Committee of the Children’s Hospital of Fudan University, and informed consent was obtained from all patients.
Data source and pretreatment
The cancer stat facts of the National Institute of Health (National Cancer Institute) were used to obtain the thyroid cancer case and death numbers in the US, including the rates of new cases and deaths, 5-year relative survival, new cases by age, race/ethnicity, and sex.
The RNA sequencing (RNA-seq) and survival data were obtained from the UCSC Xena database of TCGA Thyroid Cancer (THYROID CANCER) (
TISIDB (
The UALCAN (
Distribution analysis of thyroid cancer
We searched for thyroid cancer in the NCI’s Annual Report to the Nation on the Status of Cancer and obtained the Cancer Stat Facts: Thyroid Cancer (
Association between oncogenes expression and thyroid cancer
The relationship of candidate oncogenes’ expression with thyroid cancer was analyzed using the database of human protein atlas, UALCAN, and the UCSC Xena web tool. The heat map, hierarchical clustering, and violin plots of oncogenes were created using R language by implementing the R packages “ggplot2”, “ggpubr”, and “survival” (with a cut-off
Statistical analysis
The difference in clinicopathological features among the three subtypes was analyzed using the Chi-square test. The differences in the expression levels among the three subtypes were determined using ANOVA. Two-group comparisons were performed using the student T-test. The Pearson correlation coefficient was used for correlation analysis. R was used for statistical analyses. Statistical significance was defined as
Results
Female sex is one of the risk factors for thyroid cancer. A. Estimated number of deaths and new thyroid and other cancer types in the US in 2019. B. The distribution of thyroid cancer cases over the clinical stages. C. 5-year relative survival rate among thyroid cancer stages (SEER 17 2012–2018, all Races, both sexes by SEER combined summary of stage). D. New cases in SEER 12. Death rates from the US Mortality sheet (All races, both sexes, age-adjusted), 1975–2019. E. The rates of thyroid cancer cases and death per 100,000 persons by age. F. Female-to-male ratios of new thyroid cancer cases based on ethnicity. G. Female-to-male ratios of the death rate among thyroid cancer cases based on ethnicity. H. Male and female thyroid cancer incidence and mortality from SEER 12. Death rates from the US Mortality sheet (All races, Age-Adjusted), 1956–2019. (Data source 
Patient samples information from the SEER database
Patient samples information from the SEER database
Data source: SEER 22 2015–2019, Age-Adjusted.
We examined the distribution of the most common cancers in the United States using the National Cancer Institute database. Thyroid cancer ranked 13
We also analyzed new and death cases as a function of age using 10-year bins. The ratio of new cases started to increase around the age of 20, reaching a peak of 21.1% between 45–65 years, then decreasing along with aging (green circle) (Fig. 1E and Table 2). In contrast, the percentage of death cases showed a distinct trend, keeping a base level during the early years and rapidly increasing between 45 and 54 years, with the highest level of 28.4% between 75–84 years (red square). These trends could be related to the menopausal transition, which most often begins between 45 and 55 years, indicating that menopause may be a detrimental factor for thyroid cancer (Fig. 1E). A sex imbalance in the overall survival (OS) and mortality from thyroid cancer patients has been reported worldwide [8, 22]. Interestingly, when analyzing the sex imbalance of thyroid cancer based on ethnicity, the female-to-male ratio of new cases per 100,000 persons was always larger than one in all ethnicities (Fig. 1F). We further analyzed the sex bias of death rate based on ethnicity, and the overall mortality rate was higher in females compared to male patients in almost all ethnicities except for Non-Hispanic Whites (Fig. 1G). Based on the 2017–2019 WHO data, the long-term global trend of thyroid cancer since 1955 was approximately 2 folds higher in women during their lifetime compared to men. Similarly, the female population had a higher mortality rate than males until 1996–2006, when the sex gap slowly narrowed and reversed. Indeed, the female mortality rate ratio declined from 1.1 in 1955 to 0.44 in 2017, and the male mortality rate increased from 0.36 in 1990 to 0.49 in 2017 (unknown reasons) (Fig. 1H).
Unfavorable prognostic genes in thyroid cancer. A. Lollipop plot displaying the unfavorable prognostic gene levels (vertical axis) and their position on the chromosomes. B–U. mRNA expression levels of candidate prognostic genes in normal and thyroid tumor samples. Normal, 
Selected thyroid cancer genes captured by the HPA database
Data source: Human Protein Atlas proteinatlas.org.
Previous studies suggested that the severity of thyroid cancer is associated with an increased level of prognostic genes [23, 24, 25]. Here, we referred to 185 genes associated with unfavorable prognosis in thyroid cancer in the human protein atlas database (
The levels of prognostic genes are dependent on the thyroid cancer
The expression of prognostic genes is dependent on thyroid cancer. A–G. Levels of ALDH1B1 (A), COPZ2 (B), ANTXR1 (C), GUF1 (D), SENP6 (E), PAPSS2 (F), and PDLIM3 (G) in Solid Tissue Normal normal, Primary Tumor Solid, and Metastatic samples based on thyroid tumor types. Solid Tissue Normal normal (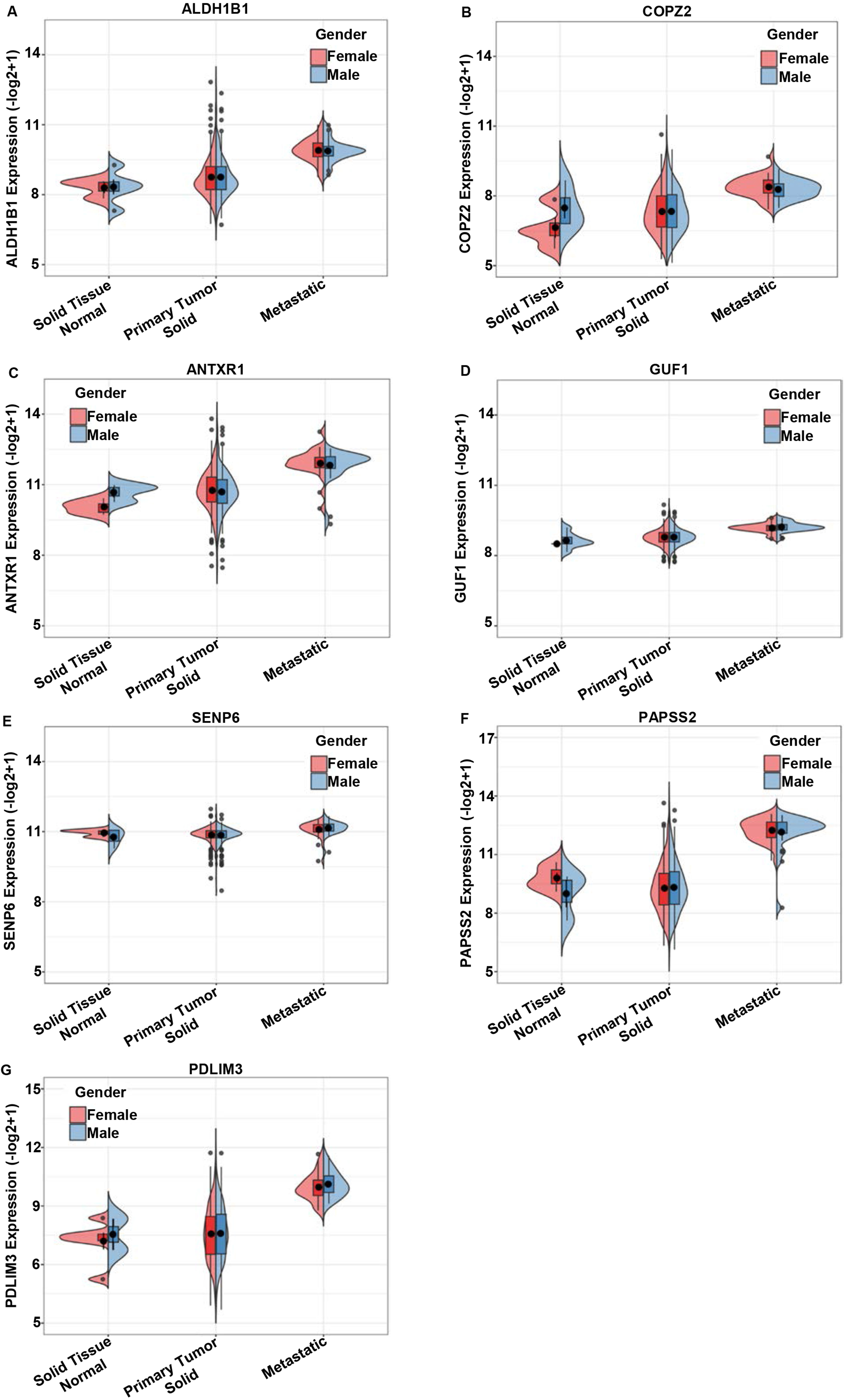
It has been reported that thyroid cancer had striking female predominance, and females have three- to four-fold higher incidence than males [26]. Gender differences in thyroid cancer are mostly confined to the detection of small PTCs, and the natural causes behind gender differences in thyroid cancer are likely due to mixed effects, including sex hormones, genetics, and the immune system [27]. These issues are often associated with thyroid disorders in female patients; however, little is known about whether prognostic genes display a sexual pattern in thyroid cancer patients. We sought to determine the expression differences of prognostic genes between male and female patients and whether thyroid tumor expands sex differences in gene expression. The top 7 genes, based on the
Then, we examined the sex-related difference in the expression of these prognostic genes (PAPSS2, PDLIM3, COPZ2, ALDH1B1, ANTXR1, GUF1, and SENP6) in normal solid tissue, primary tumor, recurrent tumor, and metastatic tumor (Fig. 3A–G). No significant difference was detected; the reason may be complicated, as these genes are all located on non-sex chromosomes. According to our observations, only genes that can escape XCI may show a sexually dimorphic expression pattern [28, 29, 30]. Even though thyroid tumors showed a dramatic sex difference in the occurrence and death rate, the hormones and life habits may contribute to this difference.
Prognostic gene expression in thyroid tumor stages. A–G. Levels of ALDH1B1 (A), COPZ2 (B), ANTXR1 (C), GUF1 (D), SENP6 (E), PAPSS2 (F), and PDLIM3 (G) in different stages of thyroid cancer. Normal (
The correlations of prognostic genes with the pathological stages of thyroid cancer were explored to examine their clinical relevance. Our results showed that the expression of ALDH1B1 could be a reliable predictor of thyroid tumor stages, showing significant differences among all 4 stages except Stage1 vs. stage3 (
Prognostic gene expression in thyroid tumor subtypes. A–G. Levels of ALDH1B1 (A), COPZ2 (B), ANTXR1 (C), GUF1 (D), SENP6 (E), PAPSS2 (F), and PDLIM3 (G) in different subtypes of thyroid cancer based on tumor histology. Normal (N) (
Determining the PTC subtype is critical in predicting patient prognosis and designing proper treatment plans. Using TCGA Thyroid Cancer data, we showed that, compared to the control tissues (N), patient tissues with classical thyroid papillary carcinoma (CTPC), tall thyroid papillary carcinoma (TTPC), follicular thyroid papillary carcinoma (FTPC), and other subtypes (O) exhibited significantly decreased expression of ALDH1B1 (Fig. 5A), ANTXR1 (Fig. 5C, GUF1 (Fig. 5D), SENP6 (Fig. 5E), PAPSS2 (Fig. 5F), and PDLIM3 (Fig. 5G) (
Additionally, the association of these prognostic genes among 6 immune and molecular subtypes was analyzed. The main clinical and molecular characteristics are depicted in Figs 5H–N as C1 (wound healing); C2 (IFN-gamma dominant); C3 (inflammatory); C4 (lymphocyte depleted); C5 (immunologically quiet); and C6 (TGF-b dominant). The results showed no significant difference within 5 types of immune and molecular subtypes (
Drugs targeting ALDH1B1 and ANTXR1 from the DrugBank database
Drugs targeting ALDH1B1 and ANTXR1 from the DrugBank database
Data source:
The association of prognostic genes expression with thyroid cancer survival. A–G. Kaplan-Meier survival curves comparing high and low expression of ALDH1B1 (A), COPZ2 (B), ANTXR1 (C), GUF1 (D), SENP6 (E), PAPSS2 (F), and PDLIM3 (G) in thyroid cancer. The high expression level of these genes is associated with unfavorable overall survival of thyroid cancer. n (high) 
We further assessed the association of the prognostic genes with the overall survival of thyroid cancer using the Kaplan-Meier curve (Fig. 6A–G). The analysis showed that shorter overall survival was significantly correlated with declined expression of ALDH1B1 (Fig. 6A), COPZ2 (Fig. 6B), ANTXR1 (Fig. 6C), PAPSS2 (Fig. 6F), and PDLIM3 (Fig. 6G) across the whole follow-up period. However, GUF1 (Fig. 6D) and SENP6 (Fig. 6E) did not affect the overall survival rate during the first 100 days. Decreased expression of these genes was associated with a lower survival rate. These observations suggest that the expression of these prognostic genes could be a detrimental factor affecting the survival of thyroid cancer.
The correlation between prognostic genes expression and immune microenvironment and modulators in thyroid cancer
Correlation Between Prognostic Genes expression and Immune Microenvironment and Modulators in Thyroid Cancer. A. Spearman correlation of prognostic gene expression with immune infiltrating cells in the Tumor-Immune System Interaction Database (TISIDB). B. Spearman correlation of prognostic gene expression with chemokine expression in TISIDB. (Data source 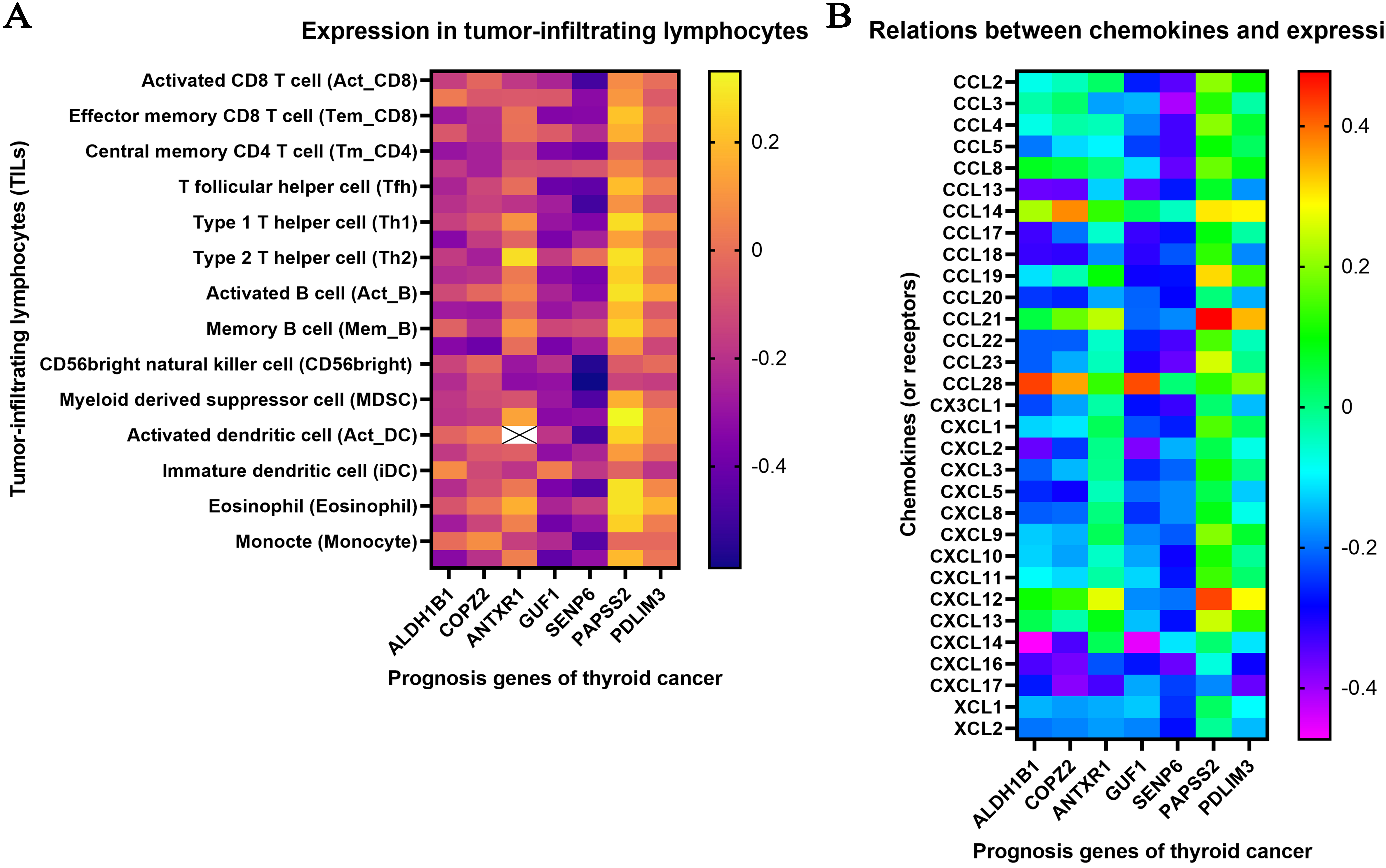
The immune system plays a critical role in thyroid cancer progression [31, 32, 33]. To further assess the relationship between the expression of prognostic genes in thyroid cancer and the immune microenvironment, we explored the correlations between these prognostic genes and tumor-infiltrating lymphocytes (TILs) using the TISIDB database. As shown in Fig. 7A (Columns 6 and 7), PAPSS2 and PDLIM3 expression is correlated with multiple types of TILs. Moreover, a negative correlation existed between diverse TILs and GUF1 and SENP6 expression (Fig. 7A Columns 4 and 5). These findings suggest that PAPSS2 and PDLIM3 might be associated with immune stimulation and inhibition. To further evaluate the relationship between chemokines and these prognostic genes, we evaluated their co-expression with immunomodulators (Fig. 7B). The expression of ANTXR1, PAPSS2, and PDLIM3 significantly increased compared to the rest of the candidate genes (Fig. 7B Column 6 and 5). However, the chemokines showed lower correlations with ALDH1B1, COPZ2, GUF1, and SENP6 expression.
Altogether, these results suggest that the potential roles of PAPSS2 and PDLIM3 in thyroid cancer are likely in an immune system-dependent manner.
Potential anticancer drugs for thyroid cancer. A–B. Prediction of drugs targeting ALDH1B1 and ANTXR1 from the DrugBank database. C. The structure of coenzyme NADH. (Data source 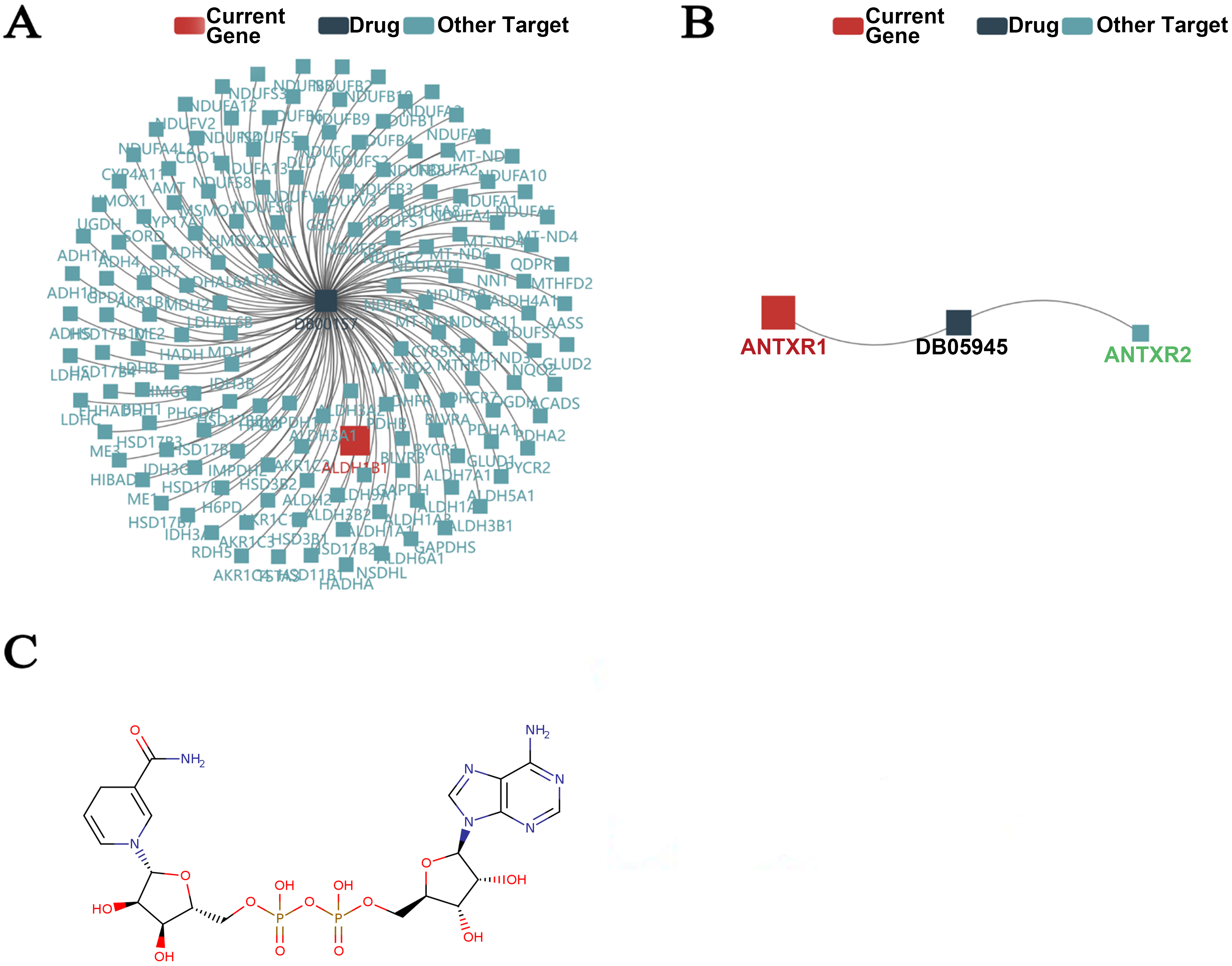
Kinase inhibitors, such as Lenvatinib (Lenvima), sorafenib (Nexavar), and cabozantinib (Cabometyx) [34, 35, 36, 37], are the most common drugs used to treat thyroid cancer. New drugs are less needed unless those treatments are ineffective or may cause severe side effects. To explore the potential mechanisms involved in thyroid cancer carcinogenesis, we used the TISIDB online tool to predict the targets of drugs collected from the DrugBank database. The results are shown in Fig. 8A and Table 4. DB00157 (NADH) has a direct interaction with ALDH1B1. On the other hand, DB05945 (MDX-1303) targeted ANTXR1 only. As a coenzyme composed of ribosyl nicotinamide 5’-diphosphate, NADH could couple to adenosine 5’-phosphate by pyrophosphate linkage (Fig. 8B and C). Some evidence suggests that NADH might be useful in treating Parkinson’s disease, chronic fatigue syndrome, Alzheimer’s disease, and cardiovascular disease. At the same time, MDX-1303 (Protein Chemical Formula unavailable) is a fully human antibody against inhalation anthrax, the most lethal form of illness in humans caused by the Bacillus anthracis bacterium. It targets the anthrax protective antigen, a protein component of these lethal toxins.
These results would accelerate the understanding and future development of drugs against thyroid cancer.
Discussion
Thyroid cancer represents 2.3% of all new cancer cases in the US, and its incidence has risen faster over the last decades [38, 39]. This study analyzed thyroid cancer burden using data from public databases and explored the associated prognosis genes expression with thyroid tumorigenesis, stages, subtypes, and survival rate. Consistent with previous studies, we identified the female sex as a risk factor for thyroid cancer. Women were overrepresented and had a higher incidence than males in both new cases and death rates (Fig. 1F and G). A sex difference in incidence and mortality was particularly noticeable in the last 50 years (Fig. 1H).
We screened genes with the most significant association with thyroid cancer. Out of 20 genes, 7 were identified based on their fold change and
The “female favorable” phenotype of thyroid cancer could be partly related to the higher induction of inflammatory factors and earlier activation of adaptive immunity by sex hormones. Previous clinical data have shown that the fluctuation of sex hormones may cause gender disparity in papillary thyroid cancer. Indeed, the peak incidence of PTC has been observed in women aged 40–49 years, around menopause [40, 41]. Estrogen is linked to a higher rate of thyroid cancer development in women; therefore, female sex hormones may play an important role in thyroid cancer development [42, 43, 44]. Consistent with these reports, our results showed a rapid increase in death cases around the age of 40–55 (Fig. 1E), while new cases remained stable between the ages of 40–60 years, then decreasing after that (Fig. 1E). Some studies also suggested that the X chromosome inactivation (XCI) theory, the higher expression level of X chromosome genes in women with 2 X chromosomes, can contribute to sex-specific phenotypes with more robust immune activity in many disorders including thyroid diseases and cancer [45, 46, 47]. Another unneglectable factor is the difference in lifestyle and thyroid cancer risk factors between male and female populations, such as BMI, physical activity, and tobacco smoking status. However, the association of hormones (e.g., estrogen), lifestyle, and XCI with thyroid cancer is inconclusive. None of the genes in our analysis showed a sex difference pattern between healthy tissues and tumor subtypes, which could be because these genes are not located on the X chromosome (Fig. 2A). Moreover, the function of these genes is not associated with immune processes.
Recently, Ljubic, et al. reported a set of genes and comorbidities associated with thyroid cancer based on PubMed and the HCUP SID California database. The authors identified five genes from PubMed analysis, including BRAF, RET, SLC5A5, RAS, and PTEN, and five genes using DisGeNET, including BRAF, RET, KRAS, NRAS, and PRKAR1A. However, genes that involve mutations include only BRAF (V600E) and NRAS(Q61R). These genes are generally oncogenic in various cancers, including thyroid cancer [48].
Our analysis supports the fact that the dysregulation of these prognostic genes is closely correlated with thyroid cancer malignancies. However, it should be mentioned that our study had some limitations to consider when interpreting the data. While the data from public databases and websites may validate each other, there could be some duplicates, and the sample size is insufficient. Therefore, the conclusion from sex difference patients requires further validation. We also did not have a chance to get the information on circulating sex hormone levels (estrogen and testosterone) from the database. Therefore, the contribution of the gonadal hormones to the thyroid cancer sex difference remains unknown. Of note, the conclusion drawn from this study is limited to the current information and data, and the conclusion may change as more data are collected in the future.
Conclusion
The present study investigated the expression variation of 7 prognostic genes in relation to sex differences, stages, and subtypes of thyroid cancer. We also analyzed the correlation of these genes in tumor immunology and their effect on the overall survival rate. Finally, we proposed potential anticancer drugs for thyroid cancer from the DrugBank database. While our study provided inconclusive results regarding the reproductive and risk factors of thyroid cancer, it may help explain the genetic susceptibility to thyroid cancer in the future.
Ethics approval and consent to participate
This study was approved by the clinical trial ethical committee of the Children’s Hospital of Fudan University. Following the approval of the ethical committee, all the methods and information in this study were performed according to the relevant guidelines and regulations.
Author contributions
Conception: Yue Huang, Yaoxin Wang, Wen-Xia Chen, and Zhengmin Xu.
Interpretation or analysis of data: Yue Huang and Yaoxin Wang.
Preparation of the manuscript: Yue Huang and Yaoxin Wang.
Revision for important intellectual content: Sining Liu, Wen-Xia Chen, and Zhengmin Xu.
Supervision: Zhengmin Xu and Wen-Xia Chen.
Funding
Not applicable.
Availability of data and materials
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author(s).
The datasets presented in this study can be found with the following database freely. UCSC Xena (
Code availability
Code used throughout this study is available upon reasonable request from the corresponding author(s).
Consent for publication
Not applicable.
Disclosure
Not applicable.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230029.
sj-docx-1-cbm-10.3233_CBM-230029.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-230029.docx
Footnotes
Acknowledgments
Not applicable.
Conflict of interest
The authors have declared that no conflict of interest exists.
